Abstract
Objective
To evaluate the in vivo effect of a single intra-articular injection of local anesthetic (LA) lidocaine on the viability of articular cartilage in the intact or osteoarthritic (OA) human knees, and to measure the synovial postinjection concentration of lidocaine in the knee
Design
This study includes 3 interconnected experiments: (A) Synovial LA concentration measurement after a 2% lidocaine injection before knee arthroscopy in 10 patients by liquid chromatography–tandem mass spectrometry (LC-MS/MS). (B) Human osteochondral explants (N = 27) from intact knees procured at autopsies were incubated for different time intervals (30 minutes, 2 hours, 24 hours) with 2% lidocaine, 0.04% lidocaine (measured), or culture medium (control), and later evaluated for cell viability by LIVE/DEAD staining. (C) Ten out of 19 matched patients scheduled for knee replacement received a single intra-articular injection of 2% lidocaine approximately 30 minutes prior to the procedure; 9 patients served as control. Osteochondral samples with OA changes were harvested during surgery and analyzed for chondrocyte viability by LIVE/DEAD staining.
Results
(A) The synovial LA concentration was significantly lower than the primary concentration injected: average 0.23 mg/mL (0.02%), highest measured 0.37 mg/mL (0.04%). (B) In vitro exposure to a reduced LA concentration had no significant influence on chondrocyte viability in intact cartilage explants (24-hour averages: control, 93%; 0.04% lidocaine, 92%; 2% lidocaine, 79%). (C) Viability of chondrocytes in OA knees was similar between 2% lidocaine injection (85%) and control (80%).
Conclusions
A single intra-articular knee injection of 2% lidocaine did not influence the chondrocyte viability neither in healthy nor in OA cartilage. A fast postinjection reduction of synovial LA concentration (more than 40 times) is the most likely protective mechanism.
Keywords
Introduction
Intra-articular application of local anesthetics (LA), either alone or in combination with other medications, has been considered a routine and safe clinical practice.1,2 LAs have been predominately used before minor joint surgeries for anesthesia, after surgeries for postoperative analgesia, and in combination with steroid injections for the treatment of joint synovitis or pain.3,4 To follow the requirements of fast track rehabilitation protocols after arthroscopic shoulder and knee procedures, continuous LA joint infusions via intra-articular catheters had been introduced. 5 However, a significant number of patients with an unusual acute chondral damage and degradation, so called joint chondrolysis, were reported.6-8 Based on those reports, subsequent in vitro studies were performed, which demonstrated a harmful effect of LA toward the articular cartilage.9,10 Even short-term exposure to a single dose of LA caused chondrocyte dysfunction and death. 11 Older groups of LA, such as lidocaine or bupivacaine, demonstrated higher cartilage toxicity than newest formulations, mepivacaine or ropivacaine. 12
Based on data above, current clinical recommendations advocate against any LA intra-articular application. 13 Conversely, there have been no known reports of acute chondrolysis after knee arthroscopies in local anesthesia, or after single intra-articular LA injections for pain management. Moreover, in our previous study we showed that single intra-articular 2% lidocaine knee infiltration for arthroscopic cartilage biopsy had no influence on bioptic chondrocyte cell viability, morphology, and their potential for growth and population doublings in cell cultures for autologous chondrocyte implantation. 14 We have therefore hypothesized that certain factors in the natural environment of the joint protect cartilage from single-dose LA toxicity. We aimed to perform a combined clinical-experimental study to evaluate the in vivo effect of a single intra-articular injection of LA lidocaine on the viability of articular cartilage in the intact and osteoarthritic (OA) human knees, and to measure the synovial postinjection concentration of lidocaine in the knee.
Methods
The study includes 3 interconnected experiments conducted either in vivo (first and third experiments) or ex vivo on human cartilage explants (second experiment). The study protocol was approved by the Republic of Slovenia National Medical Ethics Committee (Permit No. 123/02/13).
Measurement of Synovial LA Concentration After a Single Lidocaine Injection
Ten patients (8 males, 2 females; average age 42 years [range 19-55 years]) who were scheduled for a minor arthroscopic procedure in the knee under local anesthesia were enrolled in the first experiment. Their primary indications for arthroscopy were meniscal injury (5 patients), partial ligament injury (3 patients), or cartilage lesions (2 patients). None of the participants had any local or systemic illnesses or therapies that could interfere with LA turnover in the knee or in the body. Knee joints were evaluated according to International Knee Documentation Committee (IKDC) examination protocol, with a special care toward the effusion intensity. Knee arthroscopy was performed in all 10 patients with use of local infiltration anesthesia. Ten to 15 minutes prior to the procedure each area of standard arthroscopic portal was infiltrated from the skin to the joint capsule with 7 mL 2% lidocaine hydrochloride with epinephrine in concentration 1:200000 (Xylanest, Gebro Pharma, Fieberbrunn, Austria). Then the joint cavity was infiltrated with 15 mL of 2% lidocaine hydrochloride (Institutional Pharmacy of the University Medical Centre Ljubljana, Slovenia) via superolateral access. The patient was asked to move the knee from full extension to 90° of flexion 10 times to spread LA throughout the joint and to mix it with synovial fluid. All arthroscopies were performed by the same surgeon using standard inferomedial and inferolateral portals. Immediately, after the introduction of arthroscopic trocar, a sample of joint fluid was aspirated and stored at −18°C for further analysis. In a separate container, the remnants of undiluted 2% lidocaine from the same vial were stored. The exact time from the intra-articular injection until aspiration was recorded. The concentration of lidocaine in collected samples was determined by liquid chromatography–tandem mass spectrometry (LC-MS/MS). Tandem system consisted of 1100 HPLC system (Agilent, Waldbronn, Germany) and Micromass Quattro Micro API (Waters, Milford, MA, USA). Samples were adequately diluted and spiked with internal standard levobupivacaine before deproteinization with acetonitrile. The compounds were separated on Zorbax XDB-CN column (Agilent, Waldbronn, Germany, with isocratic elution (water, acetonitrile, methanoic acid). Selected reaction monitoring (SRM) was used for quantification of lidocaine in the range of linear calibration curve from 1.0 to 20.0 mg/L.
Ex Vivo Chondrocyte Viability in Synovial and Standard LA Concentrations
Cartilage samples from 10 healthy knees were procured during forensic autopsies at the Institute of Forensic Medicine, Faculty of Medicine, University of Ljubljana, Slovenia. The donors had no macroscopic signs of knee disease, previous trauma, or surgery. Osteochondral cylinders (diameter 6 mm, depth 10 mm) were harvested from femoral condyles with a coring instrument for mosaicoplasty (Helipro, Jesenice, Slovenia). The cylinders were immediately transferred to a cell culture medium Dulbecco’s modified Eagle medium (DMEM/F-12) with gentamicin (50 mg/mL) and amphotericin B (2 mg/mL) (Thermo Fisher Scientific, Waltham, MA, USA) and stored at 4°C. Twenty-seven cylinders were randomly assigned into 1 of 3 groups: 2% lidocaine, 0.04% lidocaine (i.e., maximal synovial lidocaine concentration measured during the first subexperiment), and the control group. The 0.04% concentration was prepared with 1:50 dilution of original 2% preparation (Xylocaine, Astra Zeneca, Cambridge, UK) with normal saline (0.9% NaCl). The size of the groups was predicted with use of power analysis. We tested whether lidocaine alters viability of chondrocytes by at least 15% to 20%. To detect a 15% difference (α = 0.05, β = 0.2, power = 0.80), 9 samples in each study group were required. The first 2 groups of cylinders were removed from culture medium, and completely immersed in the corresponding lidocaine solution for 30 minutes, 2 hours, and 24 hours at 20°C. The cylinders in control group remained in the culture medium for the same time intervals at 20°C.
Three cylinders were analyzed at each concentration-time point. In the preparation for the viability analysis, the osteochondral cylinders were first freed from the bone, and the remaining cartilage disks were sliced perpendicularly through the center to obtain 3 to 5 native cartilage resins with a thickness of 0.3 to 0.5 mm. Similar protocol of harvesting and resins preparation has been described elsewhere.15,16 The cartilage resins were later dyed for 30 minutes at room temperature with LIVE/DEAD Viability/Cytotoxicity Assay Kit (Molecular Probes Inc., Eugene, OR, USA), according to the manufactures recommendations. Dyed resins were scanned with a laser confocal microscope Leica TCS SP5 (Leica Microsystems GmbH, Wetzlar, Germany). The microscope uses multiphoton confocal technology for optical sectioning of tissue samples independently from sample thickness. For every sample, we determined the optical depth with highest cell concentration and captured an image of 1550 × 1550 μm (512 × 512 pixels). The quantitative cell count was performed with a computer software ImageJ, Version 1.50i (Wayne Rasband, National Institutes of Health, Bethesda, MD, USA). The ratio between viable and total cell count was calculated and compared between concentration-time points via 2-way analysis of variance (ANOVA). Tukey posttest was conducted to determine the statistical significance of differences (IBM SPSS Statistics ver. 22.0, Armonk, NY, USA).
The Influence of a Single LA Injection on the OA Cartilage In Vivo
Nineteen patients scheduled for total knee replacement due to end-stage OA of the knee were enrolled in this subexperiment. They were randomly assigned into 1 of 2 groups. (A) LA group (N = 10; mean age 70 years; 5 males and 5 females; 4 right and 6 left knees) received a single intra-articular injection of 10 mL 2% lidocaine (Xylocaine 2%, AstraZeneca) in the treated knee during the presurgical preparation period, just after spinal anesthesia became active. After knee exposure via arthrotomy (approximately 30 minutes after LA injection), cartilage samples were taken from the less damaged regions of femoral condyles. Osteochondral cylinders (diameter 6 mm, length 6-8 mm) were formed and stored in cell culture medium (DMEM/F-12) at 4°C until cell viability analysis in 24 to 72 hours. (B) Control group (N = 9, mean age 69 years; 5 males and 4 females; 3 right and 6 left knees) did not receive any intra-articular injection, osteochondral cylinders were directly incubated in DMEM/F-12 at 4°C until further analysis with confocal microscope. The preparation of cartilage samples, LIVE/DEAD staining, and viability analysis via laser confocal microscope were identical as described in the second subexperiment. Collected data were evaluated with 2-tailed T test for unpaired samples. The computer software (IBM SPSS Statistics ver. 22.0) was used for the analysis.
Results
Measurement of Synovial LA Concentration after a Single Lidocaine Injection
Samples from 8 out of 10 patients were used for further analysis; 2 samples were discarded due to small volume of aspirate not sufficient to perform LC-MS/MS analysis. The average lidocaine concentration in an original vial marked as 2% was 16.47 mg/mL (range 13.59-17.09 mg/mL; SD 1.18), that is, 1.65%. The average difference between the declared vial concentration 2% and measured concentration was therefore −0.35 mg/mL. The synovial LA concentration 10 to 15 minutes after application was distinctively reduced 0.23 mg/mL (range 0.06-0.37 mg/mL; SD 0.12), that is 0.02%. The highest measured synovial lidocaine concentration of 0.04% (0.37 mg/mL) was used in the second subexperiment of the study. Synovial lidocaine concentration was on average 73-fold lower (range 46-310 fold, SD 93.6) than the vial lidocaine concentration injected into the knee. There were no differences in measured synovial lidocaine concentration between the knees with no (IKDC normal) or minor joint effusion (IKDC nearly normal); 0.23 (SD 0.13) versus 0.21 mg/mL (SD 0.08). The details are given in Table 1 .
Patients and Their Synovial Fluid Samples Characteristics Used in the Measurement of Synovial Lidocaine Concentration.
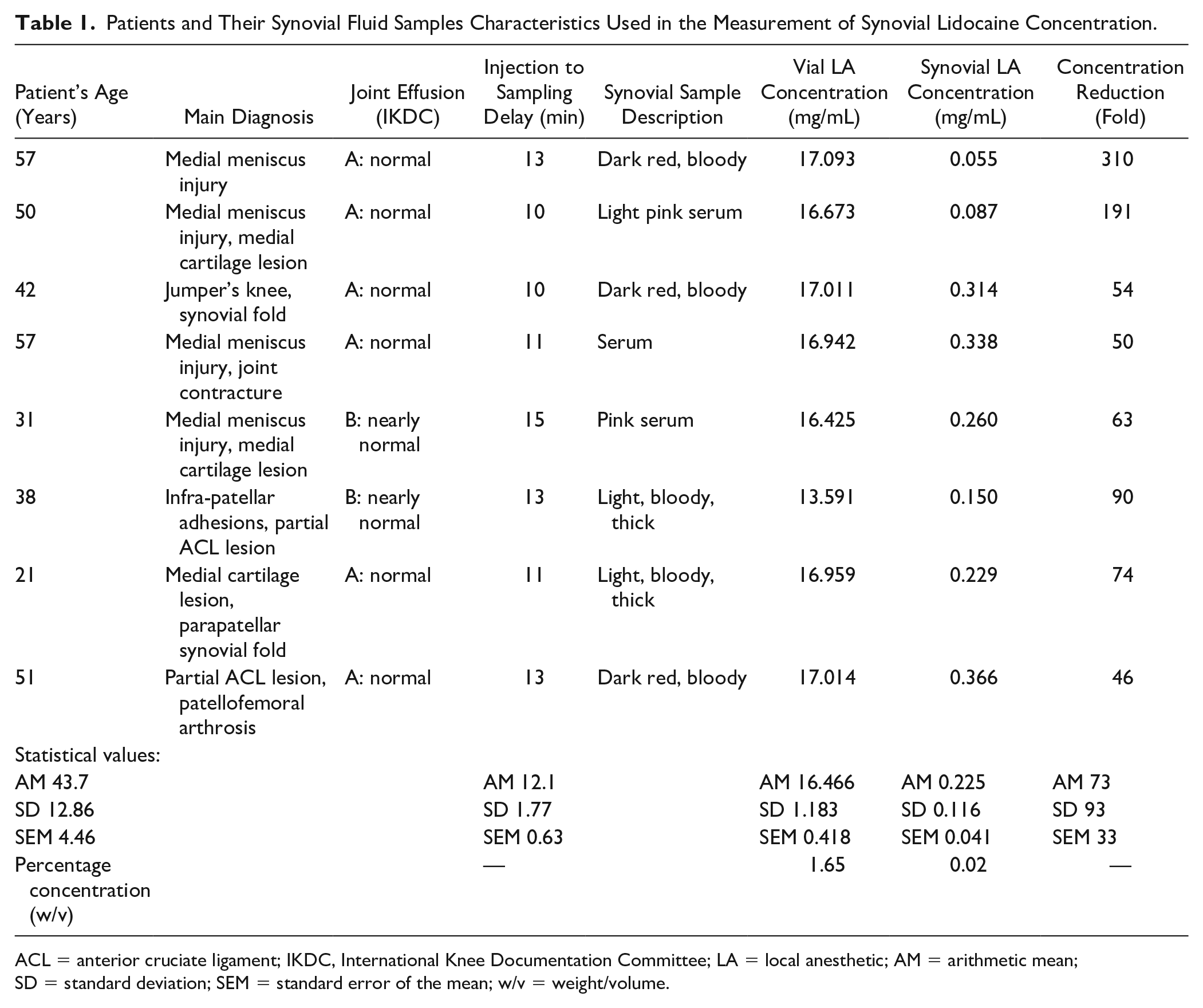
ACL = anterior cruciate ligament; IKDC, International Knee Documentation Committee; LA = local anesthetic; AM = arithmetic mean; SD = standard deviation; SEM = standard error of the mean; w/v = weight/volume.
Ex Vivo Chondrocyte Viability in Synovial and Standard LA Concentrations
Chondrocyte viability in control, synovial LA (0.04%), and vial LA (2%) ex vivo samples demonstrated progressive time- and concentration-dependent decrease ( Fig. 1 ). The chondrocyte viability values after of LA exposure were as follows: (a) 30 minutes—control 97% (SD 0.015), synovial LA 97% (SD 0.006), vial LA 94% (SD 0.035); (b) 2 hours—control 97% (SD 0.006), synovial LA 97% (SD 0.021), and vial LA 91% (SD 0.026); (c) 24 hours—control 98% (SD 0.006), synovial LA 92% (SD 0.038), and vial LA 79% (SD 0.047). Two-way ANOVA showed a statistically significant time (P = 0.012), concentration (P < 0.0001), and time-concentration (P < 0.0001) interactions. Tukey posttest revealed significant differences between vial 2% lidocaine toward synovial 0.04% lidocaine or control samples at 24 hours. Refer to Figure 1 for details.
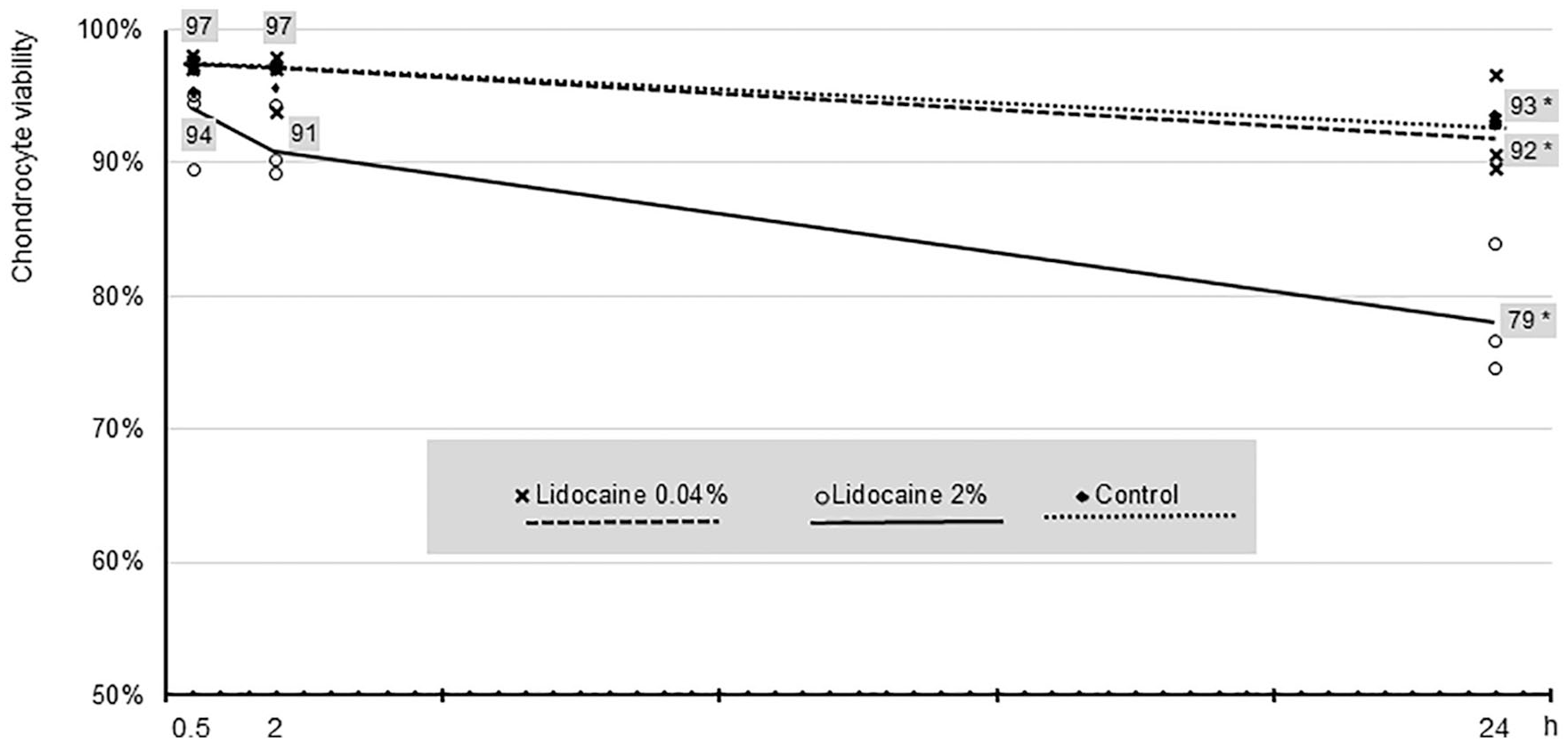
The influence of different lidocaine concentrations (vial 2%, synovial 0.04%, and control) on the chondrocyte viability in human cartilage explants at different time intervals. Two-way analysis of variance showed a statistically significant concentration-time interaction (P < 0.0001). Tukey posttest revealed significant differences between vial 2% lidocaine toward synovial 0.04% lidocaine and control samples at 24 hours*.
The Influence of a Single LA Injection on the OA Cartilage In Vivo
No statistical differences in chondrocyte viability of OA cartilage samples removed during total joint replacement in patients with or without preoperative intra-articular LA injection were detected: LA group 85% (SD 0.10) versus control group 80% (SD 0.20); P = 0.44. The details are given in Table 2 .
Chondrocyte Viability after an In Vivo Single Intra-Articular 2% Lidocaine Injection versus Control Group in the Osteoarthritic Cartilage Samples Harvested during Total Knee Replacement Surgery. a
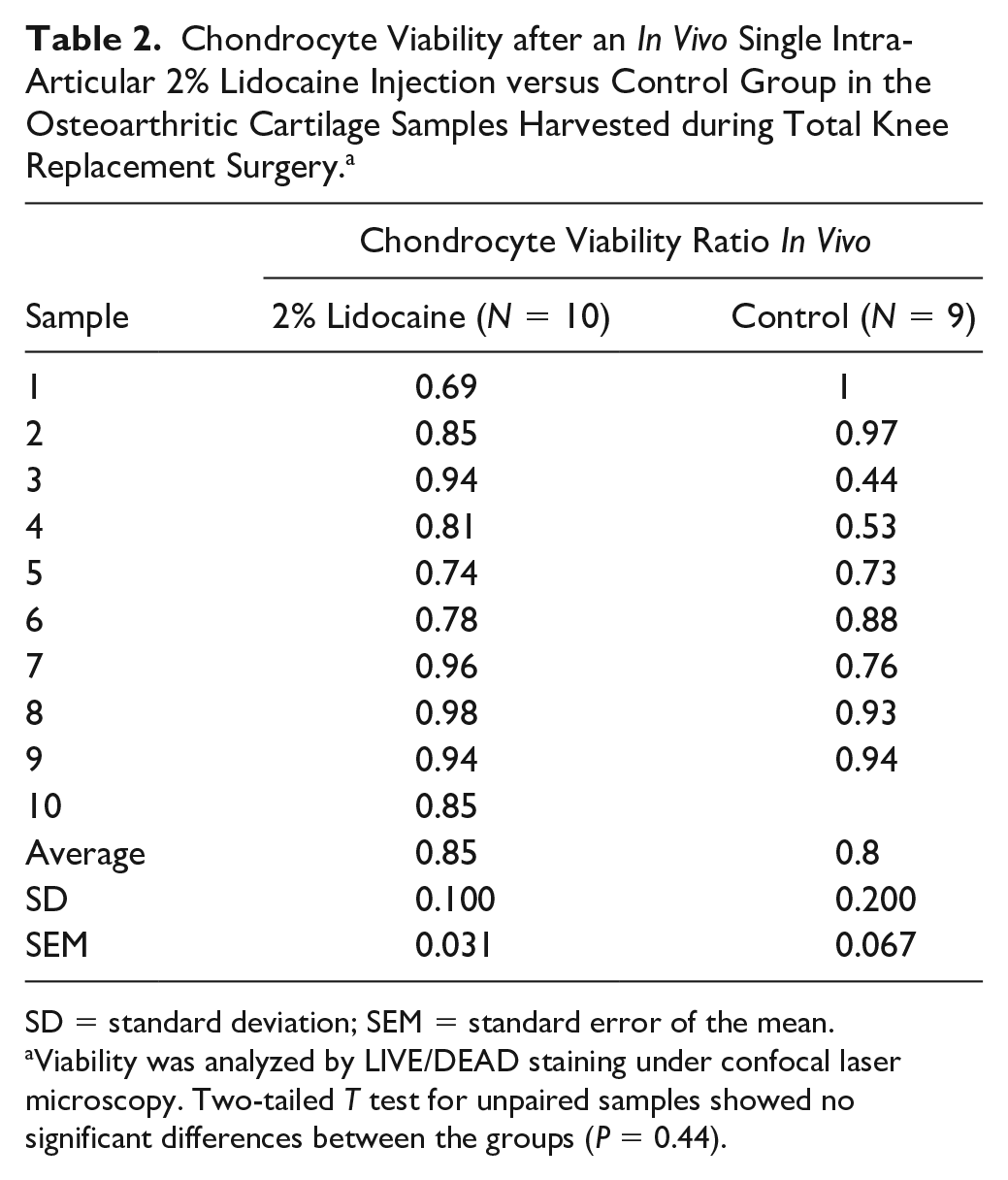
SD = standard deviation; SEM = standard error of the mean.
Viability was analyzed by LIVE/DEAD staining under confocal laser microscopy. Two-tailed T test for unpaired samples showed no significant differences between the groups (P = 0.44).
Discussion
The most important findings of this combined in vivo and laboratory study were the following: A single intra-articular knee injection of 2% lidocaine did not influence the chondrocyte viability, neither in healthy nor in OA cartilage, and fast postinjection reduction of synovial lidocaine concentration (more than 40-times). The results confirm the assumptions that in vivo intra-articular LA toxicity is negligible in contrast to the LA toxicity reported in previous in vitro studies.
Chu et al. 17 first reported a 99% mortality rate and apoptosis of chondrocytes after exposure of chondrocyte cell cultures to bupivacaine, versus 20% mortality of the normal saline control group. Furthermore, Karpie and Chu 18 demonstrated over 30% chondrocyte death after bovine cartilage exposure to 1% lidocaine in vitro for 1 hour. Chu et al. 9 again revealed 60% to 99% chondrocyte mortality in human and bovine cartilage cores submerged in 0.25% and 0.5% bupivacaine for 30 minutes. Dragoo et al. 19 applied 1% lidocaine, 0.25% bupivacaine, and 0.5% bupivacaine continuously for 24 hours on chondrocyte cultures and found high cell death rates of 7.9% for 1% lidocaine within 3 hours of exposure and no significant death rates for bupivacaine (0.25%) or ropivacaine (0.5%). A study by Breu et al. 12 on explants of healthy and OA human cartilage treated in vitro with different LA formulations, bupivacaine, mepivacaine, and ropivacaine, demonstrated a concentration- and time-dependent chondrotoxic effect. The effect of newer ropivacaine was significantly lower than the effect of older LA mepivacaine and bupivacaine. OA cartilage was more sensitive to the toxic effect than the healthy one. The toxic effect was initiated in the surface layers, where it caused disruptions in membrane integrity, followed by mitochondrial DNA changes, and alterations in cell nuclei. The same was also confirmed in a more recent study by Jacob et al. 20 They found structural cell defects in chondrocyte cultures of human cartilage. Again, newer ropivacain was less toxic than older bupivacaine and lidocaine. 20 A review from Jayaram et al. 21 studied 16 relevant articles on human cartilage trials from 1990 to 2018, all of them were performed in in vitro setting. Lidocaine alone or in combination with corticosteroids showed significant toxicity in standard concentrations of 1% and 2%. 21
None of the studies above was able to detect the toxic effects in low and very low LA concentrations. Chu et al. 9 clearly showed that toxic effect of reduced concentrations of bupivacaine (<0.125%) was similar to the effect of normal saline in the control group. We already mentioned the complex study by Breu et al., 12 low concentration of bupivacaine or ropivacaine exhibited no toxic effect. Recently, Busse et al. 22 tested different medications that are used in clinical practice as intra-articular injections. Among others, they tested lidocaine and bupivacaine on human chondrocytes and tenocytes. The effect was dose dependent, with the dilution of 1:2 and 1:10 showing strong toxic effect and the dilution of 1:100 only moderate toxic effect. 22
Similarly, weak or no toxicity was found in in vivo models. Gomoll et al. 23 found only minimal changes in cartilage tissue 3 months after the administration of 0.25% bupivacaine in rabbit shoulder in vivo. Additionally, they found increased synthesis and concentration of proteoglycans, which may indicate increased reparatory processes. Piat et al. 24 assessed the in vivo effect of 2% lidocaine and 0.5% bupivacaine in knee joints of horses. Reduced values of biochemical markers of degradation have been identified in the cartilage and increased values of the markers of the cartilage matrix synthesis, which indicates possible secondary anabolic effect. 24 Erden et al. 25 did not detect any inflammatory changes in the knee joint or subcutaneous tissue of rats after injection of 0.25% bupivacaine. A review from Kreuz et al. 13 was looking at the evidence on the toxicity of a single intra-articular LA application toward the articular cartilage. Based on 12 studies, they confirmed dose- and time-dependent LA chondrotoxicity. Higher toxicity was established for lidocaine and bupivacaine, but less toxicity were observed for mepivacaine and ropivacaine. A detailed insight into the included studies reveals remarkable discrepancy: 9 in vitro studies confirmed chondrotoxic effect, 2 in vivo animal studies revealed no LA toxicity, only reduced chondrocyte density in 1 study. The only human clinical-laboratory study showed no harmful effect of LA on cartilage biopsies used for autologous chondrocyte implantation. 14 Results from in vivo studies concur with a hypothesis that the natural knee environment exhibits protective mechanism to reduce or neutralize the chondrotoxic effect of LA.
Single intra-articular injections of LA can be used either as small volumes (up to 5 mL) in combination with corticosteroids for the management of chronic pain or inflammation, 26 or high volumes (15-30 mL) in the perspective of anesthesia for knee arthroscopy or postoperative pain management.27-29 In all instances, a decrease in intra-articular LA concentration immediately after its application is expected due to volume distribution, dilution in the synovial fluid, and fast diffusion of LA across the synovial membrane into the bloodstream. 30 Additional factor for LA removal from the knee joint is fluid flow through the joint during arthroscopy, and postoperative bleeding and effusion when LA is used in postoperative setting. 31
We have unequivocally confirmed the hypothesis of LA concentration drop with LC-MS/MS measurements in the first subexperiment. In the aspirates removed before the knee arthroscopy in local anesthesia, we found an approximately 50-fold decrease (maximum-recorded concentration 0.04%) in synovial LA concentration. This concurs also with the results of Breu et al., 12 who demonstrated that ex vivo effect of bupivacaine concentration of 0.125% or less was not toxic to the cartilage. Since we removed the aspirates 10 to 15 minutes post joint injection, we would expect certain effect of undiluted LA to the cartilage, but the real contact time is very short as the concentration apparently falls very fast to nontoxic levels. Chemical bonding to other ingredients of synovial fluid is also very likely. We do not have available data on LA binding to proteins in the synovial fluid. The analogy from blood, where 60% to 80% of LA is bound to plasma proteins 32 suggests that a large proportion of LA is similarly binding to synovial proteins. As the protein concentration in human joint fluid is approximately 50% of that in plasma 33 it is likely that a part of LA also binds to other tissues and their proteins in the joint, for example, articular capsule, menisci, ligaments, and synovial tissue.
Toxic effect of LA toward chondrocytes in vitro may also be attributable to the methodology used in those studies, since chondrocyte cultures or free-floating cartilage explants were used.9,10,12 Chondrocytes in these studies were therefore not protected with dense cartilage matrix. Moreover, in a healthy articular cartilage the injected substance interacts only with the very superficial layer, a dense lamina splendens, which prevents or slows down inflow of molecules into the tissue. Brecelj et al. 34 studied the influence of radioisotope 90yttrium on the human cartilage explants and demonstrated a chondrocyte viability increase by 1% for every 100 µm from the surface. Breu et al. 12 noted higher resistance toward LA toxicity when cartilage explants, instead of chondrocyte cultures were used for in vitro testing. Cell death in the explants was observed only in the superficial cartilage layer of 25 to 50 μm after 0.5% ropivacaine or 2% mepivacaine and up to 200 μm after 2% bupivacaine exposure. 12
Our second ex vivo subexperiment was conducted on osteochondral cylinders, and the very central part of cartilage portion was used for viability analysis. This would prevent the results bias of subchondral or lateral toxic effect, and the possible artifacts at the resection surfaces. A similar protocol of osteochondral harvesting, substance exposure, and sampling was used already in previous studies.15,16 Our subexperiment did not show increased toxic effect of LA in vivo toward the OA cartilage. This suggests that fast reduction of intra-articular LA concertation provides superior protection against LA toxicity than the integrity of cartilage superficial layers. Nevertheless, the viability results in OA cartilage were rather heterogeneous (from 44% to 100%) which is attributable to variable intra-articular conditions in OA knees. The number of patients/samples in the first part was relatively small and additionally we had to remove 2 measurements due to outlying results.
In the second subexperiment, we had to pool the cartilage samples without connection to individual donors to compensate different time intervals from sample procurement to experiment execution and analysis. This could lead to heterogeneity of cartilage condition among samples. Regardless of that risk, we obtained statistically significant results. With complicated logistics and transportation means, we could induce artifacts in observed samples. We took extra care to keep the samples at the same temperature, protected from light, and without mechanical stresses at all times. The experiment and analysis protocol were uniform for all samples; that may lead to a similar extent systemic error for all samples. All our experiments are based on the LIVE/DEAD cell viability analysis, which is an established methodology in cartilage studies.10,12,15 LIVE/DEAD viability assays offer adequate detection of dead cells (intrusion of ethidium homodimer-1 into the cell cytoplasm and binding to nucleic acids of disrupted cell nucleus), but apoptotic cells may be missed. Again, the results of viability analysis were reported as relative values, therefore apoptotic cells would be missed in all study groups to the same extent.
Conclusions
Single intra-articular knee injection of 2% lidocaine did not influence the chondrocyte viability, neither in healthy nor in OA cartilage. A fast postinjection reduction of synovial LA concentration (>40 times) can be proposed as a protective mechanism against the toxicity of a single intra-articular injection of lidocaine.
Footnotes
Authors’ Note
The work was conducted at four institutions interchangeably: Department of Orthopedic Surgery, University Medical Centre Ljubljana, Slovenia; Valdotra Orthopedic Hospital, Ankaran, Slovenia; Institute of Forensic Medicine and Institute of Pathophysiology, Faculty of Medicine, University of Ljubljana, Slovenia; and Institute of Physiology, Medical Faculty, University of Maribor, Slovenia.
Author Contributions
KR, TM, SP, AA, GK, AS, and MD conceived and designed the research, interpreted the experimental results, and approved the final version of the manuscript. KR, TM, SP, GK, and MD performed the experiments, analyzed the data, and drafted the manuscript. KR, SP, and MD prepared the figures. KR, TM, SP, AA, GK, AS, and MD edited and revised the manuscript.
Acknowledgments and Funding
The authors gratefully thank Rudi Mlakar from the laboratory for confocal microscopy, Institute of Physiology, Medical Faculty, University of Maribor, Slovenia, for his help on the scanning and quantification of the cartilage samples. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: TM and SP are supported by grants from the Slovenian Research Agency P3-0043, J7-8276, and J3-9263. MD is supported by grants from the Slovenian Research Agency P3-0043 and by the University Medical Centre Ljubljana (Institutional research funding grant number 20170143). MD, TM, and SP are supported by grant from the Slovenian Research Agency P3-0043; TM and SP also by the Grants J7-8276, and J3-9263 by the same institution.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study protocol was approved by the Republic of Slovenia National Medical Ethics Committee (Permit No. 123/02/13).
Informed Consent
Written informed consent was obtained from all subjects before the study.
Trial Registration
Not applicable.
