Abstract
Objective
Recent studies have implicated the cyclin dependent kinase inhibitor, p21, in enhanced tissue regeneration observed in MRL/MpJ “super-healer” mice. Specifically, p21 is downregulated in MRL cells and similar ear hole closure to MRL mice has been observed in p21−/− mice. However, the direct implications of p21 deletion in endogenous articular cartilage regeneration remain unknown. In this study, we investigated the role of p21 deletion in the ability of mice to heal full-thickness cartilage defects (FTCDs).
Design
C57BL/6 and p21−/− (Cdkn1atm1Tyj) mice were subjected to FTCD and assessment of cartilage healing was performed at 1 hour, 3 days, 1 week, 2 weeks, and 4 weeks post-FTCD using a 14-point histological scoring system. X-ray microscopy was used to quantify cartilage healing parameters (e.g., cartilage thickness, surface area/volume) between C57BL/6 and p21−/− mice.
Results
Absence of p21 resulted in increased spontaneous articular cartilage regeneration by 3 days post-FTCD. Furthermore, p21−/− mice presented with increased cartilage thickness at 1 and 2 weeks post-FTCD compared with uninjured controls, returning to baseline by 4 weeks post-FTCD.
Conclusions
We report that p21−/− mice display enhanced articular cartilage regeneration post-FTCD compared with C57BL/6 mice. Furthermore, cartilage thickness was increased in p21−/− mice at 1 week post-FTCD compared with uninjured p21−/− mice and C57BL/6 mice.
Introduction
Unlike nearly all other tissues within the body, articular cartilage is both avascular and aneural and is thought to contain only 1 cell population (terminally differentiated chondrocytes).1,2 These properties of articular cartilage are thought to play a significant role in the inability of cartilage to repair itself after injury, with a complete restoration of the tissue and structural properties after injury rarely observed within the animal kingdom. 3 This lack of intrinsic repair after cartilage injury is a major contributor to degenerative joint diseases, such as osteoarthritis (OA). 4 While amphibians such as salamanders do have the capacity to regenerate cartilage, this ability is almost unheard of in mammals. 5 Defying these odds, the The Murphy Roths Large (MRL) mouse strain has the unique ability to spontaneously heal injured tissues including, but not limited to, articular cartilage.6-12 There have been a number of investigations into why these MRL mice have increased cartilage regeneration abilities, and it has been shown that one of the strains used to generate the MRL strain, the LG/J (Large) strain, demonstrates significant cartilage repair ability.13,14 Furthermore, it has been shown that the LG/J mice are also protected against posttraumatic OA, and a number of genes have been identified that correlated with this (cartilage healing/protection against OA) phenotype. 13 In the MRL model, analysis of fibroblasts has revealed decreased p21 gene expression and therefore a G1 cell cycle checkpoint deficiency, and this is thought to be partly responsible for the tissue regeneration observed in MRL mice. 15 This hypothesis has been validated in a p21 knockout (−/−) mouse model, where complete closure of through-and-through ear (auricular cartilage) hole wounds similar to the MRL mouse has been observed. 15 Additional studies into the mechanisms behind p21−/− mice auricular cartilage regeneration suggests a muted inflammatory response in where p21−/− mice demonstrate decreased expression of Sdf1, which results in a decreased recruitment of Cxcr4+ leukocytes to the injury site. 16
While the ear hole injury model established a functional role for p21 in tissue regeneration, increased susceptibility to OA in p21−/− mice has also been reported.17,18 In these studies, after destabilization of the medial meniscus (DMM) injury, p21−/− mice presented with mid-zone matrix loss within the cartilage, while C57BL/6 (wild-type) mice retained more of the articular cartilage matrix. 17 Furthermore, Kihara et al. observed increased subchondral bone destruction and cartilage matrix degradation in p21−/− mice after DMM injury. 18 Both studies out of the Kuroda lab observed decreased proteoglycan staining, a component of hyaline cartilage, in mice after DMM injury independent of p21 expression, suggesting that the loss of p21 does not protect cartilage from degenerative processes like OA.17,18 However, this observation is in conflict with previous studies demonstrating that the loss and/or decrease of p21 increases the chondrogenic potential of adult 19 and pluripotent 20 stem cells, and that p21−/− mice demonstrate in vivo cartilage (auricular) regeneration, 15 which is a heritable trait highly correlated with articular cartilage regeneration. 21 Since DMM is a model of OA and not cartilage repair/regeneration, it remains unknown if p21−/− mice can spontaneously heal full-thickness cartilage defects (FTCDs).
Despite its many limitations, conventional histopathology remains one of the most popular techniques used in preclinical studies to assess articular cartilage repair. Histological staining methods utilize 2-dimensional (2D) tissue sections, making it difficult to study whole-joint diseases such as OA. To view the entire joint, methods such as MRI (magnetic resonance imaging) can be employed, which has high soft tissue contrast and, unlike histopathology, allows for 3-dimensional (3D) visualization of the different joint tissues. 22 Previous studies have utilized MRI to evaluate morphological differences in whole joints after articular cartilage injury 23 ; however, the image resolution of MRI is low (e.g., voxel size can be in the 100s of µm) and visualization of cartilage tissue in small animal models is therefore limited.24,25 Like MRI, micro-computed tomography (µCT) is another 3D imaging method; however, µCT is best suited for imaging and quantifying hard tissues such as subchondral bone rather than soft cartilaginous tissues. 26 This is an important consideration when studying OA disease as the physiology of subchondral bone changes in relation to cartilage destruction remains disputed, with some studies suggesting a thickening of the subchondral bone plate before cartilage breakdown 27 and other studies observing subchondral bone thinning before cartilage destruction. 28 The earlier reported study by Kihara et al. used µCT to visualize subchondral bone destruction in mice after DMM and could not detect differences in subchondral bone density between p21−/− and C57 mice, suggesting the voxel size in µCT images (20 µm) might have masked any differences between the 2 mouse strains. 18
Recently, a new technology has been developed, 3D X-ray microscopy (XRM), that has advantages over both MRI and µCT in that it can assess cartilage and subchondral bone simultaneously within the same joint. 29 Furthermore, XRM technology can acquire images at higher contrast and higher resolution than MRI and µCT (upper spatial resolution of 70 nm) allowing for superior visualization of bone and joint microarchitecture. 29 Previous studies have successfully employed high-resolution 3D XRM technology to study calcified (hard) and soft tissues in rabbit knee joints30,31; however, to our knowledge, no studies have utilized XRM to quantify spontaneous cartilage repair and assess subchondral bone changes after injury in a mouse model. The ability to image soft tissues is clinically valuable to understanding disease progression and small animal models are critical players in advancing clinical practices. Therefore, in this study, we sought to determine if the p21−/− mouse model demonstrates enhanced articular cartilage regeneration ability after FTCD. Using XRM, we quantified articular cartilage healing in C57BL/6 and p21−/− mice and compared these findings to Safranin-O histology to identify the type of newly formed cartilage post-FTCD in both mouse strains.
Methods
Ethics Statement
All animal studies were performed in accordance with the recommendations in the Canadian Council on Animal Care Guidelines. The University of Calgary Health Sciences Animal Care Committee approved all animal protocols and surgical procedures used in this study (AC16-0043).
Animal Strains and Sample Size
C57BL/6 and p21−/− (B6;129S2-Cdkn1atm1Tyj) mice were purchased from the Jackson Laboratory (Bar Harbor, ME). p21−/− mice were back-crossed to the C57BL/6 background for 10 generations before use (these mice are referred to as p21−/−). Furthermore, to control for differences in endogenous repair capacity between the C57BL/6 and 129 stains, we took mice from the p21−/− × C57BL/6 back-crossings in the second generation that did not carry a mutant copy of the p21 allele and backcrossed them to C57BL/6 mice for an additional 8 generations (these mice are referred to as C57BL/6).
FTCDs were surgically induced in the left knee of 8-week-old mice (n = 60) per time-point as detailed here: C57BL/6 (3D XRM: n = 15; Histology: n = 15) and p21−/− (3D XRM: n = 15; Histology: n = 15). A further n = 6 per group were employed as uninjured controls (3D XRM: n = 3; Histology: n = 3).
Cartilage Defect Model
Animals were administered an intraperitoneal injection of buprenorphine (0.05 mg/kg) prior to surgery and anaesthetized under isoflurane (Baxter) anesthesia (1.5% v/v O2) for the duration of the surgical procedure. FTCDs were performed using a modified version 23 of the established protocol by Eltawil et al. 32
Using a scalpel a small incision was made on the medial side of the left knee. A depth stopped 26-G needle (diameter = 450 µm, length to stopper = 600 µm) was used to gently displace the patella and expose the trochlear groove of the femur. A slight pressure, combined with a twisting motion, was applied at the contact with the trochlear groove to make a circular wound that penetrated no farther than 600 µm into the underlying subchondral bone. The needle was gently removed and the skin closed with a sterile wound clip after the FTCD was made.
Histological Scoring of Cartilage Repair
Animals were sacrificed at 1 hour, 3 days, 1 week, 2 weeks, and 4 weeks post-FTCD. Intact mouse femurs were dissected and fixed in neutral buffered formalin (Sigma) for 5 days prior to being decalcified in CalEx (Fisher Scientific) for 10 days. After decalcification was complete, samples were processed for paraffin sectioning. Serial sagittal sections (10 µm) were co-stained with Safranin-O and fast green, and graded based on a previously published scoring system by Fitzgerald et al. 11 The parameters of the scoring system include cell morphology (0-4), matrix staining (0-3), surface regularity (0-3), thickness of cartilage (0-2), and integration with native cartilage (0-2). On this scale, uninjured native articular cartilage is 14, while the absence of cartilage is 0. Blinded grading of all images was performed by 2 independent observers.
3D X-Ray Microscopy
Isolated mouse femurs were placed upright in polymide tubing with PBS surrounding the long bone shaft, sealed by a plastic wrap and secured in the device holder. Low-energy (40 kVp voltage, 3 W power) XRM scans were performed using a 4× objective. The exposure time of each projection was 3 seconds, and 2,001 projections were collected per sample rotation. Assessment of joint microarchitecture was achieved at an isotropic nominal resolution of 1 voxel = 4.4 µm.
3D XRM Data Analysis
The 3D XRM data sets of the isolated femurs were imported into Amira software (Version 6.0.1; FEI, Portland, OR) for visualization of femur morphometry and FTCDs. Manual segmentation of cartilage from underlying subchondral bone was performed within a standardized region of interest (diameter = 0.53 mm). The selection of a segmentation threshold for cartilage and subchondral bone parameters was done qualitatively by the same operator. Cartilage volumes were quantified to monitor changes in cartilage morphometry post-FTCD. Outcome measures included cartilage volume (Cg.V [mm3]), cartilage surface area (Cg.S [mm2]), cartilage surface-to-volume ratio (Cg.S/Cg.V [mm-1]), and mean cartilage thickness (Cg.Th [mm]). Cartilage thickness was computed using the thickness transform 33 and rendered using direct volume rendering. Morphometric analysis was performed directly on the segmented cartilage volumes in Image Processing Language (IPL Version 5.42; SCANCO Medical, Brüttisellen, Switzerland).
Histological Imaging
Slides stained with safranin-O and fast green were imaged under brightfield on an Axio Scan.Z1 Slide Scanner microscope (Carl Zeiss) using a Plan-Apochromat objective (10×/0.8 M27).
Statistical Analysis
GraphPad Prism software (Version 7.0) was used to summarize the variables (mean ± standard deviation) and perform standard descriptive statistics, significance reported at P ≤ 0.05. Statistical analysis was performed by 2-way ANOVA followed by Tukey’s post hoc testing. Statistical methods were reviewed by a biostatistician (Dr. Guomin Ren) at the University of Calgary.
Results
p21−/− Mice Display Endogenous Articular Cartilage Regeneration Post-FTCD
To directly examine the role of p21 in articular cartilage regeneration, histological samples from C57BL/6 mice were compared to p21−/− mice at 1 hour, 3 days, 1 week, 2 weeks, and 4 weeks post-FTCD. Articular cartilage healing scores demonstrated a significant difference on p21 deletion, with p21−/− mice showing enhanced regeneration by 3 days post-FTCD compared with C57BL/6 mice ( Fig. 1 ). In agreement with articular cartilage healing scores, p21−/− mice displayed robust proteoglycan positive matrix deposition compared with C57BL/6 mice, as observed by safranin-O staining in the cartilage defect area by 2 weeks post-FTCD (C57: Fig. 2M-R ; p21−/−: Fig. 3M-R ).
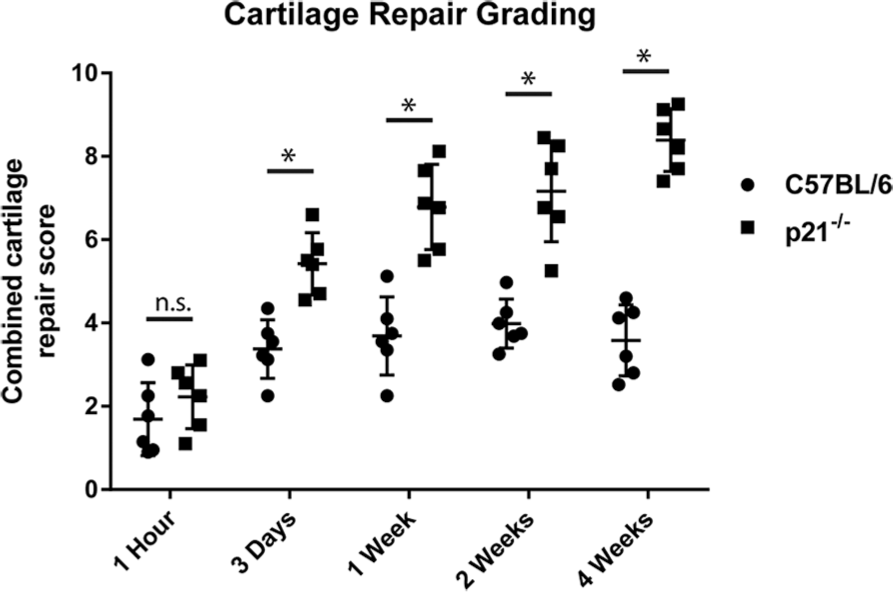
Grading of articular cartilage healing post-FTCD. p21−/− mice demonstrated significantly increased articular cartilage healing scores compared with C57BL/6 mice as early as 3 days post-FTCD (n ≥ 6 for each animal strain/time-point. Data were analyzed by analysis of variance (ANOVA), *P ≤ 0.05.
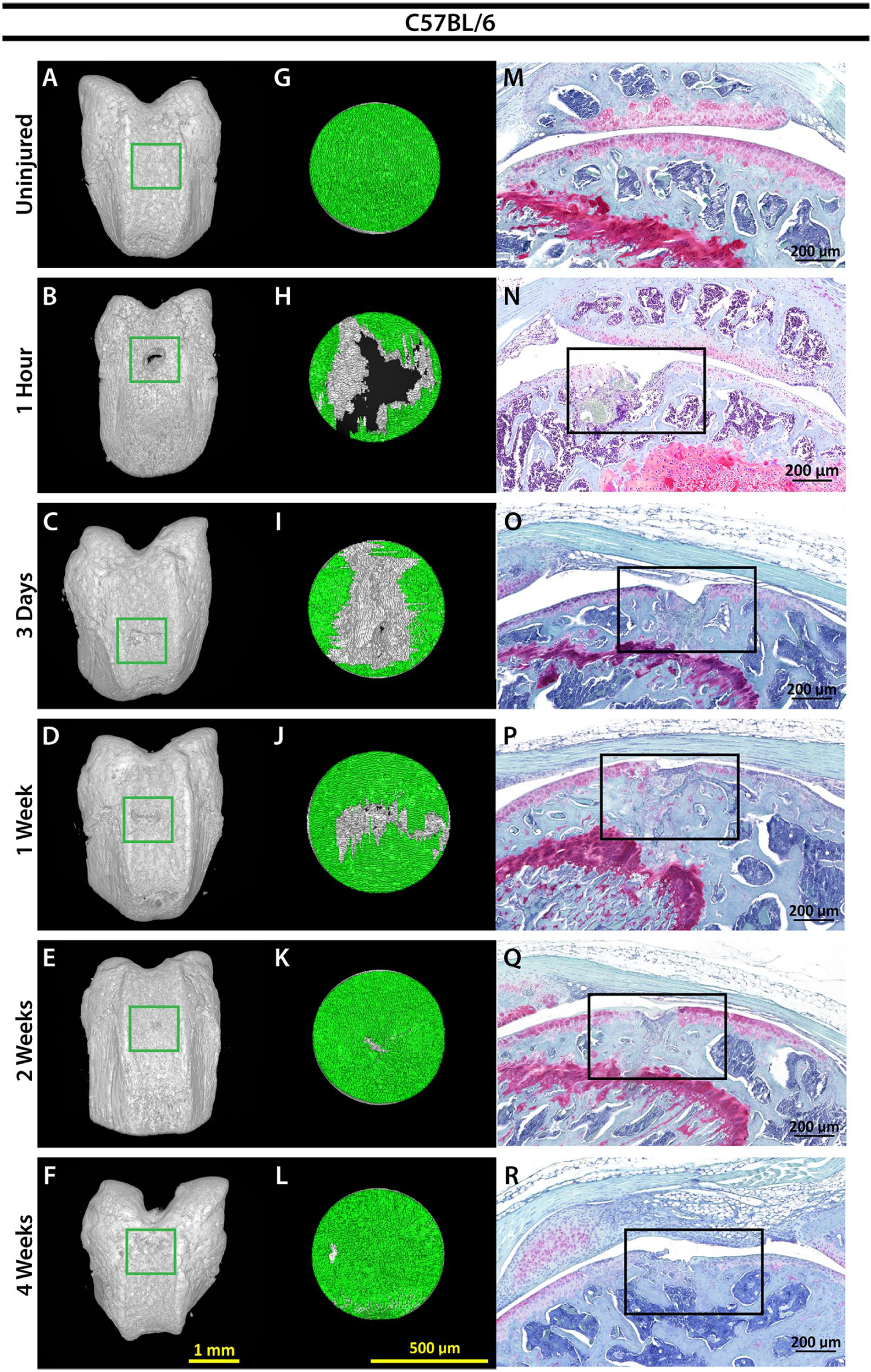
Imaging of cartilage healing in C57BL/6 mice. 3D XRM images of C57BL/6 mouse femur at indicated time points, scale bar = 1 mm, green box = location of FTCD (
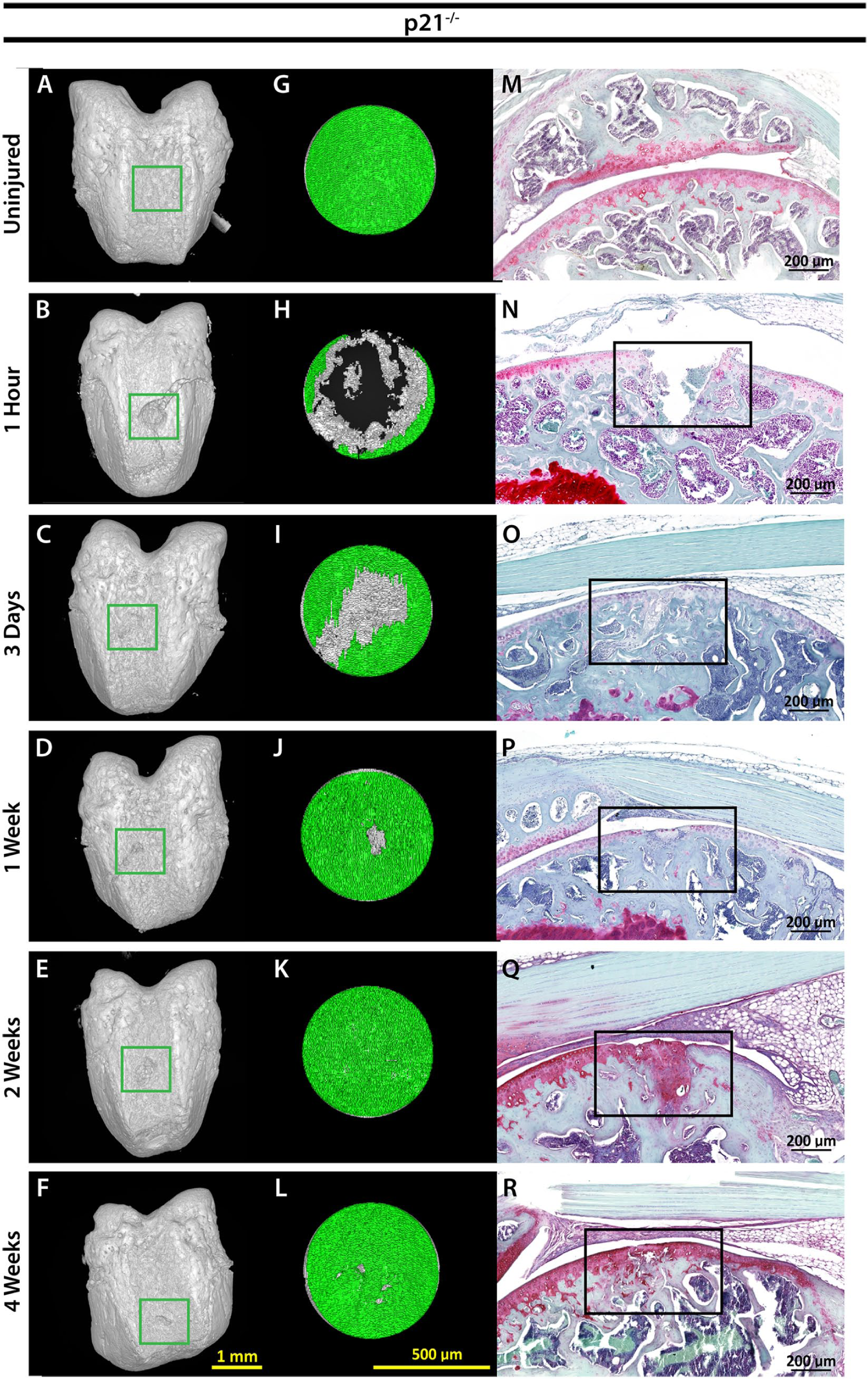
Imaging of cartilage healing in p21−/− mice. 3D XRM images of p21−/− mouse femur at indicated time points, scale bar = 1 mm, green box = location of FTCD (
p21−/− Mice Exhibit Thicker Cartilage Post-FTCD
To quantify structural changes in joint morphology post-FTCD, 3D XRM assessment of C57BL/6 and p21−/− mouse femurs was performed (C57: Fig. 2A-F , p21−/−: Fig. 3A-F ) within a standardized region of interest (C57BL/6: Fig. 2G-L , p21−/−: Fig. 3G-L ). By 1 week post-FTCD, p21−/− mice demonstrated significantly increased cartilage thickness (mm) compared to C57BL/6 mice within the FTCD region of interest (0.0226 ± 0.0037 mm; and 0.0151 ± 0.0038 mm, respectively) ( Fig. 4 ). Compared with uninjured p21−/− mice (0.0130 ± 0.0011 mm), a significant increase in cartilage thickness was observed in p21−/− mice at 1 week (0.0226 ± 0.0037 mm) and 2 weeks post-FTCD (0.0231 ± 0.0054 mm) ( Fig. 4 ). By 4 weeks post-FTCD, no significant difference in cartilage thickness was present between C57BL/6 and p21−/− mice (0.0156 ± 0.0017 mm and 0.0156 ± 0.0026 mm, respectively) ( Fig. 4 ). A cartilage thickness map of the newly regenerated soft tissue within the FTCD region of interest in C57BL/6 and p21−/− mice was generated and presented as a virtual color-scaled image ( Fig. 5 ). Furthermore, it could be observed from this imaging modality that both fibrocartilage and hyaline-like cartilage repair/regeneration in C57BL/6 or p21−/− mice, respectively, healed from the periphery to the middle of FTCD site.
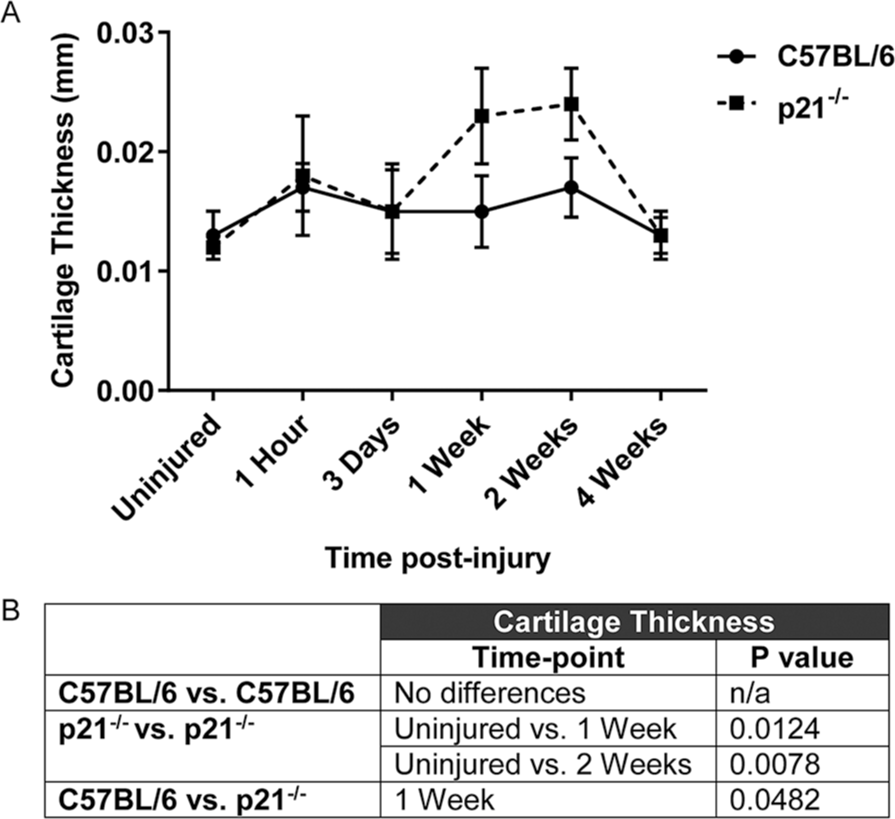
Evaluation of cartilage thickness (mm) in uninjured C57BL/6 (solid line) and p21−/− (dashed line) mice and at 1 hour, 3 days, 1 week, 2 weeks, and 4 weeks post-FTCD (
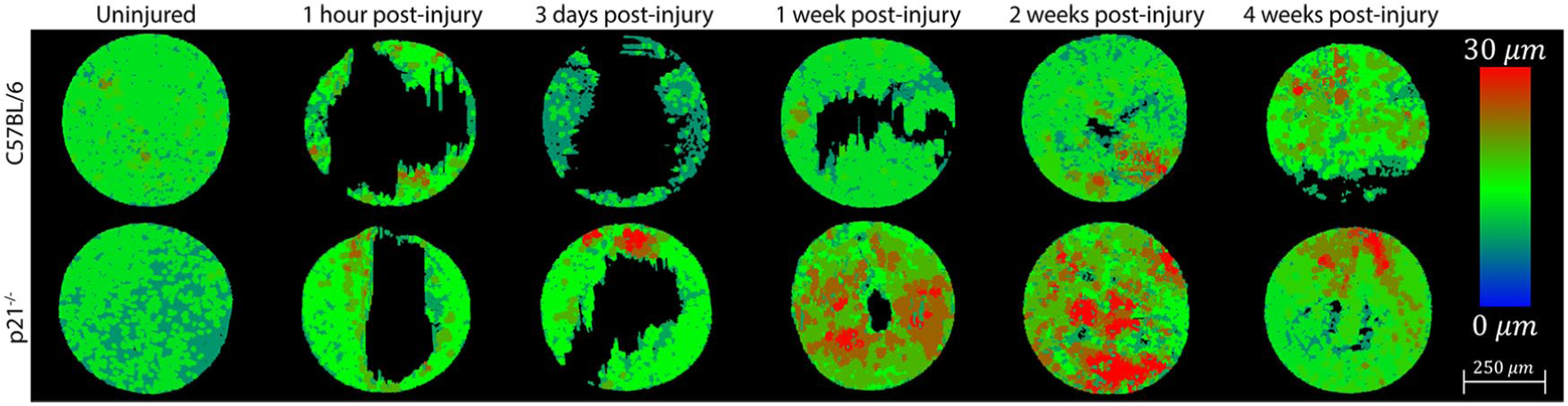
Evaluation of cartilage morphology in uninjured C57BL/6 (Upper) and p21−/− (Lower) mice and at 1 hour, 3 days, 1 week, 2 weeks, and 4 weeks post-FTCD. Representative topographic maps showing the thickness (µm) of newly formed cartilage post-FTCD within a standardized region of interest (n = 3 for each strain/time-point). Color key: 0 µm = Blue and 30 µm = Red, scale bar = 250 µm.
The ratio of cartilage surface area to volume was examined in C57BL/6 and p21−/− mice pre- and post-FTCD to assess the surface regularity of the newly formed cartilage layer ( Fig. 6 ). A high surface area/volume ratio is an indicator of surface irregularity. No differences in surface area/volume ratio were observed in or between C57BL/6 and p21−/− mice at any time point examined post-FTCD ( Fig. 6 ).
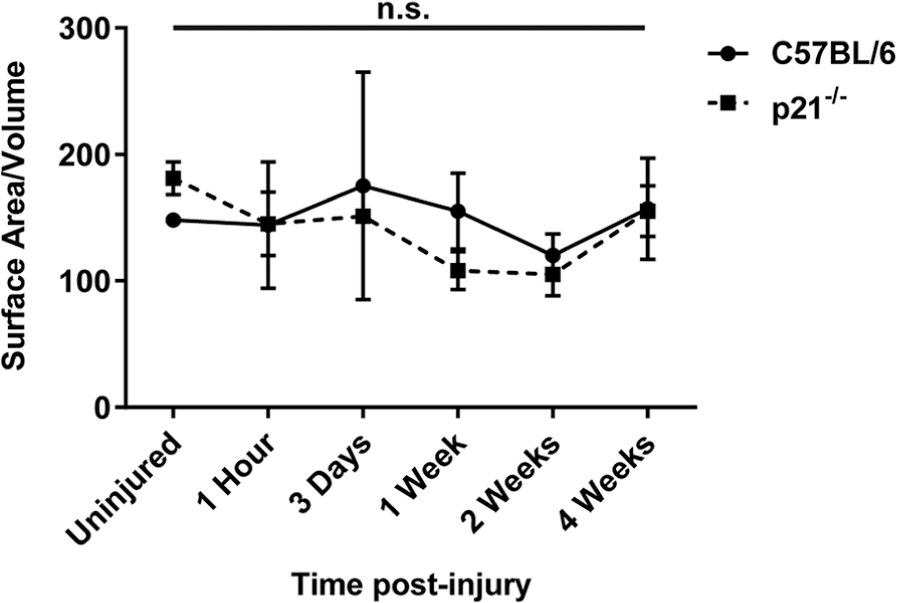
Ratio of cartilage surface area to volume in uninjured C57BL/6 (solid line) and p21−/− (dashed line) mice and at 1 hour, 3 days, 1 week, 2 weeks, and 4 weeks post-FTCD. A summary table of significance for cartilage thickness indicating no significance (ns) is shown. Data were analyzed by analysis of variance with Tukey post hoc testing (*P ≤ 0.05).
Discussion
The superior healing ability of MRL mice is well documented6-11 and has been linked to a deficiency in p21, a cell cycle regulator. 15 Enhanced healing in the absence of p21 has also been observed in tissues that normally regenerate (e.g., bone), as p21−/− mice display an increased rate of bone healing after tibial fracture compared with C57BL/6 mice. 34 However, it remained undetermined if the absence of p21 would induce an articular cartilage regeneration phenotype similar to what is observed in MRL mice. In the current study we have shown that, while both C57BL/6 and p21−/− mice heal full-thickness (e.g., penetrating the underlying subchondral bone) cartilage injuries, C57BL/6 mice repair cartilage defects with fibro-like cartilage devoid of proteoglycans, whereas p21−/− mice regenerate a thicker hyaline-like cartilage, rich in proteoglycan content. Furthermore, p21−/− mice generated thicker articular cartilage in the defect site 1 week post-FTCD (compared with uninjured controls) and this response was not observed in C57BL/6 mice. However, a limitation of this study was that 3D XRM cannot distinguish between cartilage subtypes (e.g., hyaline-like vs. fibro-like) and therefore we must assume that the increased cartilage thickness in p21−/− mice obtained from 3D XRM analysis corresponds to the hyaline-like characteristics of cartilage observed from 2D histological assessment.
The observation of increased cartilage thickness in p21−/− mice in response to cartilage injury and compared with C57BL/6 mice at 1 week post-FTCD, as well as the superior quality (hyaline-like vs. fibro-like) of newly formed cartilage in the absence of p21, coupled with previous reports that p21−/− mice heal bone fractures faster than C57BL/6 mice by 1 week, raises questions regarding the role of the underlying bone during articular cartilage repair/regeneration. Specifically, we observed hyaline-like cartilage within the injured subchondral bone in p21−/− mice but not within C57BL/6 mice. This cartilage within the bone appeared to be responsible for the increased thickness measurements observed, yet by 4 weeks post-injury, this abnormally located cartilage was replaced by subchondral bone tissue. It is therefore possible that the superior cartilage healing observed in p21−/− mice compared to C57BL/6 mice may be a consequence of altered bone healing, and potentially as a result of an altered endochondral ossification process. These hypotheses are in line with our previous observation of increased fracture repair in p21−/− mice. 34 While new bone tissue was observed in the FTCD site of both mouse strains at 3 days post-FTCD, we were unable to quantify the parameters of this newly formed tissue due to technical constraints. It would therefore be of interest to determine if the same quality of bone is being produced between the two strains of mice. If there are differences in the quantity and/or quality of subchondral bone repair between p21−/− and C57BL/6 mice, then it would be of interest to conditionally knockout p21 in the osteogenic lineage to determine if the loss of p21 in bone directly impacts the type of cartilage generated post-FTCD.
As stated earlier, our study did not quantify subchondral bone changes in C57BL/6 and p21−/− mice post-FTCD; however, a previous study by Ward et al. investigated the bony changes that occur after intraarticular fracture in C57BL/6 and MRL mice and found no differences in bone density and/or subchondral bone thickness in MRL mice, whereas C57BL/6 mice displayed decreased bone density and increased subchondral bone thickness. 35 Whether subchondral bone healing is enhanced in MRL mice compared with C57BL/6 mice and a contributing factor in the superior ability of MRL mice to regenerate articular cartilage remains unknown, as no comparisons between mouse strains were made in that study. What the previous study does suggest is that the proper restoration of subchondral bone microarchitecture must occur in order for mice to regenerate articular cartilage. Therefore, additional experiments directly examining subchondral bone changes in mouse strains which demonstrate articular cartilage regeneration (such as the MRL and p21−/− mice) are required to determine if superior articular cartilage healing in p21−/− mice post-FTCD is a consequence of enhanced/differentially regulated subchondral bone healing.
Little is known about articular cartilage regeneration since it typically does not occur in mammals. However, cartilage repair through the production of a fibrocartilage-like patch follows the wound healing response observed in numerous other tissues in the body.36,37 The wound healing response is one of the oldest and most-conserved physiological responses in the animal kingdom, as restoring tissue integrity/homeostasis can be the difference between life and death. Wound healing in mammals is mediated primarily by immune cells and inflammatory signaling molecules that can regulate other tissue resident cells, including adult stem cells, to mediate closure of the wound through formation of a scar.38,39 Since the fibrocartilage patch that occurs in C57BL/6 mice (and humans) post-FTCD is analogous to scar tissue, it could be hypothesized that the body believes it is essential to close the wound as quickly as possible. While this is necessary in vascularized tissues to maintain barrier integrity, this might not be the case for the articular cartilage since it is not vascularized, nor is it exposed directly to the circulatory system. Therefore, it may be possible that the deletion of p21 is interfering with the normal wound healing/fibrotic response, and while this may have negative repercussions in certain types of injuries, it appears to be beneficial with regard to cartilage repair.
Previous studies have shown that partial-thickness cartilage defects (e.g., not penetrating the underlying subchondral bone) do not regenerate in healing mouse strains (such as the MRL mouse).11,32 This suggests that the articular cartilage regeneration mechanism at least partially involves subchondral bone remodeling and/or cell type(s) within the bone marrow. This is supported in clinical practice, as one of the most common surgical techniques performed to stimulate new cartilage formation in patients with cartilage injuries (e.g., microfracture) involves releasing bone marrow and possibly bone marrow stem cells into the joint space. Like subchondral bone, the synovium is another vascularized tissue, rich in stem cells, and studies have shown that mesenchymal stem cells (MSCs) from the synovium migrate to injury sites and possess the ability to differentiate into chondrocytes in vivo to aid in repair. 40 Furthermore, we have previously shown that cartilage injuries in MRL mice have increased numbers of MSCs and that synovial MSCs from MRL mice injected into knee joints home to sites of cartilage injury. 41 After migration of MSCs to cartilage injuries, do MSCs release trophic factors to aid in articular cartilage healing and/or do MSCs differentiate into new cartilage in healer mouse strains? Future studies using lineage reporter mice to track the fate of MSC/progenitor cell populations during endogenous articular cartilage regeneration are necessary to determine the cell type(s) and/or factors responsible for the superior healing phenotype observed in some mouse strains, such as the MRL and p21−/− mouse.
In conclusion, we report that p21−/− mice display enhanced articular cartilage regeneration post-FTCD compared with C57BL/6 mice. Furthermore, we have demonstrated that cartilage thickness is increased in p21−/− mice at 1 week post-FTCD compared with uninjured p21−/− mice and C57BL/6 mice. Future studies examining the role of MSC/progenitors and trophic factors in the context of p21 deletion will be necessary to determine the mechanism by which p21−/− mice spontaneously regenerate articular cartilage and to determine if cell cycle targeting has potential as a safe and effective therapeutic strategy for treating cartilage injuries.
Footnotes
Author Contributions
Conception and design: CLJ, RJK. Analysis and interpretation of the data: CLJ, BAB, JA, RJK. Drafting of the article: CLJ, RJK. Critical revision of the article for important intellectual content: CLJ, BAB, JA, RJK. Final approval of the article: CLJ, BAB, JA, RJK. Provision of study materials: CLJ, RJK. Obtaining of funding: RJK. Collection and assembly of data: CLJ, BAB, JA, RJK.
Acknowledgments and Funding
The authors would also like to thank the University of Calgary Animal Resource Centre for their assistance with mouse husbandry and Catherine Leonard for her assistance with genotyping. We would also like to note that figures, tables, and text from the current study was also included in the first authors PhD thesis (Christina Jablonski), archived by the University of Calgary in 2019. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grants through Natural Sciences and Engineering Research Council (NSERC) of Canada (RJK # RGPIN-2014-04586), Canada Foundation for Innovation (RJK), Grace Glaum Professorship (RJK), and Canadian Institutes for Health Research (RJK # 201309MOP-311542-DIB-CBBA-170939) supported the work. CLJ was supported by NSERC Create and Biomedical Engineering Studentships. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The University of Calgary Health Sciences Animal Care Committee approved all animal protocols and surgical procedures used in this study (AC16-0043).
Animal Welfare
All animal studies were performed in accordance with the recommendations in the Canadian Council on Animal Care Guidelines.
