Abstract
Objective. To evaluate minimum biosecurity parameters (MBP) for arthroscopic matrix-encapsulated autologous chondrocyte implantation (AMECI) based on patients’ clinical outcomes, magnetic resonance imaging (MRI) T2-mapping, Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) score, and International Cartilage Repair Society (ICRS) second-look arthroscopic evaluation, laying the basis for a future multicenter study. Design. Pilot clinical study. We analyzed the logistics to perform AMECI to treat focal chondral lesions in different hospitals following strict biosecurity parameters related to tissue and construct transportation, chondrocyte isolation, and cell expansion. Patient progress was analyzed with patient-reported outcome measures, MRI T2-mapping, MOCART, and ICRS arthroscopic second-look evaluation. Results. Thirty-five lesions in 30 patients treated in 7 different hospitals were evaluated. Cell viability before implantation was >90%. Cell viability in construct remnants was 87% ± 11% at 24 hours, 75% ± 17.1% at 48 hours, and 60% ± 8% at 72 hours after implantation. Mean final follow-up was 37 months (12-72 months). Patients showed statistically significant improvement in all clinical scores and MOCART evaluations. MRI T2-mapping evaluation showed significant decrease in relaxation time from 61.2 ± 14.3 to 42.9 ± 7.2 ms (P < 0.05). Arthroscopic second-look evaluation showed grade II “near normal” tissue in 83% of patients. Two treatment failures were documented. Conclusions. It was feasible to perform AMECI in 7 different institutions in a large metropolitan area following our biosecurity measures without any implant-related complication. Treated patients showed improvement in clinical, MRI T2-mapping, and MOCART scores, as well as a low failure rate and a favorable ICRS arthroscopic evaluation at a mid-term follow-up. Level of Evidence. 2b.
Keywords
Introduction
Matrix-associated autologous chondrocyte transplantation (MACT) or third-generation autologous chondrocyte implantation (ACI) is a restorative treatment option for focal chondral lesions in the knee. Chondrocytes seeded onto scaffolds have overcome several problems related to first- and second-generation ACI such as periosteal patch hypertrophy, extensive suturing, and cell leakage; furthermore, it has allowed implantation to be arthroscopic, reducing the morbidity, surgical time, recovery, and complications related to surgery.1 -4 Since its introduction into clinical practice in 1998, MACT has shown positive clinical and imaging outcomes, as well as the formation of hyaline-like cartilage.1 -3,5 -8
We have previously described an all arthroscopic matrix-encapsulated autologous chondrocyte implantation (AMECI) technique for cartilage repair, with evidence of hyaline-like tissue formation in porcine models, 9 as well as positive cell viability before and after arthroscopic implantation in equine models. 10 Clinical studies with AMECI have shown safety and efficacy in young patients in a controlled scenario.3,6 Even before our technique was first described in 2014, 3 several surgeons had been trained in order to reproduce this procedure elsewhere. Due to the positive findings in previous studies, we have decided to take our technique to a multicenter scenario.
Multicenter studies arise from the need to test intervention reproducibility under the usual conditions in which it will be applied. 11 Unlike single-center clinical trials, they have greater external validity since the intervention outcomes are not dependent on a specific surgeon or hospital. 12 Despite the countless benefits of these studies, 13 several things must be established in order to carry out a multicenter study. Surgeons must be trained and “ideally” certified to perform the intervention correctly, strict biosecurity measures must be followed to ensure quality and reproducibility, and the tested procedure must have shown positive results in previous studies. 14 Pilot studies help gather any missing part, as well as to analyze feasibility prior to performing a large-scale trial. 15 Adequate biosecurity measures for new tissue engineering techniques in cartilage repair are fundamental for success and reproducibility in places far away from the “good manufacturing practices” (GMP) laboratory. Unfortunately, due to proprietary information, biosecurity measures for chondrocyte and construct transportation are vaguely described in MACT multicenter studies.5,8,16-20 The purpose of this study was to evaluate minimum biosecurity parameters (MBP) for our technique (AMECI) based on patient-reported outcome measures (PROMs), magnetic resonance imaging (MRI) T2-mapping, the Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) score, and International Cartilage Repair Society (ICRS) second-look arthroscopic evaluations in a pilot multicenter study. We hypothesize that after following MBPs for AMECI, patients will have a significant improvement in PROMs, cartilage quality on T2-mapping, MOCART scores, and second-look arthroscopic evaluations, with a low failure rate and no logistics implant-related complications, laying the basis for a future multicenter study in Latin America.
Methods
This pilot study was performed in a multicenter clinical setting with 7 different hospitals, including the National Institute of Rehabilitation “Luis Guillermo Ibarra Ibarra” (INRLGII), where the GMP laboratory for cellular expansion and implant construction is located. All hospitals were located in Mexico, in a radius of no more than 50 miles away from the GMP facility. The procedures were standardized and performed by fellowship-trained orthopedic surgeons. All surgeons were previously trained in the arthroscopic implantation technique through a comprehensive program provided by the INRLGII where they received an instructional video, surgical manual, and the postoperative rehabilitation protocol. Patients were enrolled in any of the 7 participating hospitals, from January 2012 to December 2017. Inclusion criteria were patients 18 to 50 years of age, with symptomatic full thickness-cartilage lesion(s) ICRS grade III-IV diagnosed by MRI, on either femoral condyle, trochlea, or patella. Failed conservative treatment was not a mandatory inclusion criterion if patients were willing to undergo surgical treatment; however, all 7 participating hospitals are third-level referral hospitals with most of the patients being refractory to conservative measures before enrollment. All patients had bilateral knee anteroposterior, lateral and Merchant x-ray views, as well as a full-length weightbearing anteroposterior view prior to enrollment. Any misalignment in the mechanical axis of more than 25% away from the neutral line (either in varus or valgus) was considered for alignment osteotomy, 21 and any tibial tuberosity–trochlear groove (TT-TG) distance greater than 20 mm at computed tomography (CT) scan was considered for Fulkerson osteotomy. 22 Exclusion criteria included any systemic disease, arthritis or previous total meniscectomy, previous surgical chondral lesion repair, infection, or tumor in the knee. The study was performed following the Declaration of Helsinki and was approved by the local ethics committee and the internal review board, with approval number INRLGII: 67/17. Written informed consent was obtained from all patients.
Surgical Technique
We used a novel 100% arthroscopic technique to repair chondral defects with AMECI. Two surgical procedures were performed, both under regional anesthesia. During the first procedure, an arthroscopic cartilage biopsy was obtained, and the chondral lesion was debrided and measured. The second procedure, 6 to 8 weeks later, consisted on the arthroscopic chondrocyte implantation as previously described in detail.3,23,24 Any concomitant ligament, meniscus, or knee misalignment problem was addressed during the first procedure to avoid an intense inflammatory response in the implantation surgery since inflammation has been described as a nocive factor for cartilage, promoting degeneration and ossification.25-29
Cartilage Biopsy
During routine arthroscopy, chondral lesion(s) was/were identified, debrided, and measured. Two to 3 osteochondral biopsies were obtained from a nonweightbearing area on the lateral edge of the intercondylar notch with a 4-mm osteochondral graft harvester (COR; DePuy Mitek, Raynham, MA) ( Fig. 1a and b ). During the biopsy surgery, a 120-mL blood sample was obtained from the patient for autologous serum extraction.
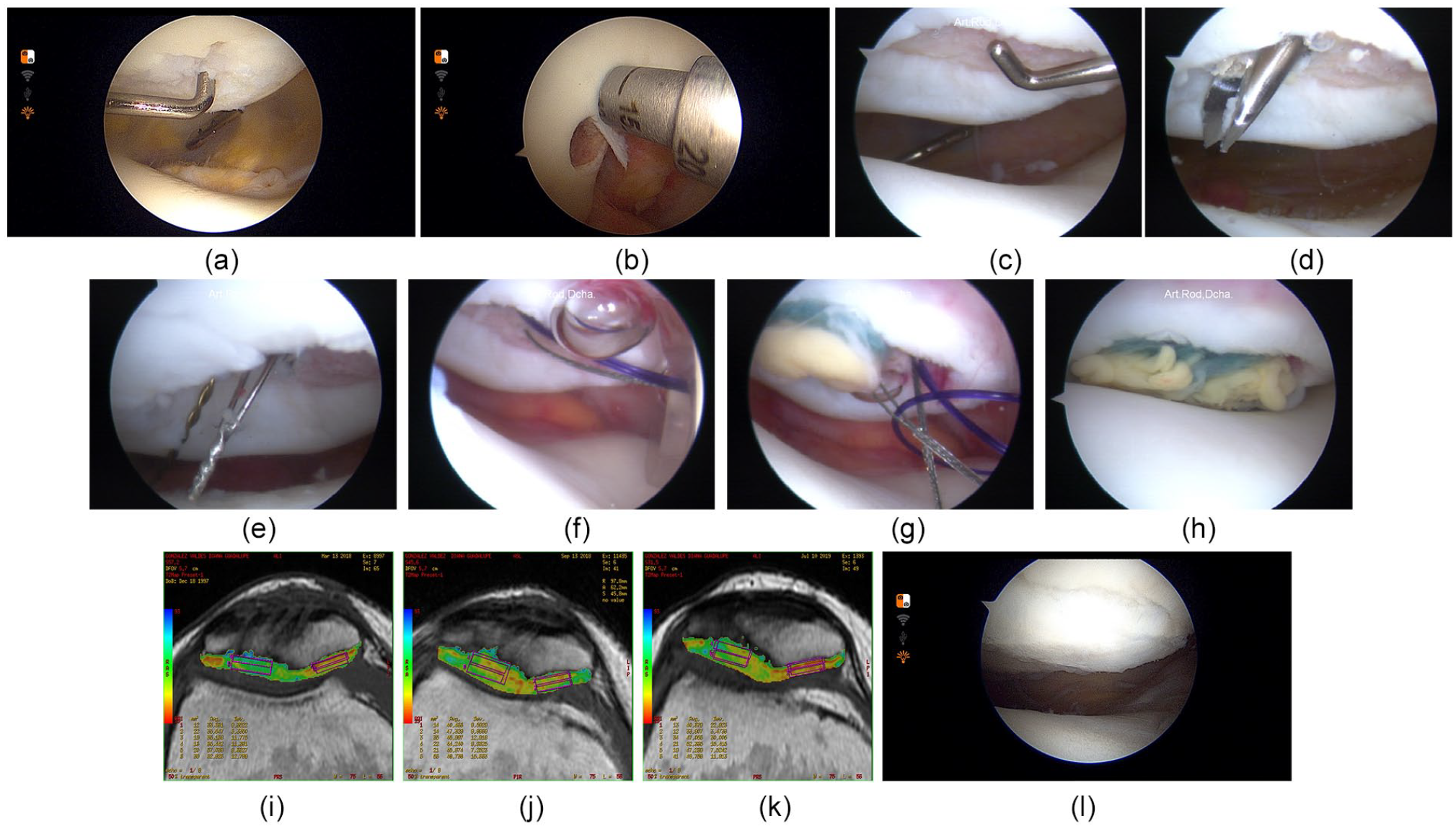
Surgical technique for arthroscopic matrix-encapsulated autologous chondrocyte implantation (AMECI), T2-mapping outcomes, and second look.
Arthroscopic Implantation on Femoral Condyles and Trochlea
The chondral lesion was debrided using spoons and curettes, trying to form 8-mm circular lesions with healthy cartilage borders. A bioabsorbable mini anchor (DePuy Mitek, Inc., Raynham, MA) loaded with a 0 PDS suture was placed at the center of the defect. In a side surgical table, 2 needles (16G) were passed through the polymer (polyglycolic acid + chondrocytes) (Neoveil Sheet, Gunze Medical Division, Tokyo, Japan), and the anchor sutures were passed through the needles. Using the anchor sutures in the polymer, a low-profile sliding arthroscopic knot was made to introduce the polymer into the joint through a clear 10-mm cannula (Smith & Nephew, Inc., Andover, MA) with the water pump decreased to gravity pressure. Implant stability was checked by dynamic arthroscopic evaluation. The procedure was repeated for each implant until the whole lesion was covered, as described in detail previously.3,23,24
Arthroscopic Implantation on the Patella
The chondral lesion was debrided with a curette, trying to form 8-mm circular lesions ( Fig. 1c ). A tibial guide for anterior cruciate ligament reconstruction (ACL guide, DePuyMitek, Inc., Raynham, MA), was placed inside the joint. A 2-cm-long mini approach was performed on the patella. The patella was drilled through the ACL guide with a 2 mm Steinmann pin through the joint in a “retro-drilling” way until the pin was visualized at one side of the chondral lesion. Following the same steps, a second perforation was made on the opposite side of the cartilage lesion, with no more than 4 mm between each pin ( Fig. 1d ). A suture shuttling device named Chia Percpasser (DePuy Mitek, Inc., Raynham, MA) was introduced through each perforation made by the pins in the patella ( Fig. 1e ). Both Chia Percpasser were recovered inside the joint and extracted toward one of the portals. At the same time, on a side surgical table, an assistant mounted the 8-mm cell-polymer construct with a 0 PDS suture using two 16G needles. Both ends of the PDS were mounted to the loop of each Chia Percpasser. The sutures were recovered by pulling the Chia from the anterior aspect of the patella at the extraarticular level ( Fig. 1f ). Each polymer bound to the PDS suture was introduced to the joint using a clear 10-mm cannula, decreasing the water pump pressure to gravity pressure ( Fig. 1g ). Once the polymer was firmly placed at the bottom of the chondral defect, a PDS knot was made at the anterior cortical surface of the patella. The process was repeated in case of a bigger lesion. Implant stability was verified by dynamic arthroscopic vision ( Fig. 1h ).
Tissue Handling and Transport
Cell Transportation and Biosecurity Measures
For patients to be scheduled for the procedure at institutions other than the INRLGII, surgeons had to request the biopsy kit 72 hours in advance. The biopsy kit included an insulated cooler, 4 to 6 gel ice packs, twenty 6-mL test tubes (Vacutainer) for blood samples, a test tube rack, and a sterile 30-mL polypropylene conical tube with 10 mL of culture medium Dulbecco’s modified Eagle medium-F12 medium (DMEM) + 10% antibiotic/antimycotic solution (Dulbecco’s modified Eagle Medium F12 GIBCO, Grand Island, NY). Osteochondral specimens obtained at biopsy surgery were placed inside the 30-mL conical tube with medium, and transported inside the cooler to the GMP lab, maintaining the temperature at an average of 5.5°C ± 2.2°C and humidity at an average of 75.82%±7.6%. All biopsies were delivered to the lab in less than 6 hours after obtention. For implantation surgery, once the cell-polymer constructs were ready, they were transported to the hospital in a similar 30-mL polypropylene sterile conical tube with 10 mL of culture medium + 10% antibiotic/antimycotic solution. The tube with the constructs was placed inside an insulated cooler with no ice at a temperature of 19.7°C ± 4.7°C, and humidity of 50.1% ± 15.5%. All constructs were implanted in approximately 2 to 5 hours after leaving the GMP lab. All biosecurity measures followed in this pilot multicenter study are shown in Table 1 .
Minimum Biosecurity Parameters (MBP) Recommended for Arthroscopic Matrix-Encapsulated Autologous Chondrocyte Implantation (AMECI).
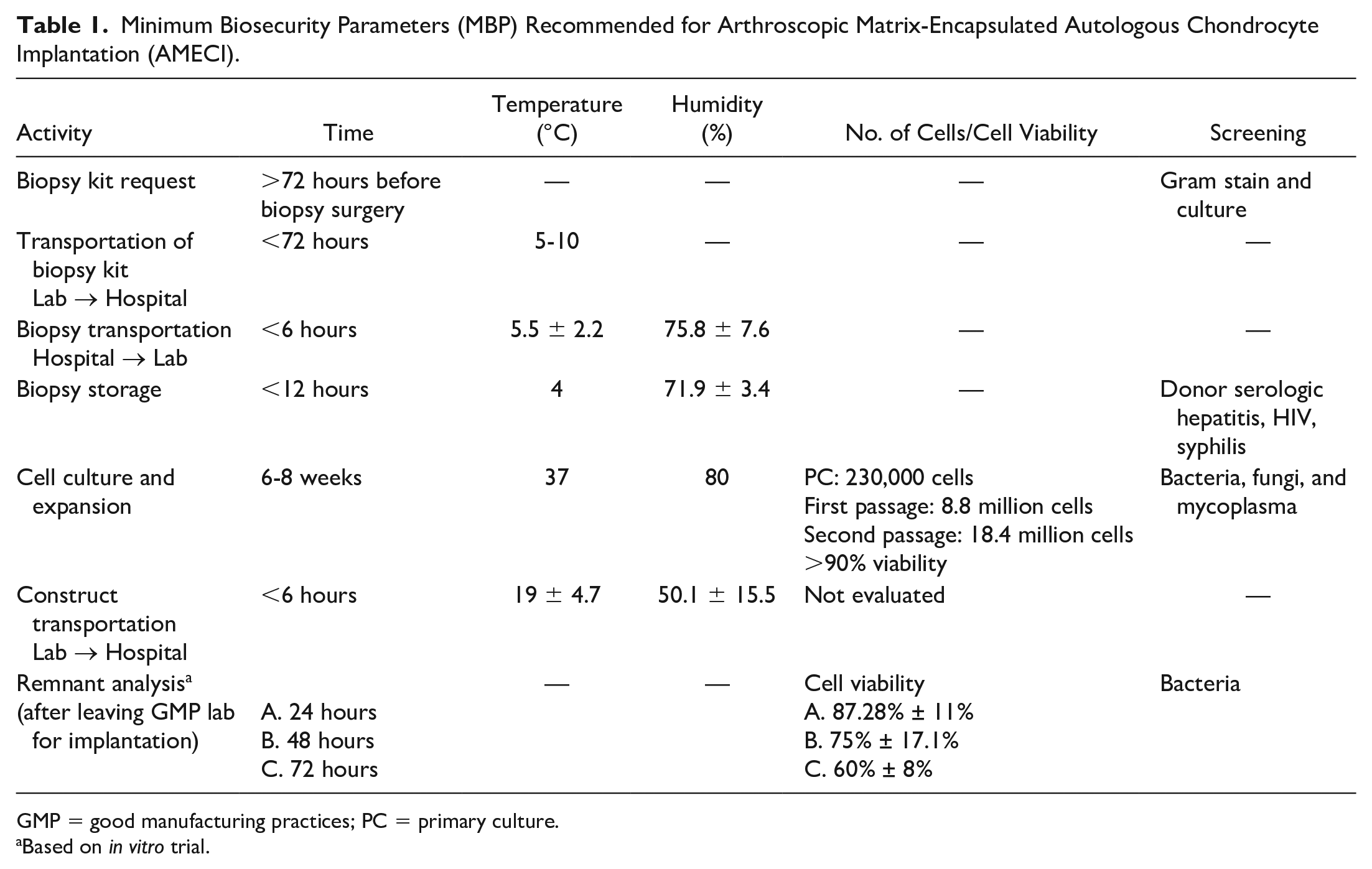
GMP = good manufacturing practices; PC = primary culture.
Based on in vitro trial.
Chondrocyte Isolation, Expansion, and Construct Formation
Cartilage and blood samples were kept in the transportation tubes inside the cooler with gel ice packs and then at 4°C until isolation, normally performed within the first 12 hours after arriving to the lab. For security measures, serologic hepatitis, HIV, and syphilis screening was performed to all patients from their blood sample before processing the biopsy specimens. All procedures were performed under sterile conditions in a laminar flow hood in a class-100 clean room with positive pressure. The transported medium was removed, and samples were washed 3 times with sterile phosphate buffered saline + 10% antibiotic/antimycotic agents. Cartilage was separated from the osteochondral plug, chopped into small fragments with a scalpel and digested in class II collagenase (Worthington) for 4 to 5 hours. The enzymatic digestion was carried out at 37°C and constant agitation of approximately 200 rpm. Isolated chondrocytes were counted, and viability was assessed by trypan blue stain using a hemocytometer. Samples of the cell suspension were sent to an independent laboratory for microbiological evaluation (bacteria, fungi, mycoplasma). In the case of a positive screening test or culture, cell expansion was suspended. After quality control was verified, cells were seeded onto a T25 culture flask (Primaria Falcon) at a density of 10,000/cm2 with culture medium (DMEM), 1% antibiotic-antimycotic agents and 10% autologous serum. The culture flask was placed in an incubator at 37°C, 5% CO2, and 80% humidity; culture medium was changed every 2 to 3 days. When 100% confluence was achieved, chondrocytes were detached with trypsin and re-seeded in culture flasks until passage 2. Cells could be cryopreserved before passage 2 if the implantation surgery was not scheduled within the next 8 weeks. At the beginning of passage 2, 33% of the cells were seeded onto a Petri dish with conventional culture medium supplemented with ascorbic acid (60 µg/mL) to induce monolayer formation, while the remaining cells were expanded in T75 flasks at a concentration of 25,000 cell/cm2. Once chondrocytes in passage 2 reached 100% confluence, cells in the T75 culture flask were separated and centrifuged for 10 minutes at 1500 rpm to obtain a pellet of chondrocytes. Chondrocytes expanded in monolayer were detached from the Petri dish bottom using a cell scraper. An 8-mm diameter polyglycolic acid scaffold disc (Neoveil Sheet, Gunze Medical Division, Tokyo, Japan) was placed over this monolayer and the pellet of chondrocytes was carefully placed on top of the scaffold. Finally, the cell-polymer scaffolds were enveloped with the cell-matrix monolayer using sterile surgical forceps. 10 The constructs were cultured for 1 week to allow cell adherence and matrix production. Constructs were sent to the implantation facility on the same day of the second surgical procedure in sterile containers with culture medium.
Rehabilitation Protocol
After surgery, all patients underwent rehabilitation following the same protocol. Pain control medications such as nonsteroidal anti-inflammatory drugs (NSAIDs) were indicated for 5 to 7 days period, Acetaminophen or low-intensity opioids were allowed as needed for pain after biopsy or implantation surgery. No intra-articular injections were allowed for the following 2 years after the implantation surgery. After the biopsy procedure, partial weightbearing assisted with crutches was indicated for 2 weeks, followed by full weightbearing. If patients underwent another concomitant procedure, a different rehabilitation protocol was followed depending on the assessed problem. After the implantation surgery, isometric quadriceps exercises were started at day 1 postoperatively (PO). Patients with defects in the tibiofemoral joint had touchdown weightbearing assisted with crutches, and used a hinged knee brace for 6 weeks, allowing progressive knee mobilization since day 1. Patients with patellofemoral joint lesions had partial weightbearing assisted with crutches and a knee brace locked in full extension for walking during the first 6 weeks. Regardless of the defect location, the range of motion was increased progressively following the continuous passive motion (CPM) protocol. Passive motion exercises with CPM were done 4 to 6 hours daily, intermittently, starting on day 3 PO at 0° to 40°. Each week, 10° of flexion was increased until complete range of motion was obtained at 6 to 8 weeks approximately. Progressive open-chain strengthening was initiated after a first isokinetic evaluation at 4 months PO. Unloader braces were not indicated, instead, patients were instructed to use crutches for nonweightbearing or partial weightbearing. Patients returned to high-demand sports activities after 12 months and when isokinetic evaluation reported 90% of the strength of the contralateral extensor and flexor muscles of the knee.3,30
Evaluation
Viability and Cell Density
Cell density and viability was assessed in all patients using trypan blue stain in a hemocytometer after chondrocyte isolation, before passage 1, and before passage 2 during cell culture. Spare cell-polymer constructs that were not used for implantation in patients were analyzed for cell viability with the LIVE/DEAD kit (Calceín AM/Ethidium homodimer -1, Molecular Probes, Invitrogen) at 24 hours after leaving the lab for implantation. One patient’s remnants were also evaluated for cell viability at 48 and 72 hours. Polymer remnants were kept in the same conditions as they were transported from the lab to the operating room for implantation.
Clinical Evaluation
PROMs were used to evaluate symptoms and functionality. Lysholm, Tegner, subjective International Knee Documentation Committee (IKDC), Knee injury and Osteoarthritis Outcome Score (KOOS) with its 5 subscales, and Kujala scores were documented preoperatively and at final follow-up.
Second-Look Arthroscopy
A second-look arthroscopy was made at 12 to 18 months PO in patients who agreed and gave written consent. An experienced arthroscopic surgeon, different from the implantation surgeon, carried out the evaluation using the ICRS Cartilage Repair Assessment (CRA)31,32 by watching the surgical video. No histologic analysis of repair tissue was performed.
Magnetic Resonance Imaging Evaluation
For imaging evaluation, we used T2-mapping and the MOCART 2.0 score.33,34 T2-mapping was taken preoperatively, at 12 months PO and at final follow-up. MOCART scores were taken at 3 and 12 months PO, and at final follow-up. MRI was performed on a 1.5-T clinical imaging system (GE Healthcare, Milwaukee WI), using an 8-channel HD knee array (GE Healthcare). Standard morphologic MRI evaluation was performed using a fast spin-echo sequence in the axial, sagittal, and coronal planes. Images were acquired with a repetition time of 1800 to 1450 ms, echo time of 30 to 40 ms, echo train length of 6, and spatial resolution of 256 mm (frequency), 256 mm (phase), 3 mm at 2 excitations.
T2-mapping (FuncTool 4.5.1, GE Healthcare, Little Chalfont, Buckinghamshire, UK) was performed to assess the biochemical integrity of native and repaired cartilage. The color map is coded to capture T2 values ranging from 25 to 91 ms. Quantitative T2-mapping was performed using a multislice multiecho pulse sequence. Eight echoes were sampled: sequential multiples of the first echo time (10-11 ms) at a repetition time of 800 ms and in-plane resolution of 384 mm (frequency), 256 mm (phase), 3 mm at 2 excitations. Data sets were analyzed (FuncTool 4.5.1; GE Healthcare). T2 values were calculated taking 6 regions of interest (ROIs). ROI 1 and 2 were 2-mm2 rectangular areas located in the healthy native cartilage next to the implanted region. ROI 1 was positioned in the deep layer of the cartilage while ROI 2 was placed on the superficial layer. ROI 3 was a 4-mm2 rectangular area that included ROI 1 and ROI 2, making this the average score. ROIs 4, 5, and 6 were sized, shaped, and placed in the same manner as the previous ones but over the repaired cartilage. The values of the 6 ROIs were recorded ( Fig. 1i-k ). ROI 3 was used as the “Native ROI” and ROI 6 was used as the “Repair ROI.”
The qualitative evaluation of cartilage repair was performed by an independent cartilage expert radiologist using the MOCART 2.0 score and the visual examples presented in the original article. Two planes were used to grade the MOCART score.
Treatment Failure
Failure was defined as the need for reoperation due to symptoms caused by the primary defect as used in previous studies.2,35 Second-look arthroscopy for outcome evaluation was not considered a treatment failure.
Statistical Analysis
All continuous data were expressed in terms of mean ± SD; the categorical data were expressed as frequencies and percentages. Shapiro-Wilk test was performed to assess the normality of continuous variables. Initial PROM, T2-mapping, and MOCART (3 months PO) were compared with final follow-up scores using Student t test if the distribution was normal, and nonparametric tests if not. P values <0.05 were considered statistically significant. Statistical analysis was done using SPSS software version 20 (IBM Corp.).
Results
Viability and Cell Density
The average cell count with trypan blue stain starting the primary culture was 230,000 cells, 8.8 million cells starting the first passage, and 18.4 million cells starting the second passage with a viability of >90%. Six patients had spare constructs after implantation surgery and were returned to lab for viability evaluation after 24 hours with the LIVE/DEAD kit obtaining an average of 87.28% ± 11%. Remnants of 1 patient were also evaluated at 48 and 72 hours with average cell viability of 75% ± 17.1% and 60% ± 8%, respectively. Remnants or culture medium were also cultured and tested for bacteria contamination 48 hours after leaving the lab for implantation, documenting no cell-polymer construct contamination ( Table 1 ).
Patient Data and Baseline Characteristics
Between 2012 and 2017, a total of 30 patients were included in the study from 7 different hospitals in Mexico. Out of the 30 patients, 1 patient was lost during follow-up. We report on 35 chondral lesions found and treated in 29 different patients with a mean final follow-up of 37 months (12-72 months). The demographic characteristics of the population are shown in Table 2 .
Demographic Characteristics of the Population (Patients N = 29; Lesions = 35).

BMI = body mass index; ACL = anterior cruciate ligament; MM = medial meniscus, LM = lateral meniscus; LRR = lateral retinaculum release.
Clinical Outcomes
There was a statistically significant improvement in all patients from preoperative scores to final follow-up ( Fig. 2 ) with a weighted improvement of 25.6 points in Lysholm, 2.7 in Tegner, 20.7 in IKDC(S), 20.4 in Kujala, 33.4 in KOOS-SR (sports and recreation), 35.1 in KOOS-QoL (quality of life), 25.8 KOOS-P (pain), 17.4 KOOS-S (symptoms), and 23.9 in KOOS-ADL (activities of daily living).
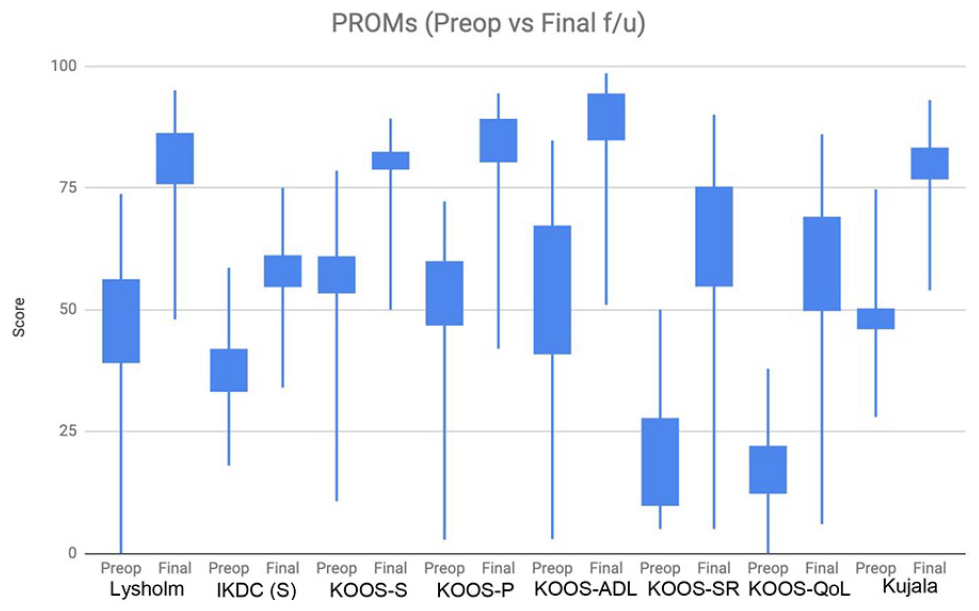
Patient-reported outcome measures (preoperative versus final follow-up).
Magnetic Resonance Imaging Evaluation
Quantitative MRI T2-mapping evaluation with the ROIs ( Fig. 1i-k ) and MOCART scores are shown in Table 3 . The repair ROI showed a statistically significant decrease from 61.2 ± 14.3 to 42.9 ± 7.2 at final follow-up (P < 0.05). Native ROI had no significant changes from preoperative to final follow-up values (P = 0.755). MOCART scores increased from 45.4 ± 15.7 at 3 months PO to 70.14 ± 12.6 at final follow-up with a significant P value (P < 0.05). Complete volume fill of cartilage defect was found in more than 60% of the repair lesions, and complete integration into adjacent cartilage or <2-mm interface in 88%.
T2-Mapping and MOCART Evaluation.
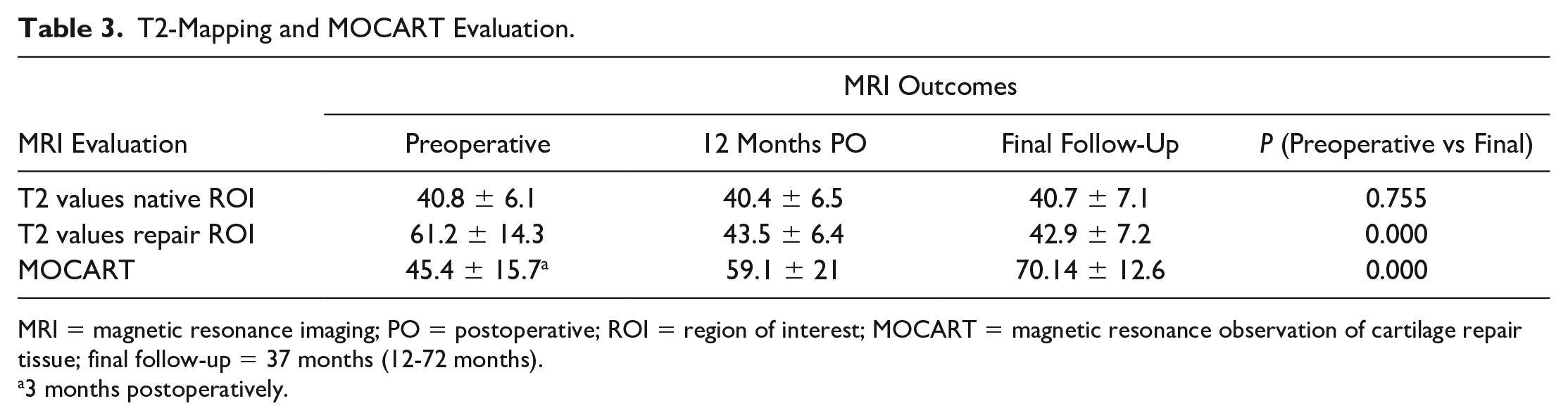
MRI = magnetic resonance imaging; PO = postoperative; ROI = region of interest; MOCART = magnetic resonance observation of cartilage repair tissue; final follow-up = 37 months (12-72 months).
3 months postoperatively.
Second-Look Evaluation
Twelve patients accepted an arthroscopic second look at 12 to 18 months postoperative. Out of the 12 patients, 10 (83%) had a grade II “near normal” cartilage repair, with a mean score of 8.7 ± 1.9 ICRS CRA ( Fig. 1l ).31,32
Failures
There were 2 failures in patients from INRLGII: one with a patellar lesion who underwent reoperation for graft debridement, chondroplasty, and lateral retinaculum release after 18 months of implantation and the other with a lateral femoral condyle lesion and an uncorrected marginal alignment in valgus who underwent reoperation after 72 months PO for chondroplasty and subsequent multimodal nonoperative approach. There were no infections, neoplasia, or knee replacements in patients in this study up to the final follow-up. All patients, including these 2 failures, were included in the final analysis.
Discussion
We report on 30 patients (35 lesions) treated with AMECI for focal cartilage lesions in the knee. All patients showed a statistically significant improvement in all clinical scores, quantitative MRI evaluation with T2-mapping, and MOCART scores at final follow-up (mean 37 months; range 12-72 months). Second-look arthroscopic evaluation was considered “near normal” by the ICRS Macroscopic Evaluation Score in 10 out of 12 who accepted the intervention. ROI values of the repair cartilage decreased over time to values almost identical to the native cartilage at T2-mapping evaluation. This behavior accompanied by a favorable macroscopic appearance could suggest the formation of hyaline-like cartilage. Only 1 patient was lost to follow-up after 6 months PO evaluation. The patient was having a favorable clinical and imaging progress with neither adverse events nor significant differences with the other patients evaluated. Our construct viability was more than 90% before implantation. Viability of remnants from a small sample of our patients was 87% at 24 hours (n = 6) and 75% at 48 hours (n = 1) after leaving the lab for implantation surgery, meaning the construct could be transported and implanted in centers at a 24- to 48-hour trip range without having major cell loss and with no contamination. A more detailed positive evaluation of our construct viability before and after arthroscopic implantation was previously described in a preclinical study using cells transduced with adenoviral vector with green fluorescent protein (AdGFP). 10 In our institution, we have conducted a pilot study, 3 a randomized controlled trial, 6 and this pilot multicenter study using AMECI, reporting positive clinical, imaging and second-look arthroscopic outcomes in more than 60 patients. Comparing the results obtained in this pilot multicenter study with our previous studies,3,6 Lysholm, Tegner, and IKDC(S) scores at 36 months PO were similar to the ones observed here. T2-mapping scores in all studies showed decreasing values of the repair cartilage ROIs and constant native cartilage values. MOCART scores showed an increase in overall grades ending with a mean grade at follow-up greater than 70 points in all studies.
Micheli et al. 36 published the first multicenter study using ACI in 2001. They evaluated 50 patients from 19 different centers who had positive outcomes after the surgery, demonstrating the safety and efficacy of ACI. Chondrocytes were isolated and cultured in the same facility, the implantation technique was standardized, and strict biosecurity measures (previously described by Mayhew et al. 37 ) were followed to ensure reproducibility. Marcacci et al. 16 published the first multicenter study using matrix-induced autologous chondrocyte implantation in 2005, showing positive clinical outcomes in 141 patients from 11 different centers. To date, there are only 7 published multicenter studies for MACT,5,8,16-20 in which clinical outcomes, MRI MOCART, and second-look evaluation were assessed after matrix-based ACI techniques similar to the present study ( Table 4 ). However, none of these multicenter studies evaluating MACT outcomes has described their biosecurity parameters in detail.
Matrix-Associated Autologous Chondrocyte Transplantation: Multicenter Studies.

IKDC = International Knee Documentation Committee; KOOS = Knee injury and Osteoarthritis Outcome Score; ADL = activities of daily living; QoL = quality of life; SR = sports and recreation; VAS = visual analogue scale; WI = weighted improvement; MOCART = Magnetic Resonance Observation of Cartilage Repair Tissue.
Schuette et al. 2 in a recent systematic review of mid- to long-term clinical outcomes after MACT showed an increase in both Tegner and KOOS scores with a weighted improvement (averaging their tibiofemoral and patellofemoral results) of 2.6 points in Tegner, 34.5 in KOOS-SR, 31.2 in KOOS-QoL, 21.4 KOOS-P, 18.5 KOOS-S, and 18 in KOOS-ADL. We obtained similar clinical outcomes at medium-term follow-up, as shown in Table 4 . In 2018, Ogura et al. 38 came up with the minimal clinically important differences (MCID) after MACT. Ninety-two patients were evaluated with at least 2 years of follow-up after surgery, and concluded that patients had to have a minimum increase in specific values in order to present a clear symptomatic improvement, as follows: Improvement between 4.2 and 10.5 points in the Lysholm score, between 10.8 and 16.4 for IKDC, between 3.6 and 8.4 for KOOS-S, between 11 and 18.8 for KOOS-P, between 9.2 and 17.3 for KOOS-ADL, between 12.5 and 18.5 for KOOS-SR, and between 12.8 and 19.6 for KOOS-QoL. In our prospective pilot/multicentric study, patients showed a clinical improvement that surpassed the thresholds established by Ogura et al., as shown in Table 4 .
Niethammer et al. 39 in a prospective study evaluated patient progress with MRI T2-mapping at 36 months PO after matrix-based ACI. They observed that T2 relaxation time values of the repaired tissue decreased from 41.6 ms at 6 months PO, to 39.3 ms at 12 months PO, to 30.9 ms at 36 months PO. They did not find statistically significant differences between native cartilage and repair tissue at 36 months PO. Similar to them, we found a constant decrease in relaxation time values in the repair tissue, reaching close to native cartilage values at final follow-up ( Table 3 ).
Based on our results, the efficacy of our biosecurity measures seems to be reliable and reproducible, suggesting that other groups could reproduce the results with our technique by following our established Minimum Biosecurity Parameters (MBP), as well as to lay the basis for a future multicenter study in Mexico and eventually other Latin American countries.
As limitations of our study we consider that viability and cell density at time of implantation was not evaluated, we propose this as a mandatory biosecurity parameter for a future larger multicenter study. Our arthroscopic implantation technique requires the use of suture anchors placed in the subchondral bone, as well as drilling through bone to secure constructs in patellar lesions. This implicates damage to the subchondral bone with subsequent bleeding, with the potential of generating repair tissue with mixed cell types. Despite this, anchors help us recognize the lesion location during MRI evaluation, making sure the correct region is being analyzed. Another limitation is that histological analysis of repair tissue was not performed after the second-look arthroscopic evaluation because biopsy of repair tissue in asymptomatic patients was not allowed by the local ethics committee and the internal review board.
Strengths of this study are as follows: evaluations were made by independent clinical investigators with no commercial funding or conflict of interest, it was performed in a real-world scenario with regular independent orthopedic practices and no controlled scenario, and the patients were evaluated in a multimodal way with few studies having such a complete evaluation ( Table 4 ).
In conclusion, the study suggests that by following our established MBP for AMECI, favorable clinical, imaging, and second-look arthroscopic outcomes, may be obtained, laying the basis for a future large-scale multicenter study in Mexico and eventually other Latin American countries.
Footnotes
Acknowledgments and Funding
We thank grant support from the National Council of Science and Technology (CONACYT SALUD 2017-02-290541). We also thank Arturo Almazán, MD, and Jorge Aziz, MD, for being part of this study as certified surgeons for AMECI. Francisco Cruz, MD, who contributed on developing the AMECI technique for lesions in the patella, Reynaldo Arredondo, MD, Amara Gantier, MD, and Andrés Cabal, MD, that contributed in the clinical and radiological follow-up of patients during their fellowship. Finally, thanks to Jorge Zamora-Quezada, MD, for his scientific and ethical support to first author EV in this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was performed following the Declaration of Helsinki and was approved by the local ethics committee and the internal review board (approval number INRLGII: 67/17).
Informed Consent
Written informed consent was obtained from all patients.
Trial Registration
Not applicable.
