Abstract
Objective
Given the potential applications of combined biologics, the authors sought to evaluate the in vitro effect of combined platelet-rich plasma (PRP) and hyaluronic acid (HA) on cellular metabolism.
Design
Bone marrow–derived mesenchymal stem cells (BMSCs) and chondrocytes were obtained from the femurs of Sprague-Dawley rats. An inflammatory model was created by adding 10 ng/mL interleukin-1-beta to culture media. Non-crosslinked high-molecular-weight HA, activated-PRP (aPRP), and unactivated-PRP (uPRP) were tested. Cellular proliferation and gene expression were measured at 1 week. Genes of interest included aggrecan, matrix metalloproteinase (MMP)-9, and MMP-13.
Results
Combined uPRP-HA was associated with a significant increase in chondrocyte and BMSC proliferation at numerous preparations. There was a trend of increased chondrocyte aggrecan expression with combined PRP-HA. The greatest and only significant decrease in BMSC MMP-9 expression was observed with combined PRP-HA. While a significant reduction of BMSC MMP-13 expression was seen with PRP and HA-alone, a greater reduction was observed with PRP-HA. MMP-9 chondrocyte expression was significantly reduced in cells treated with PRP-HA. PRP-alone and HA-alone at identical concentrations did not result in a significant reduction. The greatest reduction of MMP-13 chondrocyte expression was observed in chondrocytes plus combined PRP-HA.
Conclusions
We demonstrated a statistically significant increase in BMSC and chondrocyte proliferation and decreased expression of catabolic enzymes with combined PRP-HA. These results demonstrate the additive in vitro effect of combined PRP-HA to stimulate cellular growth, restore components of the articular extracellular matrix, and reduce inflammation.
Introduction
Articular cartilage pathology represents a clinical challenge for orthopedic surgeons due to its inherent avascularity and poor potential for spontaneous healing.1-3 As a result, research to improve cartilage homeostasis has focused on biologic adjuncts, such as platelet-rich plasma (PRP) and hyaluronic acid (HA). 4
In vitro studies show that PRP enhances the proliferation and synthetic capability of bone marrow–derived mesenchymal stem cells (BMSCs) and chondrocytes.5-8 PRP administration may accelerate the formation of cartilage repair tissue and/or improve repair tissue quality toward native (hyaline) cartilage tissue. 5 The catabolic intraarticular environment existing in the presence of pathologic processes of cartilage may also be influenced by the administration of PRP. The presence of catabolic cytokines, such as interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF-α), increases the activity and levels of matrix metalloproteinases (MMP) and elastases by both synovial cells and chondrocytes. These enzymes breakdown the articular extracellular matrix (ECM) and can cause progression of osteoarthritis (OA) and inhibit the production of regenerative tissue during cartilage repair. 9 Previous studies have demonstrated that PRP inhibits the catabolic effects of inflammatory cytokines on chondrocytes while enhancing chondrogenesis and ECM synthesis.8,10,11 In diseases of chronic articular inflammation, the concentration and average molecular weight of HA have been shown to decrease significantly, resulting in decreased synovial fluid viscosity 12 and, possibly, cartilage disruption. 13 The therapeutic effects of intraarticular HA supplementation are thought to be mediated by increased lubrication of joint synovial fluid, a protective effect against the cytotoxicity of reactive oxygen species and an anti-inflammatory effect.14,15 However, HA-alone has been shown to have limited efficacy in OA patients and may require adjunctive agents to enhance its efficacy. 16
Evidence suggests that PRP and HA act to modulate the OA environment in similar ways but by different mechanisms. As a result, the combination of PRP and HA might be a better clinical option for the treatment of OA than either PRP or HA alone. 17 Prior in vitro studies have demonstrated the synergistic anti-inflammatory and chondrogenic effects of combined PRP-HA. 16 The authors concluded that the synergistic effect was the result of different molecular mechanisms. Furthermore, a more recent basic science study demonstrated that HA increased the release of growth factors from platelets in PRP. 18 As such, there are numerous potential molecular and cellular mechanisms by which PRP and HA can act in concert. These studies offer scientific rationale for the clinical findings (albeit limited) that support the use of combined PRP-HA to OA. Clinical evidence supporting the combined administration of PRP-HA for knee OA is limited. Lana et al. published a randomized clinical trial comparing PRP-HA to PRP-alone and HA-alone for the treatment of mild to moderate knee OA. In their study, combined PRP-HA had better outcomes than HA-alone at 1 year and PRP-alone at 3 months. Their results also suggested that combined PRP-HA would also provide better functional outcomes in the first 30 days posttreatment. 19
Given the potential clinical application of combined biologics, the authors sought to evaluate the in vitro effect of combined PRP and HA on BMSC and chondrocyte metabolism. We hypothesized that combined administration of PRP and HA to culture media would increase cellular proliferation, reduce the expression of inflammatory cytokines, and increase aggrecan expression.
Materials and Methods
BMSCs and chondrocytes were obtained from the femurs of 12-week-old Sprague-Dawley rats (SDRs) using a standardized protocol.
BMSCs were isolated from the femurs of 6 adult male SDRs by needle aspiration of bone marrow and plating of resultant cell fraction in standard BMSC culture medium: low glucose Dulbecco’s modified Eagle’s Medium (DMEM), 10% (v/v) fetal bovine serum (FBS; Crystalgen), 1% (v/v) penicillin/streptomycin/amphotericin (PSA; Corning).
Chondrocytes were isolated from femoral cartilage of 6 adult male SDRs. The harvested cartilage was washed 4 times in phosphate-buffered saline (PBS) containing 2% PSA (penicillin/streptomycin/amphotericin), with 5 minutes in solution each time, and then placed into 0.05% hyaluronidase in PBS for 10 minutes. Cartilage was then minced into fine pieces (<1 mm3), placed in PBS, and centrifuged at 24°C and 1,000 rpm for 1 minute. Following discarding of PBS and hyaluronidase, tissue was placed in 0.2% trypsin solution without ethylenediaminetetraacetic acid (EDTA; Gibco) and digested while being agitated in a 37°C water bath for 10 minutes. Following discarding of trypsin, cartilage tissue was digested in 0.2% type II collagenase solution while being agitated in a 37°C water bath for 90 minutes, then centrifuged at 24°C and 1000 rpm for 10 and 5 minutes, with a standard chondrocyte culture medium wash (DMEM with Ham’s F-12 [DMEM/F-12], 10% FBS, 1% PSA).
Cells were cultured in vitro in T150 cell culture flasks in standard culture media at 37°C, 21% O2, and 5% CO2 with media change every 3 days to obtain BMSCs and chondrocytes after the first passage. Cells were passaged at 95% confluency after a PBS wash and detachment with 0.05% trypsin-EDTA (Gibco) and cultured to the fifth passage for experimentation.
While a homogenous mixture of PRP and HA can be prepared for intraarticular injection via proprietary products, the concentrations required for this assay necessitated separate preparation. HA was supplied in 2 mL vials at a concentration of 20 mg/mL (Regen Lab SA, Le Mont-sur-Lausanne, Switzerland). The HA gel was contained in a phosphate buffer in a non-crosslinked high-molecular-weight (1,550 kDa) preparation. 20
PRP was prepared according to manufacturer protocol (Regen Lab SA). Blood for PRP preparation was obtained via direct cardiac puncture from 12-week-old SDRs. Tubes were spun for 5 minutes at centrifugal force of 1,500g. This produced 4 to 5 mL of autologous PRP with a platelet concentration factor of 1.6-fold and concurrent reduction in white blood cells (>85% depletion) and red blood cells (>99.7% depletion). 21 To create activated-PRP (aPRP), 100 nM CaCl2 was added to PRP samples prior to plating. Unactivated-PRP (uPRP) was combined with 0.2% heparin solution per manufacturer protocol prior to plating.
BMSCs and chondrocytes were plated at a standard concentration (3.6 × 104 cells/well) in 24-well plates. Negative control samples were cultured with media alone. PRP was added to experimental media in multiple concentrations: 1%, 5%, 10%, 20%. HA was added to media in multiple concentrations: 0.25 mg, 0.5 mg, 0.75 mg, 1.0 mg. Combined PRP-HA samples consisted of 1% PRP with 0.25 mg or 0.5 mg of HA and 5% PRP with 0.25 mg or 0.5 mg HA. All control and experimental samples were prepared in quadruplicate.
IL-1β was added to cell cultures at a concentration of 10 ng/mL to simulate an osteoarthritic environment. Prior studies have shown that adding IL-1β to cell cultures is a useful model to reproduce an arthritic environment.10,22
Media was changed twice during the experimental period: day 3 and day 6.
Cellular proliferation was measured using a MTS formazan assay (Promega). 23 Chondrocyte and BMSC gene expression was measured using quantitative real-time polymerase chain reaction (qRT-PCR). RNA extraction was completed with a High Pure RNA isolation kit (Roche). qRT-PCR analysis of gene expression found in chondrocytes and BMSCs was completed with primers for MMP-9 and MMP-13. qRT-PCR analysis of gene expression found in chondrocytes was completed with primers for aggrecan. Cellular proliferation and gene expression were compared to negative control samples. Negative control values were obtained for each 24-well plate to account for potential minor variations in growth conditions.
Blood and cells were obtained from SDRs through Institutional Animal Care and Use Committee approved protocols as part of their scheduled necropsy.
Statistical Analysis
Statistical analysis was conducted using Stata 13.1 (College Station, Texas). The Mann-Whitney U nonparametric test was used to compare the effect of the various preparations (HA-alone, PRP-alone, combined PRP-HA) to media-alone.
Results
BMSC Proliferation
uPRP-alone at 1% (P < 0.001), 5% (P < 0.001), 10% (P < 0.001), and 20% (P < 0.01) was associated with a significant increase in BMSC proliferation ( Fig. 1A ). aPRP-alone at 5% (P < 0.01) and 10% (P < 0.001) was associated with a significant increase in BMSC proliferation ( Fig. 1B ). aPRP-HA ( Fig. 1D ) and HA-alone ( Fig. 1E ), at any concentration, was not associated with a significant increase in BMSC proliferation (P > 0.05). Combined uPRP-HA ( Fig. 1C ) was associated with a significant increase in BMSC proliferation with 5% uPRP + 0.25 mg HA (P < 0.0001) or 0.5 mg HA (P < 0.0001).
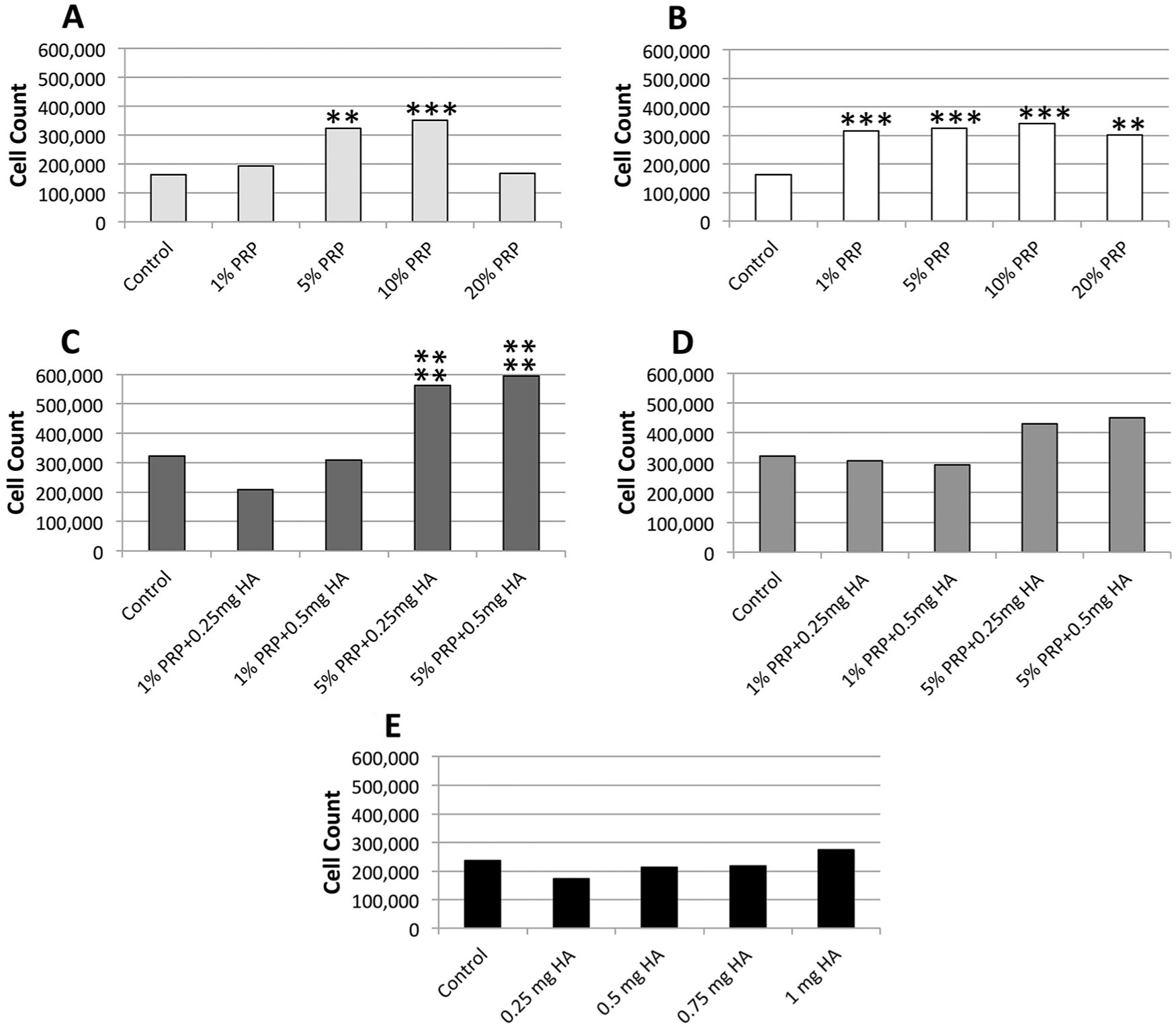
BMSC proliferation at 1 week. The addition of both uPRP-alone (
Chondrocyte Proliferation
uPRP-alone was associated with a significant increase in chondrocyte proliferation at 1% (P < 0.01), 5% (P < 0.001), and 10% (P < 0.01) ( Fig. 2A ). aPRP-alone was associated with a significant increase in chondrocyte proliferation at 1% (P < 0.01), 5% (P < 0.05), and 10% (P < 0.05) ( Fig. 2B ). HA-alone was associated with a significant increase in chondrocyte proliferation at 0.5 mg (P < 0.05) ( Fig. 2E ). Combined uPRP-HA resulted in significantly increased chondrocyte proliferation with 1% PRP + 0.25 mg HA (P < 0.01) or 0.5 mg HA (P < 0.01) and 5% PRP + 0.25 mg HA (P < 0.05) ( Fig. 2C ). aPRP + HA was associated with a significant increase (P < 0.05) in chondrocyte proliferation at 1% PRP + 0.25 mg HA ( Fig. 2D ).
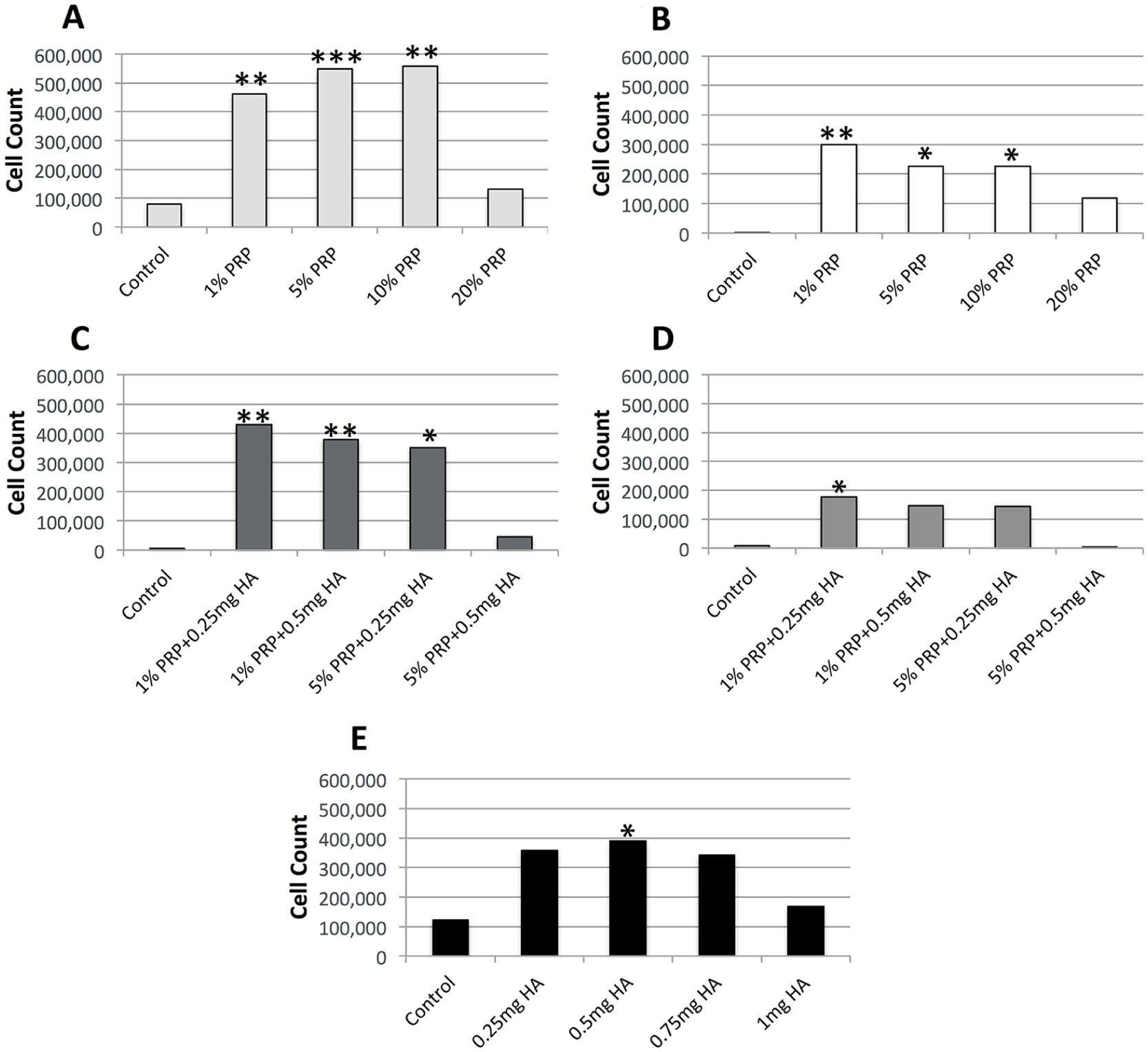
Chondrocyte proliferation at 1 week. Combined uPRP-HA (
Aggrecan Gene Expression
There was a trend of increased aggrecan expression with combined PRP-HA ( Fig. 3 ). The largest increase in expression was observed at 20% uPRP and 5% uPRP + 0.5 mg HA. Aggrecan expression increased with greater concentrations of uPRP.
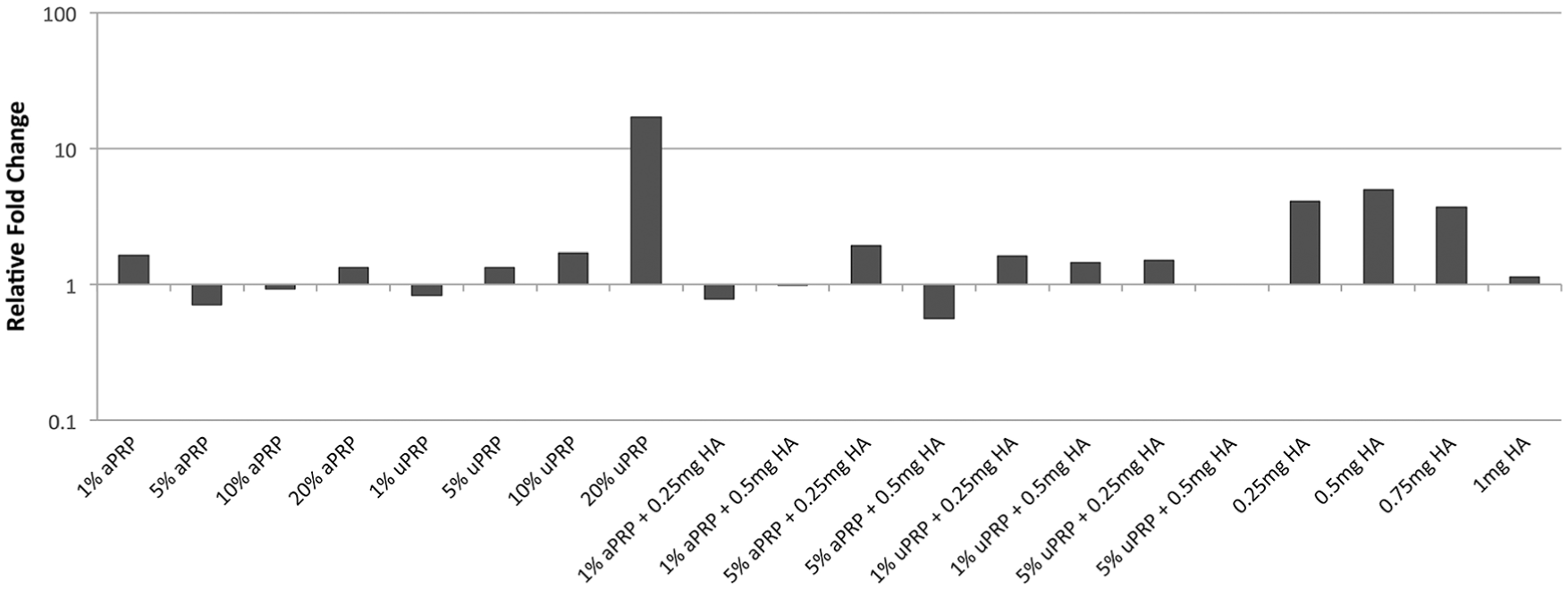
Chondrocyte aggrecan gene expression at 1 week. There was a trend of increased aggrecan expression in chondrocytes cultured in the presence of combined PRP-HA.
Inflammatory Model and MMP Expression
The greatest and only significant (P < 0.001) decrease in BMSC MMP-9 expression was observed with 5% uPRP + 0.5 mg HA ( Fig. 4 ). Similar results were observed with BMSC MMP-13 expression ( Fig. 4 ). While a significant reduction of BMSC MMP-13 expression was seen with 1% aPRP (P < 0.05) and 1.0 mg HA (P < 0.01), a greater and more significant reduction was observed with combined 5% uPRP + 0.5 mg HA (P < 0.0001), 1% aPRP + 0.5 mg HA (P < 0.01), and 5% aPRP + 0.5 mg HA. Chondrocyte MMP-9 expression was significantly reduced in cells treated with various combinations of PRP-HA ( Fig. 5 ): 1% uPRP + 0.25 mg HA (P < 0.0001), 5% uPRP + 0.5 mg HA (P < 0.0001), and 5% aPRP + HA (P < 0.0001). PRP-alone and HA-alone at these concentrations did not result in a significant reduction. All samples tested resulted in a significant reduction in chondrocyte MMP-13 expression ( Fig. 5 ). The greatest reduction was observed in cells treated with combined PRP-HA.
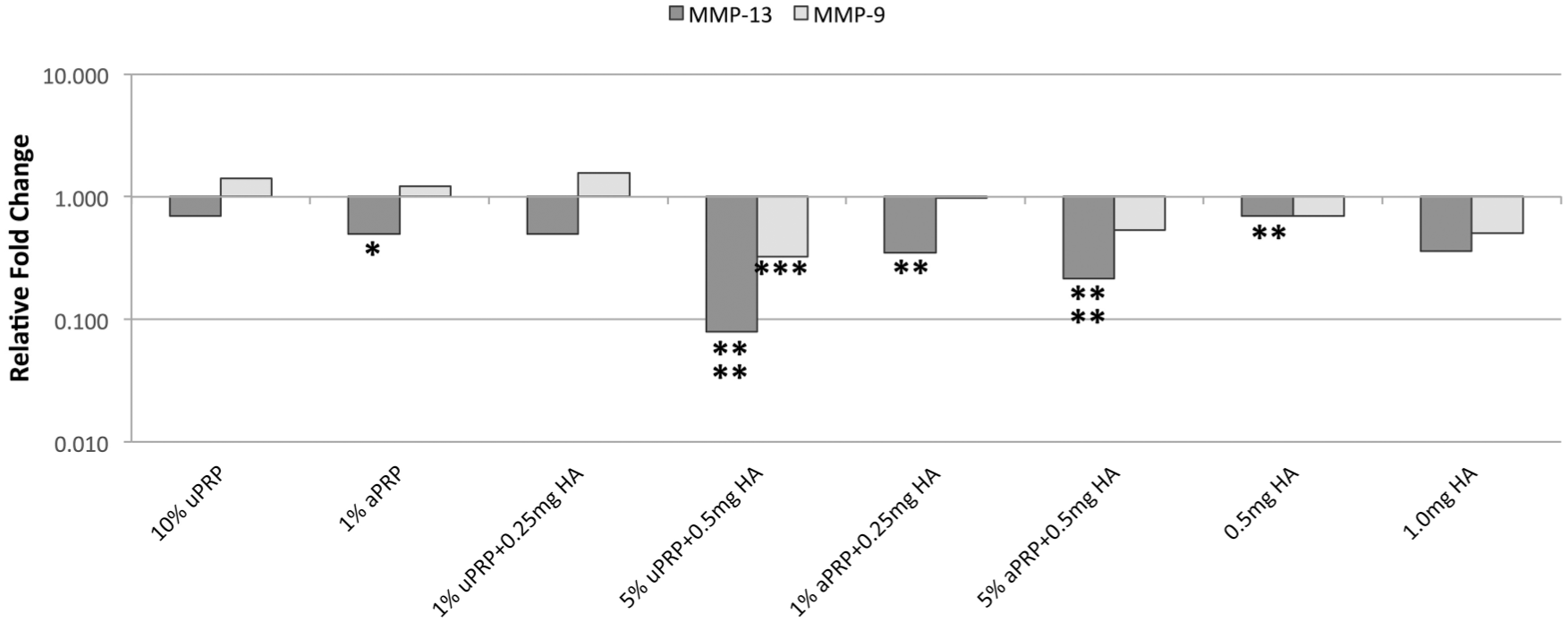
BMSC MMP-9 and MMP-13 gene expression at 1 week. The greatest and only significant reduction in MMP-9 expression occurred in BMSCs cultures with combined PRP-HA. The greatest and most significant reduction in BMSC MMP-13 expression was observed in cells cultured with combined PRP-HA. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
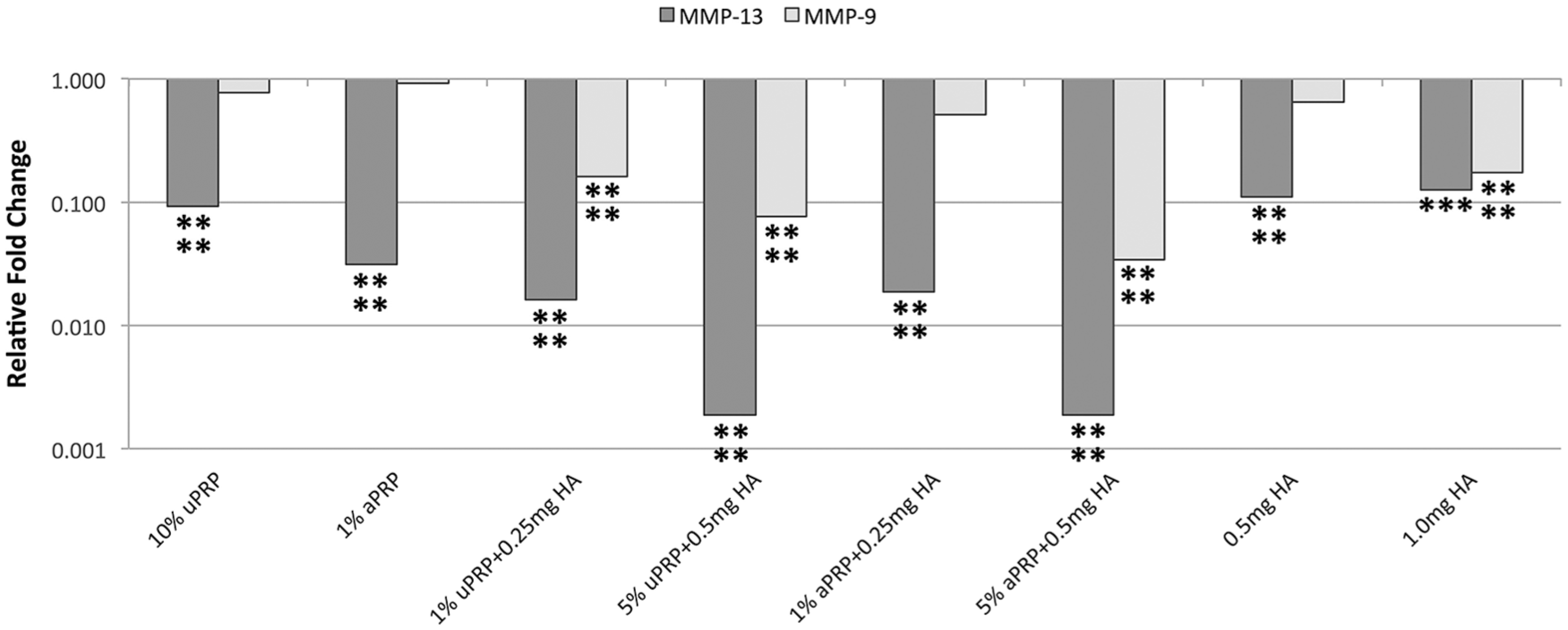
Chondrocyte MMP-9 and MMP-13 gene expression at 1 week. The greatest reduction in chondrocyte MMP-9 expression was observed in cells cultured with combined PRP-HA. All preparation resulted in significantly reduced MMP-13 expression with the largest reduction occurring in chondrocytes cultured with combined PRP-HA. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
Discussion
The primary goal of this study was to investigate the effect of combined PRP-HA on BMSC and chondrocyte proliferation. Our results demonstrated a statistically significant increase in BMSC and chondrocyte proliferation at numerous concentrations of combined PRP-HA. We also showed a statistically significant increase in proliferation of both cell types with activated and unactivated PRP-alone. This is consistent with prior in vitro studies that show PRP-alone enhances cellular proliferation.5-8,24-26 Secondarily, we sought to determine if combined PRP-HA affected chondrocyte and BM-MSC gene expression. Aggrecan, a key component of the articular ECM, endows cartilage with unique load bearing properties and is reduced in OA. While not significant, we observed a trend of increased aggrecan expression in chondrocytes in the presence of PRP-HA. This suggests that combined PRP-HA may act to promote regeneration of the articular ECM in vivo.
IL-1β, a potent inflammatory and catabolic cytokine, was added to cellular cultures to create an inflammatory and osteoarthritic environment. 22 Prior work has demonstrated that IL-1β upregulates MMP gene expression and activity. 27 MMPs are a family of zinc-dependent endopeptidases that play a crucial role in the degradation of collagen and bone in the development of OA.28,29 In this study, we looked at the expression of MMP-9 and MMP-13 in both chondrocytes and BMSCs following culture with IL-1β. The only significant reduction in BMSC MMP-9 expression occurred with combined PRP-HA. PRP-alone and HA-alone did not significantly reduce BMSC MMP-9 expression. While BMSC MMP-13 expression was significantly reduced with HA-alone, the combined products (at lower concentrations) resulted in a greater reduction of MMP-9 expression. Similar results were obtained with chondrocyte MMP expression. The reduction observed with the combined product was increased compared with the same concentrations of HA-alone and PRP-alone. These results support the additive effect of combined PRP-HA to reduce the expression of enzymes that contribute to the pro-inflammatory, catabolic phenotype observed in OA.
Interestingly, we observed a greater increase (absolute value) in cellular proliferation when uPRP was used (alone and with HA). Activation of PRP forms a gel-like clot or putty that can be useful in surgical applications. However, the formation of this clot may impede growth factor diffusion and result in reduced growth factor delivery. The gel formed by aPRP may have also impaired diffusion within cell culture.
We also observed a decrease in proliferation at the highest concentrations of PRP-alone. Previous in vitro analysis regarding the proliferation of BMSCs shows similar results, with decreasing proliferation at higher concentrations of PRP. 30 A precise dose combination and timely delivery of growth factors are critical for optimal proliferation.31,32 Perhaps, high growth factor concentrations may lead to unwanted differentiated cell phenotypes, as well as downregulation of surface receptors rendering cells insensible to such factors. 30 This could explain why the higher PRP concentrations, by containing excessive amounts of growth factors, were less effective.
Commercially available PRP preparation systems vary widely with regard to the composition of factors and concentration of platelets in the final PRP product. 33 The variability of PRP content, especially with regard to leukocyte and platelet concentrations, may affect clinical treatment outcomes. 34 We utilized a preparation system that prepares leukocyte-poor PRP with a platelet concentration factor of 1.6×. While the ultimate goal is to prepare a clinically efficacious product, careful consideration was taken to prepare optimal conditions for this in vitro investigation. As previously discussed, high concentrations of growth factors in vitro may have deleterious effects on cellular metabolism. Graziani et al. found that platelet concentrations greater than 2.5× resulted in decreased proliferation of fibroblasts and osteoblasts in vitro. 35 Furthermore, tenocytes treated with high concentrations of platelets in vitro resulted in decreased proliferation and collagen production with a simultaneous increase in MMP production. 36 Yoshida et al. found that 1× PRP significantly outperformed 5× with respect to collagen gene expression, apoptosis prevention, and cell metabolism proliferation when studying anterior cruciate ligament (ACL) fibroblasts in 3-dimensional culture. 37 The authors noted the importance of specifying platelet concentration factors and cautioned that platelet concentrations used in vitro do not predict the optimal platelet concentrations for clinical applications. This may be further amplified when considering the addition of HA and the potential interaction of HA on platelet growth factor release. 18
The rationale for the use of PRP is that the supraphysiological release of platelet-derived factors at the direct site of cartilage injury or disease may stimulate the natural healing cascade. 38 Platelet activation and the subsequent release of the aforementioned growth factors may promote cartilage matrix synthesis; increased cell growth, migration, and phenotype changes; and also facilitate protein transcription within chondrocytes.39,40 Platelets have also been found to have anti-inflammatory activity via inhibition of the nuclear-factor-kappa-light-chain-enhancer of activated B cells (NF-κB) pathway. 11 The transcription of numerous degradative cytokines including IL-1β, TNF-α, and IL-6 are under the upstream control of NF-κB. The contents of platelet α-granules inhibit this catabolic pathway and prevent the detrimental effects on articular cartilage.17,41 PRP has also been shown to have anti-nociceptive and anti-inflammatory properties via the inhibition of MMP-13 expression. 17 Recent in vivo studies suggest that the clinical benefit of intraarticular PRP might be due to its anti-inflammatory effects. 42
The basic science literature provides numerous mechanisms for which intraarticular HA may provide clinical benefit. However, the predominant mechanism remains unclear. 43 Chondroprotection is the most frequently reported mechanism within the literature, followed by proteoglycan and glycosaminoglycan synthesis, anti-inflammatory, mechanical, subchondral, and analgesic actions. 44 HA affects the intraarticular environment by binding to CD44 resulting in suppression of catabolic cytokines IL-1β and IL-6, and subsequent therapeutic effects of their suppression. 45 Additional mechanisms for the effectiveness of HA treatment in OA include Intracellular Adhesion Molecule binding, mechanical improvements attributed to shock absorption and lubrication, an increase in cartilage/bone interface type II collagen turnover, as well as analgesic effects through interaction with nerve endings and joint nociceptors.46-49 Yatabe et al. demonstrated that HA can inhibit IL-1β-induced MMP-1, MMP-3, and MMP-13 via activation of chondrocytes. 50 Prasadam et al. showed that the anabolic and immunomodulatory effects of HA are mediated by the inhibition of MMPs. 51 In vitro, HA has been shown to stimulate chondrocyte metabolism and increase ECM deposition. 52 However, HA-alone has been shown to have limited efficacy in certain OA patients and may require adjunctive agents (i.e., PRP) to enhance its efficacy. 16
Support for the combined administration of PRP-HA for the treatment of OA comes from the work of Sundman et al. 17 The authors compared the anti-inflammatory and matrix restorative effects of PRP and HA using synovium and cartilage harvested from patients undergoing total knee arthroplasty. They found that both PRP and HA significantly reduced the concentration of TNF-α in culture media. TNF-α is a recognized mediator of acute inflammation and activator of MMPs in the articular environment. 53 Furthermore, elevation of TNF-α in synovial fluid is common in OA joints and has been shown to correlate with patient pain scores. 54 The authors suggest that PRP and HA act to modulate the OA environment in similar ways but by different mechanisms. They conclude that the anti-nociceptive and anti-inflammatory activity of PRP support its use in joints with early OA to modulate disease progression. Given the potentially positive effects of HA on cartilage matrix gene expression, Sundman et al. suggest that a combination of PRP and HA might be better than PRP or HA alone for the treatment of OA.
Basic science evidence for the combined administration of PRP-HA for OA comes from Chen et al. 16 The authors showed that combined PRP-HA rescues inflammatory cytokine-inducted degeneration via chondrogenic signaling recovery. Using an ACL-transection animal model, they also showed that intraarticular PRP-HA can attenuate cartilage degeneration. Furthermore, their results showed combined PRP-HA exerted significantly increased anti-inflammatory effects and enhanced regeneration compared with PRP- and HA-alone. These results support the synergistic effect of combined PRP-HA. The authors concluded that the synergistic effects were the result of different cellular mechanisms: HA-dependent Erk1/2 pathway and the PRP-dependent Smad2/3 pathway. Iio et al. studied the influence of HA on platelet growth factor release and offer an additional mechanism by which PRP and HA act synergistically. 18 They found that the amount of growth factors released by platelets in PRP was increased by the addition of HA. While the exact mechanism is unclear, the authors propose that HA may affect the selective permeabilization and population of α-granules.
This study has a number of limitations. The lack of standardization regarding PRP preparation, techniques, and reporting limits the generalizability of any PRP-related study. 55 Many factors influence the clinical yield of PRP: blood draw method, centrifugation technique, temperature, use of anticoagulants, patient-specific factors, and activation. 33 We utilized direct cardiac puncture to obtain blood for PRP at the time of a previously scheduled necropsy. Variability in blood draw method has the potential to influence the yield and condition of platelets obtained. As discussed, the methods in this investigation were optimized for in vitro analysis. Careful consideration should be made when applying the results of any in vitro study to clinical application. Furthermore, it is unclear if the specific preparation system used (and corresponding concentration factor and leukodepletion) optimize our results. As a result, future work should focus on comparing these factors and how they affect the efficacy of combined PRP-HA in vitro. There is significant variability in the literature regarding the reporting of PRP components, that is, concentration factors versus actual platelet amount. We classified our platelet concentrations based on the concentration factor of a commercially available PRP system. However, significant variation in the baseline platelets can occur between individuals and even within individuals. 56 As such, reporting actual platelet and leukocyte amount may improve (but certainly not optimize) the generalizability of results from PRP research. Cells and blood for PRP were obtained from SDRs using a system designed for human use. The implications of using equipment and PRP preparation tubes with animal cells is unknown at this time.
Conclusions
We observed a statistically significant increase in BMSC and chondrocyte proliferation with combined PRP-HA. Our results also demonstrated an increase in aggrecan expression in chondrocytes when cultured with PRP-HA. Through an in vitro OA model, we showed that combined PRP-HA significantly reduces the expression of degradative enzymes involved in the pathogenesis of cartilage and bone destruction. These results demonstrate the additive in vitro effect of combined PRP-HA to stimulate cellular growth, restore components of the articular ECM and reduce inflammation. As a result, the combined administration of PRP-HA may have greater clinical efficacy than either product alone. Additional studies are needed to assess the optimal composition and clinical efficacy of combined PRP-HA.
Footnotes
Authors’ Note
All work was completed at the Feinstein Institute for Medical Research (Manhasset, NY).
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support: RegenLab SA (Le Mont-sur-Lausanne, Switzerland) provided study materials and also this study was funded by the department of or thopedic surgery, Northwell Health.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AMS: The Spine Journal: Editorial or governing board. JBN: None. NAS: Arthroscopy Association of North America: Board or committee member; Biomet: Paid consultant; Sports Medicine Arthroscopy Review: Editorial or governing board; Wolters Kluwer Health—Lippincott Williams & Wilkins: Publishing royalties, financial or material support; Zimmer: Research support. DAG: ICRS: Board or committee member; RegenLab SA: Paid consultant. DAG is a paid consultant for RegenLab SA and does not directly benefit from the sale of commercial products.
Ethical Approval
Ethical approval was not sought for the present study because this was an in vitro study only.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because no animals were part of our study.
