Abstract
Objective
To enhance the in vitro integration of self-assembled articular cartilage to native articular cartilage using chondroitinase ABC.
Design
To examine the hypothesis that chondroitinase ABC (C-ABC) integration treatment (C-ABCint) would enhance integration of neocartilage of different maturity levels, this study was conducted in 2 phases. In phase I, the impact on integration of 2 treatments, TCL (TGF-β1, C-ABC, and lysyl oxidase like 2) and C-ABCint, was examined via a 2-factor, full factorial design. In phase II, construct maturity (2 levels) and C-ABCint concentration (3 levels) were the factors in a full factorial design to determine whether the effective C-ABCint dose was dependent on neocartilage maturity level. Neocartilages formed or treated per the factors above were placed into native cartilage rings, cultured for 2 weeks, and, then, integration was studied histologically and mechanically. Prior to integration, in phase II, a set of treated constructs were also assayed to provide a baseline of properties.
Results
In phase I, C-ABCint and TCL treatments synergistically enhanced interface Young’s modulus by 6.2-fold (P = 0.004) and increased interface tensile strength by 3.8-fold (P = 0.02) compared with control. In phase II, the interaction of the factors C-ABCint and construct maturity was significant (P = 0.0004), indicating that the effective C-ABCint dose to improve interface Young’s modulus is dependent on construct maturity. Construct mechanical properties were preserved regardless of C-ABCint dose.
Conclusions
Applying C-ABCint to neocartilage is an effective integration strategy with translational potential, provided its dose is calibrated appropriately based on implant maturity, that also preserves implant biomechanical properties.
Keywords
Introduction
Translational cartilage tissue engineering aims to generate neotissue with biomimetic properties for the purpose of in vivo implantation. In addition to achieving biomimetic properties, one of the major impediments to cartilage regeneration is the inability of implants to integrate with adjacent, native tissue; this poses a serious, clinical problem.1-4 Evaluating one of the salient standards of care shows that cartilage allografts suffer from incomplete integration, which hampers their long-term durability and efficacy.5,6 Because tissue engineered cartilage is designed to be similar if not identical to native tissue, like with allografts, barriers to neocartilage integration are also anticipated. 7 Cartilage integration strategies are necessary for both native tissue repair and for deploying tissue-engineered cartilage in the clinical setting.
Cartilage has unique intrinsic properties that hinder integration, including (1) lack of highly metabolic cells at the interface, 8 (2) repulsive negative charges induced by cartilage glycosaminoglycans (GAGs), 6 (3) a dense extracellular matrix (ECM), 9 and (4) a limited number of stabilizing crosslinks at the interface. 10 Chondroitin sulfate, an abundant ECM component in cartilage, contributes to the paucity of cells at the interface because it has been shown to impede migration of chondrocytes. 11 Chondroitin sulfate is also a negatively charged molecule that results in repulsion between engineered and native cartilage.6,12 With regard to ECM, juvenile articular cartilage integrates better than adult tissue because fully mature cartilage contains denser ECM and chondrocytes that are less metabolically active, both factors that impede integration. 13 The maturation and development of articular cartilage has been modulated by removal of GAG and has been proposed as a method of improving collagen network interaction, and, therefore, crosslink development.14-16 The observation that chondroitin sulfate and tissue maturity can hinder cartilage integration suggests that the 4 factors above can be targeted by focusing on GAG-specific agents and tissue maturity in the development of cartilage integration strategies.
In terms of improving cartilage integration, chondroitinase ABC (C-ABC), a catabolic enzyme that digests chondroitin sulfate, and to a lesser degree other GAGs such as dermatan sulfate and hyaluronic acid, 17 has been explored previously for engineered cartilage integration because it could target each of the four integration hindrances described above.4,18 For example, removal of chondroitin sulfate could encourage cell migration, 11 transiently decrease repulsive negative charge and physical ECM density,14,16 and, due to decreased steric hindrance, lead to more collagen remodeling and pyridinoline (PYR) crosslinking at the interface.15,19 A variety of cartilage integration strategies employing catabolic enzymes have been explored;4,9,20-23 however, to the best of our knowledge, catabolic enzymes have not been used with biomimetic neocartilage. Furthermore, the effects of catabolic enzymes have not been quantified on neocartilage properties. For example, collagenase has been explored for cartilage integration and has shown promise.20,21 However, digesting cartilage matrix at the implant edge using collagenase could also damage PYR crosslinks, which have been shown to enhance self-assembled cartilage properties and integration.7,10 To avoid disrupting these beneficial crosslinks while still targeting integration hindrances, C-ABC could be used in lieu of collagenase. Self-assembled articular cartilage, which has been shown to have many properties on par with those of native articular cartilage, has the potential for more complete integration than scaffold-based cartilage constructs because it contains highly metabolic cells and a propensity for remodeling since it is composed strictly of ECM formed from its own cells.24,25 C-ABC integration treatment (C-ABCint) in combination with these inherent properties could significantly enhance self-assembled articular cartilage integration, especially when combined with lysyl oxidase like 2 cocktail (LOXL2), an enzymatic treatment that mediates PYR crosslinking. C-ABCint dose would be selected to only affect the construct periphery, and, thus, would not compromise construct mechanical integrity.
Using both C-ABCint and bioactive agents, this study targeted the aforementioned integration hindrances in both immature and mature self-assembled articular cartilage. In phase I of this study, it was hypothesized that a combination of C-ABCint and bioactive agents (TCL; TGF-β1, C-ABC, and LOXL2) applied to constructs could enhance integration between native and engineered articular cartilage. Because it has been shown previously that tissue maturation influences cartilage integration, in phase II, the combined effects of construct maturity and C-ABCint concentration were examined. In phase II, it was hypothesized that the effective C-ABCint dosage would be higher for mature constructs and lower for immature constructs, but that no dose of C-ABCint would affect construct mechanical properties.
Methods
Cell and Tissue Harvest
Primary articular chondrocytes and articular cartilage explants were isolated in a sterile fashion from juvenile bovine stifle joints as previously described. 26 Briefly, juvenile bovine hind limbs were obtained from a commercial provider of animal specimens (Research 87, Boylston, MA). Subsequently, the stifle joints were dissected in a sterile fashion to reveal the articular cartilage surfaces of the distal femur. To obtain primary articular chondrocytes, articular cartilage from both the femoral condyles and trochlear groove were minced and digested in a 0.2% w/v collagenase type II solution (Worthington Biochemical Corporation, Lakewood, NJ). Osteochondral explants were isolated from the femoral condyles using an 8-mm dermal biopsy punch. These explants were then trimmed to 1.5 mm such that only the most superficial articular cartilage remained.
Construct Formation and Culture
Chondrocytes were seeded at a high density (4.5 × 106 cells/100 µL medium) in chemically defined chondrogenic medium (CHG; Dulbecco’s modified Eagle’s medium with high glucose/GlutaMAX [Life Technologies, Grand Island, NY], 1% penicillin-streptomycin-fungizone [Lonza, Basel, Switzerland], 1% nonessential amino acids [Life Technologies], 1% ITS+ Premix [BD Biosciences, San Jose, CA], 50 μg/mL ascorbate-2-phosphate [Sigma-Aldrich, St. Louis, MO], 40 μg/mL
Construct Integration
Using a sterile dermal biopsy punch, 4-mm discs were taken from constructs, which were then treated with C-ABCint for 2 hours. Experiments were carried out in 2 phases. In phase I, a 2-factor, full factorial study was conducted with 4-week constructs of 2 culture conditions (CHG or TCL) and C-ABCint (none or 0.15 U/mL). For phase II, a 2-factor, full factorial study was conducted with constructs of 2 different maturities (3-week or 5-week) and 3 C-ABC concentrations (0.05 [low], 0.15 [medium], and 0.25 [high] U/mL). Following treatment, C-ABCint activity was quenched with zinc sulfate (1 mM) for 10 minutes, and elimination of residual, inactive enzyme was achieved by sequentially washing constructs with medium. Subsequently, neocartilage discs were press-fit into native bovine articular cartilage explant rings with an 8 mm outer diameter, 4 mm inner diameter, and 1.5 mm thickness, thus, creating engineered-native composites ( Fig. 1 ). In both phases, engineered-native composites were cultured in CHG for an additional 2 weeks on an orbital shaker (20 rpm for week 1 and 50 rpm for week 2) and then processed for subsequent assays.
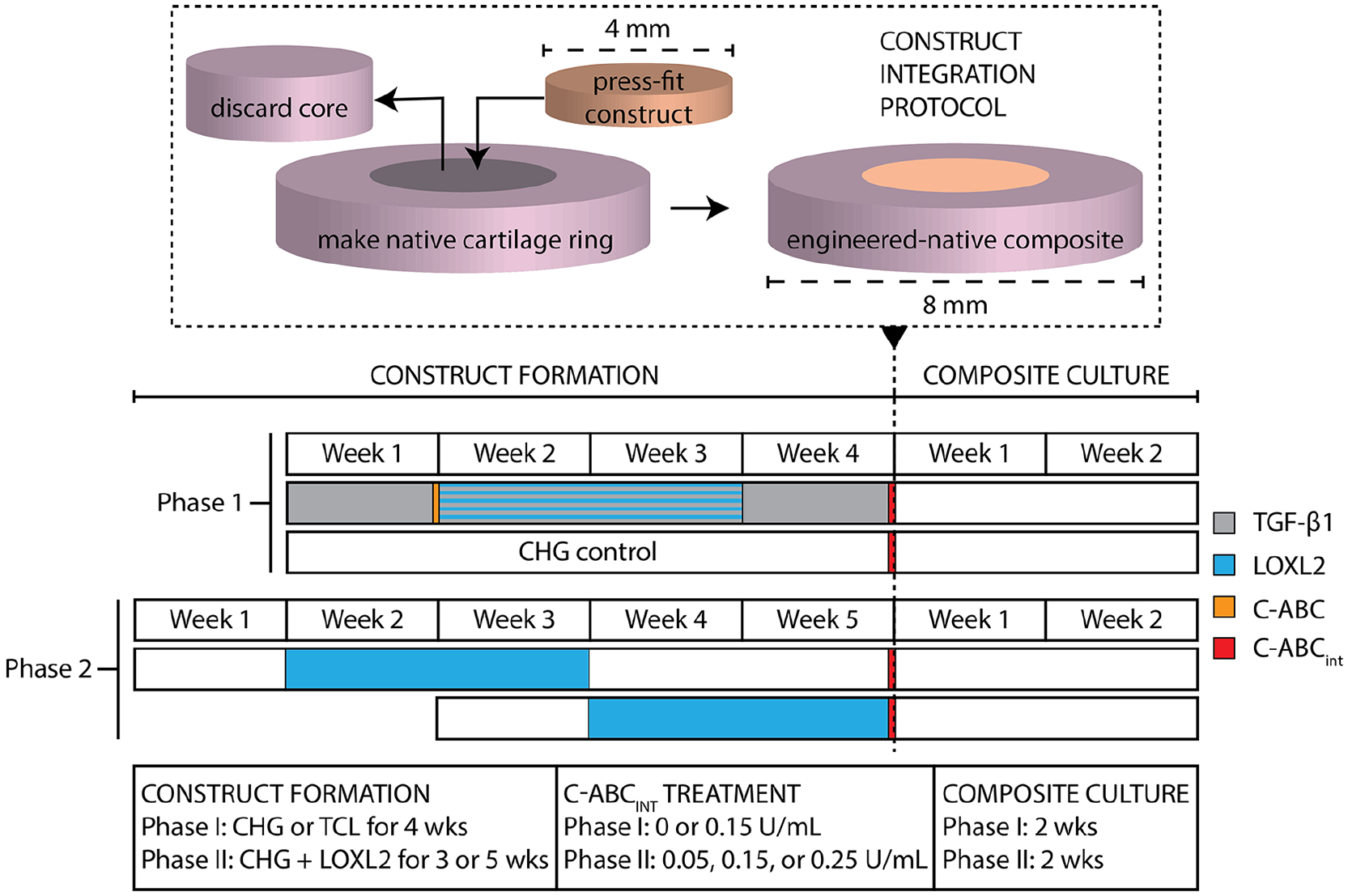
A schematic diagram describes the integration of engineered cartilage to native cartilage. Construct formation in phase I lasted for 4 weeks and involved either TCL or CHG, while in phase II, all constructs were formed with CHG and LOXL2, but were cultured for either 3 or 5 weeks prior to integration. The timeline depicts when these construct formation agents were applied. In phase I, C-ABCint was either applied at 0.15 U/mL or not at all directly before integration, whereas in phase II, 0.05 (low), 0.15 (medium), or 0.25 (high) U/mL was used. Engineered-native composite culture lasted 2 weeks for each phase before subsequent assays. TCL, transforming growth factor–β1 (TGF-β1), C-ABC, and lysyl oxidase like 2 (LOXL2); CHG, chemically defined chondrogenic medium; C-ABCint, chondroitinase ABC integration treatment.
Histology of Constructs and Composites
Self-assembled constructs and engineered-native composites were fixed in 10% neutral-buffered formalin for histological evaluation. Fixed samples were processed, embedded in paraffin, and sectioned at a thickness of 5 μm. Sections were then stained with hematoxylin and eosin (H&E), safranin O/Fast Green (Saf-O), and picrosirius red as previously described. 27 Sections stained with Saf-O were also rehydrated and stained with additional Fast Green counterstain for 10 minutes to determine GAG diffusion from the sections.
Biochemical Analyses of Constructs
Self-assembled constructs were weighed before and after lyophilization, and then digested in papain. DNA content was measured with Picogreen (Thermo Fisher Scientific, Waltham, MA), total collagen content was measured via a modified hydroxyproline assay as previously described, 26 and GAG content was determined using a dimethylmethylene blue dye-binding assay kit (Biocolor, Newtownabbey, Northern Ireland). PYR collagen crosslink quantification was performed via a liquid chromatography–mass spectrometry (LCMS) assay. 28 Samples were hydrolyzed in 6 N HCl at 105°C for 18 hours, after which acid was removed using centrifugal evaporation. Dried hydrolysates were resuspended in 25% (v/v) acetonitrile and 0.1% (v/v) formic acid in water, centrifuged at 15,000 × g for 5 minutes, and the supernatant was transferred to a vial compatible with the LCMS system. Liquid chromatography was performed on a Cogent Diamond Hydride HPLC Column (2.1 mm × 150 mm, particle size 2.2 μm, pore size 120 Å, MicroSolv, Leland, NC). The elution gradient used 0.1% (v/v) formic acid in water as solvent A and 100% acetonitrile as solvent B. The 5-minute elution gradient ran at 300 μL/min (0 minutes 25% B, 2 minutes 25% B, 2.2 minutes 5% B, 3 minutes 25% B). Mass spectrometry was performed on a Quadrupole Mass Detector (ACQUITY QDa, Waters, Milford, MA) in ESI+ MS scan mode. The quadrupole range was set to 150-450 m/z with cone voltage 12.5 V. MassLynx software version 4.1 with TargetLynx was used to quantify PYR in 10 μL injections of self-assembled construct hydrolysate by integrating the extracted ion chromatogram of double-charged PYR (m/z = 215.1) and comparing to a PYR standard (BOC Sciences, Shirley, NY) prepared and run in the same fashion.
Construct and Composite Mechanical Testing
Tensile testing was conducted on dog-bone-shaped construct specimens as previously described. 7 To test the integration, engineered-native composites were trimmed into strips containing engineered tissue on one side and native tissue on the other. The ends of each strip were then glued to paper tabs, which were then gripped by a uniaxial testing machine (Instron 5565, Norwood, MA). A pull-to-failure test was conducted at a rate of 1% strain per second. From these experimental data, Young’s modulus (Ey) and ultimate tensile strength (UTS) values were determined for both constructs and for the engineered-native interface of composites. Creep indentation compressive tests were conducted on self-assembled constructs using a flat, porous indenter tip and a constant load as previously described. 29 A linear biphasic model and finite element analysis were used to obtain the aggregate modulus, permeability, and Poisson’s ratio from the experimental creep curves.30,31
Statistics
All statistical analyses were performed using Prism 8 (GraphPad Software, San Diego, CA). Quantitative data were assessed using a 2-way analysis of variance (ANOVA) with a post hoc Tukey’s HSD (honestly significant difference) test at a significance level of α = 0.05. Significance of ANOVA factors is denoted with Greek characters. Two-way ANOVA factor interactions were analyzed to determine if the effect of one factor is dependent on the level of the other. Significance among particular groups is illustrated by a connecting letters report with Latin characters (i.e., bars that do not share the same Latin character(s) are statistically significant). Synergism of factors was defined as the case in which the combined application of 2 factors led to a greater increase in a property than the sum of the effects of those 2 treatments individually.
Results
Phase I: C-ABCint and TCL Enhanced Cartilage Integration Synergistically
C-ABCint improved visible congruence of engineered constructs to native tissue, both in terms of gross morphology and H&E staining ( Fig. 2A and B ). Gaps between the native cartilage and neocartilage were visible in control composites ( Fig. 2A ), but not in composites treated with C-ABCint ( Fig. 2B ). As an individual factor, the effect of C-ABCint significantly enhanced interface Ey (P = 0.0009) and interface UTS (P = 0.0001), but TCL was not a significant factor for interface Ey (P = 0.05) or UTS (P = 0.98) ( Fig. 2C and D ). For the CHG group, C-ABCint increased interface Ey from 0.20 ± 0.21 to 0.69 ± 0.13 MPa (P = 0.2) and significantly increased interface UTS from 0.057 ± 0.046 to 0.24 ± 0.080 MPa (P = 0.004). Similarly, for the TCL group, C-ABCint significantly improved interface Ey from 0.36 ± 0.39 to 1.25 ± 0.78 MPa (P = 0.02) and interface UTS from 0.076 ± 0.090 to 0.22 ± 0.090 MPa (P = 0.04). The combined, beneficial effect of TCL and C-ABCint on interface Ey (1.05 MPa) was greater than the sum of the individual effects of those factors (0.65 MPa). Thus, TCL and C-ABCint synergistically increased interface Ey.
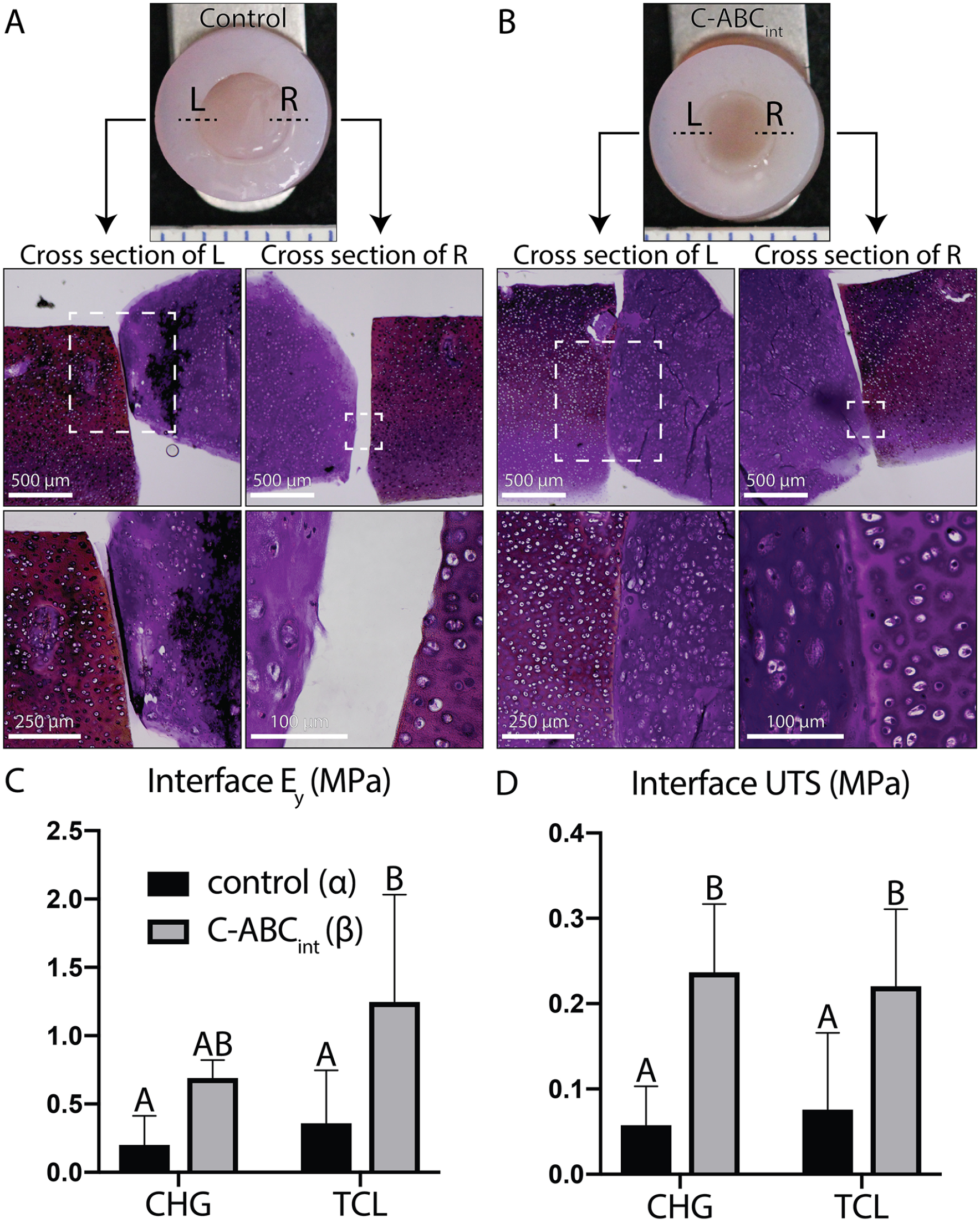
C-ABCint and TCL synergistically enhanced cartilage integration (phase I). (
Phase II: Construct Mechanical Properties Were Preserved Regardless of C-ABCint Concentration, but Local, Histologic Effects of C-ABCint Were Observed for Both Immature and Mature Constructs
For constructs assayed prior to integration, C-ABCint as a factor did not significantly affect any quantitative morphological, biochemical, or mechanical properties of constructs. C-ABCint did affect construct histological properties, however ( Fig. 3 ). Saf-O staining intensity in general was lower for 3-week constructs than 5-week constructs. Both construct maturity levels demonstrated a reduction in Saf-O at the neocartilage edge as C-ABCint concentration increased from the low dose to the high dose. Picrosirius red staining was not affected by C-ABCint at any concentration but was more intense at the edge of 5-week constructs in comparison to 3-week constructs.
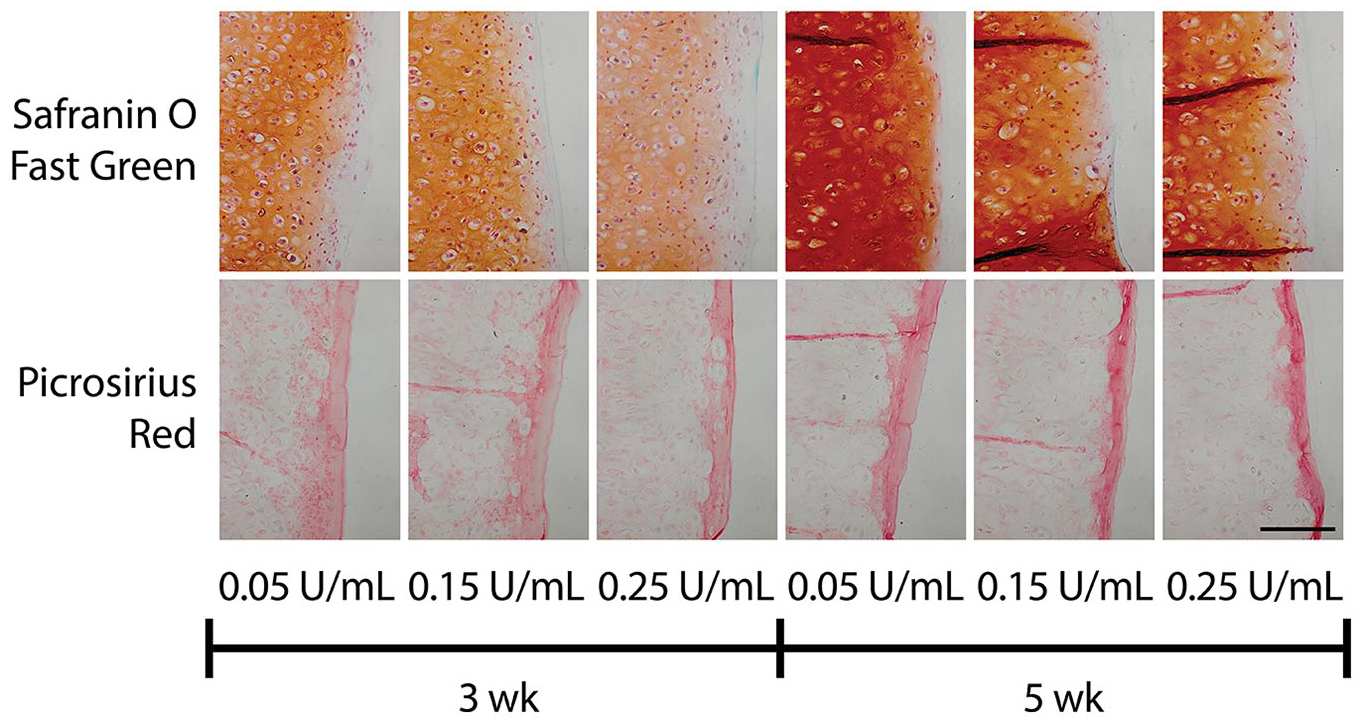
A higher C-ABCint dose was required for local GAG depletion in 5-week constructs due to increased collagen density at the edge (phase II). Histology of constructs demonstrated differences in construct staining intensity and distribution based on construct maturity and C-ABCint dose. Safranin O/Fast Green staining of constructs illustrated local effects of higher doses of C-ABCint at the edge of both immature and mature constructs. Picrosirius red staining demonstrated higher intensity staining of collagen at the edge of 5-week constructs. The scale bar represents 100 µm and applies to all images. C-ABCint, chondroitinase ABC integration treatment; GAG, glycosaminoglycan.
As a factor, greater construct maturity (i.e., 5-week constructs vs. 3-week constructs) significantly increased construct wet weight (WW) (P < 0.0001) and construct thickness (P < 0.0001) (Supplementary Table S1). Increased construct maturity did not lead to differences in biochemical contents per WW or per dry weight (DW) (Supplementary Table S1), but caused collagen/DNA (P < 0.0001), GAG/DNA (P < 0.0001), and PYR/DNA (P = 0.002) to significantly increase ( Fig. 4A-C ). Qualitatively, increased Saf-O (GAG) and picrosirius red (collagen) staining intensity for 5-week constructs versus 3-week constructs corresponded to these biochemical data ( Fig. 3 ). For all constructs, picrosirius red staining was primarily localized to the edge of the construct. Construct maturity as a factor significantly influenced Ey (P = 0.01), as 3-week constructs had an Ey of 1.70 ± 0.24, 1.91 ± 0.46, 1.59 ± 0.34 MPa, and 5-week constructs had an Ey of 2.21 ± 0.53, 2.22 ± 0.31, 2.20 ± 0.70 MPa (low, medium, and high doses of C-ABCint, respectively) ( Fig. 4D ). There were no significant differences between individual groups, however. Similarly, construct maturity as a factor significantly affected UTS (P = 0.049) ( Fig. 4E ). While tensile properties were significantly affected by construct maturation, aggregate modulus was not affected over the levels of maturities examined (P = 0.3) ( Fig. 4F ). Comprehensive construct properties can be found in Supplementary Table S1.
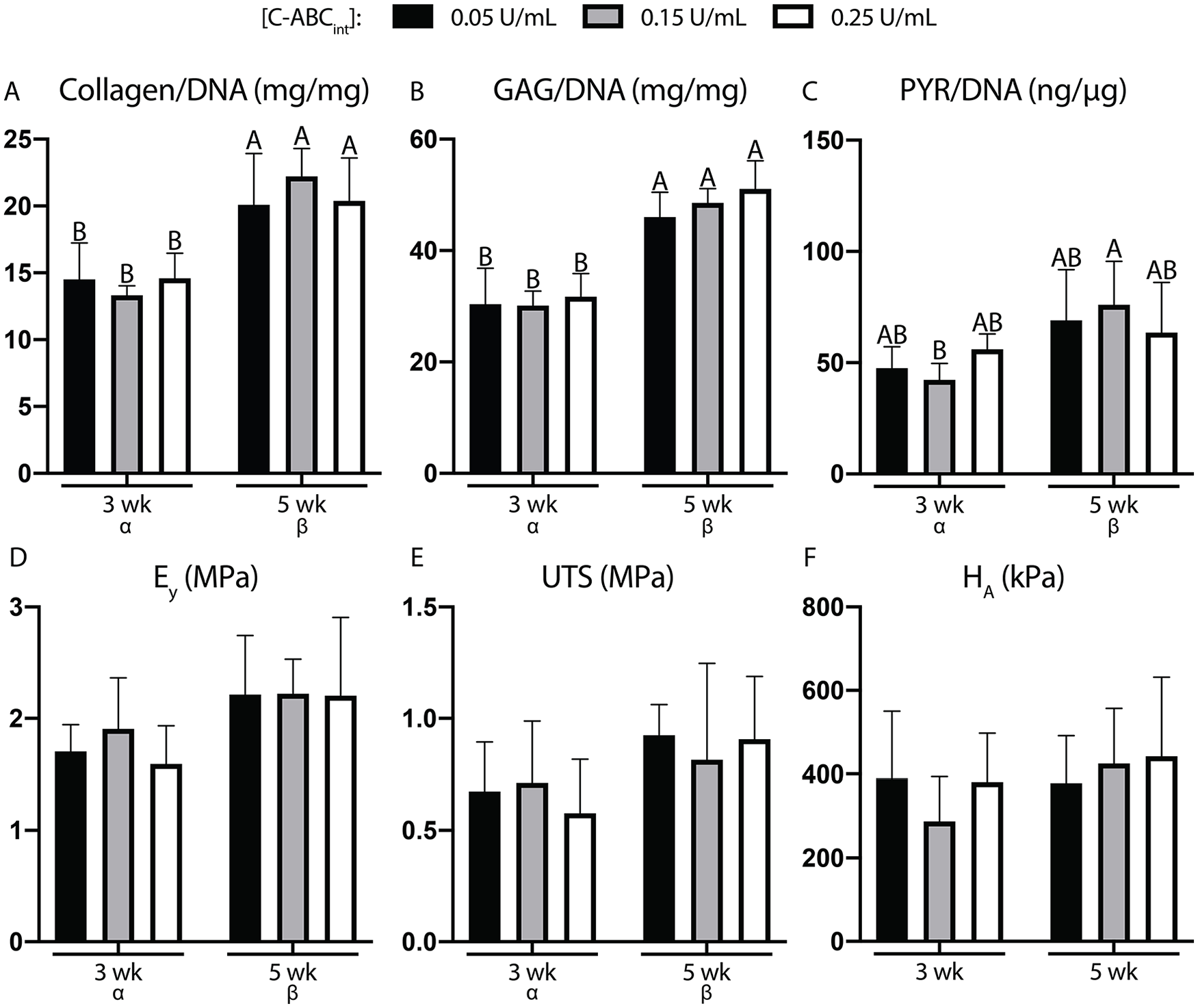
Properties of engineered constructs were found to be preserved regardless of C-ABCint dose (phase II). Biochemical contents (
Phase II: Effective C-ABCint Dose to Enhance Cartilage Integration Is Greater for Mature Constructs than for Immature Constructs
Saf-O and picrosirius red staining of the interface indicated that for 3-week constructs treated with either the low or medium dose of C-ABCint and for 5-week constructs treated with either the medium or high dose of C-ABCint, the native tissue and neotissue were in apposition (i.e., interface was closed), and this interface stained for both collagen and GAG ( Fig. 5 ). Conversely, in the 3-week construct maturity and high C-ABCint dose group, Saf-O and picrosirius red staining indicated a discontinuity at the interface. Flattened cells were also present at this interface as observed in the Saf-O stain. In the 5-week construct and low C-ABCint dose group, there was a distinct gap between the native and engineered tissue illustrating a lack of integration.
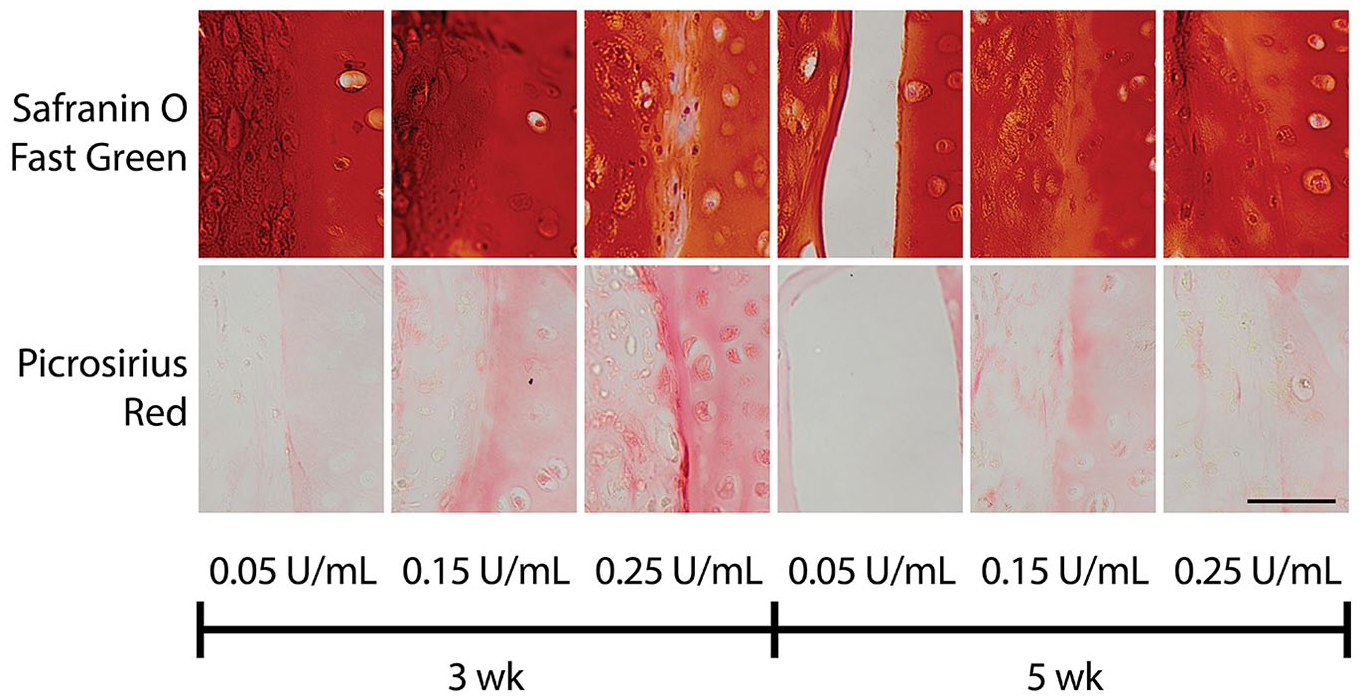
Effective C-ABCint dose to achieve integration for immature constructs was lower than for mature constructs (phase II). Histology of the engineered-native composites showed GAG content (Safranin O/Fast Green) and collagen content (picrosirius red) at the interface. In each image, the engineered construct is on the left and the native tissue is on the right. Staining illustrated tissue apposition and development of matrix across the interface in groups with 3-week constructs treated with either 0.05 (low) U/mL or 0.15 (medium) U/mL C-ABCint and groups with 5-week constructs treated with either 0.15 (medium) U/mL or 0.25 (high) U/mL C-ABCint. The scale bar represents 50 µm and applies to all images. C-ABCint, chondroitinase ABC integration treatment; GAG, glycosaminoglycan.
The interface Ey and UTS of engineered-native composites in phase II were significantly dependent on the interaction between the factors of construct maturation and C-ABCint, as the P values for the 2-way ANOVA interaction term were 0.0004 and 0.003, respectively ( Fig. 6A and B ). As individual factors, neither construct maturation nor C-ABCint concentration had a significant effect on interface Ey or UTS. For 3-week construct maturity, the low and medium doses of C-ABCint showed trends of increased interface Ey by 4.5-fold (P = 0.08) and 4.3-fold (P = 0.08), respectively, relative to the high dose of C-ABCint ( Fig. 6A ). For 5-week construct maturity, the medium and high doses of C-ABCint increased interface Ey 10.5-fold (P = 0.04) and 11.4-fold (P = 0.01), respectively, over the low dose of C-ABCint. Interface Ey for 3-week construct maturity combined with the low dose of C-ABCint was 1.02 ± 0.46 MPa. Interface Ey for 5-week construct maturity combined with the high dose of C-ABCint was 1.07 ± 0.36 MPa. These 2 groups demonstrated the best interface Ey. Across all groups, 3-week construct maturity treated with the low dose of C-ABCint led to the highest interface UTS (0.20 ± 0.11 MPa), which was significantly greater than the interface UTS of 3-week constructs treated with the high dose of C-ABCint (P = 0.02), and 5-week constructs treated with the low dose of C-ABCint (P = 0.01) ( Fig. 6B ). Gross morphologically ( Fig. 6C ), gaps were not present between neocartilage and native cartilage in groups that integrated effectively, but existed for the 3-week construct maturity and high C-ABCint dose group as well as the 5-week construct and low C-ABCint dose group (shown with arrows).
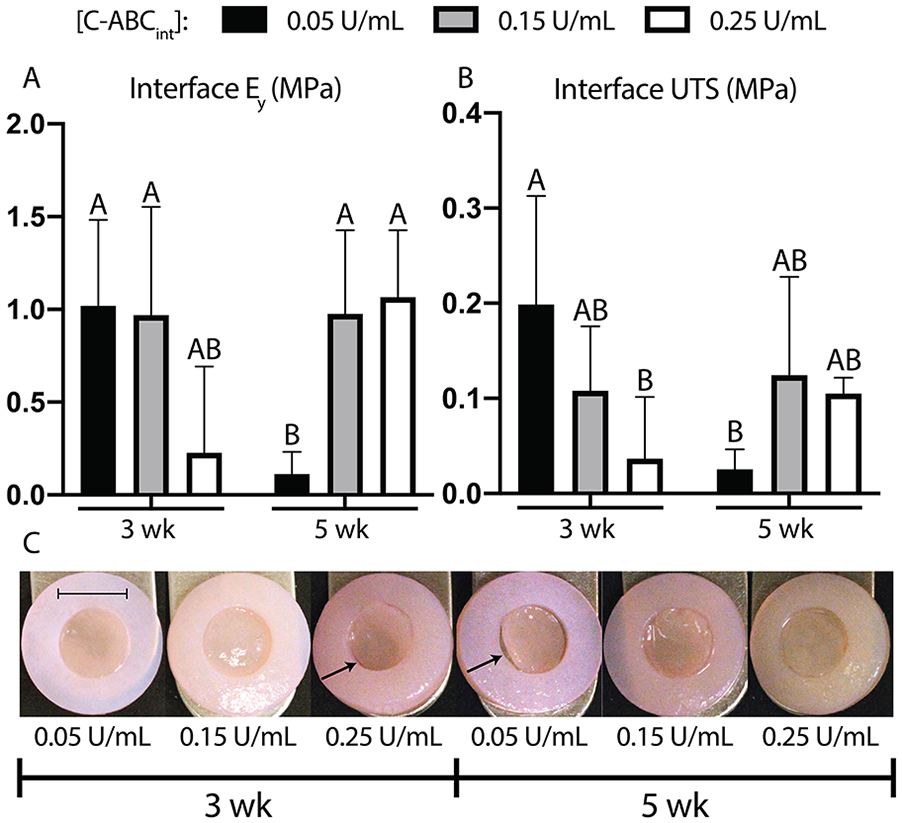
C-ABCint dose required for enhanced mechanical integration was higher for mature constructs than for immature constructs (phase II). Interface mechanical properties and gross morphology demonstrated the dependence of C-ABCint dose on construct maturity. (
Discussion
Toward restoring load-bearing function of articular cartilage, strides have been made in enhancing the mechanical properties of tissue-engineered neocartilage. While both the compressive and tensile properties of self-assembled articular cartilage have approached those of native tissue,32,33 an effective cartilage integration strategy is still lacking. For either new or existing cartilage therapies, cartilage-to-cartilage integration is crucial to the in vivo function, durability, and, ultimately, translational success of implants. An integration strategy that employs a catabolic agent should be designed to integrate cartilage as quickly as possible while using a minimal amount of that agent to avoid damaging implant properties. This study examined the ability of C-ABCint to enhance the integration of neocartilage to native cartilage in an in vitro defect model. C-ABCint dosages and neotissue maturation levels were modulated with the intent of identifying effective treatments that do not affect implant mechanical properties and, yet, are effective toward enhancing neotissue integration to native cartilage explants. In phase I, the hypothesis that C-ABCint combined with TCL would enhance the integration of self-assembled cartilage to native cartilage was confirmed. Indeed, TCL and C-ABCint together increased cartilage integration synergistically. In phase II, the hypothesis that C-ABCint at a lower dose would improve integration of immature self-assembled cartilage, but that a higher dose would be required for more mature constructs, was also supported. C-ABCint at a low dose enhanced the integration of immature constructs by up to 4.5-fold, while at a high dose, it improved mature self-assembled cartilage integration by up to 11.4-fold. Furthermore, it was confirmed that C-ABCint did not affect implant mechanical properties at any of the doses selected for testing. Ultimately, the results from this study demonstrate that C-ABCint can enhance both immature and mature self-assembled construct integration without compromising construct integrity.
C-ABCint demonstrated the capacity to enhance the integration of immature self-assembled articular cartilage. Specifically, 3-week constructs treated with either low or medium doses of C-ABCint experienced better integration than the high dose. It has been shown that that there is a positive correlation (R2 = 0.7773) between normalized integration strength and rate of change of equilibrium modulus of mesenchymal stem cell–seeded, hydrogel cartilage constructs, supporting the notion that tissues which are still developing, and therefore are more immature, integrate more effectively. 34 However, since 3-week constructs are less mature and still developing, if they are treated with a high dose of C-ABCint, they do not integrate effectively. This finding is likely due to excessive GAG removal by C-ABCint, and an inability for that GAG to recover within 2 weeks. Indeed, GAG content and compressive properties following a 2 U/mL C-ABC treatment have been shown to require at least 2 weeks to recover in self-assembled cartilage constructs. 16 Thus, for the purpose of cartilage integration within a short timeframe, using a minimal amount of C-ABCint is desired.
C-ABCint also demonstrated the capacity to enhance the integration of mature self-assembled articular cartilage, provided that a higher concentration of C-ABCint is applied to the neocartilage. Greater maturity and mechanical robustness of engineered tissue appears not to inhibit self-assembled articular cartilage integration. In other words, a more biomimetic self-assembled construct can be implanted with C-ABCint. It is well established that mature cartilage does not integrate effectively, 6 and success has been elusive for mature tissue-engineered cartilage implants as well.4,6,9,10 For example, it has been shown that cartilage constructs cultured for 5 days integrate more effectively than those cultured for 5 weeks. 35 Conversely, other studies have shown that construct maturity does not affect cartilage integration 36 or that precultured (i.e., mature) cartilage hydrogel constructs integrate more effectively than hydrogel constructs formed in situ. 37 We found that by increasing C-ABCint dosage, mature self-assembled cartilage constructs could be integrated as well. Indeed, a low dose of C-ABCint led to minimal GAG removal, likely due to increased collagen density at the construct edge ( Fig. 3 ). The medium and high doses, however, allowed for greater C-ABCint diffusion into the neotissue, and, therefore, more efficient GAG removal ( Fig. 3 ). Catabolic enzymes like C-ABC have been shown to improve tensile characteristics of engineered and native articular cartilage by removing ECM components like chondroitin sulfate that could interfere with the collagen network’s ability to remodel and develop.14,16 A similar mechanism is possibly responsible for enhancing interface tensile properties in engineered-native composites, provided the dose of C-ABCint is high enough to remove sufficient GAG. Given this capacity to improve integration of mature self-assembled cartilage constructs, C-ABCint dosage could be tailored to enhance the integration of a variety of engineered cartilage constructs, such as highly biomimetic cartilage implants.
C-ABCint was able to enhance cartilage integration without affecting whole construct properties. Other groups have shown that catabolic enzymes like C-ABC can be used to improve engineered cartilage integration4,9,21-23; however, this study illustrates how it can enhance cartilage integration without diminishing construct properties. No dose of C-ABCint evaluated in this study affected any construct mechanical properties ( Fig. 4 ). Construct mechanical integrity must remain intact if it is to withstand the loads present within a synovial joint and subsequently integrate. 38 C-ABC at high concentrations is known to decrease compressive properties of constructs transiently,16,25 but many studies investigating catabolic enzymes as cartilage integration agents have not assessed implant mechanical properties following treatment.4,9,21-23 This lack of data hampers evaluation of the translational potential of these strategies. This study identified C-ABCint as an effective integration treatment that does not adversely affect self-assembled construct properties.
C-ABCint and TCL synergistically improved the integration of self-assembled cartilage constructs to native tissue. C-ABCint treatment increased the interface Ey and UTS relative to untreated controls by more than 6-fold and 4-fold, respectively, and worked synergistically with TCL. Notably, the interface Ey of TCL- and C-ABCint-treated engineered-native composites reached levels more than 5-fold higher than previously shown by our group, 7 while the interface UTS was on par with other integration models that extended composite culture from 2 weeks to 4 weeks.9,39 Additionally, the best integration Ey values shown in this study are approaching the Ey of self-assembled articular cartilage shown in this study and others,32,40 indicating a developing, mechanically stiff interface. The reduction of GAG-induced steric hindrance at the periphery of implants via C-ABCint possibly enabled more effective interfacial collagen crosslinking due to increased collagen network remodeling and interaction, 15 which would explain the discrepancy between interface Ey and UTS values in groups receiving TCL but with or without C-ABCint treatment. Prior work has demonstrated the beneficial effect of LOXL2 alone on integration of self-assembled articular cartilage, 10 and this effect is enhanced with the addition of C-ABCint. In addition to TCL, C-ABCint could potentially be combined with a variety of cartilage construct treatments to further improve integration to native articular cartilage.
Another mechanism through which C-ABCint could enhance integration of self-assembled articular cartilage is by encouraging chondrocyte migration via reduction of GAG negative charge density at the construct edge.11,12 C-ABC-mediated chondroitin sulfate removal has been shown to increase chondrocyte migration in vitro. 11 Importantly, in that study, migrating chondrocytes regained their rounded, chondrocytic phenotype once they had migrated into adjacent tissue. The hypercellularity of self-assembled neocartilage would provide a substantial number of potentially migratory cells that could be released following C-ABCint treatment. Trypsin has also been investigated as a GAG-depleting, integration enhancer. 9 However, trypsin pre-treatment of chondrocyte-seeded hydrogels only improved integration strength when it was used in concert with HB-IGF-1 (heparin-binding insulin-like growth factor–1), which can effect both chondrocyte motility and anabolism.9,41 Self-assembled constructs subjected to C-ABCint did not require a motility agent for improved integration strength, suggesting that the cells within this neotissue could have greater migration potential than those in a hydrogel. It is possible that certain, inherent characteristics of self-assembled constructs, such as hypercellularity, 42 and metabolic activity of juvenile bovine ACs,13,32 led to C-ABCint being a more effective enhancer of integration than if it were applied to native articular cartilage or scaffold-based cartilage constructs in the same way. Furthermore, enzymatic treatment of the native tissue ring was not necessary to facilitate integration, suggesting that the properties of self-assembled constructs treated with C-ABCint are sufficient for enhanced cartilage integration. Ultimately, the characteristics of self-assembled articular cartilage lend themselves to transient, small-scale depletion of GAG via C-ABCint to facilitate cartilage integration, as the implant has demonstrated the capacity to secrete collagen at the wound interface and recover GAG content subsequently, suggesting the development of a homogeneous, contiguous interface.
While promising, the results of this study could be further enhanced in a variety of ways. In general, in vitro methods of cartilage integration have yet to produce cartilage integration strength that approaches the UTS of native, intact articular cartilage. Additional culture time after forming the composites of engineered and native tissue beyond the 2 weeks examined in this experiment could further enhance integration. Our group is interested in extending in vitro composite culture duration in future integration studies to assess longer-term effects of treatments since previous work has shown temporal differences in in vitro integration. 34 Also, for phase II, LOXL2 and 0.15 U/mL C-ABCint treatment were carried forward due to their positive effect on cartilage integration in phase I. TGF-β1 and C-ABC as bioactive factors were omitted to more closely examine the effects of construct maturation time and varying concentrations of C-ABCint had on both interfacial and construct properties. However, it would be interesting to explore the effect of modulating C-ABCint dose on constructs treated with TCL or other bioactive stimuli, since additional combinations could further enhance interface properties. Additionally, measuring specific ECM components present at the interface, through LCMS techniques, for example, could verify the presence of cartilaginous repair tissue and exclude the possibility that fibrocartilage instead has formed at the interface.
Other methods of enhancing cartilage integration, such as additional enzymatic treatments,15,22 chondrocyte motility agents, 9 or other bioactive factors, 39 could also yield improved in vitro cartilage integration strength. However, we expect that the in vivo environment and associated stimuli are required to elicit the integration strength desired for stability, durability, and long-term efficacy of an implant. 23 Ectopic in vivo implantation has been shown to improve integration strength by 230% between native and engineered cartilage relative to in vitro controls. 7 In particular, mechanical stimulation could further enhance integration. Fluid-induced shear stress has previously been shown to enhance cartilage integration by 1.89-fold. 43 Composites in this study were subjected to fluid-induced shear stress since they were placed on an orbital shaker during culture, but this stimulus could be rigorously investigated further to determine its impact on in vitro self-assembled cartilage integration. Due to joint articulation, the orthotopic, in vivo environment would provide this, as well as other, mechanical stimuli that could further enhance integration, so long as the implant is protected sufficiently until it is stable within a defect. In this study, interface Ey approached those of intact self-assembled constructs within 2 weeks of in vitro composite culture. Thus, upon in vivo implantation, a period of 2 weeks or less may be sufficient for construct stability within the host articular cartilage environment. We are interested in testing this strategy in an orthotopic, large animal, in vivo defect model as this model would be more representative of the integration potential of self-assembled articular cartilage treated with C-ABCint, and would represent progress toward the clinical translation of self-assembled articular cartilage.
In this study, C-ABCint enhanced integration of both immature and mature self-assembled articular cartilage to native tissue. While its potential is promising, C-ABCint could be combined with other treatments, such as additional growth factors, motility agents, or catabolic factors to further enhance its integration capacity. Ultimately, the straightforward nature of applying a GAG-removal agent at the periphery of a self-assembled articular cartilage implant could be readily implemented into a clinical setting and should be investigated further as this type of neocartilage construct nears clinical translation.
Supplemental Material
Supplemental_Material_Table_S1 – Supplemental material for Chondroitinase ABC Enhances Integration of Self-Assembled Articular Cartilage, but Its Dosage Needs to Be Moderated Based on Neocartilage Maturity
Supplemental material, Supplemental_Material_Table_S1 for Chondroitinase ABC Enhances Integration of Self-Assembled Articular Cartilage, but Its Dosage Needs to Be Moderated Based on Neocartilage Maturity by Jarrett M. Link, Jerry C. Hu and Kyriacos A. Athanasiou in CARTILAGE
Footnotes
Supplemental Material
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH grant R01AR067821. JML was also supported by the NSF Graduate Research Fellowship Program (DGE-1321846). Ben Bielajew’s assistance with PYR quantification is greatly appreciated.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not sought for the present study because human subjects were not involved in this work.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because live animals were not used (i.e., all animal tissues were obtained from a commercial provider of animal specimens).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
