Abstract
Objective
Syndecan-4 plays a critical role in cartilage degradation during osteoarthritis (OA). The aim of this study was to investigate the expression and localization of syndecan-4 in different OA joint tissues.
Design
Syndecan-4 mRNA levels were quantified by reverse transcription–polymerase chain reaction in human OA primary cells. Syndecan-4 was localized by immunohistochemistry in knee, hip, or shoulder OA bone/cartilage biopsies. Syndecan-4 was quantified by immunoassay in chondrocytes culture supernatant and cell fraction.
Results
Using immunochemistry, syndecan-4 was observed in chondrocytes clusters in the superficial zone of OA knee, but not in OA hip or shoulder cartilage. No significant difference was detected in syndecan-4 expression level in sclerotic compared with nonsclerotic osteoblasts or in inflamed synoviocytes compared to normal/reactive ones. Differentiated hypertrophic chondrocytes from knee, but not from hip cartilage, expressed more syndecan-4 than nonhypertrophic cells. Using an immunoassay for the extracellular domain of syndecan-4, we found 68% of the syndecan-4 in the culture supernatant of OA chondrocytes culture, suggesting that a large majority of the syndecan-4 is shed and released in the extracellular medium. The shedding rate was not affected by hypertrophic differentiation state of the chondrocytes or their joint origin.
Conclusions
Even if chondrocytes clusters are seen in OA knee, hip and shoulder cartilage and hypertrophic differentiation appears in knee and hip OA articular chondrocytes, syndecan-4 synthesis only increased in knee. These findings suggest the presence of biochemical difference between articular cartilage according to their location and that syndecan-4 could be a biochemical marker specific for knee OA.
Introduction
Syndecan-4 is one of the 4 vertebrate syndecans. Syndecans are transmembrane heparan sulfate proteoglycans that regulates cell-matrix interactions. Though their glycosaminoglycan chains, syndecans interact with a variety of extracellular matrix molecules as well as with growth factors and cytokines. 1 They are involved especially in embryonic development, tumorigenesis, and angiogenesis. 1 Syndecans are expressed by chondrocytes 2 but their role in cartilage homeostasis or degradation remain poorly documented. It has been demonstrated that syndecans facilitate the binding of FGFs to their receptors during chondrogenesis. 2
Syndecan-4 mRNA were significantly increased in knee OA chondrocytes cultures compared with healthy 3 and are correlated with vascular endothelial growth factor expression. 4 Syndecan-4 is involved in endochondral ossification 5 and is specifically expressed by chondrocytes producing type X collagen both in human and murine OA cartilage. 6 These findings suggest that in OA, syndecan-4 expression could be associated with hypertrophic chondrocyte differentiation. The loss of syndecan-4 in genetically modified mice and intra-articular injections of syndecan-4-specific antibodies into wild-type mice protect from proteoglycan loss and thereby prevent cartilage damage in a surgically induced OA model 6 through a marked decrease in ADAMTS-5 activity. 6 Syndecan-4 controls the activation of ADAMTS-5 through a direct interaction, and through regulating mitogen-activated protein kinase (MAPK)-dependent synthesis of matrix metalloproteinase-3 (MMP-3). 6 Syndecan-4 is also able to bind MMP-13. 7 Finally, tumor necrosis factor–α and interleukin-1β increase syndecan-4 expression by chondrocytes. 8
The aim of this study was to investigate the expression and localization of syndecan-4 in different tissues, especially the subchondral bone, synovial membrane and articular cartilage, from OA hip, knee, or shoulder. We have also compared, using well-characterized in vitro models, the expression of syndecan-4 by sclerotic (SC) and nonsclerotic (NSC) osteoblasts,9-11 inflamed and normal/reactive synoviocytes12,13 and nonhypertrophic and hypertrophic chondrocytes. 14
Methods
Subchondral Osteoblasts Cell Culture
All tissues used in this study were obtained after approval by the Faculty of Medicine Ethics Committee of the University of Liège (number B70720108313, reference 2010/43) and written informed consent was obtained from all subjects. Tibial subchondral bone plates were obtained from 5 OA men during total knee replacement (TKR). On dissection, the femoral, tibial, and patellar articular surfaces were evaluated for the gross morphological modifications of cartilage according to the Collins diagram. 15 Different grades were considered: 0, normal white cartilage in all areas examined; I, the presence of a yellow-gray area with some superficial fibrillation on one or more articular surfaces; II, irregular surface with deep fibrillations on one or more articular surfaces; III, subchondral bone denudation on one or more articular surfaces, in less than 50% of the most damage compartment; and IV, subchondral bone denudation on more than 50% of the articular surface at least in one tibial plateau. All cartilage samples showed grade IV OA lesions. Patient characteristics are listed in Table 1 . Bone explants were prepared as previously described10,11 (http://dx.doi.org/10.17504/protocols.io.mkmc4u6). Briefly, after careful elimination of trabecular bone and overlying cartilage, OA subchondral bone plate was dissected to separate NSC from SC zones. We have considered as SC bone only the subchondral bone zones with a thickness greater than 2 mm and either denuded or overlaid by fibrillated cartilage. Also, we have considered only the subchondral bone zones with a maximal thickness of 1 mm as NSC bone. 10 Osteoblasts from SC or NSC subchondral bone were then obtained by outgrowth from explants. At confluence, primary cells were collected by trypsinization, seeded (20,000 cells/cm2) in 12-well plates (12-well companion plates, Falcon, BD Biosciences) and grown for 3 days in Dulbecco’s modified Eagle medium (DMEM) containing 10% fetal bovine serum (FBS), 100 U/mL penicillin, 100 µg/mL streptomycin, 10 mM HEPES, and 2 mM glutamine. The structural difference between SC and NSC bone explant was confirmed histologically as previously reported. 10 Furthermore, before inclusion in the proteomic analysis, phenotype difference between SC and NSC osteoblasts was checked using multiple biomarkers as already published.10,11
Patient Characteristics.
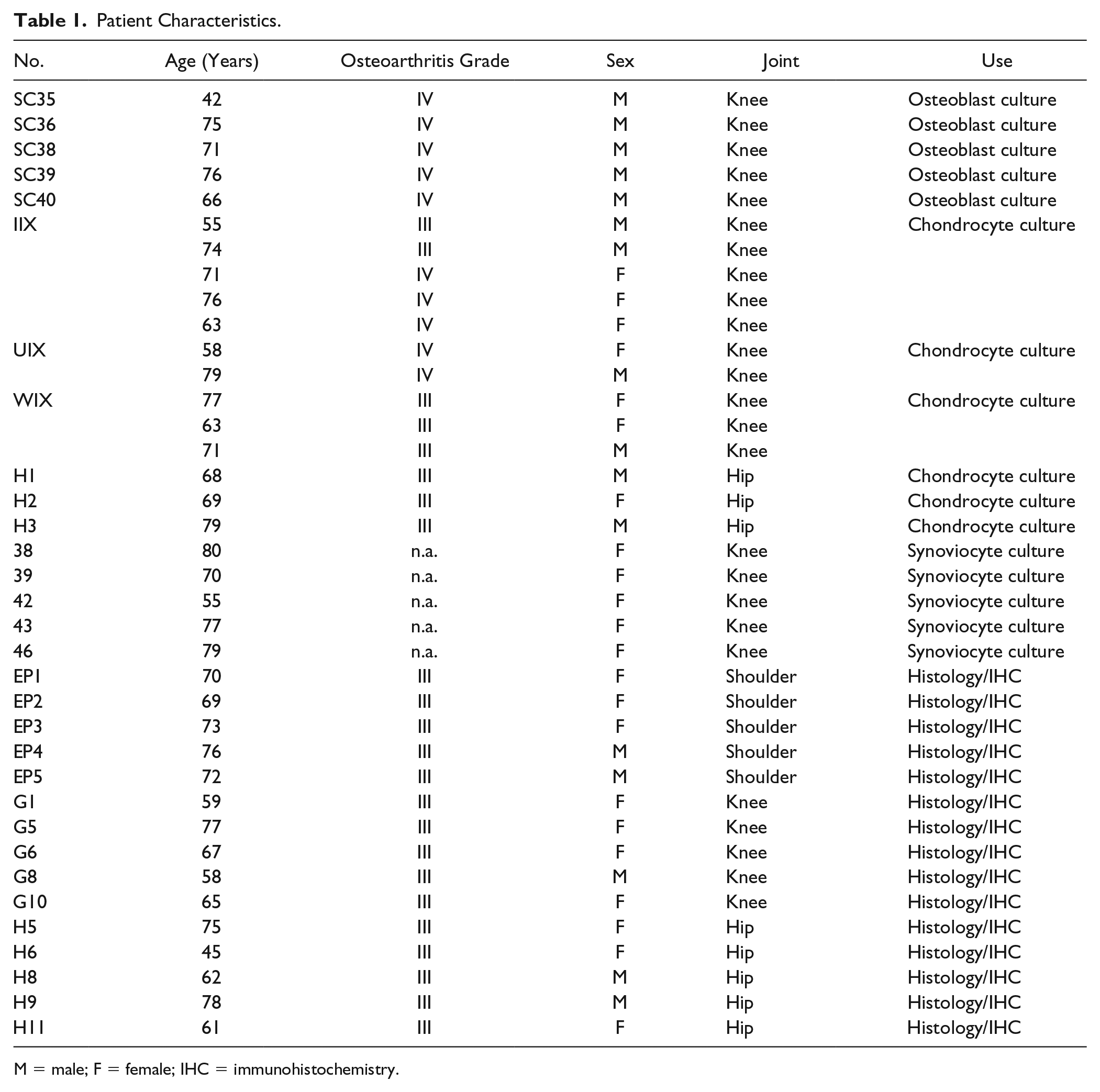
M = male; F = female; IHC = immunohistochemistry.
The osteoblasts were used at two states of differentiation: preosteoblast or mature osteoblasts. After this first 3 days in 10% FBS media, they were considered as preosteoblasts, characterized by low alkaline phosphatase and no alizarin red staining (mineralization). After 2 additional weeks of culture in a culture media composed of DMEM containing 100 units/mL penicillin, 100 µg/mL streptomycin, 10 mM HEPES, 2% Ultroser G (a serum substitute, Pall, France), 2 mM glutamine, 10−8 M 1,25-dihydroxyvitamin D3 (Sigma-Aldrich, Bornem, Belgium), 50 µg/mL ascorbic acid (Sigma-Aldrich), and 20 µg/ml proline (Invitrogen, Merelbeke, Belgium), they are considered as mature osteoblasts. 10 Mature osteoblasts had a high alkaline phosphatase level and mineralization ability while preosteoblasts did not express alkaline phosphatase and did not mineralize extracellular matrix. Cells were rinsed and then cultured in a bovine serum albumin (BSA)/FBS-free medium for 72 hours. The nutrient media used was DMEM supplemented with 1% ITS (Lonza, Belgium), 10 mM HEPES, 100 U/mL penicillin, 100 µg/mL streptomycin, 2 mM glutamine (Lonza, Belgium), 50 µg/mL ascorbic acid (Sigma-Aldrich, Belgium), 20 µg/mL proline (Invitrogen, Belgium). ITS is a premixed cell growth system containing in one ml: 0.625 mg insulin, 0.625 mg transferrin, 0.625 µg selenious acid. These conditioned 72-hour supernatants were used to perform the secretome analysis. 16 Cells were collected and RNA extracted for reverse transcription–polymerase chain reaction (RT-PCR) analysis.
Chondrocyte Culture in Alginate Beads
Alginate beads were prepared as previously described. 17 Each culture was realized using a pool of chondrocytes coming from 1 to 5 different OA patients (age between 55 and 79 years). Patient characteristics are listed in Table 1 . Experiment has been reproduced 3 times with different cartilage samples, for each joint location (hip or knee).
Chondrocytes in alginate beads were cultured for 28 days in DMEM supplemented with 100 U/mL penicillin–100 µg/mL streptomycin solution, 10 mM HEPES, 20 µg/mL proline, 50 µg/mL ascorbic acid, 2 mM glutamine, and 10% FBS. Chondrocyte culture media were changed twice a week. The first 72-hour conditioned supernatants (day 0 to 3) were used to perform the secretome analysis of nonhypertrophic chondrocytes, and the last 72-hour conditioned supernatants (day 25 to 28) were used to perform the secretome analysis of hypertrophic chondrocytes. Hypertrophic phenotype was determined using a panel of biomarkers (alkaline phosphatse, NTPPPH, ColX, MMP-13, RUNX2) as previously described. 14 Cell pellet used for enzyme-linked immunosorbent assay (ELISA) and RT-PCR were collected at day 3 (nonhypertrophic) and at day 28 (hyperptrophic). 14
Isolation and Culture of Synovial Cells
Synovial tissue samples from 5 patients with knee OA (mean age 70 years) were obtained at the time of total knee joint replacement. Patient characteristics are listed in Table 1 . All subjects provided written informed consent, and ethical approval (Ethics Committee Agreement of Catholic University of Louvain, no. B403201111664) was granted for this study. The inflammation status of the synovial membrane was characterized by the surgeon according to macroscopic criteria established by Ayral et al. 18 These criteria included synovial vascularization, villi formation, and hypertrophic aspect of the tissue and allowed the classification of the synovial membrane into 3 grades: normal, reactive, and inflamed. For the purposes of this study, synovial samples were grouped as normal/reactive or inflamed.
Normal/reactive and inflamed synovial biopsy samples were separately digested as previously described
13
with type IA collagenase from Clostridium histolyticum (1 mg/mL; Sigma-Aldrich) in complete medium, consisting of DMEM supplemented with 10 mM HEPES, 100 U/mL penicillin, 100 μg/mL streptomycin, 2 mM-
Quantitative Real-Time RT-PCR
RNA from 1 × 106 cells was isolated by RNeasy mini kit total RNA isolation system (Qiagen, Belgium) and PCR was performed by using the Rotor Gene (Qiagen, Belgium)- SYBR premix Ex Taq (Takara, Belgium). The PCR template source was either 3 ng first-strand cDNA or purified DNA standard. PCR program comprised an initial denaturation step at 95°C for 10 seconds followed by 40 cycles of denaturation at 95°C for 5 seconds and annealing/extension at 60°C for 25 seconds and an ending melting step from 65°C to 96°C with a 1°C increase each second. The following primer sequences were used to amplify the desired cDNA: hypoxanthine-guanine phosphoribosyltransferase (HPRT) forward 5′-TGTAATGACCAGTCAACAGGG-3′ and reverse 5′-TGCCTGACCAAGGAAAGC-3′, syndecan-4 forward 5′- GAGCCCTACCAGACGATGAG-3′ and reverse 5′-GCACCAAGGGATGGACAACT-3′, and COL10A1 forward 5′-GGGAGTGCCATCATCG-3′ and reverse 5′-AGGGTGGGGTAGAGTT-3′. HPRT was used as an internal standard and the ratio of genes to HPRT were calculated. After HPRT normalization for each gene, relative expression was calculated.
Histology and Immunohistochemistry
Cartilage/bone biopsies were obtained from patients undergoing either total knee, hip, or shoulder joint replacement for OA (5 patients of each joint) and aged between 45 and 78 years. Patient characteristics are listed in Table 1 . For knee, biopsies were taken in the middle of the medial tibial plate and for hip at 4 cm of the foveal ligament insertion. For shoulder, biopsies where taken in the macroscopically damaged area. For each joint, the sample section extended horizontally from one edge of the joint to the other and in depth from the articular surface to below the articular bone plate, as recommended by Pritzker et al. 19
Samples were fixed in 4% paraformaldehyde, decalcified in DC2 (Labonord, Belgium) overnight, dehydrated, and wax embedded. Five micrometers thick tissue sections were cut with a microtome, and either safranin-O stained or processed for immunohistochemistry (IHC).
Syndecan-4 was immunolocalized in tissues using mouse monoclonal antibody (5G9, Santa Cruz Biotechnology Inc., Germany). A skin section was used as positive control. To set up the syndecan-4 IHC, we tried several demasking methods, including chondroitinase ABC, hyaluronidase, pepsin, tryspin, citrate, both with and without Triton X-100 treatment. We never observed staining in bone with these treatments. In cartilage, better staining was obtained with hyaluronidase pretreatment (3 hours, 37°C). So, we used this method with the 5 OA knees, hips, and shoulder. Briefly, tissues sections were deparaffinized in xylene and rehydrated in distilled water. Slides were then demasked with hyaluronidase (type I-S 1 mg/mL in phosphate buffered saline pH 6, Sigma) for 3 hours at 37°C. The blocking of endogenous peroxidase was performed with 3% H2O2 in water over 20 minutes and the nonspecific binding sites were blocked with normal goat serum (1:10, Jackson) supplemented with 1% BSA and 0.1% Tween 20 for 45 minutes. Anti-syndecan-4 antibody at dilution 1:50 was applied and incubated overnight at +4°C. A negative control with normal mouse IgG (1:50, DAKO) was included in the experiment. The tissue sections were then incubated with HRP (horseradish peroxidase) link (Envision+, DAKO) 40 minutes. Peroxidase was revealed by the 3,3′-diaminobenzidine tetrahydrochloride reaction (DAKO). Sections were finally counterstained with hematoxylin, dehydrated and mounted. Pictures were taken with Leica DM1000 LED microscope.
Syndecan-4 ELISA
Syndecan-4 was quantified into culture supernatant and cell pellet by specific sandwich ELISA against the ectodomain of syndecan-4 (IBL International, Hamburg, Germany), according to manufactory instructions (dilution 1/4).
Statistical Analysis
For gene expression and protein levels obtained by immunoassays, the results (mean ± SD) were expressed as HPRT (hypoxanthine phosphoribosyltransferase)-normalized gene expression or as concentration of protein per microgram of DNA. A paired t test was performed for each experiment (GraphPad Prism 6.0).
Results
Histology and Immunohistochemistry
Histological observation of OA knee, hip, and shoulder subchondral bone and cartilage stained with safranin-O allow the observation of different features according to the localization ( Fig. 1A , C , and E ). Beside the proteoglycan loss, important fibrillations were observed in knees while “flamed” were observed in hip and shoulder cartilage ( Fig. 1C-E ). Chondrocytes clusters were seen in superficial fibrillated layer in knee, and larger clusters were located in deep or middle layers in hip and shoulder.
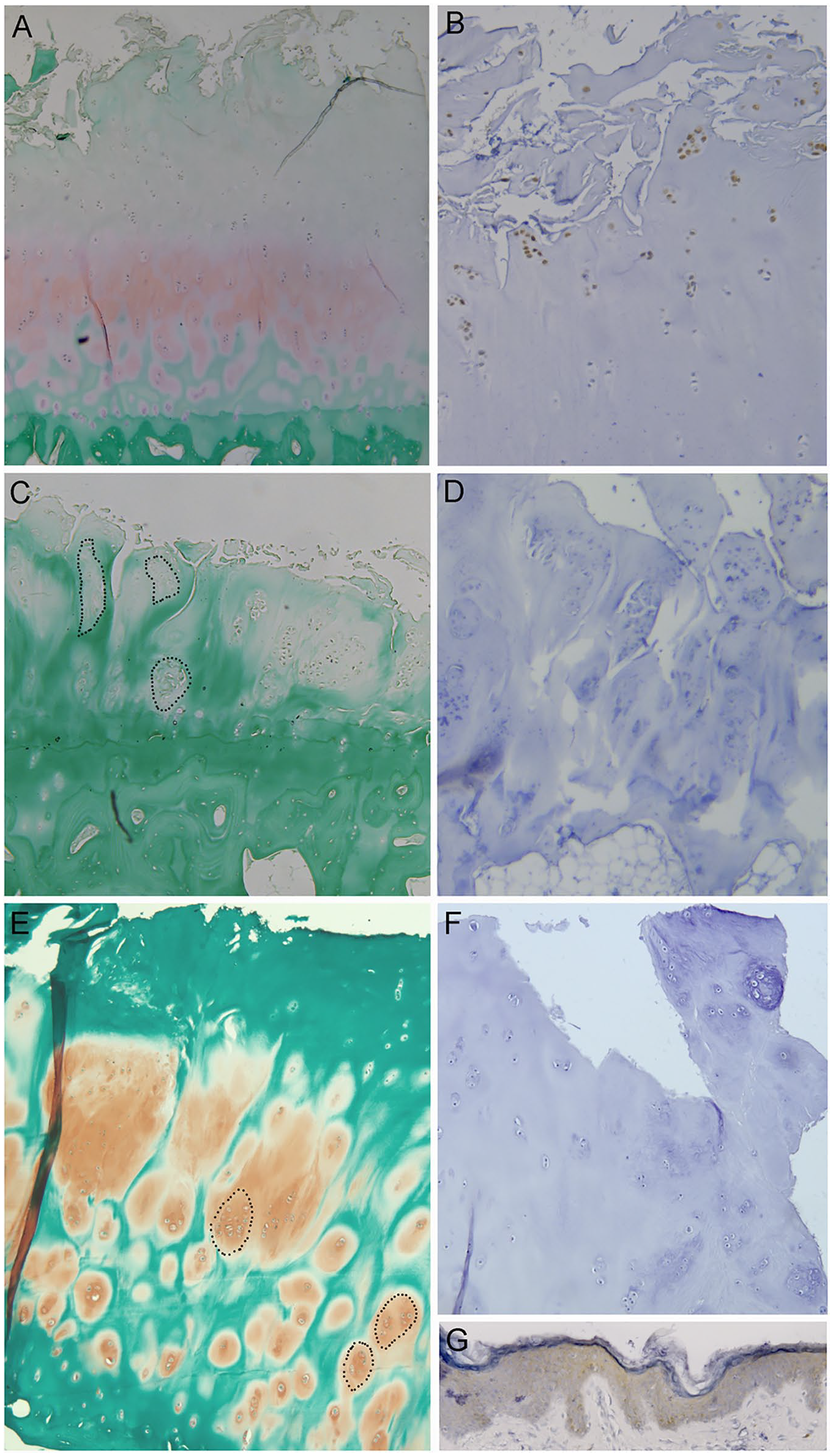
Safranin-O fast green and syndecan-4 immunohistochemistry (IHC) in knee osteoarthritis (OA) (
We observed a strong syndecan-4 staining in OA knees chondrocyte clusters located in the surface layer around the fibrillations ( Fig. 1B ). In contrast, no staining was detected in hip and shoulder OA ( Fig. 1D and F ).
Syndecan-4 Gene Expression in Osteoblasts, Synoviocytes, and Chondrocytes
NSC preosteoblasts expressed 0.1370 ± 0.0165 and mature NSC osteoblasts expressed 0.1692 ± 0.0385 syndecan-4 copy per HPRT copy. No significant difference between preosteoblasts and mature osteoblasts or between SC and NCS osteoblasts was observed ( Fig. 2 ).
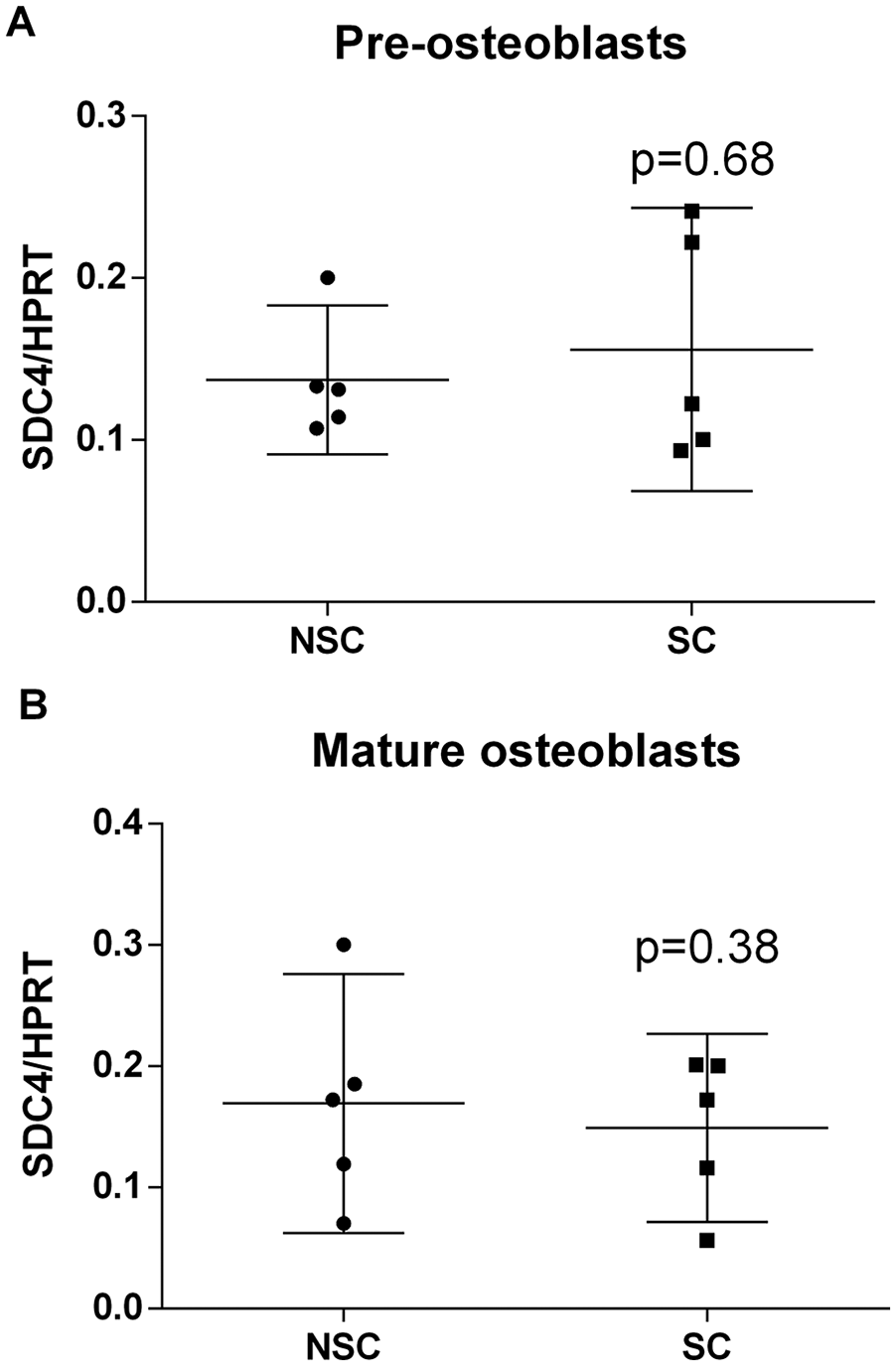
Syndecan-4 mRNA levels in (
Inflamed synoviocytes expressed a higher level of syndecan-4 mRNA (0.36 ± 0.07, Fig. 3 ) than normal/reactive synoviocytes expressed 0.25 ± 0.02 syndecan-4 copy per HPRT copy, but no significant difference between group was observed.
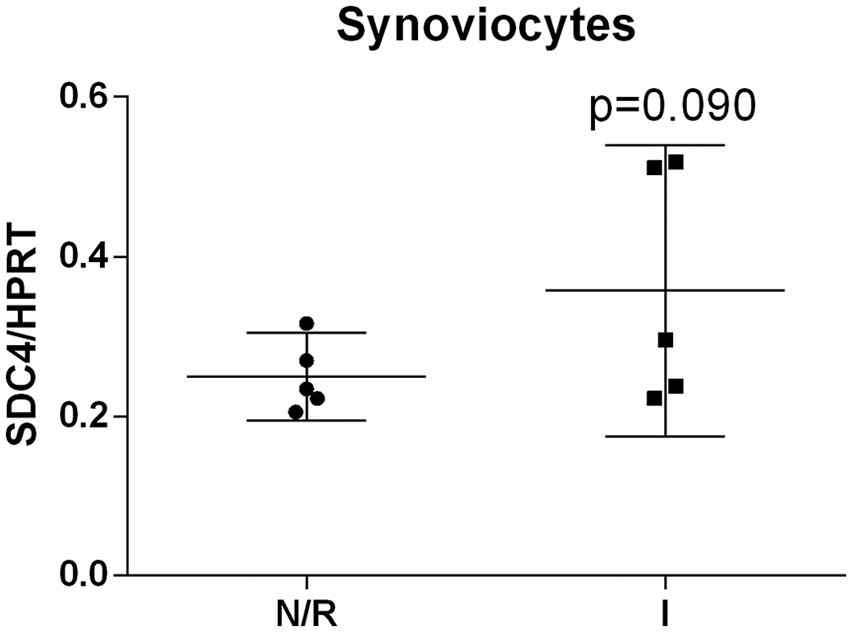
Syndecan-4 mRNA levels in normal/reactive (N/R) and inflamed (I) synoviocytes. Syndecan-4 (SDC4) was normalized with HPRT expression. Data are means ± 95% CI from 5 patients. Mean values comparisons of were performed by paired t test.
In alginate beads and in 10% FBS culture medium, we have already demonstrated that knee OA chondrocytes become hypertrophic after 28 days of culture. 14 We have compared expression of type X collagen (COL10A1) and syndecan-4 in nonhypertrophic chondrocytes (day 3) or hypertrophic chondrocytes (day 28). COL10A1 increased in hypertrophic chondrocytes in both knee and hip chondrocytes (6.4-fold, P = 0.0671 for hip and 3.7-fold, P = 0.0028 for knee, Fig. 4A ). No difference between hip and knee chondrocytes was observed. In contrast, we observed a significant increase of syndecan-4 expression with hypertrophy (3.5-fold, P = 0.0102, Fig. 4B ) in knee, but not in hip chondrocytes.
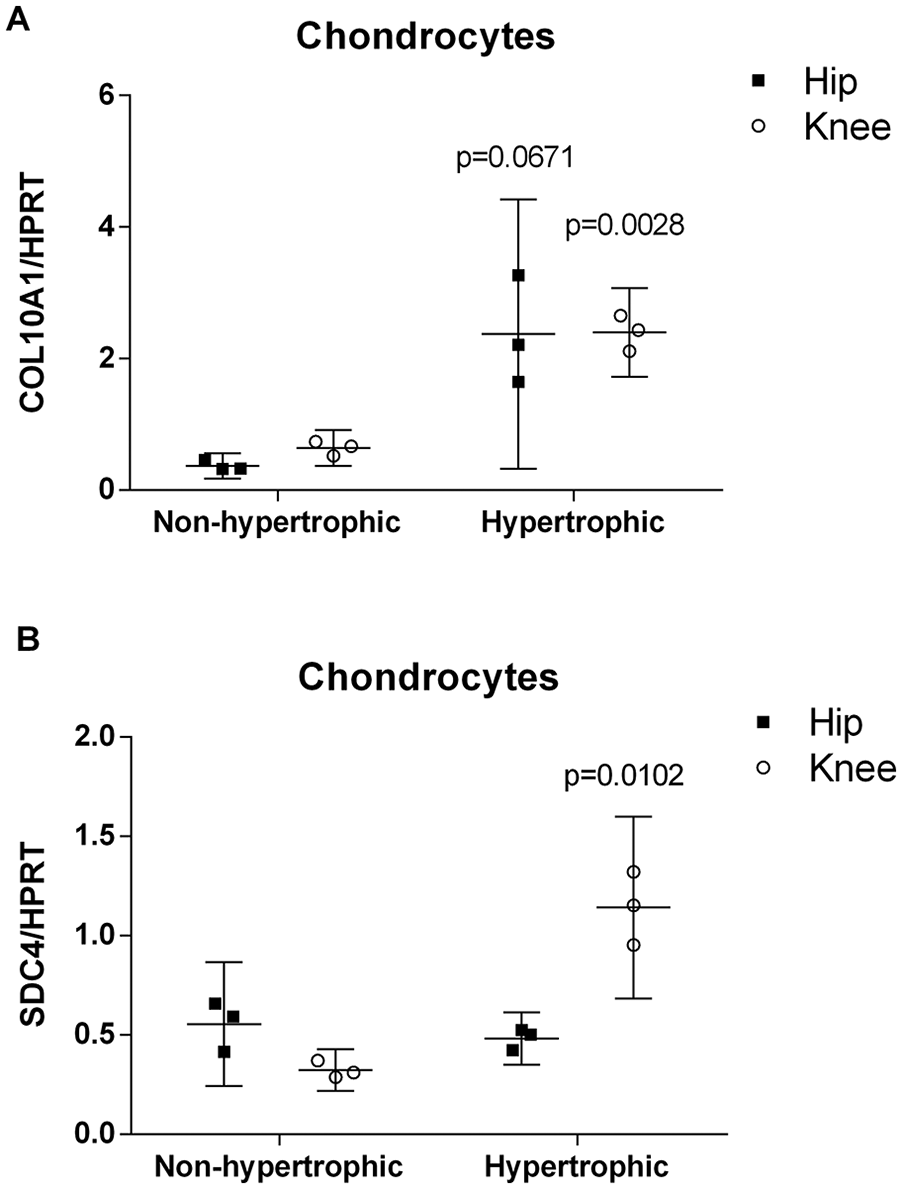
COL10A1 (
Syndecan-4 Production Quantified by Immunoassay
Using an immunoassay specifically measuring the syndecan-4 ectodomain, we have demonstrated that a large part of the ectodomain was found in culture supernatants, indicating that most of the syndecan-4 ectodomain were released. Indeed, the mean rate of syndecan-4 ectodomain was 68% ± 9% in the culture supernatants. This ratio did not vary according to joint localization or hypertrophic chondrocyte differentiation. Total (supernatant plus cells fraction) syndecan-4 level was increased by 2.8-fold in knee hypertrophic chondrocytes culture (P = 0.0014, Fig. 5 ), compared to nonhypertrophic knee chondrocyte while no difference was observed between nonhypertrophic and hypertrophic hip chondrocytes.
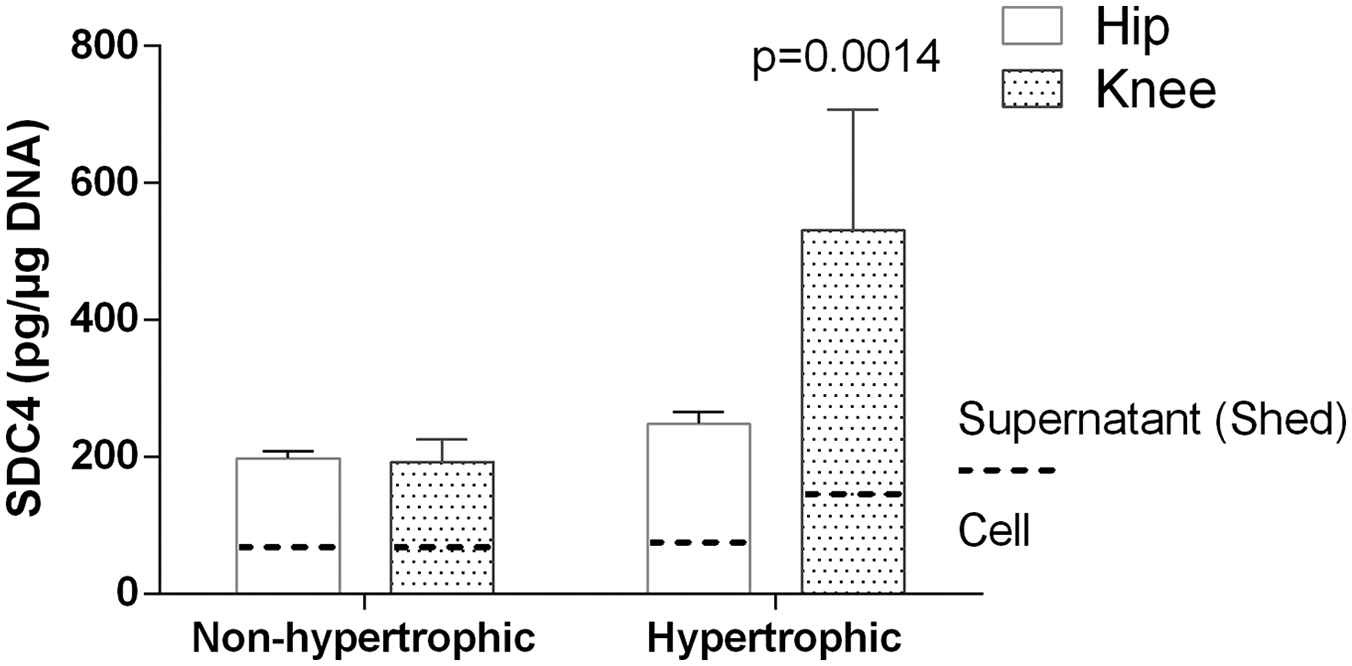
Syndecan-4 found in ELISA in cell pellet and culture supernatant of nonhypertrophic and hypertrophic chondrocytes culture from osteoarthritis (OA) hip or knee. Syndecan-4 was normalized with DNA. Data are means ± 95% CI from 3 patients. Mean values comparisons were performed by paired t test.
Discussion
So far, little information is available regarding syndecan-4 distribution in joint tissue, its expression according joint localization or the influence of OA condition on its expression. Primarily, in vitro or animal in vivo OA studies focused on knee or hip joint, but mainly on knee. This article gives important information on the role of syndecan-4 in OA physiopathology. First, we demonstrated that syndecan-4 is detectable in chondrocytes clusters located in the superficial layer of knee cartilage but not in hip or shoulder OA cartilage. Secondary, we confirm that syndecan-4 is overexpressed by hypertrophic cartilage compared with OA nonhypertrophic chondrocytes. In contrast, syndecan-4 is not influenced by synovium inflammatory condition or subchondral bone sclerosis. More precisely, synoviocytes coming from the inflammatory zone of the synovial membrane do not overpressed syndecan-4 compared with synoviocytes coming from non-inflammatory zone. This result is consistent with a previous study in where syndecan-4 was not found in chronically inflamed synovium. 20 In the same way, osteoblasts coming from the SC area of subchondral bone do not overexpressed syndecan-4 compared with neighboring osteoblasts of the NSC area. This is the first time that syndecan-4 is study on primary cells coming from different joint tissues and living in different pathological conditions. This study opens some interesting perspectives.
These findings are relatively unexpected and suggest that the hypertrophic differentiation involves syndecan-4 in knee but not hip and shoulder. Indeed, chondrocytes clusters are observed in knee, hip and shoulder cartilage, even if the histological aspect of the OA articular cartilage differ from joint to joint (fibrillations/fissures, flamed aspect). Risk factors for OA differ across different joints. Whereas knee OA is associated with meniscectomy, obesity, muscle weakness, and major injury, hip OA is commonly associated with congenital and developmental defects such as acetabular dysplasia, obesity, and abnormal loading. 21 Although knee and hip OA risk factors suggest biomechanical etiopathogenesis, this histological difference between OA joints could be explained by the mechanical conditions that are different in shoulder, hip, and knee. Knees have to support high mechanical load and OA lesions appear first in the load-bearing region. On the contrary, in hip OA, cartilage degeneration initially occurs in the fovea below the femoral head and in medial/ lateral areas of the femoral head- areas with low exposure to little loading or minimal contact. Once this occurs, the contact area and motion increase frictional shear stress that facilitate OA onset 22 . This important difference emphasizes the variance in the OA process occurring in these 2 joints.
As syndecan-4 is known to play a role in mechanotransduction23,24 and is a mechanosensitive gene, 25 we can suspect that the presence of syndecan-4 may determine the OA physiopathological pathway. As mechanical forces also influence the homeostasis of cartilage by regulating expression and activities of MMP-3 and ADAMTS-5,26-28 syndecan-4 is a mechanotransducer candidate involved in this process, responsible for the degradation of extracellular matrix. If it is the case, this function is probably related to the transmembrane and not the shed form of the syndecan-4.
Interestingly, the soluble ectodomain released during the shedding process regulates growth factor activity and proteolytic balance, meaning that this part of syndecan-4 is an actor of the OA physiopathology. 29 This domain triggers inflammation, chemoattraction, and fibrosis in different tissues, 30 but its role in cartilage degradation remains poorly documented. Shedding of the syndecan-4 occurs constitutively and is accelerated under certain conditions, such as acute inflammation, trauma, or mechanical stress.29,31-33 A variety of chemokines and cytokines in acute inflammation can regulate the shedding process of syndecan-4. Recently, it was shown that advanced glycation end-products induced syndecan-4 shedding in a dose- and time-dependent manner.34,35 Several enzymes are involved in the shedding, mainly plasmin, thrombin, MMP-2, -9, -14, ADAMTS1, and ADAM17, cleaving the ectodomain at different sites 36 . In this study, we shown for the first time that the rate of shedding of syndecan-4 in articular chondrocytes is around 68% and do not vary according the hypertrophy state of the chondrocytes. Therefore, syndecan-4 is mostly present in the shed configuration in joint. It would be interesting to perform an epitope analysis of the syndecan-4 in the synovial fluid to know which proteases are involved syndecan-4 shedding in the OA context, and how this shedding is involved in the pathology. Indeed, knockout and neutralization studies have demonstrated 6 that syndecan-4 show a deleterious role, through ADAMTS-5 activation and MMP-3 production, but it is still unknown if this is the result of the shed or the unshed activity of the syndecan-4.
One limitation of our study is that we compared joints from different patients. Interesting would be to compare syndecan-4 expression in hip, knee, and shoulder coming from the same patients, to verify that changes in syndecan-4 expression is joint dependent and not patient dependent. Another important limitation is that synovial tissue, bone, and cartilage come from different patients and not from the same patient. The reason is that our study is a retrospective study including tissue samples coming from our biobank. Based on these encouraging results, a prospective study comparing syndecan-4 expression in all joint tissues from the same joint and patient could be planned.
Conclusions
We highlighted some joint-to-joint discrepancy in the presence of both the shed and the transmembrane syndecan-4 during OA. Syndecan-4 is overproduced by hypertrophic knee articular chondrocytes, which could lead to a distinct function of this important regulator of cell-matrix interaction proteoglycan. Study of the precise function of shed vs unshed syndecan-4 in cartilage remain to be investigate.
Footnotes
Acknowledgments and Funding
The authors want to thank François Sevrin and Laurence Pesesse for their technical help, and Dr Delcour for the biopsies. Christelle Sanchez, Jessica Bertrand, Thomas Pap, and Yves Henrotin are members of the D-BOARD European Consortium for Biomarker Discovery European Union’s Seventh Framework Program grant agreement no. 305815 (
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All tissues used in this study were obtained after approval by the Faculty of Medicine Ethics Committee of the University of Liège (number B70720108313, reference 2010/43) and ethical approval was granted by the Ethics Committee Agreement of Catholic University of Louvain (No. B403201111664).
Informed Consent
Written informed consent was obtained from all subjects before the study.
Trial Registration
Not applicable.
