Abstract
Objectives
The utilization of viral vectors to deliver genes of interest directly to meniscus cells and promote long-term modulation of gene expression may prove useful to enhance meniscus repair and regeneration. The objective of this study was to optimize and compare the potential of lentivirus (LV) and adeno-associated virus (AAV) to deliver transgenes to meniscus cells in both intact meniscus tissue and isolated primary cells in monolayer.
Design
Porcine meniscus tissue explants and primary meniscus cells in monolayer were transduced with LV or self-complementary AAV2 (scAAV2) encoding green fluorescent protein (GFP). Following transduction, explants were enzymatically digested to isolate meniscus cells, and monolayer cells were trypsinized. Isolated cells were analyzed by flow cytometry to determine percent transduction.
Results
LV and scAAV2 showed a high transduction efficiency in monolayer meniscus cells. scAAV2 was most effective at transducing cells within intact meniscus tissue but the efficiency was less than 20%. Outer zone meniscus cells were more readily transduced by both LV and scAAV2 than the inner zone cells. Higher virus titers and higher cell density resulted in improved transduction efficiency. Polybrene was necessary for the highest transduction efficiency with LV, but it reduced scAAV2 transduction.
Conclusions
Both LV and scAAV2 efficiently transduce primary meniscus cells but only scAAV2 can modestly transduce cells embedded in meniscus tissue. This work lays the foundation for viral gene transfer to be utilized to deliver bioactive transgenes or gene editing machinery, which can induce long-term and tunable expression of therapeutic proteins from tissue-engineered constructs for meniscus repair and regeneration.
Introduction
Menisci are fibrocartilaginous tissues that are essential for normal biomechanical function of the knee. The meniscus can be divided into 2 zones: the vascularized outer zone and the avascular inner zone. The outer zone is composed predominantly of type I collagen and fibrochondrocytes, while the inner zone is composed of predominantly type II collagen, proteoglycans, and chondrocyte-like cells. Meniscal tears, particularly in the inner zone, have a very low healing potential. Whenever possible, repair is preferred over partial or total meniscectomy because the loss of tissue results in osteoarthritis development. Therefore, new strategies are needed to promote meniscus repair and induce regeneration of functional meniscus tissue.
Tissue engineering strategies that use a combination of cells, biomaterials, and bioactive molecules have shown initial successes, but long-term controlled delivery of therapeutic factors has been challenging because of the short half-lives of bioactive molecules. 1 One solution is the utilization of viral vectors to deliver genes directly to meniscus cells to promote long-term expression of therapeutic proteins. Lentivirus (LV) and adeno-associated virus (AAV) can transduce nondividing cells and transduced genes can be expressed long term; thus, LV and AAV are promising viral vectors for potential clinical applications. 1 Therefore, the objective of this study was to optimize and compare the potential of LV and AAV to deliver transgenes directly to meniscus cells in both the context of intact meniscus tissue and isolated primary cells in monolayer.
Methods
LV encoding enhanced green fluorescent protein (eGFP) under the control of the EF-1α promoter was produced in HEK293T/17 cells by calcium phosphate precipitation, using the second-generation packaging system (psPAX2 and pMD2.G). 2 LV supernatants were filtered and concentrated using Amicon Ultra-15 100K filters, and biologically titered in HeLa cells by flow cytometry. 3
rAAV2/scAAV-CMV-GFP, and rAAV5/Hpa-TRS-SK (sc-CMV-eGFP) were purchased from the Gene Therapy Vector Core at the University of North Carolina, Chapel Hill. Viral particles (vp)/mL were determined by the core facility using dot blot, and we determined biological titer using HeLa cells.
Medial menisci were harvested from the knees of skeletally mature 2- to 3-year-old female pigs obtained from a local abattoir and explants (5 mm diameter × 2 mm thickness) were harvested from the midline of these menisci. To simulate a full-thickness defect, a 3 mm diameter core was removed and reinserted. 4 Explants were either undigested or digested with 0.05 mg/mL type 1 collagenase (Worthington) for 6 hours 5 to promote penetration of the virus. LV at 3 titers ranging from 1.3 × 106 to 5.2 × 106 transducing units (TU; n ≥ 5/group) in culture media (DMEM-HG [Dulbecco’s modified Eagle medium, high glucose; Gibco], 10% fetal bovine serum [Corning], 1% nonessential amino acids [Sigma], 1% HEPES buffer [ThermoScientific], 1% penicillin-streptomycin-fungizone [Gibco], 40 μg/mL proline [Sigma-Aldrich], and 37.5 μg/mL ascorbic acid 2-phosphate [Sigma-Aldrich]) containing 4 µg/mL polybrene (Sigma) or self-complementary AAV2 (scAAV2) at 6 × 1010 vp (n = 3/group) in DMEM-HG was applied to cover explants. Nontransduced controls contained media without virus. After 90 minutes at 37°C, meniscus culture media was added up to 1 mL and after 20 hours fresh culture media was added. On post-transduction day 7, tissue explants were cut in half, and fluorescent images were collected of both the tissue surface and cross-section to visualize GFP+ cells. Then, explants were digested with pronase and collagenase 6 to isolate the meniscus cells for flow cytometry.
Meniscal cells were isolated from the inner one-third and outer two-thirds of medial porcine menisci, using enzymatic digestion. 6 Freshly isolated cells were seeded onto 24-well plates at densities of 20,000 and 50,000 cells/well. After 48 hours, the media was removed and replaced with the following viruses based on prior work in cartilage 7 and mesenchymal stem cells 8 : (a) 1.17 × 107 TU (high titer) or 3.35 × 106 TU (low titer) LV in culture media with or without 4 µg/mL polybrene (n = 6/group); (b) scAAV2 or scAAV5 at 100,000 vp/cell (high titer) or 50,000 vp/cell (low titer) in culture media with 4 μg/mL polybrene (n = 4/virus); or (c) scAAV2 at 3,000 vp/cell (low titer), 10,000 vp/cell (medium titer), or 35,000 vp/cell (high titer) (n = 6/group) in culture media with or without 4 µg/mL polybrene. Nontransduced controls did not contain virus. After 20 hours, fresh culture media was added and replaced every 3 days. On post-transduction day 5, cells were placed on a fluorescent microscope, and images were taken in both the brightfield and FITC channels to show all cells and GFP+ cells, respectively. Next, cells were trypsinized and analyzed by flow cytometry.
In order to detect GFP+ cells by flow cytometry (Accuri), isolated cells were fixed in 1% paraformaldehyde for 24 hours. Cells were gated for whole cells and singlets, then nontransduced controls were used to set the background and 10,000 events were counted. For viability measurements, cells were stained with a fixable live/dead stain kit (Invitrogen) prior to fixation. Factorial analysis of variance and Newman-Keuls post hoc tests and t tests were performed to determine significant differences with α = 0.05 (Statistica).
Results
Following viral transduction of the meniscus tissue, GFP+ cells were visible along the tissue surfaces of the meniscal explants. However, by flow cytometry only 2% ± 2% (mean ± SEM) of cells in the meniscus tissue were GFP+ in the lowest LV titer group, and the highest titer yielded only 4% ± 5% GFP+ cells. Furthermore, collagenase digestion to promote viral penetration into the matrix did not improve LV transduction efficiency of cells in the tissue. On the other hand, scAAV2 transduced 17% ± 3% of meniscus cells in the context of the meniscus tissue.
Monolayer meniscus cells were efficiently transduced by LV (
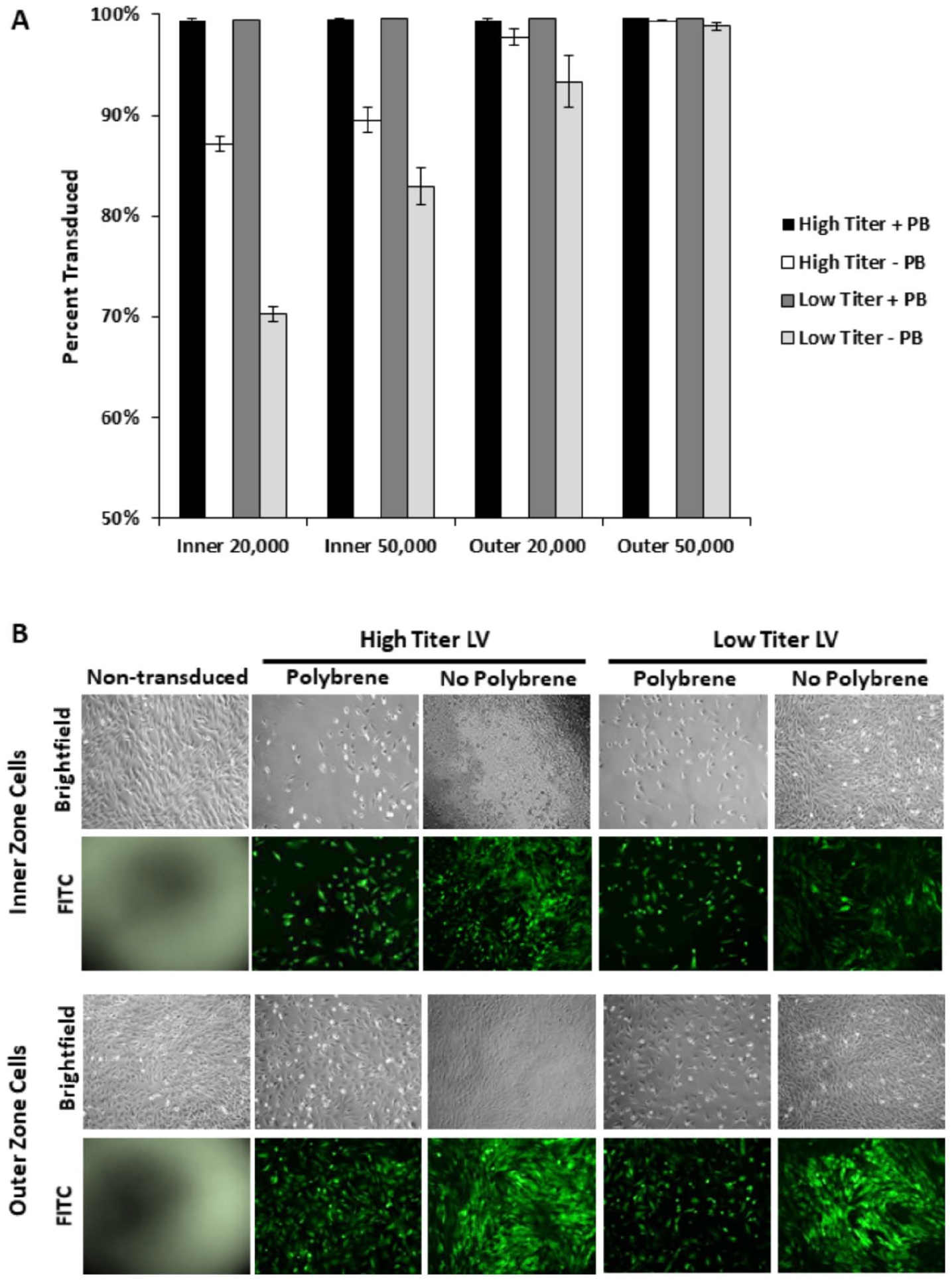
Lentiviral transduction of primary inner and outer zone meniscus cells. (
Monolayer meniscus cells were transduced by both scAAV2 and scAAV5. However, scAAV2 on average resulted in 72% ± 4% transduced cells, while scAAV5 resulted in only 50% ± 6% transduction (P < 0.01). Therefore, a more thorough analysis was performed with scAAV2.
scAAV2 more efficiently transduced outer zone meniscus cells than inner zone cells (
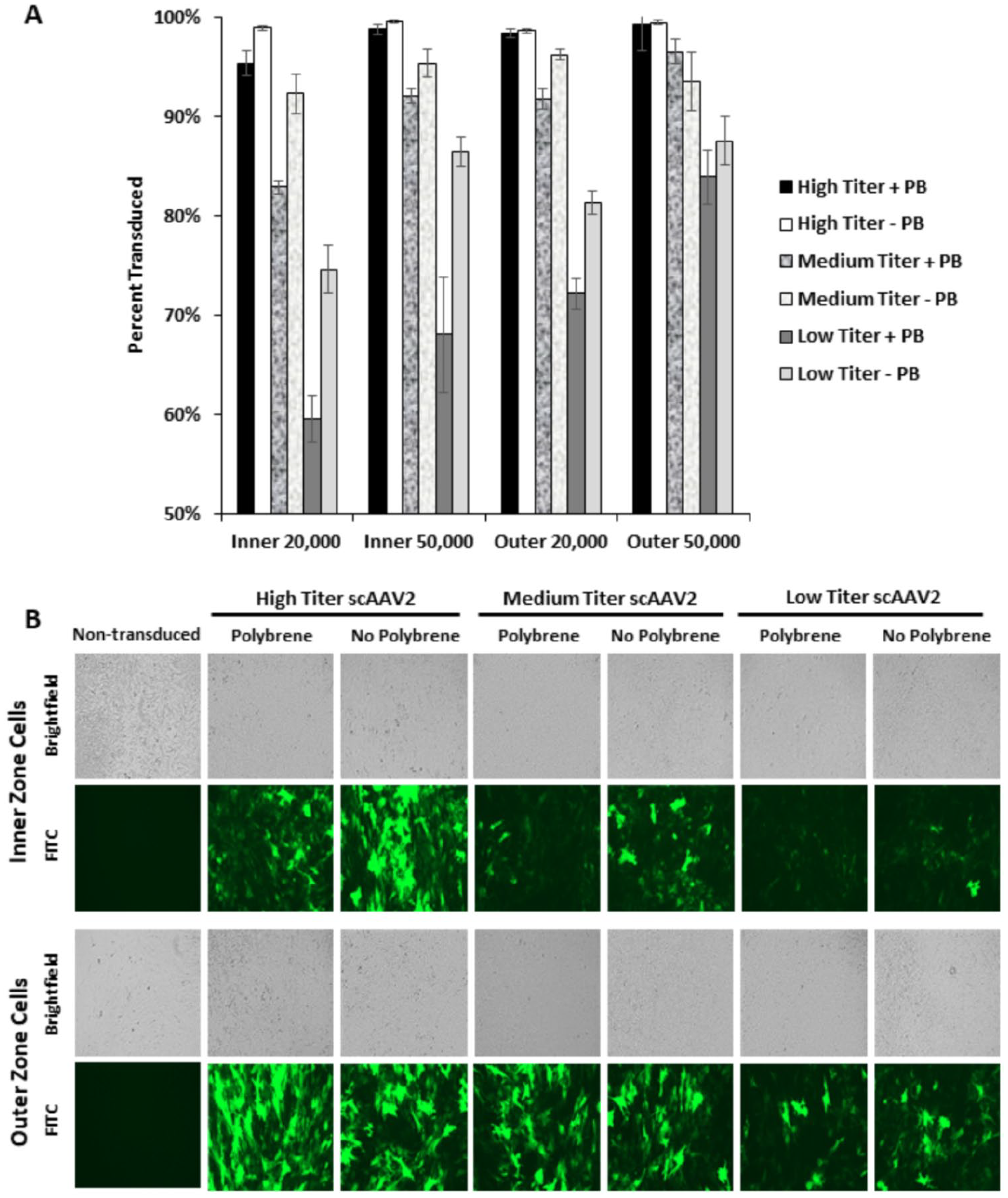
Self-complementary adeno-associated virus serotype-2 (scAAV2) transduction of primary inner and outer zone meniscus cells. (
Discussion
LV and scAAV2 showed a high transduction efficiency in isolated inner and outer zone meniscus cells, but only scAAV2 was able to modestly transduce cells within intact meniscus tissue. Prior work has shown that the fascia covering the meniscus is transduced when LV is injected into the joint, 9 a finding consistent with our observation of GFP+ cells at the tissue surface. This suggests that the extracellular matrix impedes virus penetration throughout the meniscus tissue. Prior work by Cucchiarini et al. has shown that rAAV2 can transduce the edges of injured meniscus tissue, 10 and that localized overexpression of growth factors may be able to promote tissue healing, 11 which is consistent with our findings. However, our results indicate that monolayer transduction of meniscus cells is necessary for efficient and uniform genetic modulation of meniscus cells.
Both inner and outer zone meniscus cells were 99% transduced by LV in the presence of polybrene, independent of cell density or viral titer. Meniscus cells were also efficiently transduced by LV even without polybrene, particularly in the outer zone. Polybrene neutralizes the electrostatic repulsion between the cell membrane and LV particles to promote cellular transduction, but it can reduce cellular proliferation. 12 This may have caused the reduced cell density in the samples transduced with LV in the presence of polybrene. Thus, successful LV transduction in the absence of polybrene reveals that it is not required to efficiently transduce meniscus cells for studies in which cellular growth and proliferation are important. Furthermore, polybrene suppressed transduction by scAAV2, indicating that polybrene is detrimental to scAAV transduction of meniscus cells.
While greater transduction efficiency is expected with a higher virus titer, cell viability was slightly reduced at the maximum scAAV2 dose. This indicates that the high titers necessary to achieve near-universal transduction may induce some cellular toxicity; however, the effect was modest with 94% of cells remaining viable. The higher viral titers are also likely to result in multiple transductions per cell, which is evidenced by the very bright GFP+ cells present in the transduction with high titer scAAV2. The lower titer transductions contain many more dimly GFP+ cells, which may only contain a single copy of transgene.
It is interesting to note that outer zone meniscus cells were more efficiently transduced than inner zone cells by both LV and scAAV2. Beyond the extracellular matrix and vascularity differences between the inner and outer zones of the meniscus, this suggests other cellular properties and possibly cell surface receptors can influence the ease of transduction. To directly compare the transduction efficiency of LV and scAAV, we biologically titered the scAAV2 and found we were using 6.5 TU/cell (high titer) and 0.56 TU/cell (low titer). For the LV experiments, we used 585 TU/cell (high titer) and 168 TU/cell (low titer) for 20,000 cells. In subsequent experiments, we have found that both inner and outer zone meniscus cells are approximately 90% transduced by 20 TU/cell of LV, indicating that the LV and scAAV2 are nearly equally efficient at transducing primary meniscus cells.
Gene therapy is an attractive possibility to enhance meniscus repair by combining cells that are overexpressing growth factors or other genes of interest with biomaterials. 1 Viral gene delivery has been reported using a retroviral vector, 13 adenovirus,14,15 and AAV 10 transduction of meniscus cells. However, to the best of our knowledge, our work is the first to report LV transduction of meniscus tissue and primary meniscus cells. Prior studies in cartilage tissue engineering have shown that LV can be used to deliver therapeutic genes in a controlled and tunable manner. 8 In the future, viral transduction of meniscus cells can be utilized to deliver or edit bioactive genes and induce long-term and tunable expression of therapeutic proteins from tissue-engineered constructs for meniscus repair and regeneration.
Footnotes
Authors’ Note
Research materials related to this article can be accessed by contacting the corresponding author.
Acknowledgments and Funding
The authors would like to acknowledge Jarrett Link, Taylor Waanders, and Lucas Lyons for technical assistance. This study was supported in part by National Institutes of Health grants AR073221, AG028716, AG15768, AG46927, a VA Rehabilitation Research Service Merit Review Award, the Arthritis Foundation, and an Orthopaedic Research and Education Foundation grant with funding provided by the Musculoskeletal Transplant Foundation.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Farshid Guilak is a founder and shareholder of Cytex Therapeutics, Inc. The other authors declare no potential conflicts of interests.
