Abstract
Objective
Fibrin has been used as a standard material for scaffold fixation during cartilage repair surgery. Most of the commercially available fibrin preparations need an additional method for scaffold fixation, most often with sutures, thus damaging the surrounding healthy cartilage. There is therefore a need to find alternatives to this method. In our study, we have investigated the potential possibility to use mussel adhesive protein as such an alternative.
Methods
In this study, hydrophobic plastic was coated with the mussel adhesive protein Mefp-1 as well as with other cell adhesives (poly-lysine, fibronectin, and collagen). Human keratinocytes and chondrocytes were seeded on these substrates at 37°C in culture medium, followed by analysis of attachment and proliferation by crystal violet staining and metabolic labelling. Performance of Mefp-1 and fibrin as tissue glues were estimated by tensional force resistance measurement of moist porcine dermis (as a correlate to scaffold) glued to dermis, cartilage, or bone at 37°C.
Results
Mefp-1 supported maximal cell attachment at a coating density of approximately 1 µg/cm2. This was at least as good as the other adhesives tested. In addition, it supported cell proliferation at least as good as regular tissue culture plastic over a 7-day period. Measurement of tensional force resistance showed that Mefp-1 performed equally well as fibrin when porcine dermis was glued to cartilage and bone at the same concentration. Separation of the moist tissues after 15-minute incubation required a force of approximately 1 N/cm2 for both compounds.
Conclusions
Mefp-1 show properties that qualify it as a compound that potentially could replace fibrin as a tissue glue for scaffold fixation. Given the possibilities to modify this protein by bioengineering, it is likely that the properties can be further improved.
Introduction
Numerous cartilage repair surgery techniques have been developed in the past decade, some of them using scaffolds as a cell-carrier or cell-free scaffolds. There are several methods for scaffold fixation such as transosseus fixation, fixation with sutures, bioresorbable pin fixation, and biological glue fixation with fibrin. Of all these techniques, the only one with no damage to the surrounding tissue, whether it was bone or cartilage, is the fibrin fixation. This type of fixation is often being done in combination with other techniques, suture fixation mostly. Furthermore, fibrin is produced from human serum and this includes a potential risk for virus diseases transmission, despite the most modern techniques for virus detection. The use of fibrin in internal tissue is associated with the potential risk of thromboembolism, the fibrin glue may also absorb prematurely, before adequate tissue repair is achieved. 1 In addition, handling of fibrin is quite delicate (must be stored frozen, due to risk for premature curing), and strength is not sufficient in all situations. 2 Moreover, in an in vitro cartilage repair model, we found that chondrocytes could not migrate into a fibrin network, thus disabling it for use as a scaffold in cartilage repair. 3 Thus, given the scarce number of alternatives, there is plenty of room for new approaches in this area. In particular, novel methods are warranted for scaffold fixation during cartilage repair surgery that would give more efficient scaffold attachment and minimal damage to the surrounding tissue.
The blue mussel, Mytilus edulis, is known for its ability to adhere to hard surfaces in wave-swept seashores. 4 This rapid and permanent adhesion is mediated by a structure called byssus, which is composed by a bundle of filaments with a flattened, adhesive plaque tip in the end 5 ( Fig. 1 ). This remarkable property of the mussel enables adherence to nearly all types of inorganic and organic surfaces in aqueous environments. 6 The mussel foot forms the byssus whenever the mussel is under any type of stress within 2 to 5 minutes. 7 This mechanism is enabled by a number of glands called phenol glands in the foot, which secrete different proteins that are essential to create the byssus 3 ( Fig. 1 ). An important group of these proteins are polyphenols called mussel adhesive proteins (MAPs) or Mytilus edulis foot proteins (Mefps). Until date, 8 Mefps have been identified, and they are in turn part of 20 more or less well characterized proteins in the byssus. 8 The first Mefp that was discovered (Mefp-1) is the main component of the cuticle covering and protecting all parts of the byssus as well as the plaque. 9 In previous in vitro studies, Mefps have been used as a coating for attachment of cells. Successful attachment to Mefp-coated surfaces has been reported for various adherent cells, for example, neuronal cells, 10 T47D human breast cancer cells, 11 epiphyseal cartilage and osteosarcoma cells, 12 and nonadherent cells such as T-lymphocytes. 13 These studies also indicate that MAP is not toxic to cells. The aim of this study was to further investigate the properties of Mefp-1 in comparison to other adhesives that would be relevant to scaffold fixation in orthopedic surgery.
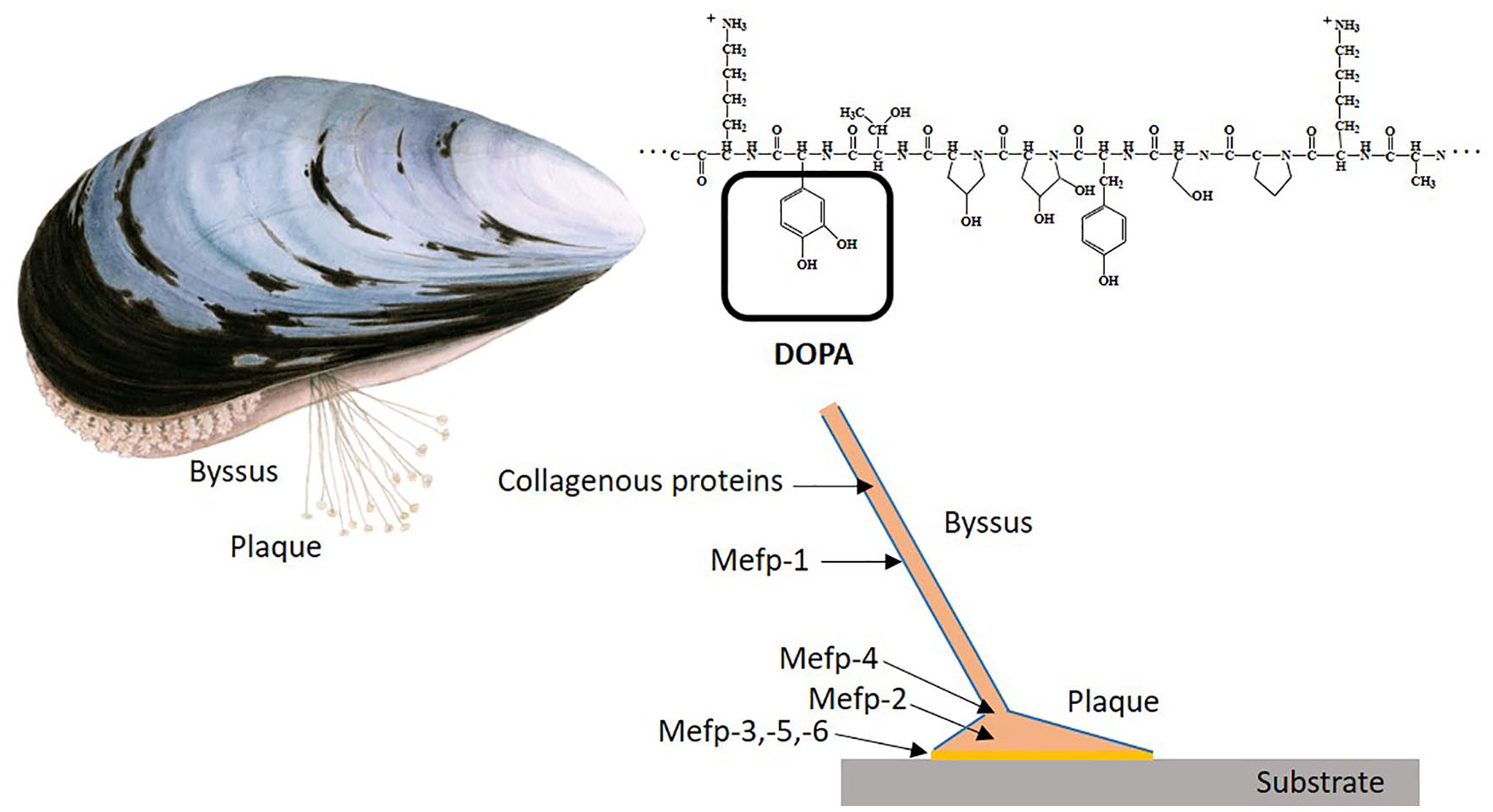
Adhesive mechanism of the blue mussel Mytilus edulis. The byssus is produced by the mussel foot and contains various adhesive proteins that adhere firmly to rock or other substrates in the environment. It is generally believed that the strong adhesion property of all the Mefps is dependent on the high content of polyphenols (DOPA) as well hydroxylation of prolins. Mefp-1 is covering the entire byssus including the plaque. Blue mussel illustration by Maj Persson. Byssus structure and Mefp-1 repeat adapted from Silverman et al. 4 and Haemers et al., 18 respectively.
Materials and Methods
Cell Culturing
The immortalized human keratinocyte cell line HaCaT (CLS Cell Lines Service GmbH, Eppelheim, Deutschland) and chondrocyte cell line CHON001 (CRL 2846, ATCC/LGC Standards AB, Boras, Sweden) were subcultured in tissue culture flasks (Sarstedt AB, Helsingborg, Sweden). Culture medium was DMEM/high glucose 4.0 mM (Fisher Scientific, Gothenburg, Sweden) supplemented with 10% FBS (fetal bovine serum) and 0.1% Gentamicin (both from GIBCO Life Technologies, Stockholm, Sweden). Cells were removed from the culture plastic surface by incubation in 0.25% (HaCaT) and 0.05% (CHON001) trypsin-EDTA (GIBCO Life Technologies) at 37°C for 5 minutes. To inactivate the trypsin, cells were suspended in medium containing serum and centrifuged for 5 minutes, 400 × g (HaCaT) or 200 × g (CHON-001). Cells were then resuspended in fresh medium and plated in tissue culture flasks or multiwell plates. All experiments were performed 3 times with triplicate sampling.
Coating of Hydrophobic Plastic and Plating of Cells
Coating of hydrophobic plastic (Sarstedt AB) with Mefp-1 (Biopolymer AB, Gothenburg, Sweden), poly-D-lysine, fibronectin, collagen, and bovine serum albumin (BSA) was performed as described below. A 1 mg/mL Mefp-1 stock solution in 1% citric acid was diluted in 0.1 M Na2CO3 (Sigma-Aldrich Sweden AB, Lund, Sweden) to the concentrations indicated (concentration curve), or 30 µg/mL (as standardized concentration). Forty microliters of this solution was added to each well of a 96-well plate (0.32 cm2), and plate incubated for 1 hour at 37°C, in a humidified incubator with 5% CO2. The poly-D-lysine (BD Biosciences, Stockholm, Sweden) was dissolved in sterile H2O to obtain a stock solution of 10 mg/mL. This was diluted to a final concentration of 40 µg/mL in PBS (phosphate-buffered saline; Fisher Scientific). Fibronectin (BD Biosciences) was dissolved in sterile H2O to a concentration of 500 µg/mL and further to a final concentration of 30 µg/mL in PBS. Bovine collagen I (SigmaAldrich) was diluted in PBS to a final concentration of 75 µg/mL. Ninety-six-well plates (50 µL/well) were coated with poly-D-lysine, fibronectin, and collagen for 1 hour at room temperature with the indicated concentrations according to the manufacturers’ instructions. After 1 hour, the wells were washed twice with 50 µL of PBS, the cells resuspended in culturing medium, and plated at a concentration of 5,000 cells/well in hydrophobic 96-well plates with and without precoating with the 4 adhesives as described. Cells were also seeded in regular cell culture plates (Sarstedt AB). The final volume was 50 µL/well. Cells were allowed to attach to the well surface at 37°C, 5% CO2 in a humidified incubator.
Analysis of Cell Attachment
Cultures were terminated by fixation with methanol after various culture times as indicated. Methanol was discarded after 20-minute fixation and the cells were stained with crystal violet solution (Sigma-Aldrich). After 20 minutes of staining, wells were washed in running water for 2 minutes, plates emptied, and replenished with 30 µL of water to each well. Cells were photographed under a light microscope. The water was discarded and 50 µL of 33% acetic acid (VWR International AB) was added to the wells. Dissolution was allowed to proceed for 15 minutes at room temperature. Fifty microliters from each well was transferred to 96-well plates (Sarstedt AB) and absorbance measured at 540 nm using a Multiskan Ascent Microplate Photometer (Thermo Scientific, Gothenburg, Sweden).
Analysis of Cell Growth
Cells were cultured at 37°C, 5% CO2, in a humidified incubator for various time periods as indicated. The medium was discarded and 100 µL of fresh medium and 20 µL of Celltiter-blue reagent (CellTiter-Blue Cell Viability Assay, Promega Biotech AB, Nacka, Sweden) was added to the wells. The plates were incubated in 37°C for 4 hours. The content of each well was transferred to clear 96-well plates (Sarstedt AB) and samples analyzed at 560Ex/590Em nm using a FLUOstar-OPTIMA Microplate Reader (BMG Labtech, Varmdo, Sweden). To allow interpretation of the relative values obtained, fold change was calculated to enable comparison of the measurements between the different sampling dates. The reference value of one was assigned to the well which contained medium only (blank).
Adhesives as Tissue Glue: Measurement of Tensional Strength
In order to investigate the performance of Mefp-1 (Biopolymer AB, Gothenburg, Sweden) and fibrin (Tisseel, Baxter Medical AB, Kista, Sweden) a force measurement instrument (Mark-10, Art No: ESM301E, equipped with Digital Force/Torque Indicator, model M5i, and Data Acquisiton Software MESURgauge, New York, NY) was used to estimate generation of tensional strength when these adhesives were used to glue dermis, bone, or cartilage. Thin pieces of polyethylene foam, porcine rib bone, or human cartilage were first glued on top of hard plastic cubes with cyanoacrylate. Moist porcine skin pieces (EZ Derm, Mölnycke Healthcare, Gothenburg, Sweden) were glued on top of the polyethylene foam to assure a good contact with the mating tissue ( Fig. 2A and B ). Contact surface was approximately 0.7 cm2. Fifteen microliters of adhesive at the indicated concentrations, prepared as described by manufacturers, was applied to the (moist) tissues, and immediately pressed together with the mating tissue under a weight of 800 g in an incubator at 37°C. This load was necessary for a tight contact between the polyethylene foam/dermis and the hard tissues ( Fig. 2C-E ). The cubes with glued tissue were hooked up to a Mark-10 instrument and tensile strength (Newton) measured. The velocity of pulling the tissues apart was adjusted to approximately 0.17 mm/s, with 20 readings of generated force/second ( Fig. 2F-H ).

Force measurement of tissue glue applied to various tissues. (
Statistics
Results are presented as mean values of replicates (n = 3) with the standard deviation indicated as error bars.
Results
Cell Attachment to Mefp-1-Coated Hydrophobic Plastic
Different concentrations of Mefp-1 were used to coat hydrophobic plastic, a material that poorly supports cell attachment itself. Keratinocytes or chondrocytes were seeded to the Mefp-1-coated plastic, fixed, and stained with crystal violet after 90 minutes. Resulting absorbance after dissolution in acetic acid were used to calculate relative attachment of cells. Improvement of cell attachment was seen already at a coating density of 2 µg/mL for both cell types. Attachment was maximal at approximately 8 µg/mL. This corresponds to a density of 1 µg/cm2. When comparing the 2 cell types, chondrocytes adhered better to Mefp-1, irrespective of concentration. This could, however, largely be explained by a better attachment to hydrophobic plastic only of this cell type ( Fig. 3 ). When inspecting cells under microscope, both cell types also showed a better spreading when seeded on Mefp-1, compared to hydrophopbic plastic alone ( Fig. 4 ).
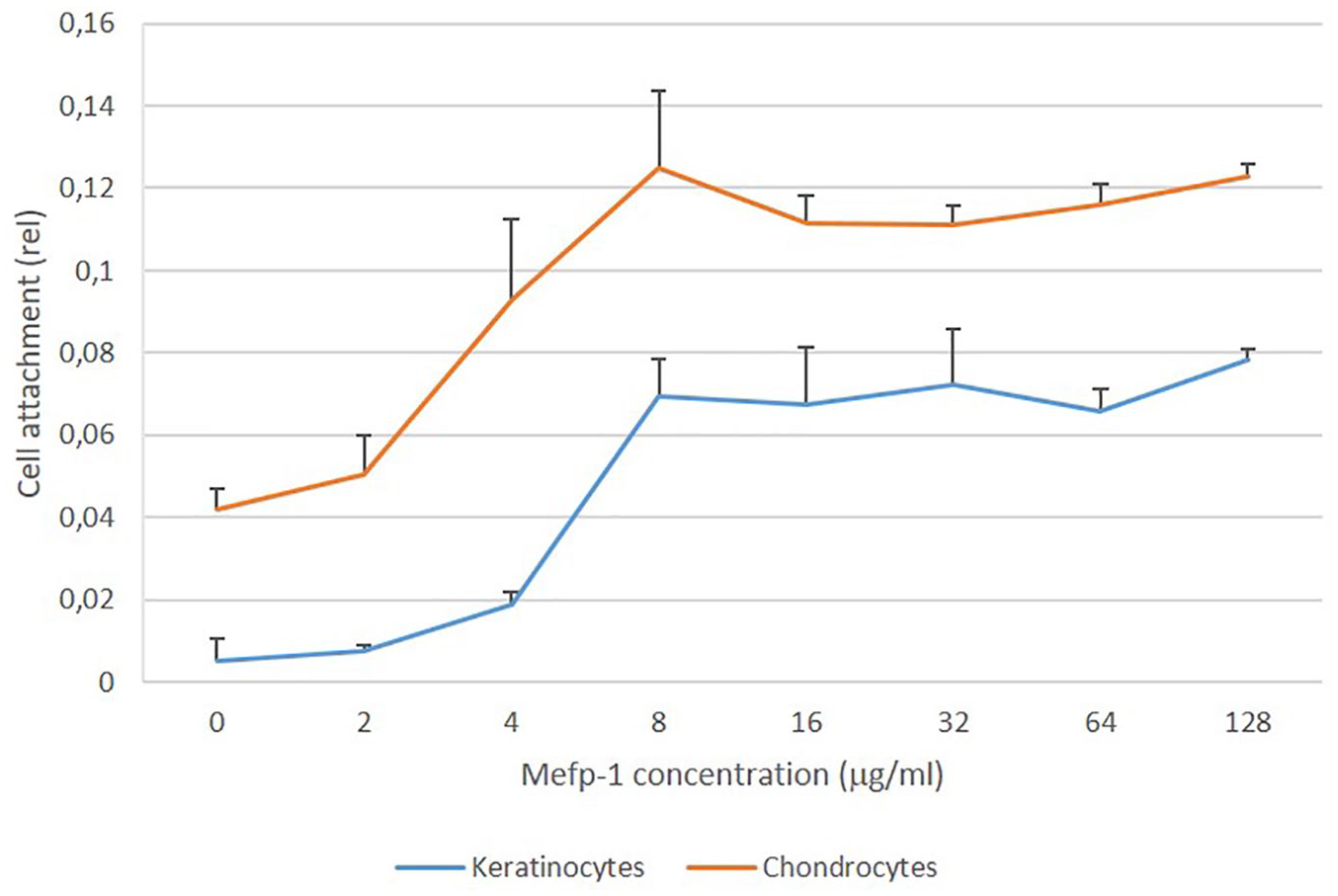
Cell attachment to hydrophobic plastic coated with different concentrations of Mytilus edulis foot protein-1 (Mefp-1; n = 3).
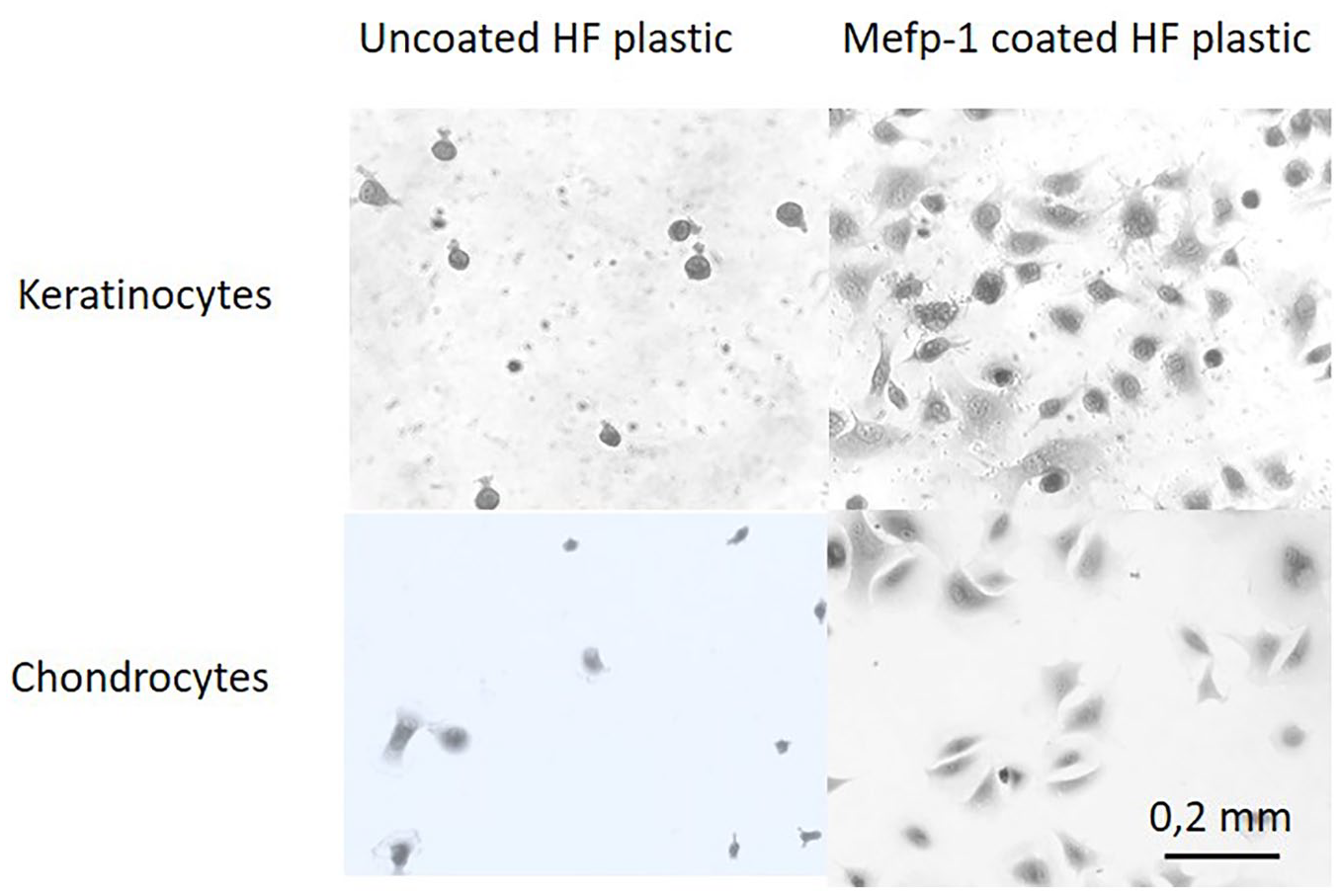
Microscopic images of keratinocytes and chondrocytes attached for 90 minutes to uncoated or Mytilus edulis foot protein-1 (Mefp-1) coated (30 µg/mL) hydrophobic (HP) plastic.
Cell Attachment to Other Adhesives
Poly-lysine, fibronectin, and collagen are well-characterized cell adhesives, and these were used to coat hydrophobic plastic as described in Materials and Methods. In order to compare Mefp-1 with these adhesives, keratinocytes and chondrocytes were seeded to these substrates in the same experiment. Coating with BSA was used as a nonadhesive protein as comparison. The cells were incubated with the various substrates for different time periods up to 120 minutes, and cell attachment analyzed with crystal violet staining, acetic acid dissolution, and absorbance measurement. Mefp-1 coating supported attachment of both cell types equally well as the other adhesives, or tissue culture plastic. Uncoated hydrophobic plastic supported poorly cell attachment, and BSA further inhibited cell attachment. This was true for all time periods analyzed ( Fig. 5 ).
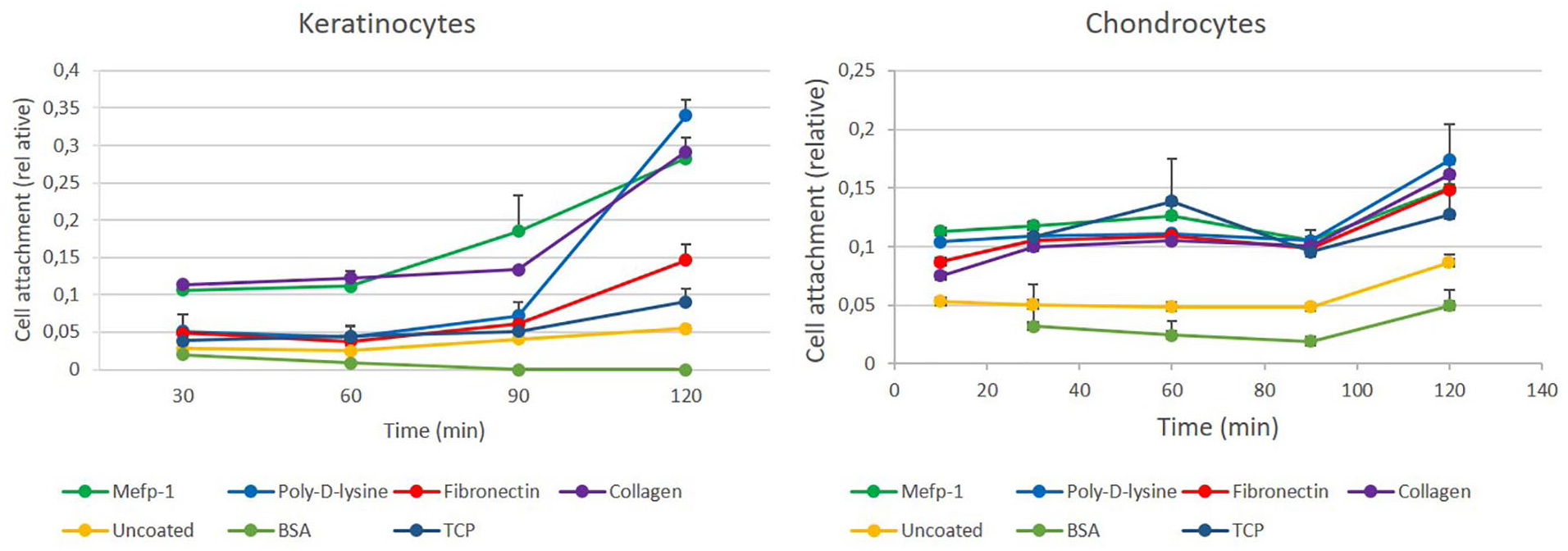
Keratinocyte and chondrocyte attachment to different cell adhesives. Coating of adhesives to hydrophobic plastic was performed as described in Materials and Methods. Mefp-1 = Mytilus edulis foot protein-1; BSA = bovine serum albumin; TCP = tissue culture plastic (n = 3).
Cell Growth on Mefp-1
In order to investigate how cells proliferate on Mefp-1, keratinocytes and chondrocytes were seeded on uncoated or Mefp-1-coated hydrophobic plastic, as well as on regular tissue culture plastic. Cells were cultured for up to 1 week and metabolic activity, reflecting relative number of living cells, were monitored at different time points. Mefp-1 supported cell growth at least as good as tissue culture plastic for both cell types over a 7-day period ( Fig. 6 ).
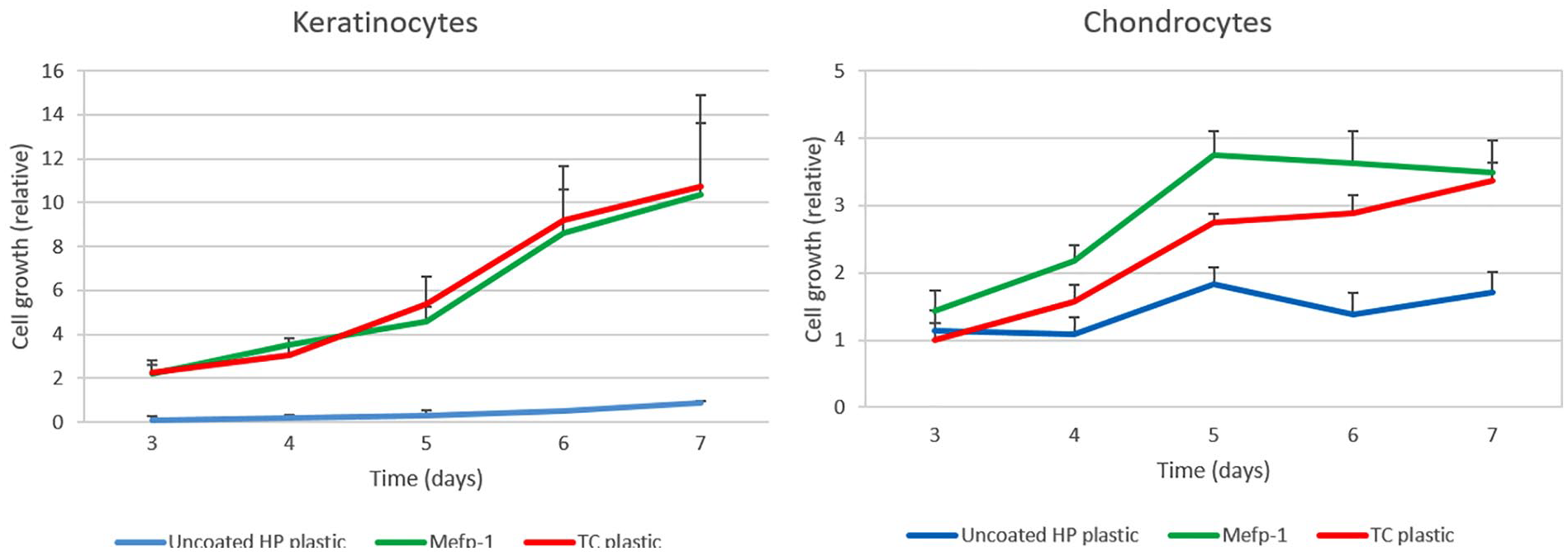
Growth of keratinocytes and chondrocytes on Mefp-1-coated hydrophobic plastic and tissue culture plastic over a 7-day period. HP = hydrophobic; Mefp-1 = Mytilus edulis foot protein-1; TC = tissue culture (n = 3).
Mefp-1 as Tissue Glue: Tensional Force Measurement
In order to estimate the capacity of Mefp-1 to act as a tissue glue, modified porcine skin pieces were glued together with other skin pieces, cartilage, or bone as described in Materials and Methods. Mefp-1 was able to glue skin to skin, cartilage, and bone with a tensional strength of approximately 1.5 N/cm2 at a concentration of 40 mg/mL ( Fig. 7A ). Pressing tissues together for 15 minutes generated higher tensional strength than pressing them together for 5 minutes ( Fig. 7B ). Longer pressing times resulted in partly dried tissue; thus, effects beyond 15 minutes could not be reliably estimated.
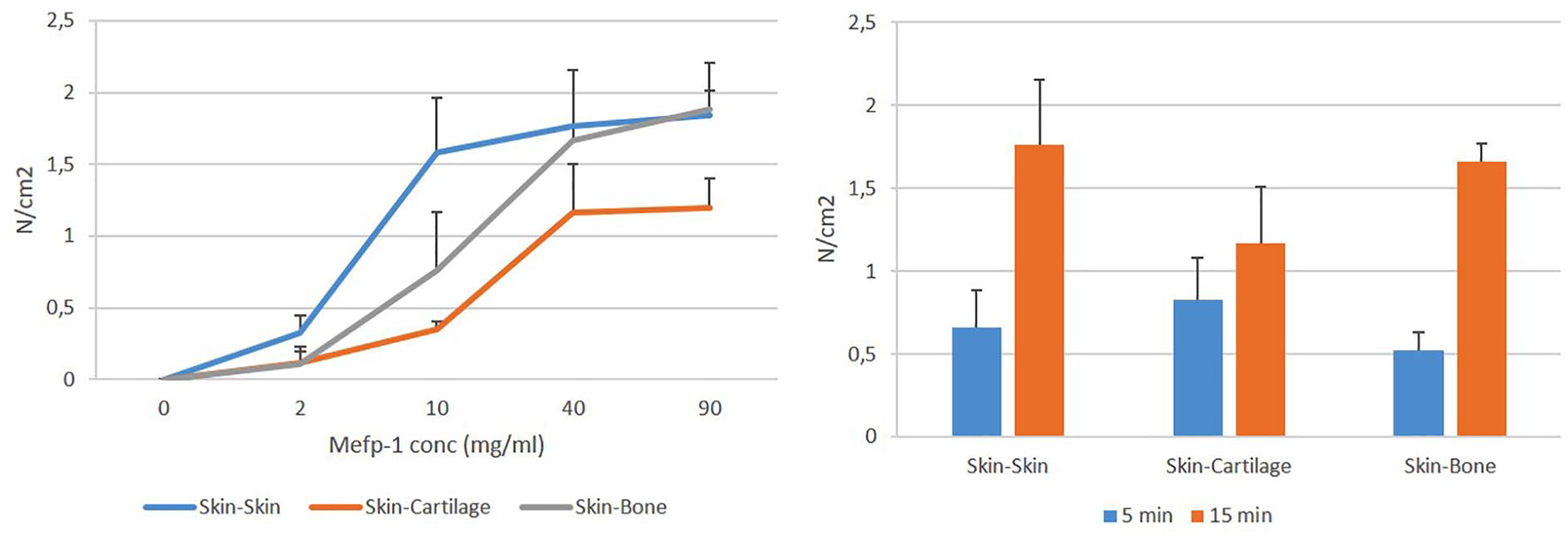
Adhesive strength of Mytilus edulis foot protein-1 (Mefp-1) to different tissues. (
Mefp-1 as Tissue Glue: Comparison with Fibrin
Next, Mefp-1 tensional force resistance was compared to that of fibrin. Skin, cartilage, and bone were glued together with 40 mg/mL of Mefp-1 or fibrin for 15 minutes at 37°C as described in Materials and Methods. The nonadhesive protein BSA and the cell adhesive bovine collagen were also measured as comparison. Tensional force resistance was measured using the Mark-10 instrument. Under these conditions, both Mefp-1 and fibrin developed a tensional force resistance of approximately 1 to 1.5 N/cm2. Collagen developed a force resistance of approximately 0.3 N/cm2 depending on the tissue type. BSA could not glue the tissues together to any extent that could be measured ( Fig. 8 ).
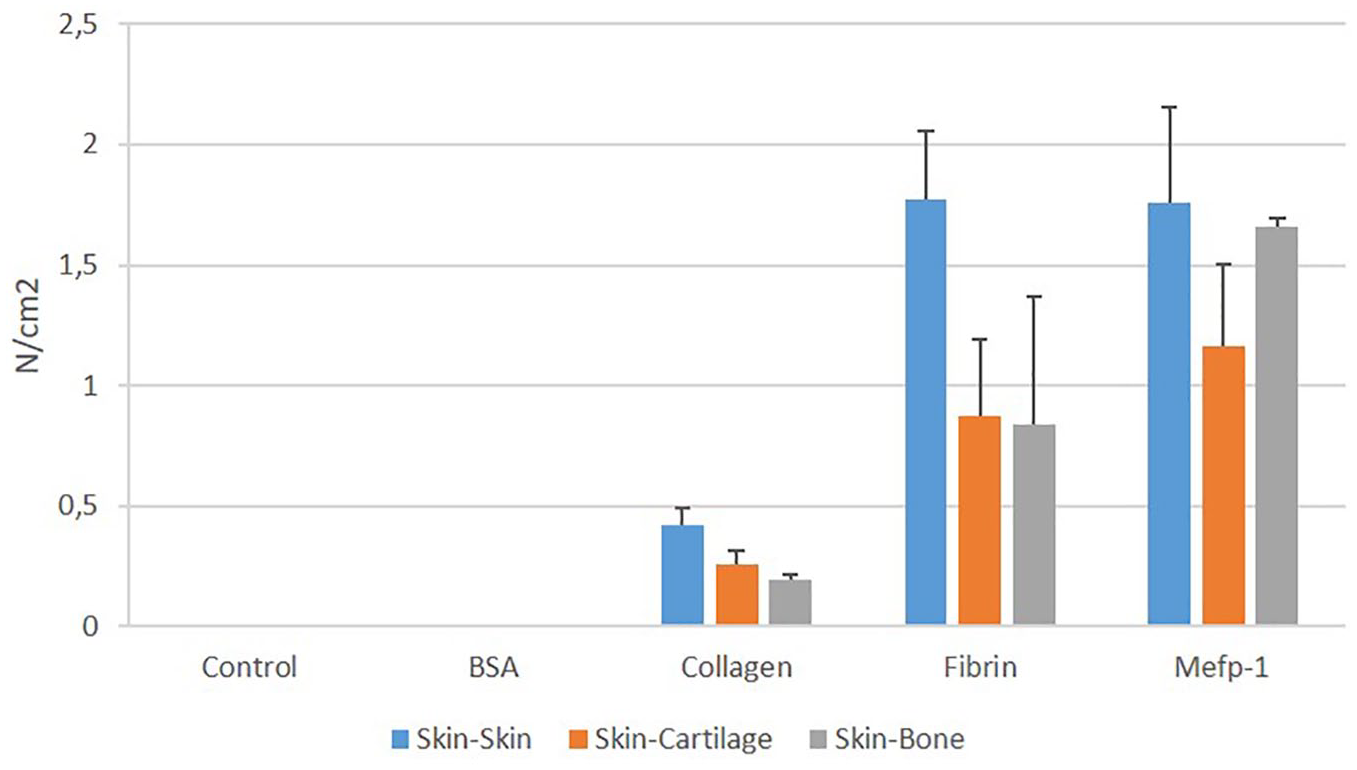
Tensional force resistance of 40 mg/mL bovine serum albumin (BSA), bovine collagen type I, fibrin, and Mytilus edulis foot protein-1 (Mefp-1) applied to different tissues. Tissues were pressed together under 800 g for 15 minutes at 37°C (n = 3).
Discussion
The results demonstrate that Mefp-1 can act as an efficient substrate for cell attachment and proliferation at low density (approximately 1 µg/cm2). Notably, Mefp-1 supported cell attachment at least as good as other well-characterized cell adhesives. At higher concentrations, approximately 40 mg/mL, it is also an efficient tissue glue, as demonstrated by experiments in which porcine dermis was attached to dermis, cartilage, or bone under moist conditions at 37°C for 15 minutes. Attachment of these tissues was found to be equally good as with fibrin (approximately 1 N/cm2 for both compounds).
The cell adhesion data in this report are in line with a previous study that showed that attachment of cartilage and osteoblast cells was increased when plastic culture plate was coated with MAP (Cell-Tak) compared to a noncoated control. 12 Here, we extend on this to compare directly with various commonly used adhesives. When it comes to tensional strength of tissues glued together with Mefp-1, there are scarce reports in the literature. One such study, however, demonstrated that whole feet mussel extracts developed a tensional strength between 2 pieces of porcine skin comparable to fibrin after prolonged curing time (beyond 6 hours). 14 This is in contrast to our results where tensional strength was developed after only 15 minutes of tissue adhesion. The reason for the discrepancy between our studies is unclear, but may relate to the fact that Ninan et al. used whole feet mussel extracts, which likely is containing a large proportion of nonadhesive proteins as well. 14 Importantly, the difficulty to maintain humidity in the tissue in these types of ex vivo experiments, even at water saturation of the environment, makes it difficult to draw relevant conclusions after prolonged attachment. It is, thus, likely that the mechanism of attachment/curing is different in our respective studies and may depend on concentration of adhesive proteins as well as other parameters, including humidity of the tissue.
While there are many appropriate methods for skin closure today, approaches for closure or fixation of internal organs are much more limited. Such organs cannot easily be stapled, or taped together. In addition, the substance used for fixation of tissues needs to be biodegradable and nonimmunogenic. With regard to the latter, it appears that purified polyphenolic proteins elicit a poor antibody response, when used to immunize rabbits, even when mixed with Freud’s adjuvant.9,15 Cell-based immunity/complement activation has also been reported to be poor (Nordic Biopharma AB, unpublished data). Thus, immunogenicity may not be an issue for further development of these proteins. Interestingly, different polyphenolic proteins have different binding properties when it comes to adhesion and cohesion. 8 For example, Mefp-5 has been reported to be a stronger adhesive protein than Mefp-1, 16 probably because of higher DOPA content. These data may help bioengineers generate novel polyphenolic proteins, with improved attachment to connective tissue. Increasing viscosity of the MAP formulation may also be beneficial. This would enable the proteins to remain in place in the tissue for longer time and, due to this, possibly increase binding efficiency. Moreover, although MAPs themselves are not scaffolds, they may improve binding and proliferation of cells in such constructs, as has been described earlier. 17 Thus, we envision that MAPs have the potential to aid in both scaffold fixation and to facilitate cell ingrowth in such constructs. Taken together, although the strength of the current preparation of MAP was similar to fibrin, there are several potential advantages of MAP compared to fibrin that could be further developed (see also limitations of fibrin described in the Introduction). Cost-effectiveness for clinical use depends on how successful this will become.
In conclusion, we found that Mefp-1 promotes cell adhesion at least as good as other well-established adhesives. At higher concentrations, it can also support adhesion of different types of soft and hard tissues comparable to fibrin. Thus, given the cell-friendly properties of Mefp-1, along with the capacity to glue various tissues together, Mefp-1 has properties that qualify it as a candidate for scaffold fixation and cell attachment in orthopedic surgery. The possibility to further modify properties of the Mefp-1 molecule by bioengineering, or by adding adjuvant (e.g., to increase viscosity), imply that further improvement can be anticipated.
Footnotes
Acknowledgments and Funding
The authors would like to acknowledge funding from The Knowledge Foundation to MI, and Örebro University to PW. We also wish to thank Sarah Gamble for proofreading the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Human cartilage was obtained from discarded tissue after complete knee or caput replacement surgery. Approval for this procedure was obtained from Swedish Ethical Review Authority (Dnr: 2019-02770).
Informed Consent
Written informed consent was obtained from legally authorized representatives before the study.
