Abstract
Exosomes are extracellular vesicles (EVs) released from cells that are a part of many biological and pathological processes, especially in intercellular communication. These vesicles are involved cell signaling, influence tissue and immune response, and serve as biomarkers for diseases. Most interesting are the exosomes that are released from mesenchymal stem cells (MSCs) for inflammation in joint diseases. Preliminary studies have demonstrated the advantages of using EVs rather than MSCs for cell free therapy. Research on exosomes have shown promising results as biomarkers for tracking the pathogenesis and prognosis of inflammatory arthritis. Therapeutically, animal studies have demonstrated immunosuppression, reversing inflammation, increasing chondrocyte proliferation, and drug delivery properties. The field of exosomes continues to develop and more basic science and clinical studies with safety and efficacy studies are needed.
Introduction
Stem cells have been at the forefront of regenerative medicine to heal tissue damage. Mesenchymal stem (or stromal) cells (MSCs) have attracted wide research attention because of their abundance and ease of collection for use in tissue engineering. Other stem cell types used include induced pluripotent stem cells (iPSCs), and embryonic stem cells (ESCs). 1 MSCs are multipotent, nonhematopoietic adult stem cells that can be isolated from a variety of different tissues, including bone marrow, 2 placental, 3 adipose tissue, 4 and umbilical cord. 5 These cells have regenerative properties, can modulate the immune response, and have the ability to differentiate into other cell types. 6 Much of the current therapeutic applications of MSCs can be linked to their immunosuppressive and anti-inflammatory properties. 7 Stem cells induce a local effect on their microenvironment, involving the release of dozens of active biological factors such as growth factors, cytokines, extracellular vesicles (EVs), and exosomes.
Osteoarthritis is one of society’s most common and influential diseases, with the knee being one of the most affected joints. This review will focus on exosomes and their potential use for inflammation in knee arthritis.
Extracellular Vesicles: Anatomy and Physiology
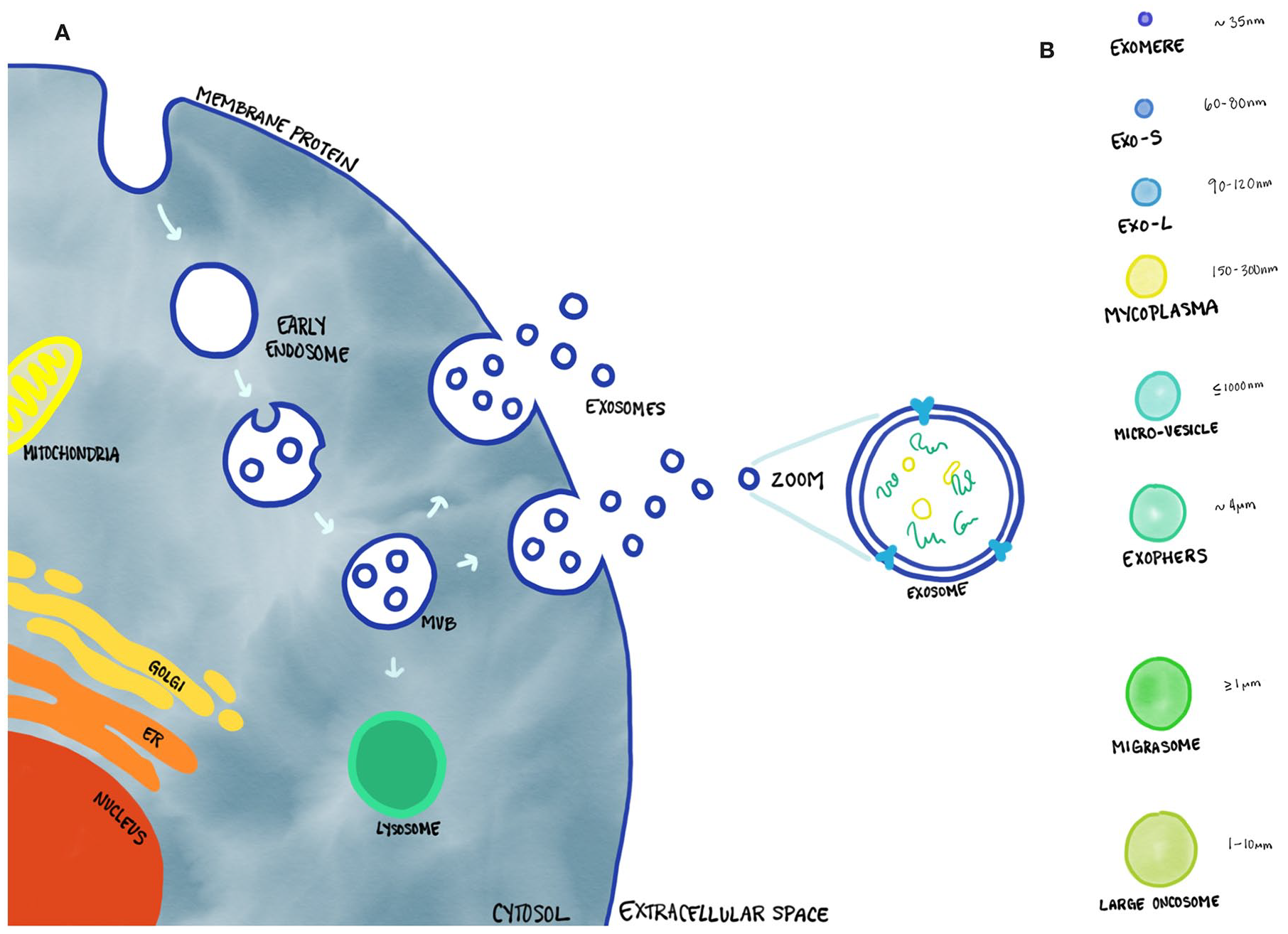
There are many different types of extracellular vesicles, including exosomes, microvesicles, membrane particles, ectosomes, exosome-like vesicles, and apoptotic bodies. 8 These vesicles not only differ by their size, but also their origins, and specific markers as shown in Table 1 . Exosomes have received a surge in both publications and attention in the past couple of years. 9 They are a subcategory of small, membrane-bound extracellular vesicles with sizes ranging from 40 to 100 nm as shown in Figure 1 . 10 These EVs were first described as “vesicles used by cells to eliminate cellular waste with no biological significance.” 11 They were in scientific obscurity until 1996, when Raposo et al. 12 discovered that these exosomes could stimulate adaptive immune responses. Since then, there has been an increase in research conducted on exosomes. Studies have now shown that these vesicles play a role in intercellular communication, immunomodulatory functions, and even have the potential for use in diagnosing and treating diseases.13-15
Characteristics of Extracellular Vesicles.
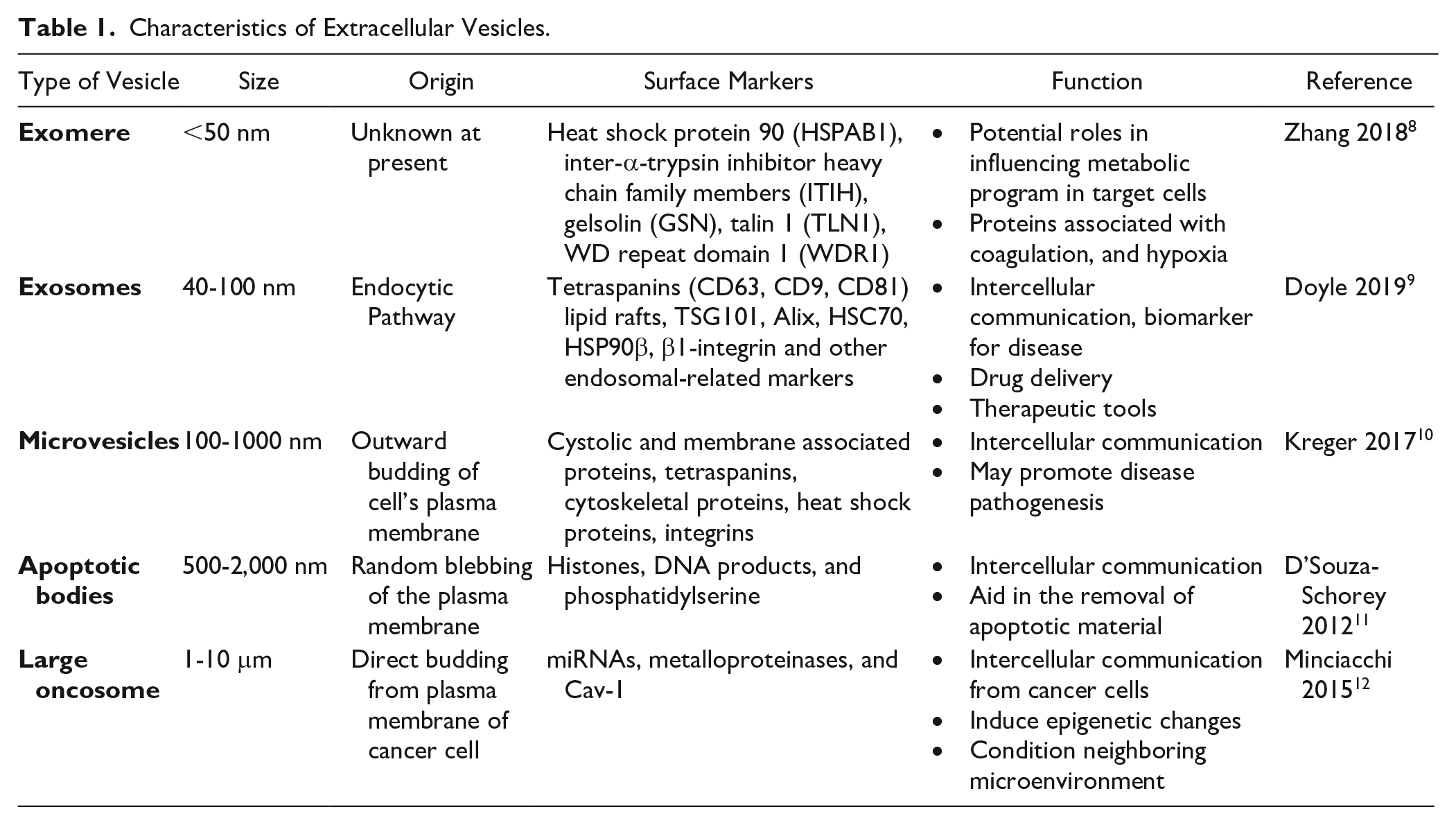
(
Exosomes and microvesicles are not exclusive to stem cells and are released by many cells throughout the body. Intercellular communication is a hallmark of multicellular organisms, and in addition to direct contact and secretion of soluble factors, cells also release these membrane bound vesicles. 16 In order to function normally, these extracellular vesicles, play a significant role in cell communication through a variety of ways. EVs can transmit information to acceptor cells by acting at the cell surface without delivery of their contents, such as during immune responses. 17 However, the main characteristics of EVs is the transference of secreted molecules. The transference of these extracellular vesicles provides local signaling through autocrine and paracrine fashion, and even affects distant cells through endocrine signaling. 18 The recipient cell’s plasma membrane fuses with the exosome and the contents are delivered into the cytosol inducing subsequent physiological changes. 19
Immune cells, cancer cells, stem cells all secrete different vesicles with vastly different cargos of information. 20 Exosomes are formed as intraluminal vesicles (ILVs) from the inward budding of the endosomal compartments called multivesicular body (MVB) as shown in Figure 1A . 21 This distinguishes exosomes from microvesicles that forms from the direct budding of the cell membrane. 22 This process allows the exosomes to encapsulate cytosolic cargo, including proteins, microRNAs (miRNA), messenger RNAs (mRNAs), lipids, and other metabolites. 23 The cargo contents of the exosomes are mediated by its microenvironment and the biological state of the parent cell. However, the differences in the biogenesis of exosomes during different physiological and pathological states are not fully elucidated. The transference of these metabolites acts in paracrine or even possibly endocrine manner affecting targeted cells. 18 The recipient cell’s plasma membrane fuses with the exosome and the contents are delivered into the cytosol inducing subsequent physiological changes. 19
In animals, exosomes can be generated and released from almost all cell types, but the yield greatly differs depending on cell type and culture condition. 24 Studies have suggested that one of the most prolific exosome producers come from mesenchymal stem cells, which release exosomes. 25 Yeo et al. 26 measured the amount of exosomes secreted by 5 different cell lines including a monocytic leukemia cell line, an embryonic kidney cell line, an epithelial cell line, a myoblast and myotube cell line, and human embryonic stem cell (hESC)-MSCs. They found that MSCs secreted the most exosomes, measured by the amount of CD81 found in the conditioned medium. 26
Secreted exosomes end up in biological fluids throughout the body, including milk, semen, urine, saliva, amniotic fluid, cerebrospinal fluid, synovial fluid, and blood plasma.27-29 EVs produced by both immune and nonimmune cells have an essential role in the regulation of immunity and inflammation.30-32
Exosome Isolation and Characterization
The physical features and chemical markers on EVs ( Table 1 ) form the basis of isolating and characterizing exosomes. The International Society for Extracellular Vesicles has provided minimal experimental requirements for definition of EVs and their functions. 32 Several methods have been developed for the isolation of exosomes from body fluids including ultracentrifugation, size exclusion techniques, filtration, immunological separation, precipitation methods, and immunoaffinity capture-based techniques. 33 There is currently no gold standard for exosome isolation with each method having its pros and cons.
For characterization, electron microscopy is commonly used to determine the size and physical characteristics of exosomes. Other approaches include nanoparticle tracking analysis (NTA) and dynamic light scattering (DLS) analysis. 34 However, due to their small diameter and complex and variable composition, these techniques have struggled to accurately count and phenotype EVs. More recent advances in technology have led to the use of using high-resolution flow cytometry to better characterize exosomes with high-throughput, single particle, analysis techniques. 35
Despite coming from different cells and various body fluids, all exosomes share a specific set of surface protein molecules, known as tetraspanins. These tetraspanins serve as exosome markers 36 and include CD81, CD63, CD93, Alix, and Tsg101. 37 Exosomes from different cells have their unique protein and RNAs that correspond to the cell and its physiological state. 38 Since exosomes released by different cells can be collected from body fluids, their cargo content represent potential disease markers and future therapeutic tools for diagnosis. A review by Wang et al. 39 showed that across 11 clinical studies, exosome biomarkers in breast cancer patients were significantly higher than in healthy controls. Understanding the exosome packaging process and content could provide valuable insights into mediating the pathological processes of disease and inflammation.
Inflammatory Response
Inflammation is a fundamental physiological response from the immune system to respond to injury and infection. It is a homeostatic process where the body is trying to heal and repair damaged tissue, as well as fight off any potential foreign invaders. At the tissue level, inflammation is characterized by redness, swelling, heat, pain, and eventual loss of tissue function. These symptoms are the result of the effect of local immune, vascular, and inflammatory cell responses to infection or injury. Inflammation can be caused for a myriad of reasons. With the immune response, the initial recruitment of innate immune cells results in the production of chemokines and cytokines, 2 protein groups of signaling molecules. They attract other immune cells to the site of infection/inflammation, resulting in inflammation, destruction of pathogens, clearing of dead cells, and tissue repair. 40
This process is necessary and beneficial when regulated in a controlled manner. If a stimulus persists, or inflammatory response is not resolved, then results can lead to cellular damage. 41 Constant inflammation, known as chronic inflammation, is a slow, long-term inflammation lasting for prolonged periods of several months to years. 42 Chronic inflammation is generally regarded as a loss of homeostasis between the host and immune cells and has been linked with the development of many diseases, including diabetes,43,44 cardiovascular disease, 45 cancer, 46 and several musculoskeletal conditions, including rheumatoid arthritis, and osteoarthritis.
Advantages of Exosome-Based Therapy
There have been many promising results in the field of stem cell–based therapies for the treatment of degenerative orthopedic conditions. The most popular stem cell treatments use allogeneic injectable MSCs taken from placenta-derived products, bone marrow aspirations, or adipose tissue. 47 These cells have a set of defined criteria by The International Society for Cellular Therapy. They must be plastic adherent, have the potential for differentiation in osteogenic, chondrogenic, and adipogenic lineage, contain cell surface expression of markers CD105, CD73, CD90 and the absence of hematopoietic markers CD45, CD34, CD14 or CD11b, CD79α or CD19 and HLA-DR. 48 However, the vast majority of clinically applied MSCs do not fulfill these criteria. Regulatory requirements in terms of quality control and assurance are lacking in the rapidly evolving field of biologics/regenerative medicine, and adequate quantifiable metrics is often lacking to reduce cost and bypass regulatory concerns of in vitro cell expansion. 49 This leads to issues of heterogeneous treatments and difficulty in measuring therapeutic outcome. Last, on a cellular level, there are concerns for the unwanted differentiation of injected MSCs, leading to possible malignant transformation or complications. The active migration of endogenous or exogenous MSCs to tumor sites have been documented in studies.50-52
MSCs have a relatively short life span in the body and at the site of injection. After an intravenous infusion, the majority of the cells have been found to migrate to the lungs and are rapidly cleared within 24 hours. 53 Barbash et al. 54 explained the high pulmonary entrapment of systemically infused stem cells are due to their large size. The size issue is a problem with intravascular administration, where injections of MSCs could lead to occlusions and blockages in the vasculature. 55 Intra-articularly, these human adipose-derived MSCs cells fair better and have shown to persist for 10 weeks inside the joint space in rats. 56 These injected MSCs show little engraftment and instead, are thought of to have therapeutic effects through the secretion of trophic factors, immunomodulatory cytokines and chemokines. 57 Additionally, there has been an issue with contamination and potentially harmful unproven treatments, documented in a brief report by Kuriyan et al. 58 Three patients went to a stem cell clinic and received intravitreal injections of autologous adipose tissue–derived stem cells. These patients subsequently suffered severe vision loss which resulted in total blindness. Ankrum et al. 59 discuss antibodies formed against donor MSCs, 60 as well as an immune rejection 61 leading to the conclusion that MSCs cannot be considered immune privileged.
Advantages of Exosome Usage
Replacing cell therapy with non-self-replicating exosomes eliminates many of the challenges faced with cell therapy such as immune activation for allogeneic transplantations, problems with size, and unwanted or malignant transformations. Preliminary safety data has reported that mice have shown no adverse immune reaction even after repeated exosome injections 62 or injections of cross species exosome. 63 Allogeneic MSCs may be perceived as foreign invaders by the innate and adaptive immune system; however, EVs are able to evade the immune system. Additionally, the small size of EVs opens up the therapeutic possibility of using exosomes as nanosized drug delivery vehicles without the toxicity associated with metal nanoparticles64,65 or the biological challenges/immune response elicited through liposomal delivery systems. 66 The unique nature of exosomes size allows them to travel throughout the body, without being trapped in the vasculature or cleared in the lungs. 67 Exosomes are able to home to target cells and injury sites, 68 directly passing on their contents across cell membranes, and the blood-brain barrier. 69 Exosome production is amenable to process optimization, including a future possibility of harvesting exosomes from immortalized MSC cell lines, ensuring reproducible and cost-effective production of exosomes. These harvested exosomes do not suffer from age-related issues like autologous stem cells 70 and their effectiveness does not decrease. Theoretically, exosome production can be fine-tuned in the laboratory, and the stem cell source for exosome production could be selected or genetically manipulated. Exosomes have also been shown to retain their efficacy and biological properties even after storage at −80°C for up to 5 months. 62 All things considered, MSC exosome therapy could potentially be a safer, cheaper, and a more effective treatment modality than cell-based MSC therapy in regenerative medicine.
Regulations
There has been no mention of exosomes anywhere on the U.S. Food and Drug Administration’s (FDA) website until December of 2019. The FDA released a public safety notification on exosome products in response to serious adverse events experienced by patients in Nebraska who were treated with unapproved products containing exosomes. In this announcement, the administration states that there are currently no FDA-approved exosome products and that exosomes used to treat diseases and conditions in humans are regulated like other drugs and biological products under the Public Health Service Act and Federal Food Drug and Cosmetic Act. 71 For exosomes to be used on humans, they are subject to FDA’s premarket review and approval requirements.
Clinical Therapeutics
Therapeutics
Exosomes have been shown to have immunostimulating effects, and research on exosomes and EVs has been used in treating inflammatory and immune disorders. EVs can have multiple properties depending on their cellular origin and target. Kim et al. 72 showed that interleukin-10 (IL-10) treated bone marrow–derived dendritic cells (DCs) can reverse the inflammation and suppress the arthritic disease. They also used antigen-presenting cells (APC)–derived exosomes to demonstrate immature DC were able to secrete exosomes and this was involved in the suppression of the inflammatory and autoimmune response. These exosomes had the same properties as the IL-10 treated DC and were able to modulate the APC and T cells in vivo, reducing inflammation and arthritic symptoms in mice. 73 Zhang et al. 74 showed that MSC-derived exosomes were able to mediate repair and regeneration in a rat model of temporomandibular joint osteoarthritis. This study showed that the MSC exosomes reduced transcription of inflammatory genes such as IL-1β, reduced presence of inflammatory IL-β+ and iNOS+ cells, and increased proliferative PCNA+ cells. This led to a reduction in inflammation and an increase in chondrocyte proliferation compared to the sham group.
There are many ways that exosomes can mediate the inflammatory response. It was discovered that the endogenous miR-155 and miR-146a were 2 key microRNA that regulated the inflammation process. 75 These 2 microRNAs were found by Alexander et al. 76 to be released in exosomes and modulate the recipient immune cells to respond to inflammatory cues in vitro and in vivo. Another way in which the body controls inflammation is through CD4+ T cells, 77 which are divided between helper T cells and regulatory T cells (Treg). Treg cells have been shown to prevent inflammatory disease as well as suppress inflammation. Okoye et al. 78 showed that not only do these Treg cells produce exosomes, but these exosomes had the therapeutic effect of suppressing conventional T cells, leading to the prevention of systemic/inflammatory diseases in the mice model. This phenomenon has also been studied by Cai et al. 79 who showed the exosomes secreted by bone marrow–derived DCs also showed potent anti-inflammatory properties and immunosuppressive properties by inducing Treg cells in a mouse inflammatory bowel disease model.
Exosomes can also be turned into drug delivery vehicles that could deliver a variety of therapeutic cargo to reduce inflammation. One such molecule is curcumin, a natural polyphenol that has anti-inflammatory, antioxidant, and chemopreventive activity. 80 Sun et al. 81 figured out a way to bypass curcumin’s poor bioavailability and incorporated curcumin in exosomes, then assessed the anti-inflammatory activity in vitro and in vivo. They showed how exosomes as a carrier could bypass the immune response in mice, showed statistically significant differences in cytokines and mortality in an lipopolysaccharide-induced septic shock model, and downregulated CD11b+gr-1+ lowering the inflammatory response. 81 This research was further studied by Zhuang et al. 82 who discovered that the method of encapsulating curcumin with exosomes could reach inflammatory microgila cells intranasally and directly deliver drugs to the central nervous system and across the blood brain barrier with the advantages of target specificity.
Rheumatoid Arthritis
Rheumatoid arthritis (RA) is a chronic autoimmune disease that causes inflammation in the joints, leading to pain, swelling, painful deformity, and immobility.83,84 Approximately 1% of the world’s population has RA, and it represents one of the most significant causes of musculoskeletal morbidity in the developed world. 85 There exist many different types of treatments to alleviate RA symptoms and/or improve disease state. 86 However, there is no known therapy to reverse RA progression.
Exosomes and microvesicles in the synovium fluid have been extensively studied in animals. The role of vesicles in RA has been identified and shown to be present in the synovial fluid of patients with RA.87,88 One study done by Sellam et al. 89 suggested that levels of circulating EVs were significantly elevated in RA disease state. These vesicles have been shown to have strong biological effects. EVs and MPs isolated from RA patients’ synovial fluid or plasma have shown proinflammatory activities and modulated the release of B-lymphocytes. 90 Additionally, these MPs have been shown to modulate fibroblast-like synoviocytes (FLS), a key effector cell in the pathogenesis of RA, 91 by increasing concentrations of IL-6, monocyte chemoattractant protein-1 (MCP-1), intercellular adhesion molecule 1 (ICAM-1), as well as vascular endothelial growth factor A (VEGF). 92 This shows that the MPs and EVs found in these diseased knees are proinflammatory, and could serve as biomarkers for inflammatory arthritis disease progression.
Studies have investigated the autoimmune aspects of RA and whether exosomes have a role in the signaling process. Skriner et al. 93 found that exosomes prepared from an arthritic patient’s synovial fluid all contained citrullinated proteins. These proteins have been implicated in causing other autoimmune diseases such as osteoarthritis, inflammatory bowel disease (IBD), and other conditions. 94 These citrullinated particles combine with immunoglobulins and antigens to form immune complexes (IC), which in turn stimulate neutrophils in joints to produce leukotrienes, further worsening the inflammation. 95 These observations point out that autoantigens on EVs are recognizable by the autoantibody and could be a factor in the cause of autoimmunity. 93
Osteoarthritis
Osteoarthritis (OA) is the most common joint disorder in the world, and represents one of the biggest causes of pain, loss of function, and disability in adults. In fact, OA is the second most common cause of work disability in the United States, and accounts for many more cases than RA. 96 These 2 diseases have a different prognosis as OA is more related to biologic processes involving cartilage degradation, while RA is through constant inflammation by the autoimmune system.
Murata et al.’s 97 research showed that OA patient’s tissue had similar expression of miRNA compared with synovial fluid, while being dissimilar to plasma miRNAs, suggesting a possible exosome–mediated miRNA transfer between the two. This idea was followed up by Kolhe et al. 98 who performed the first and only characterization of exosome miRNA from human synovial fluid in order to differentiate gender specifics in the prognosis of OA. This group found that there was a unique OA specific miRNA (miRNA-504) present in both genders.
Exosomes have also been shown to mediate new secretion and communication factors in the joint. Kato et al. 99 used exosomes isolated from IL-1β stimulated human synovial fibroblasts to induce osteoarthritic changes in chondrocytes. This research showed that exosomes were an additional signaling mechanism outside of cytokines and growth factors that could also lead to pathogenesis of disease. Further research in the field of exosomes and EVs will focus on finding biomarkers of pre- and early onset OA that will help diagnose or reverse the disease outcome.
Conclusion
The therapeutic potential of exosomes cannot be understated. There has been an increase in research focused on the physiologic and pathologic function of these vesicles. Additionally, there are numerous companies emerging as specialists in exosome technologies that are on track to bring this technology to the market.
Exosomes contain a variety of compounds like protein, miRNA, growth factors that could be used either to diagnose disease or combat it. In the field of orthopedics, exosomes have the properties to attenuate inflammation, either alone, or as a vehicle to transport other therapeutic agents. Exosomes have been shown to be able to induce inflammatory disease progression, potentially leading to better research on the mechanisms and diagnosis. Last, the cell-free nature of exosomes, make it an attractive therapeutic that has all the beneficial aspects of MSCs without inducing any potential malignant growths or immune responses.
The field of research is nascent, and most of the research data on exosomes comes from in vitro experiments and in vivo animal studies. There has been very little to no human trials done with exosomes to date. To move this technology forward, clinical trials need to be established and human safety data obtained. Exosomes represent the future of regenerative medicine and have the potential to change and help many lives.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Helen and Will Webster Foundation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
