Abstract
Objective
Articular cartilage is incapable of effective repair following injury or during osteoarthritis. While there have been developments in cartilage repair technologies, there is a need to advance biologically relevant models for preclinical testing of biomaterial and regenerative therapies. This study describes conditions for the effective ex vivo culture of the whole human femoral head.
Design
Fresh, viable femoral heads were obtained from femoral neck fractures and cultured for up to 10 weeks in (a) Dulbecco’s modified Eagle’s medium (DMEM); (b) DMEM + mixing; (c) DMEM + 10% human serum (HS); (d) DMEM + 10% HS + mixing. The viability, morphology, volume, and density of fluorescently labelled in situ chondrocytes and cartilage surface roughness were assessed by confocal microscopy. Cartilage histology was studied for glycosaminoglycan content using Alcian blue and collagen content using picrosirius red.
Results
Chondrocyte viability remained at >95% in DMEM + 10% HS. In DMEM alone, viability remained high for ~4 weeks and then declined. For the other conditions, superficial zone chondrocyte viability fell to <35% at 10 weeks with deeper zones being relatively unaffected. In DMEM + 10% HS at 10 weeks, the number of chondrocytes possessing cytoplasmic processes increased compared with DMEM (P = 0.017). Alcian blue labeling decreased (P = 0.02) and cartilage thinned (P ≤ 0.05); however, there was no change to surface roughness, chondrocyte density, chondrocyte volume, or picrosirius red labeling (P > 0.05).
Conclusions
In this ex vivo model, chondrocyte viability was maintained in human femoral heads for up to 10 weeks in culture, a novel finding not previously reported. This human model could prove invaluable for the exploration, development, and assessment of preclinical cartilage repair and regenerative therapies.
Introduction
Articular cartilage has very poor regenerative potential following injury and the repair tissue formed is mechanically weak, and has a fibro-cartilaginous, rather than the resilient load-bearing extracellular matrix (ECM) of hyaline cartilage. 1 Furthermore, the native regeneration potential of cartilage declines with age 2 and while fibro-cartilaginous repair can be observed to form within injured hyaline cartilage, it is more evident when the injury has penetrated the subchondral bone.1,3 The reasons for the production of mechanically incompetent repair tissue are not well understood, and clearly there is intense interest in developing more effective biomaterial and regenerative therapies for cartilage repair. However, current models are not optimal and usually involve preparations ranging from in vitro cultures of cells, through to osteochondral explants 4 and ex vivo and in vivo models of mainly animal (i.e., nonhuman) joints.5,6 The research and development of more effective cartilage repair and regenerative therapies would be enhanced by the ability to preclinically evaluate novel strategies in ex vivo physiological, tribological models of natural joints.
While there have been many detailed studies on explant and organ culture of animal osteochondral tissue (e.g., Lin et al. 7 ), few studies have conducted experiments directly on human tissue. The limited studies using human cartilage for experimentation have obtained the material as discarded, and frequently degenerate osteochondral tissue (e.g., during joint replacement surgery for osteoarthritis). 8 To test orthobiological treatments for human cartilage repair, the use of healthy cartilage is essential. However, this source of material is difficult to obtain and relies on collaboration between clinicians, theatre staff, and research scientists. While normal human cartilage may be obtained from amputations, trauma victims, or occasionally cadavers, such material is rarely available for wider adoption into experimental human cartilage research. Ex vivo organ culture is becoming increasingly important for basic and applied biomedical research because it is more representative of normal cellular behavior. However, obtaining a steady supply of viable human tissue, which then has to be cultured for weeks under aseptic conditions, remains a challenging research area.
Femoral neck fractures are one of the most common surgically treated injuries in elderly patients. The human femoral head is discarded during the surgery undertaken to treat femoral neck fractures and is replaced with an artificial prosthesis. While this discarded femoral head is aged, it is generally nondegenerate. We hypothesized that the human femoral head would be a viable source of normal, nondegenerate articular cartilage suitable for preclinical cartilage research and regenerative therapies. We were able to coordinate the timely, sterile collection and delivery of the discarded human femoral head from the operating theatre to the laboratory. The aim of this study was to optimize the culture conditions for fresh human femoral heads from femoral neck fractures to permit viable long-term (10 weeks) culture.
Methods
Human Femoral Heads
Femoral heads were obtained with ethical permission (Tissue Governance, National Health Service, Lothian) and patient consent from 15 patients (11 females, 4 males, mean age 75.5 years [range 56-88 years]) undergoing hemiarthroplasty or total hip replacement for femoral neck fracture (FNF). Femoral heads were carefully removed intraoperatively by a qualified orthopedic surgeon using a corkscrew device and immediately placed into a sterile container with saline (0.9% w/v; 21°C) to prevent chondrocyte death from drying.
9
Femoral heads were then transferred to a sterile container with Dulbecco’s modified Eagle’s medium (DMEM) with
Culture Conditions
Femoral heads were maintained in culture (37°C) in sealed single use sterile containers and media changed every 3 days and the container replaced with every media change. Media volume for each femoral head was ~50 to 60 mL. Male femoral heads were larger than those of females (diameter typically 6 cm vs. 4.5 cm) and required larger culture containers. The femoral heads were cultured for up to 10 weeks under the following conditions: (a) static culture in DMEM, (b) culture in DMEM with movement (DMEM + mixing), (c) as for (a) above + 10% normal human serum (HS; Merck, Feltham, UK), and (d) as for (b) above + 10% HS. A cavity was carved in the cancellous bone of the femoral head for a magnetic stirring bar using bone trimmers (
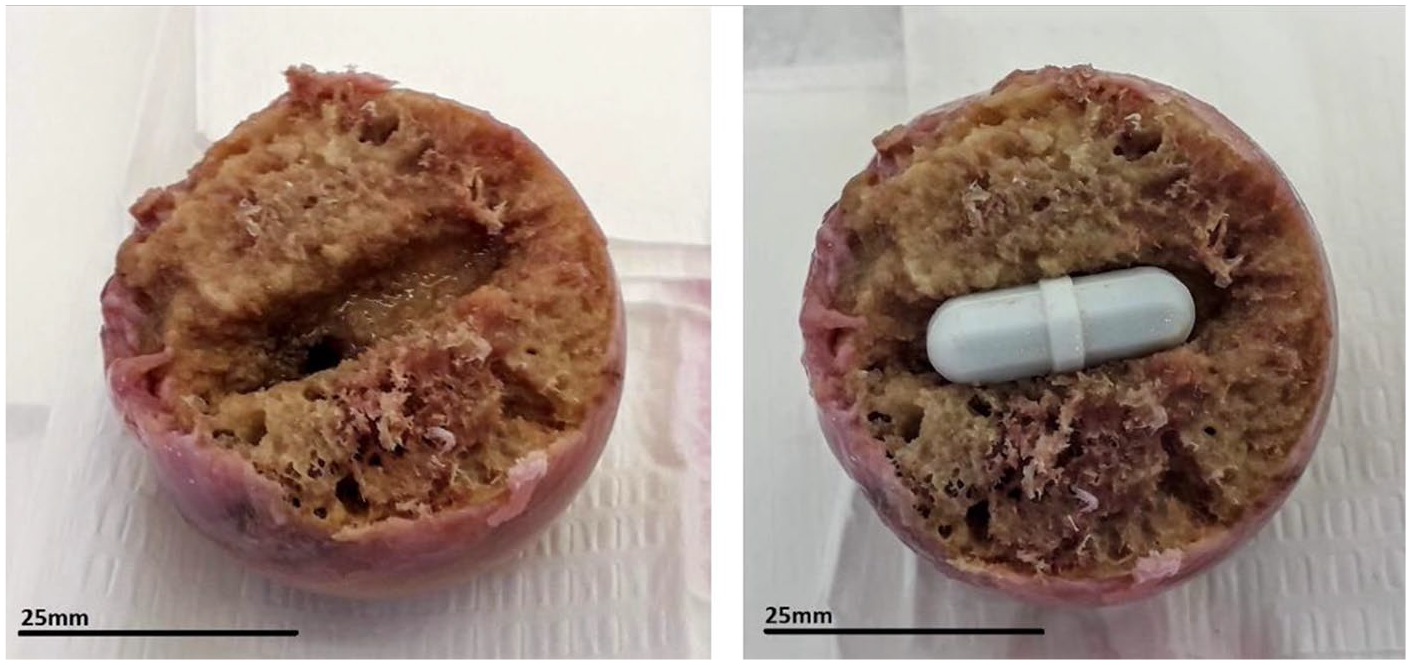
The cavity produced in the cancellous bone of the human femoral head to accommodate the magnetic bar for stirring culture experiments.
Cartilage Sampling, Fluorescent Labeling of In Situ Chondrocytes and Confocal Microscopy
Full depth cartilage explants were harvested using 3- or 5-mm diameter biopsy punches (Kai Medical, Solingen, Germany) for weekly chondrocyte viability measurements. To minimize the variability, explants were taken from random areas within the load-bearing parafoveal superior region
10
(
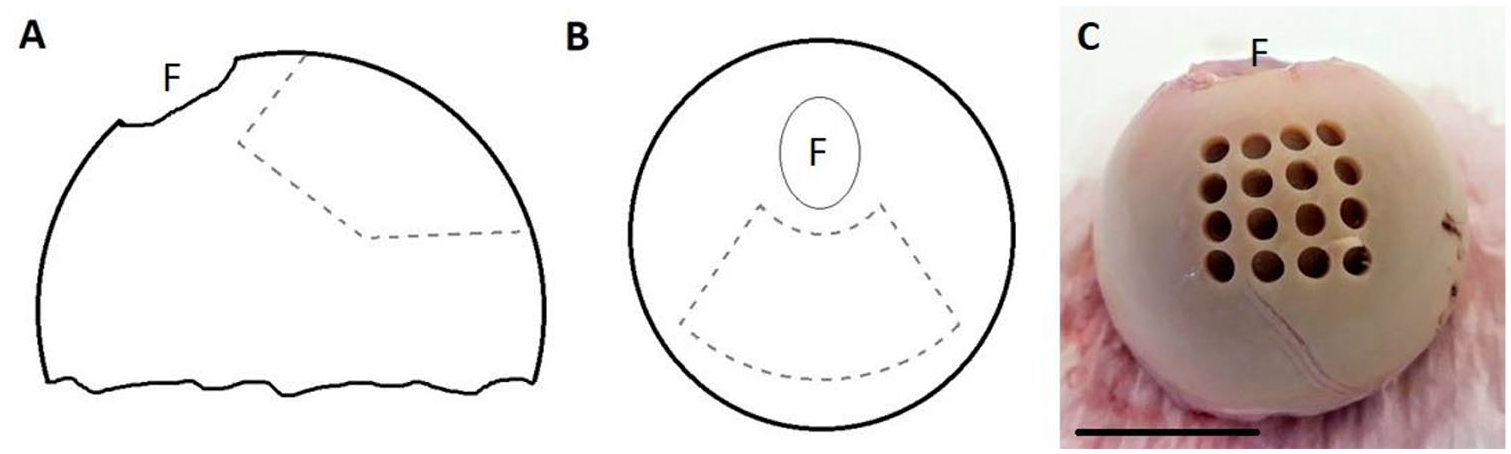
Localisation of the cartilage area on human femoral heads used for sampling. (A) Lateral view of the femoral head, fovea (F) on the upper left, (B) Top view of the femoral head, (C) Femoral head with biopsy explants taken from within the specified zone. Scale bar represents 25mm.
Measurements of Chondrocyte Viability, Density, Volume, and Morphology
Confocal projected axial views were analyzed using ImageJ/FIJI (National Institutes of Health) and IMARIS software (Zurich, Switzerland) as described. 12 Chondrocyte viability (% live cells) was calculated as: the number of CMFDA-labeled cells/(number of CMFDA-labeled cells + number of PI-labeled cells) in a given region of interest (ROI) volume. For chondrocyte density, the total number of cells (CMFDA-labeled and PI-labeled) in the ROI volume were counted in IMARIS, and results given as cells/μm 3 . Chondrocyte volumes were obtained using the IMARIS “Surfaces” algorithm. Volume calibration was performed using fluorescent microspheres (Polysciences, Warrington, PA, USA). Chondrocyte morphology was considered “normal” if cells were visualized as having a “smooth” surface and elliptical/rounded shape. “Abnormal” chondrocytes exhibited at least one CMFDA-labeled cytoplasmic process ≥2 μm long. Abnormal cells were counted manually and divided by the total number of live cells in the field of view with results presented as the % abnormal cells in the whole cell population within the ROI. 12
Histology, Cartilage Thickness, and Surface Roughness
Explants were frozen (−80°C) in a freezing medium (1:1 optimal temperature cutting compound with 30% w/v sucrose in PBS for histology.13,14 The plugs were then cut into 40 µm sections and stained with Alcian blue (Vector Laboratories Ltd, Peterborough, UK) to label cartilage glycosaminoglycans (GAGs) according to manufacturer’s instructions. For picrosirius red staining of collagens, sections of 10 µm thickness were first stained with hematoxylin for 8 minutes, then washed 3 to 4 times in distilled water. This was followed by 1-hour incubation in 0.1% picrosirius red (Direct Red 80, Sigma-Aldrich, Irvine, UK) in picric acid (VWR International, Lutterworth, UK). Sections were washed briefly 2 times in 0.5% acetic acid, dehydrated (2 washes in ethanol absolute followed by 1 wash in xylene) and mounted in resinous mounting medium. Histological sections were imaged on a Leica bright field microscope and analyzed with ImageJ/FIJI software. 15 After converting the images to greyscale 8-bit pixel depth, the same intensity threshold was set for each pair of sections (week 0 vs. week 10). The stained area above the threshold was measured and presented as a % of the total sectional area. Cartilage thickness measurements were performed on the coronal sections of cartilage using ImageJ/FIJI. For the surface roughness measurements, the ImageJ/FIJI Analyse Stripes macro was used on the images of histologically stained sections. Calculation of cartilage surface roughness was based on the deviation from an idealized smooth cartilage surface and expressed as Rq (the root mean square [RMS] deviation) in micrometers (µm).
Data Presentation and Statistical Analysis
Statistical analyses were performed using Graphpad Prism version 8.2.1 (GraphPad Software, La Jolla, CA, USA). Data were presented as (N(n)), with N representing the number of independent femoral heads and n the total number of replicates. Each data point presented on graphs and used for statistical analyses was an average (±SD or SEM as indicated) of the replicates taken from each femoral head. Unless otherwise stated, paired Student’s t tests were used to compare differences within pairs of treatment groups or time points, and analysis of variance (ANOVA) used to compare differences across several groups. A significant difference was accepted when P < 0.05.
Results
Cartilage Grading and Chondrocyte Viability
Femoral heads were macroscopically assessed using an established system. 16 For all femoral heads considered suitable, the cartilage was grade 0 over >75% of the surface, with small isolated grade 1 lesions in the parafoveal area (which were <25% of the surface area). The isolated areas of grade 1 cartilage (mild surface fibrillation) were not studied and only cartilage of grade 0 was used.
Chondrocyte viability was assessed on day 1 and any femoral heads with a viability of <80% were excluded. Out of a total of 24 femoral heads received, 5 were excluded due to low initial chondrocyte viability and a further 4 were also excluded as they developed infection during subsequent culture. In the remaining 15 femoral heads, the cell viability was 95.3% ± 5.3% (N(n) = 15(60)) on day 1. Chondrocyte viability in femoral heads cultured under static conditions in DMEM remained high (>90%) for ~4 weeks but decreased to 44.0% ± 22.3% by week 10 (N(n) = 4(16), one-way ANOVA, posttest for trend, P = 0.045,
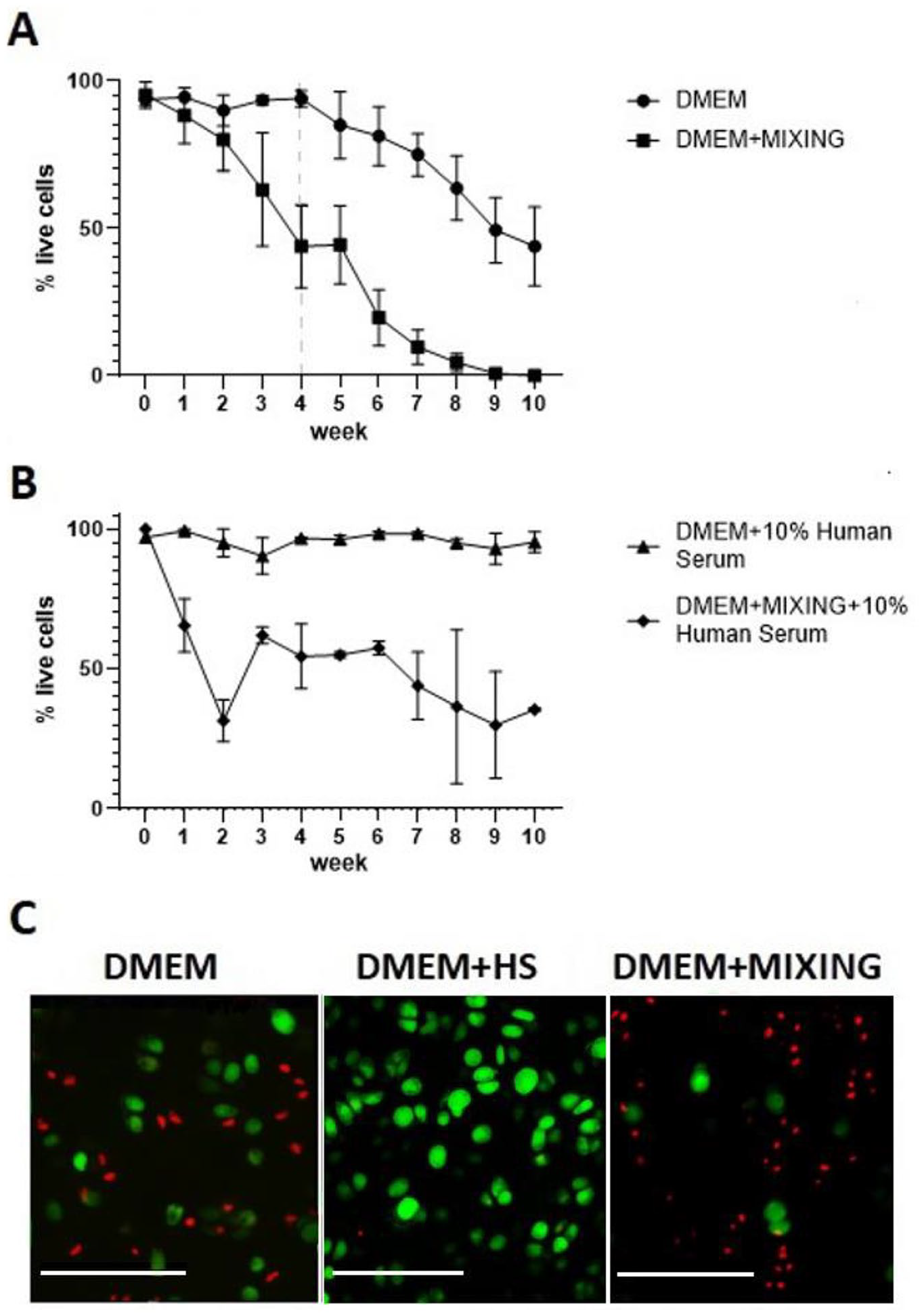
The viability of in situ human chondrocytes within femoral head cartilage cultured under various conditions. (A) DMEM N(n) = 4(16); DMEM + mixing (N(n) = 3(12)). (B) DMEM + normal human serum (HS); (N(n) = 3(12)), DMEM + mixing + human serum (N(n) = 2(8)); error bars represent S.E.M. (or S.D. for N = 2). (C) Representative images of viability staining (CMFDA green – live cells, PI red – dead cells) at week 10 for DMEM, DMEM + 10% human serum and DMEM + mixing; scale bars represent 100µm.
CLSM permits 3-dimensional imaging and quantitative analyses of chondrocyte viability within the full depth of cartilage.
17
To assess if chondrocytes within any zone were more sensitive during culture, chondrocyte viability was determined as a function of depth from the articular surface. We compared the percentage cell viability within the different zones (superficial [SZ], middle [MZ], and deep [DZ]) in coronal sections of articular cartilage at week 10 compared with baseline (week 0). There was a decrease in the SZ viability in both culture conditions (DMEM only, and DMEM + mixing; [P = 0.0425 and P = 0.016, respectively;
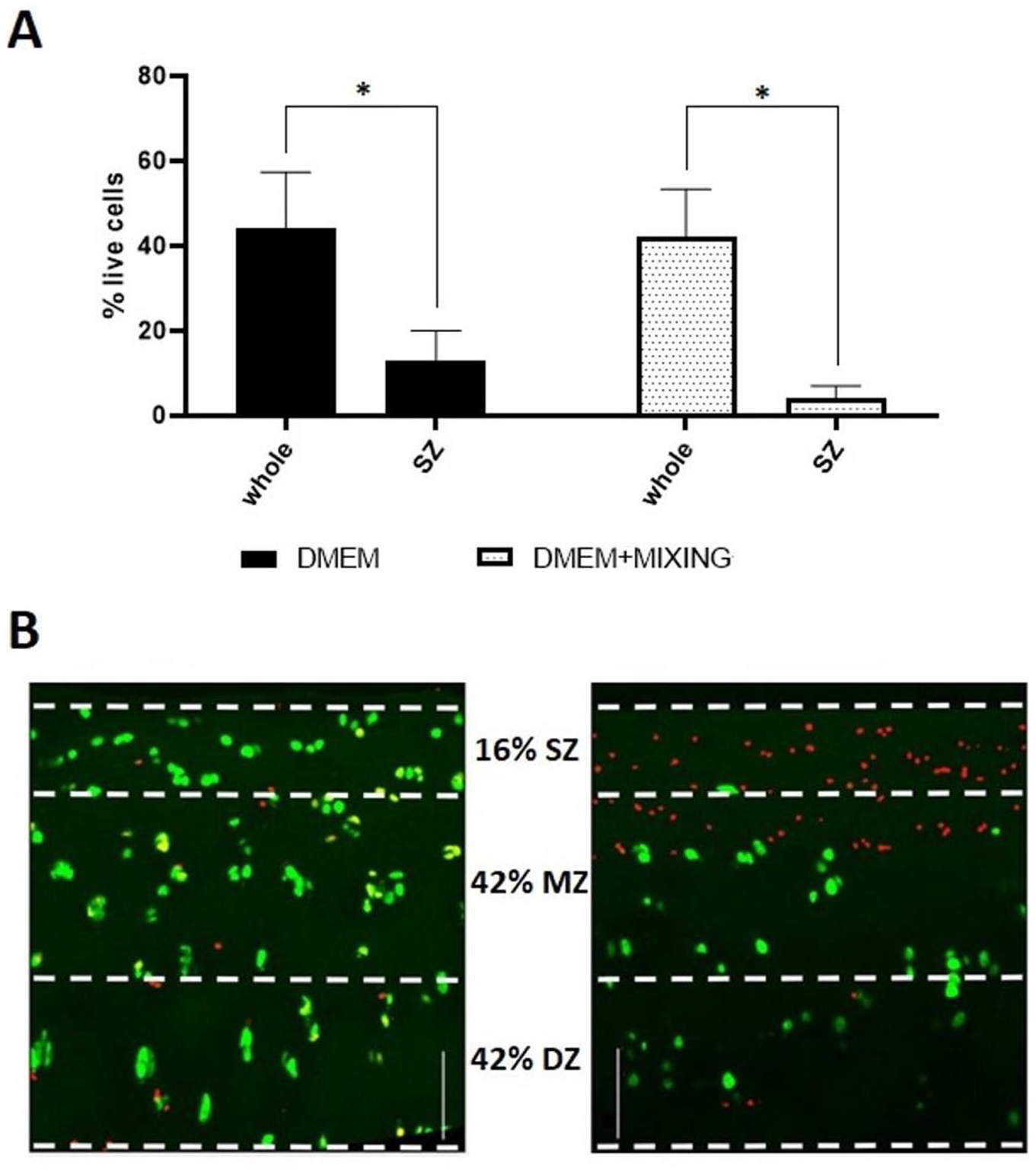
Viability of chondrocytes in various zones after 10 weeks of femoral head culture (A) Comparison of chondrocyte viability in the whole thickness vs superficial zone at week 10 of culture. DMEM N(n) = 4(16), *p = 0.0425, DMEM + mixing, N(n) = 3(12), *p = 0.016 (data shown as mean ± S.E.M.). (B) Representative coronal images of labelled chondrocytes within cartilage sections cultured in DMEM on day 0 (left panel), and after 10 weeks of culture in DMEM + mixing + HS on week 10 (right panel). The scale bars represent 100µm.
Chondrocyte Morphology
In fresh explants of macroscopically nondegenerate femoral head cartilage, a small population (8.0% ± 1.5%; N(n)=4(8)) of cells in the SZ (~100 µm from the surface) demonstrated one or more cytoplasmic processes (
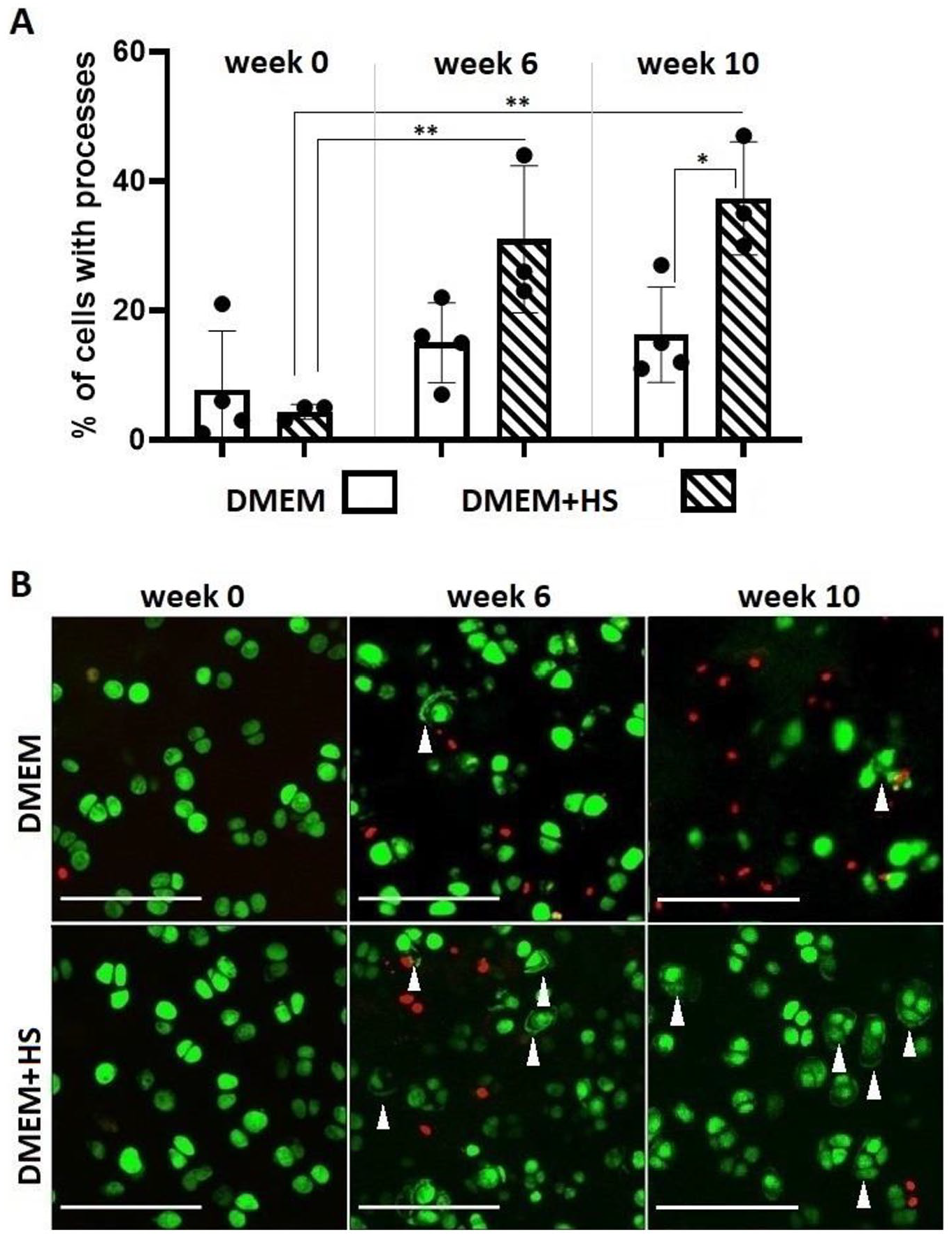
Changes to the morphology of in situ femoral head chondrocytes during culture. Panel (A) shows the % of cells with cytoplasmic processes at three time points (weeks 0, 6 and 10) in cultures with DMEM (N(n) = 4(16)) and DMEM + human normal human serum (HS) (N(n) = 3(12)). (Data shown as mean ± S.D.). Panel (B) shows representative images of cell stained with CMFDA Cell Tracker green to visualise chondrocyte morphology, and propidium iodide (PI) red to identify dead cells. The top row of images is from DMEM cultures, (left to right panels for weeks 0, 6 and 10 respectively), the bottom row of images for DMEM + human serum (left to right panels for weeks 0, 6 and 10 respectively. Note examples of chondrocytes with cellular processes indicated by white arrowheads. The scale bar represents 100µm.
Extracellular Matrix Composition
To evaluate whether extracellular matrix composition changed during culture, two histological stainings were performed on cartilage samples (week 0 and week 10) and analyzed semiquantitatively. There was a general trend of decreasing GAG staining using Alcian blue
18
under all culture conditions, but in DMEM + HS chondrocyte viability was significantly higher (
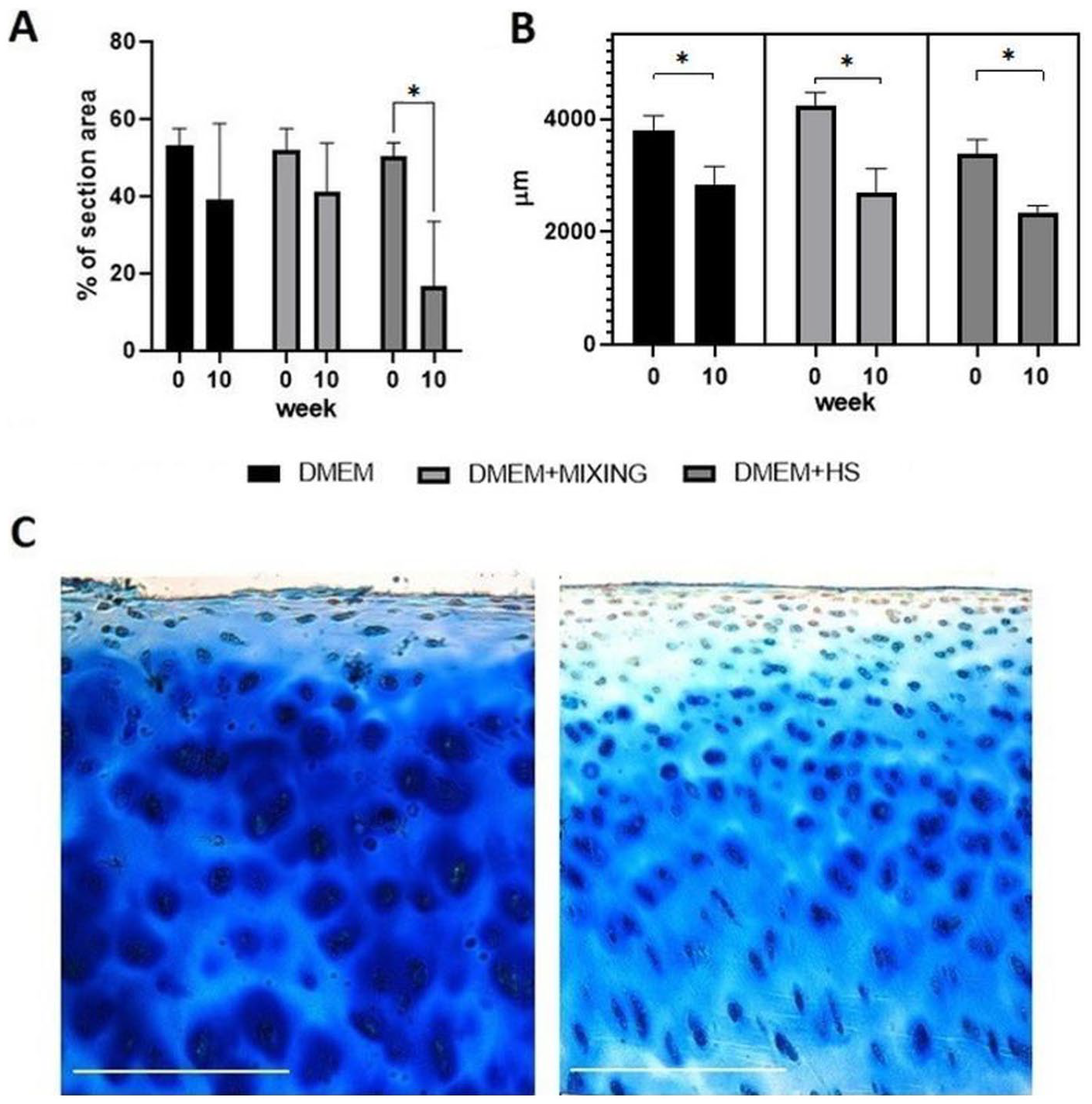
Histology of femoral head cartilage with time in culture. (A) GAG content in various culture conditions, (a) DMEM N(n) = 4(8), (b) DMEM + mixing N(n) = 4(8), (c) DMEM + 10% human serum N(n) = 3(6), *p = 0.028. (B) Cartilage thickness in various culture conditions, (a) DMEM N(n) = 4(24), (b) DMEM + mixing N(n) = 4(24), (c) DMEM + 10% human serum N(n) = 3(18), (p = 0.05; p = 0.02; p = 0.02 respectively). (Data shown as mean ± S.E.M.). (C) Representative images for Alcian blue staining of DMEM + human serum on week 0 (left panel) and week 10 (right panel).
Cartilage Thickness and Surface Roughness
In parallel with the loss of GAGs, cartilage thickness was reduced after 10 weeks in culture (
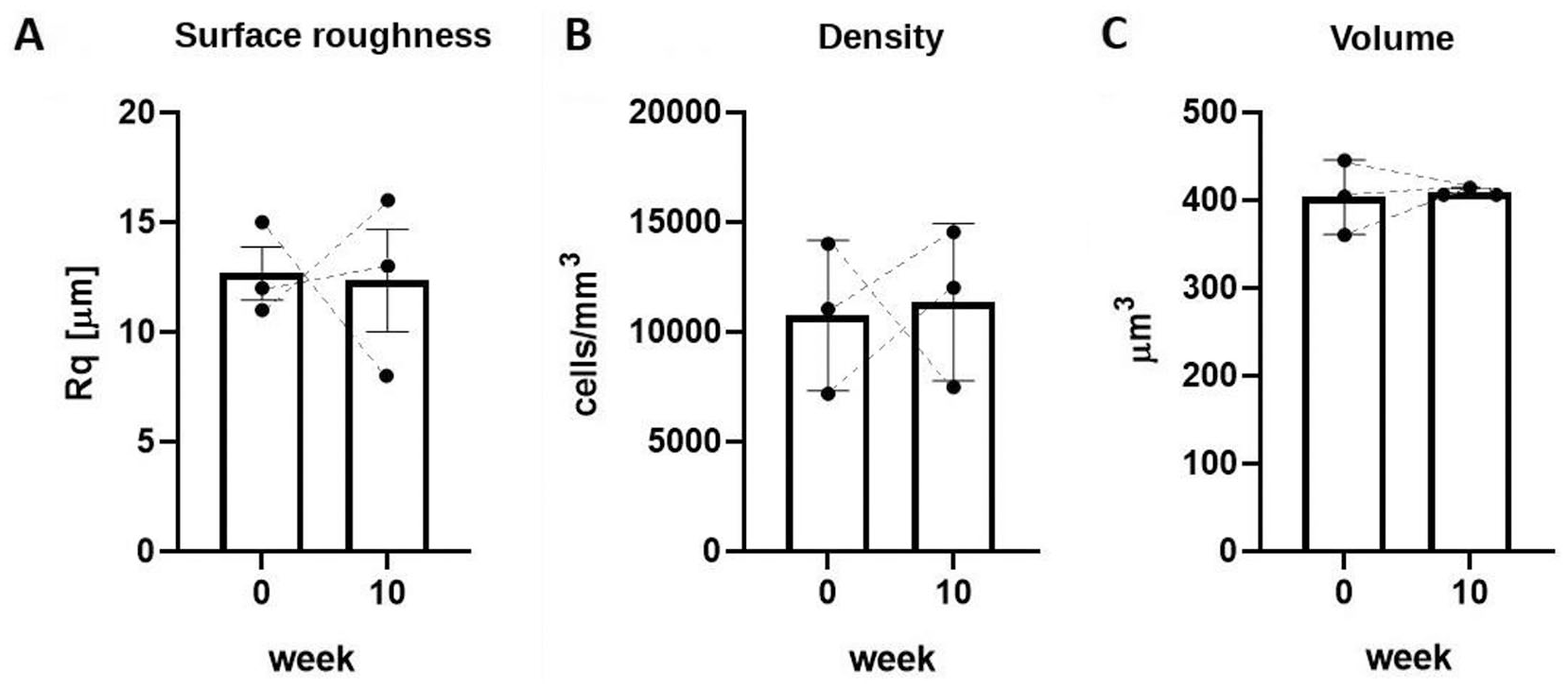
Surface roughness, chondrocyte density and volume of in situ femoral head chondrocytes with time in culture in DMEM + HS. (A) Surface roughness expressed as the root mean square (RMS) deviation)) in µm at week 0 and week 10, N(n) = 3(9), p = 0.2). (B) Cell density (total number of chondrocytes per mm3) at week 0 and week 10, N(n) = 3(60), p = 0.22. (C) Chondrocyte volume at week 0 and week 10, N(n) = 3(44), p = 0.22. Broken lines illustrate pairs of data at week 0 and week 10 for each femoral head. (Data shown as mean ± S.D.).
Chondrocyte Density and Volume
There was no difference in cell density for all the samples at week 0 (9971 ± 2389 cells/mm3, N(n) = 15(60)) compared with week 10 (11256 ± 3305 cells/mm3; P = 0.01; N(n) = 15(60);
Discussion
We have established culture conditions that maintain chondrocyte viability during ex vivo culture of human femoral heads for 10 weeks by supplementing standard culture medium with 10% normal HS. We have identified an excellent and reliable source of viable, nondegenerate human articular cartilage ideal for ex vivo experimentation. Previously, we have investigated the microscopic effects of mechanical and other forms of injury on human articular cartilage.9,19 However material was obtained from tissue discarded during knee replacement for osteoarthritis and the yield of nondegenerate tissue was often low. This was because the majority of the tissue was osteoarthritic with loss of superficial zone cells, even if macroscopically the tissue may have appeared nondegenerate. In contrast, the cartilage of the femoral head discarded after femoral neck fracture was in most cases nondegenerate and our experiments have confirmed the presence of viable cartilage tissue that is macroscopically and microscopically ideally suited for investigating cartilage repair and regeneration. Due to the excellent chondrocyte viability throughout culture, the model may also allow ex vivo validation of an optimal combination of cells, growth factors, and scaffolds that lead to the formation of repair tissue resembling the desirable hyaline articular cartilage at the microscopic level.
Current knowledge of the microscopic quality of cartilage repair tissue in humans is based on histological assessment of opportunistic biopsy specimens retrieved during “second look” arthroscopy (keyhole surgery). While these specimens have provided valuable insight into the quality of cartilage repair, with so-called “hyaline-like” composition, the information is limited by small numbers of specimens, distortion of the tissue during biopsy, variability in the site/size of biopsies and heterogeneity of the study sample. The lack of a noninvasive method of evaluating the microscopic characteristics of the quality of the cartilage repair tissue has also been recognized as a major problem limiting advances in cartilage repair and regenerative techniques by the International Cartilage Repair Society (ICRS). 20 Modern imaging techniques (e.g., CLSM) allow microscopic examination of articular cartilage by optically sectioning the tissue. We believe that the significantly easier access to normal, human cartilage ex vivo will help overcome the problems associated with in vivo biopsy, and allow detailed quantitative microscopic assessment and optimization of the quality of cartilage repair. The proposed model will significantly enhance our ability to test a wide range of preclinical therapeutic cartilage repair and regenerative strategies directly in human tissue so that the best candidate therapies can be identified for subsequent clinical study.
In our model, the large area of grade 0 cartilage (
Femoral head culture in DMEM maintained chondrocyte viability for ~4 weeks after which there was an increase in chondrocyte death (
Visualisation of fluorescently labeled in situ chondrocytes revealed the classical morphology of elliptical cells in the SZ with the more spheroidal forms in the deeper zones (
There was no evidence of chondrocyte clustering 34 under any of the experimental conditions. Nomura et al. 35 did not observe changes to chondrocyte morphology in mice subjected to hindlimb unloading when cartilage was studied by histology. This might appear to conflict with the changes to chondrocyte morphology reported here. However, the detection of the fine cytoplasmic process is not possible with their histological techniques as they involve tissue shrinkage 36 and high-resolution imaging of unperturbed in situ chondrocyes is essential for the visualization of the processes.17,31
It might be considered that a limitation in our study was that no mechanical load was applied to the femoral heads. However, this revealed that after 10 weeks there were features of the cartilage ECM and in situ chondrocytes that bear similarities to the changes observed with both in vivo and in vitro cartilage disuse atrophy. Alcian blue staining reflecting GAG content was decreased (
While there were changes to GAG labeling, there was no change to picrosirius red labeling suggesting the cartilage collagen content remained unaltered throughout culture. While picrosirius red does not discriminate between the collagen types, 43 it is possible that there were changes to collagen metabolism and/or its organization. For example, while total collagen content might not have changed significantly, it is possible that there was a decrease in the collagen type II: type I ratio reflecting chondrocyte de-differentiation. In addition, changes to collagen fiber distribution/orientation could be evident, and future studies utilising polarized light microscopy would be worth pursuing. A small decrease in collagen cross-linking, which recovers after remobilization, has been reported 44 and the immobilization of rabbit knee joint leads to a partial shift in the density of collagen composition from type II to type I. 45 However, in human cartilage, there is negligible/minimal collagen turnover over the lifetime in a healthy joint. 46
There was no change to the volume of in situ chondrocytes following 10-weeks of culture in DMEM + HS (
This study described the first successful ex vivo culture of a large human joint. In situ chondrocyte viability remained high in DMEM + HS; however, it decreased under the other conditions tested. Although chondrocyte viability was optimal, there were changes to the matrix (cartilage thinning, GAG loss, no change to collagen or surface roughness) and chondrocytes (development of cytoplasmic processes, no change to volume or density). This preclinical model may be an invaluable addition for the assessment of human cartilage repair therapies and may replace some animal studies.
Footnotes
Acknowledgments and Funding
We thank Ms. F. Stewart for excellent assistance with the provision of the femoral heads, Dr. A. Kubasik-Thayil at the IMPACT imaging facility in the Centre for expert assistance with confocal imaging, and Ms. A. Luczak for assistance with histology. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a project grant from the Chief Scientist’s Office (Scotland) TCS/18/01.
Author Contributions
Study concept and design; Amin, Simpson, Hall. Acquisition of data; Styczynska-Soczka. Analysis and interpretation of data; Styczynska-Soczka, Amin, Simpson, Hall. Manuscript preparation; Styczynska-Soczka, Amin, Simpson, Hall.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Femoral heads were obtained with ethical permission from the Tissue Governance, National Health Service, Lothian.
Informed Consent
Informed consent was obtained from all patients.
Trial Registration
Not applicable.
