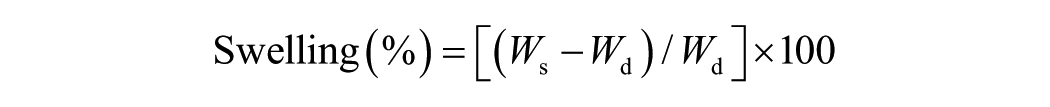Abstract
Because of poor regenerative capabilities of cartilage, reconstruction of similar rigidity and flexibility is difficult, challenging, and restricted. The aim of the present investigation was to develop cost-effective acellular xenogeneic biomaterial as cartilage substitution. Two novel biometrics have been developed using different chemical processes (Na-deoxycholate + SDS and GndHCl + NaOH) to decellularize caprine (goat) ear cartilage and further extensively characterized before preclinical investigation. Complete cell removal was ascertained by hematoxylin and eosin staining followed by DNA estimation. No adverse effect on extracellular matrix (ECM) was found by quantifying collagen and sulfated glycosaminoglycans (sGAG) content as well as collagen, sGAG and elastin staining. Results showed no drastic changes in ECM structure apart from desired sGAG loss. Scanning electron microscopy images confirmed cellular loss and unaltered orientation. Nano-indentation study on cartilage matrices indicated interesting output showing better results among decellularized groups. Increased elastic modulus and hardness indicated better stiffness and more active energy dissipation mechanism due to decellularization. Fluid uptake and retention property remained unchanged after decellularization as analyzed by swelling behavior study. Additionally, acellular materials were confirmed to be nonreactive and nonhemolytic as assessed by in vitro hemocompatibility study. In vivo study (up to 3 months) on rabbits showed no symptoms of graft rejection/ tissue necrosis, established through postoperative histology and biochemical analyses of tissue explants. With regard to size, shape, biomechanics, source of origin and nonimmunogenic properties, these developed materials can play versatile role in biomedical/ clinical applications and pave a new insight as alternatives in cartilage reconstruction.
Keywords
Introduction
Reconstruction and replacement of defective or abandoned cartilage within human body is one of the demanding challenges since ages as a consequence of its incapability for self-regeneration or repair. 1 Osteoarthritis, one of the common cartilage disorders, is the result of repetitive stress in the joint region causing irreparable damage. Although initially osteoarthritis can be treated with exercise, medications, and so on, eventually, joint replacement therapy and/or other surgical interventions are applied. But the outcome is often unsatisfactory and also restoration of its native tissue is rarely achieved. 2 Moreover, nasal and tracheal cancers, congenital disorders, such as Treacher Collins syndrome and Aperts syndrome are diseases that cause entire abscission of the organs and/ or deformed ear and nose, respectively. The application of autologous cartilage from nasal septum, 3 rib, and auricular, 4 is the gold standard technique for the reconstruction of neck and head injury. But unfortunately, certain convoluted consequences such as multiple surgeries, donor site morbidity, postoperative dysfunction, and inadequate cosmetic results at explantation and implantation site, give unsatisfactory results.4,5 Therefore it is evident from these clinical scenarios that there is an immense need to impose the existence or introduce of cartilage tissue engineering to circumvent these problems. Lately, it is being observed that natural acellular extracellular matrix (ECM) scaffolds and various decellularized xenogeneic tissues are being successfully developed as viable replacements.
The materials are acquired by treating them with some decellularizing agents (physical, chemical, and/ or enzymatic treatments) 6 which will retract the nonimmunogenic 3-dimensional (3D) structure of the ECM intact while eventually destroy the antigenic cellular part. 7 Additionally, the developed materials will conserve its unaltered biomechanical properties, which will also provide functional support on implantation without any immunogenic stimulation due to the lack of donor cellular antigens. 8 For instance, numerous studies have been reported on acellular xenogeneic tissues 9 and decellularized cartilages, that is, tracheal, articular, nasal, meniscal cartilages and intervertebral discs. 10 In contrast, application of acellular ear cartilage has been less explored in comparison to hyaline and fibrous cartilages. Ear cartilage is mainly composed of collagen types I and II and additional thick elastic fibers making it denser and thus laborious to decellularize. Furthermore, it is also advantageous to develop scaffolds from whole cartilage tissues in contrast to ECM-derived scaffolds11,12 as acellular large tissue structures can often easily be used to hold complex native structure such as nose, ear, and synovial joints.
However, considering all the aforementioned facts, the study needs to be outlined in a way that will not only assure thorough decellularization of the material but also combat the chances of disease transmission and risk of infection. Apparently, this process will also help mask the antigenicity and eliminate immunoreactions.13,14 On the other hand, the effectiveness of the decellularizing agents depends on various factors, including tissue’s cellularity, density, thickness and lipid content. 15 Additionally, it is already studied that the structure and composition of ECM get minutely altered due to these methods and agents. Therefore, in our study, decellularizing agents have been selected very carefully along with the modified methods. Some changes were introduced to reduce the degree of ultrastructure disruption of cartilage and also to maintain the biomechanical integrity of cartilage transplant matrix, chondroconductive, properties and matrix recellularization capabilities. 14 Detergents (ionic, nonionic) are one of the effective decellularizing agents that disrupt the cell membrane and detach the DNA from protein, and therefore eliminate cellular materials from tissues.15,16 Triton X- 100 (nonionic), sodium dodecylsulfate (SDS), and sodium deoxycholate (ionic) have been repeatedly used as potent decellularizing agents for the removal of cells from dense tissues and organs.15,17 Besides, guanidine hydrochloride (GndHCl), a chaotropic salt, in combination with NaOH and H2O2 is a strong decellularizing agent that disrupts the complex macromolecular structure of DNA, RNA, and protein. 18
Therefore, we conducted our study to develop a decellularized xenogeneic cartilage material and had an extensive evaluation of its basic properties, including histology, cytotoxicity, mechanics, and biochemistry. The construct might also be handled as scaffolds for cellular migration, adhesion, recellularization, and proliferation. Thus, the aim our research was to observe the effects of various decellularizing agents on full-thickness goat auricular cartilage and to judge whether these processed acellular cartilage materials are able to be utilized as successful implants in tissue engineering field, sustaining all the requirements mentioned above.
Materials and Methods
Preparation of Decellularized Cartilage Matrices
The fresh auricular cartilages of young goats from local slaughterhouse were collected and preserved in cold normal saline solution after removing hairy part. The materials were then gently washed to remove any attached tissues/blood, defatted in 95% ethanol for 24 hours at 4°C and finally rinsed in phosphate buffered saline (PBS; pH 7.4). Two different techniques were followed to achieve decellularization of cartilages. In the first group, cartilage samples were decellularized following the method of Gupta et al.19,20 with some modifications. Briefly, small pieces (2 × 2 cm) of cartilages were immersed in deionised water in a shaking condition for 12 hours. The materials were then placed in 4% Na-deoxycholate (Sigma Aldrich, St, Louis, MO, USA) solution, an ionic detergent for 4 hours at 37°C with mild shaking followed by incubating in 0.5% SDS (HiMedia Lab, India) solution for 24 hours at 37°C with agitation. The solution was changed every 6 hours. Finally the tissues were thoroughly washed (with 4-5 changes) in PBS (pH 7.4) containing 1% antibiotic antimycotic solution (HiMedia Lab, India) with agitation at room temperature and stored in PBS with antibiotic solution at −20°C for further use.
Alternatively, another group of decellularized cartilage was obtained by following the protocols 18 with some modifications. In brief, H2O2 pretreated cartilage samples were transferred to 1 N NaOH solution for 3 hours at room temperature for denaturation of DNA/RNA and inactivation of pathogens. Cartilage samples were washed with PBS containing antibiotic-antimycotic solution for 24 hours at room temperature. The materials were then transferred to 1 M guanidine hydrochloride (GndHCl) (HiMedia Lab, India) solution containing 0.05 N sodium acetate (SRL India) dissolved in an incubation buffer and incubated at +4°C for 96 hours. However, after another washing step with PBS for 24 hours, cartilage materials were again treated with 5% H2O2 solution (Merck) for 48 hours at 4°C. After a last washing step, decellularized cartilage materials were stored in sterile PBS with antibiotic solution at −20°C for further use. Tissues from both the groups were also preserved in 10% neutral buffered formalin (NBF) solution for histological analysis to check accellularity. Fresh or untreated cartilage samples were also preserved in normal saline solution to be used as control.
Histology
The intact and decellularized cartilages (Na-deoxycholate and GndHCl treated) were fixed in 10% NBF and embedded in paraffin wax. Sections (5-μm thicknesses) were prepared (Microtome, Leica, RM2235, Germany) for staining with hematoxylin and eosin (H&E), Alcian blue, Sirius red, and elastin staining by Weigert’s resorcin fuschin method, to evaluate basic tissue histoarchitecture, sulfated glycosaminoglycans (sGAG) content, orientation and distribution of collagen and elastic fibres, respectively. All the staining procedures were performed following manufacturer’s protocol. Tissue slices were observed under an upright microscope (Leica, Germany).
Scanning Electron Microscopy
Cartilage samples were examined using scanning electron microscopy (SEM) ZEISS, EVO system. For this the materials were processed by cutting the cartilage samples into small pieces and washed with Hank’s Balanced Salt Solution (HBSS). First it was soaked in 2 mL of glutaraldehyde for 24 hours. Next, samples were washed thrice with HBSS followed by washing with 50%, 70%, and 90% ethanol once and 100% ethanol twice. The samples were further washed with 1:1 ratio of ethanol and acetone and again in 100% acetone and dried completely at room temperature. Finally the samples were sputter coated with gold-palladium alloy for 120 seconds the ultrastructure of samples was imaged.
Mechanical Testing
Nanoindentation studies were performed on the cartilage sample (decellularized and native) surfaces at wet condition, using Hysitron-TI 950 TriboIndenter, equipped with fluid cell conical diamond indenter with tip radius of 1 µm. A minimum of 15 indents were performed on each sample to evaluate the hardness (H) and elastic modulus (E) in depth control mode (200 nm) at constant rate of 20 nm/s during loading and unloading, dwell at a peak depth of 10 seconds. E was determined from the unloading part of the load versus depth of penetration plot of the indents following the Oliver-Pharr method. 21 The hardness, elastic modulus, and stress relaxation of different surfaces were analyzed through nanoindentation test. Stress relaxation for visco-elastic materials is measured by the reduction in load during the dwell time at applied peak load.
Swelling Properties
The swelling property of different developed cartilage matrices were studied by calculating the differences of the dry weight (Wd) and swelled weight (Ws) at various time points under identical conditions. In brief, the dry weight of decellularized cartilage matrices along with control (untreated) were recorded (in an oven at 70°C under vacuum for overnight) followed by immersing the same matrices into PBS (pH 7.4) at room temperature until they reach equilibrium. The swelled weight of matrices was taken at various time points and the percent swelling was calculated using the equation:
Biochemical Assays
Prior to performing the biochemical estimations, intact (untreated) cartilage and decellularized cartilages were chopped and lyophilized to a dry powder having constant weight. DNA quantification was carried out using DNeasy Blood and Tissue Kits (Qiagen) following manufacturer’s protocol. In brief, minute amount of (25-30 mg) lyophilized powder of each group were taken in a 1.5-mL microcentrifuge tube. Proteinase K solution (20 µL) and 180 µL buffer ATL were added to each tube, mixed thoroughly by vortexing and incubated at 56°C until attaining complete tissue digestion. Next, 200 µL buffer AL and 200 µL of absolute ethanol was added to the digested samples with repeated mixing. The mixture was transferred into the DNeasy Mini spin column placed in a 2-mL collection tube and centrifuged at 8000 rpm for 1 minute. The flow-through was discarded and 500 µL buffer AW1 followed by 500 µL buffer AW2 was added and centrifuged for 1 and 3 minutes at 8,000 and 14,000 rpm, respectively. Finally, DNA was collected in clean 1.5 mL microcentrifuge tube by adding elution buffer (200 µL buffer AE) and centrifuging for 1 minute at 8,000 rpm. DNA was quantified by measuring absorbance at 260/280 nm in a nanodrop spectrophotometer (Eppendorf BioSpectrometer basic). For glycosaminoglycan assay, small pieces of decellularized and intact cartilage samples were digested overnight in papain solution (125 μg/mL papain, 5 mM L-cysteine, 100 mM Na2HPO4, and 5 mM EDTA, pH 6.2) at 60°C. The DMMB (1, 9-dimethylmethylene blue) assay for sGAG estimation was followed according to the manufacturer’s instructions 22 with some modifications. In brief, DMMB (Sigma Aldrich, USA) reagent solution (pH 3) was prepared according to standard protocol along with chondroitin-4 sulfate standard solution. Papain (HiMedia Lab, India) digested samples (50 µL) were taken into the 96-well microplate and 200 µL of DMMB solution was added to each sample and mixed gently by pipetting. Distilled water was used as negative control. The contents of sGAG were with a spectrophotometer at a wavelength of 525 nm (ECIL, India). The data were extrapolated following the calibration curve of standard chondroitin sulfate (Sigma Aldrich, USA) solutions. The amount of collagen was determined by using Hride Tullberg–Reinert method 23 with some modifications. Briefly, small amount of decellularized and control cartilage was digested in a pepsin cocktail (1 mg/mL pepsin, pH 3.0) (HiMedia Lab, India) at 4°C for 48 hours. Following centrifugation, the digested samples were added in a 96-well plate and dried at 37°C for 24 hours. Next, 100 μL of Sirius red dye (Sigma Aldrich, USA) solution (prepared in picric acid saturated solution to a final concentration of 1 mg/mL) was added and incubated for 1 hour with mild shaking. The wells were then washed 3 times with 0.01 N HCl and revolved the dye sample complex using 0.1 N NaOH. The absorbance was read at 550 nm using a spectrophotometer (ECIL, India). Total collagen content was estimated using a standard curve prepared with commercially available rat tail collagen type I (Sigma Aldrich, USA). Distilled water was used as negative control.
Transforming growth factor (TGF)-β and -β1 were estimated by means of specific ELISA (enzyme-linked immunosorbent assay) kits (Bioassay Technologies laboratories and Qayee Biotechnology respectively) according to manufacturers’ protocol. Briefly, the decellularized matrices were chopped into small pieces, homogenized, sonicated (5 cycles/sample; 200 W, Hielscher Ultrasonics GmbH, Germany) and centrifuged (1500 × g for 15 minutes). The supernatant was taken and further assayed according to the principle of sandwich ELISA. The biochemical estimations were performed by measuring the optical density values at 450 nm in an ELISA plate reader (ECIL, India).
In Vitro Hemocompatibility (Red Blood Cell Compatibility)
The experiment was initially conducted after collecting the fresh blood from healthy in-house animals (rabbits) of West Bengal University of Animal and Fishery Sciences (WBUAFS), Department of Veterinary Surgery and Radiology. Prior to successful outcome, fresh blood of healthy human volunteer was also drawn at R.G. Kar Medical College and Hospital, Kolkata, India with prior consent. The anticoagulated blood was centrifuged at 700 rpm for 10 minutes and the red blood cell (RBC) pellet at the bottom was collected for further use discarding the blood plasma at the top. The pellet was washed with PBS (pH 7.4) by centrifugation (at 700 rpm, 10 minutes) until getting a clear supernatant. The developed cartilage materials were then submerged in the diluted RBC (1:4 ratios in PBS) and incubated at 37°C for 2 hours. The solutions were then pipetted out and centrifuged at 1200 rpm for 5 minutes. The absorbance (A) of the collected supernatants was measured at 540 nm in an ELISA plate reader. The experiments were conducted in triplicates for each sample along with a set of positive and negative controls (distilled water and PBS respectively). Percentage of haemolysis can be determined by following formula;
In Vitro Cell viability and Cytotoxicity Assay by MTT
Cell viability and cytotoxicity assessments were carried out using MTT assay.24,25 Rabbit peripheral blood mononuclear cells (PBMC) were isolated by density gradient centrifugation 26 from freshly collected blood from healthy animals of WBUAFS animal house. The mononuclear cells were washed 2 times with sterile PBS (pH 7.4) by centrifugation at 900 rpm for 10 minutes. The pellet was resuspended with 1 mL RPMI-1640 (Sigma-Aldrich, USA) growth medium (containing 10% fetal calf serum and 50 U/mL penicillin and streptomycin). The viability of the cells was ascertained by trypan blue dye exclusion method and was found to be more than 95%. The final number of PBMC, adjusted at 106cells/mL, was seeded into each well of 96-well flat bottom tissue culture plate (Corning, USA), and incubated overnight in a humidified atmosphere containing 5% CO2 at 37°C. After 24 hours of cell seeding, Na-deoxycholate and GnDHCl-treated decellularized cartilage and untreated native extracts (homogenized and sonicated) were added to each well (in triplicate) (50 µg/mL) and 2% and 4% dimethyl sulfoxide (DMSO; negative control) and TCP (tissue culture plate) without any treatment were served as positive control. The effects were monitored over a period of 12, 48, and 72 hours using MTT assay. In brief, 100 µL of MTT (5 mg/mL) in PBS was added to each well followed by incubation for 4 hours at 37°C. Finally, 150 µL of supernatant was discarded and 150 µL of DMSO was added to each well to dissolve the blue formazan product and the absorbance was measured at 595 nm in an ELISA plate reader.
In Vivo Xenografting in Rabbit Model
The total in vivo animal trial was conducted in accordance with the “Principles of Laboratory Animal Care” from the Institutional Animal Ethical Committee (IAEC), WBUAFS, West Bengal, India (Permit No. IAEC/59(i) dated July 27, 2018) for a period of 3 months, with 2 time points (1 month [1M] and 3 months [3M]). The experiment was initiated by randomly selecting 16 healthy New Zealand white rabbits, weighing between 1.5 and 2kg of either gender and housing them under proper laboratory conditions in separate cages. All the animals were anaesthetized using an intramuscular injection of xylazine hydrochloride (5 mg/kg) (Xylaxin, Indian Immunologicals, India) and ketamine hydrochloride (25 mg/kg) (Ketalar, Parke-Davis, India). Dorsal surface of mid thoracolumbar region was shaved and wiped with povidone iodine solution, 5-mm incisions were made in the subcutaneous pockets and intact and decellularized cartilage matrices (size 2.5 cm × 1.5 cm, thickness of 1-1.5 mm) of each group were implanted ( Fig. 5.I ) into 16 animals (4 animals/group) and the incisions were sutured. At predetermined time points (1M and 3M), engrafted cartilage along with surrounding host tissues was removed ( Fig. 5.II ) and further processed for histology and ECM study (qualitative and quantitative).
Histology and Biochemical Analysis
After completion of in vivo animal experiment, collected tissue biopsies were fixed (10% NBF), paraffin embedded, sectioned (5 µm thickness) for histological evaluation. Before staining, the samples were rehydrated using a series of decreasing alcohol and xylene. Following the same procedure as mentioned above, sections were stained with H&E, Alcian blue (HiMedia Lab, India), Sirius red (Sigma Aldrich, USA), elastin staining to assess cellular and structural morphology of the host with xenografted cartilage matrices, presence of sGAG, distribution of collagen and elastic fibers respectively. Images were taken under a bright field microscope (Leica, Germany).
The ECM was analyzed further by quantifying the amount of sGAG, collagen, and DNA following previously described protocols. Tissue biopsies were collected from xenografted and healthy animals after 1 and 3 month and processed further. Tissue from normal healthy animal was also explanted for quantification of DNA, collagen and sGAG for a relative study. In brief, for proteoglycan estimation, samples were papain digested overnight and concentration was calculated following manufacturer’s instructions. 22 The amount of collagen was studied by using Hride Tullberg−Reinert method. 23 DNA was estimated by using DNeasy Blood and Tissue Kits (Qiagen) following manufacturer’s protocol as mentioned earlier.
Statistical Analysis
All the data were presented as mean ± standard deviation (n =3) and analyzed using statistical software OriginPro8 (Originlab Corporation, USA). Statistical analysis was performed by 1-way analysis of variance (ANOVA) followed by Tukey’s test. The differences between groups of *P ≤ 0.05 were considered statistically significant and **P ≤ 0.01 as highly significant.
Results
In order to understand the efficiency of the decellularizing agents, cell content and structural morphology were monitored by SEM, histological, and biochemical techniques. The SEM images ( Fig. 1 ) showed that the decellularized groups of cartilage matrices had partial collagen matrix along with moderate elastic fibers immensely interweaved within the uniformed array of collagen network as compared with thinner of native cartilage. On gross inspection, no evidence of nucleus staining was observed in the decellularized groups as compared to the native/intact matrix by H&E staining ( Fig. 2 ) and the cellular architecture was unchanged. The orientation of collagen fibers in the decellularized groups was almost intact without any alterations as assessed by Sirius red staining ( Fig. 2 ) compared with native cartilage matrix. Elastin staining by Weigert’s resorcin fuschin method 27 revealed synchronization of elastic fibers among the tissue cells and collagen matrix in both the decellularized group and native cartilage samples ( Fig. 2 ). However, sGAG was remarkably reduced during the decellularizing process as assessed by Alcian blue staining ( Fig. 2 ). The results of TGFβ and TGFβ1 depicted that the concentration of TGFβ ( Fig. 3A ) and TGFβ1 ( Fig. 3B ) were reduced due to decellularization by the chemical agents. Na-deoxycholate-treated cartilage showed significant loss whereas, GndHCl-treated group showed very less reduction compared with native cartilage (nonsignificant). Similarly, biochemical analysis revealed that DNA content was significantly reduced in decellularized groups. The decellularized groups showed a reduction of DNA by an average ~88% (P ≤ 0.05) compared with the native cartilage ( Fig. 4a , Table 1 ). The total sGAG content was also reduced by an average ~73% (P ≤ 0.01) in decellularized groups ( Fig. 4b , Table 1) as compared with the native sample. However, the total collagen concentration was not affected by the decellularization process as insignificant statistical changes ( Fig. 4c , Table 1 ).
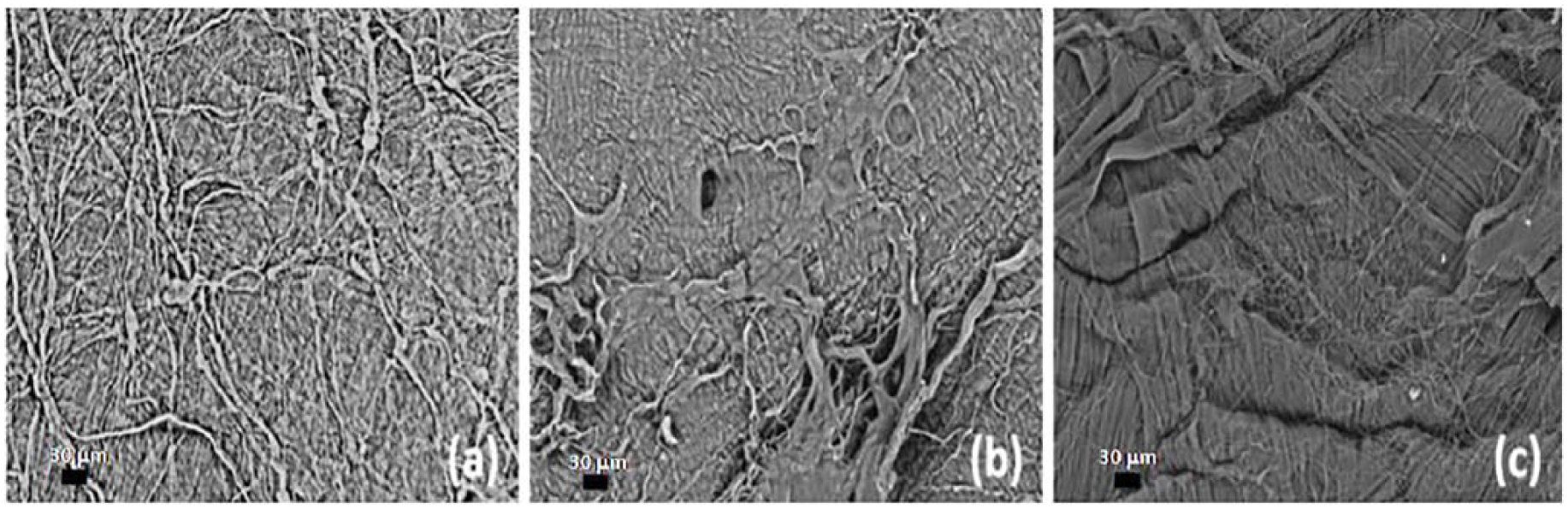
Scanning electron Microscopy image of (a) native (b) Na-deoxycholate-treated, and (c) GndHCl-treated cartilage.
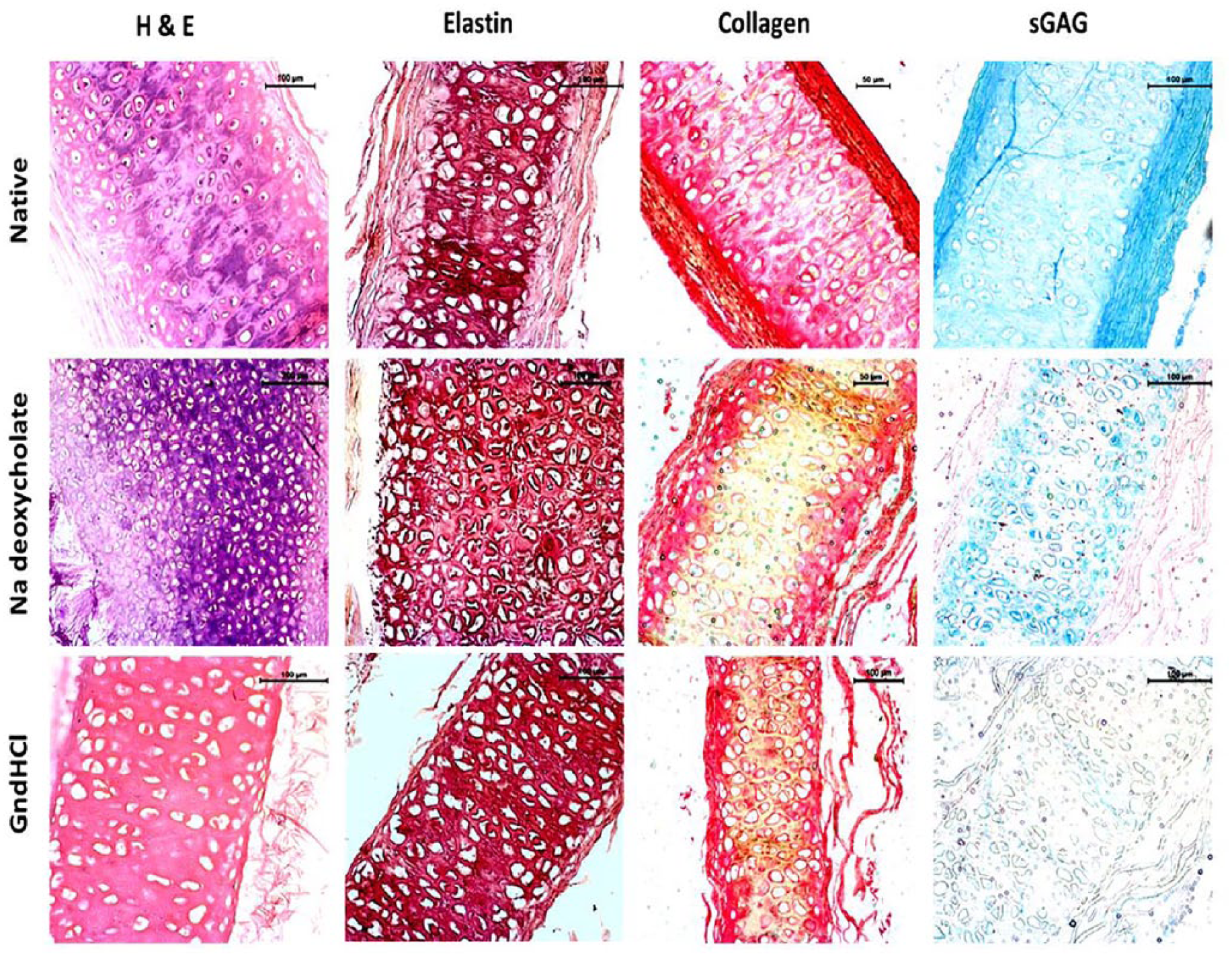
Histological images of fresh/native and decellularized cartilage samples. Native, Na-deoxycholate- and GndHCl-treated cartilages stained with hematoxylin and eosin (H&E), Sirius red for collagen staining, Elastin staining by resorcin fuschin and Acian blue staining for sulfated glycosaminoglycans (sGAG). Scale bar: 50 µm, 100 µm, 200 µm.
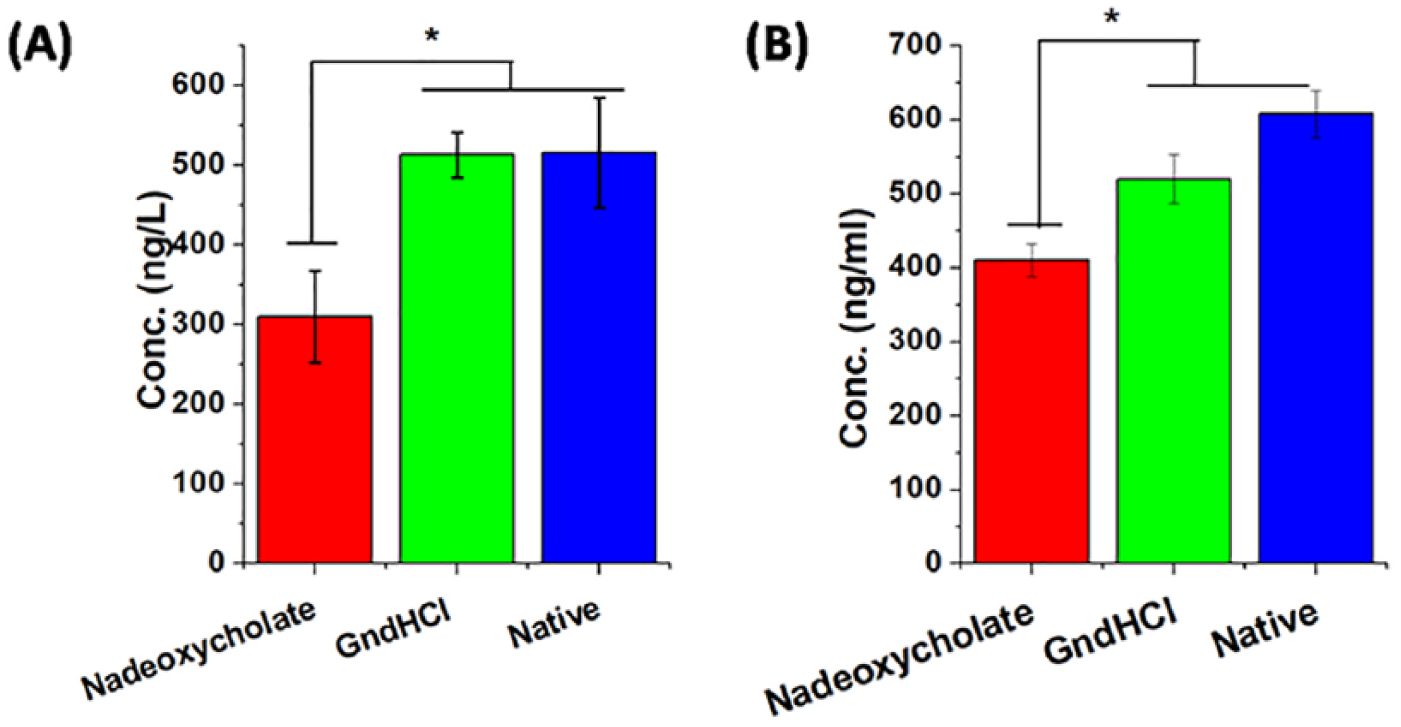
Concentration of transforming growth factor (TGF) β (
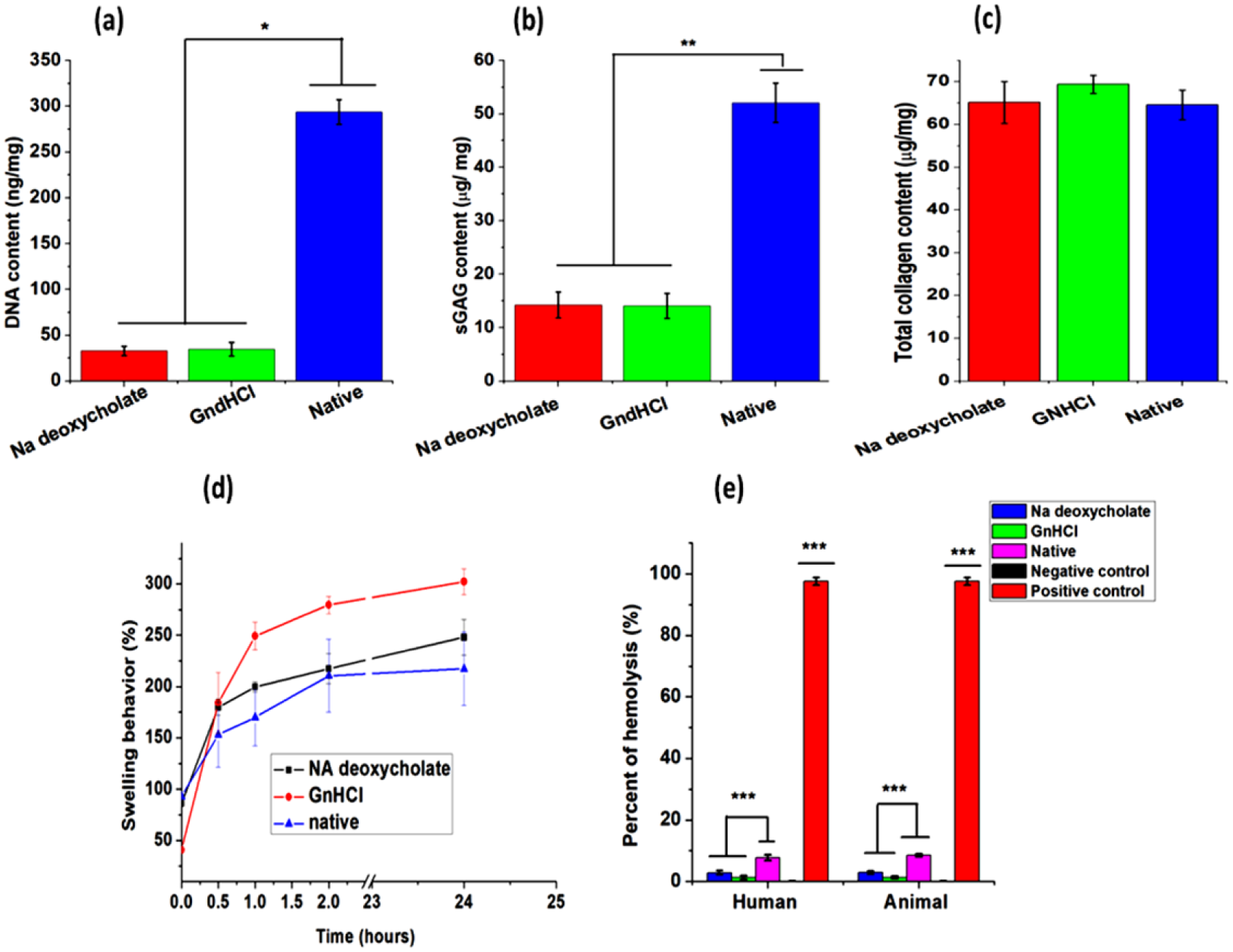
Biochemical assay of fresh and decellularized cartilage. DNA assay (
DNA, Collagen, and Sulfated Glycosaminoglycan (sGAG) Content in Fresh and Decellularized Cartilage.
Mechanical Testing
The mechanical property of decellularized and native caprine cartilage revealed interesting output indicating better results among the former groups ( Fig. 5 , Table 2 ). Elastic modulus and hardness were found to be expanded by ~62.96% and ~119.25% and 40% and 80% for Na-deoxycholate- and GndHCl-treated groups, respectively. The increased property indicated better stiffness and more active energy dissipation mechanism most likely as a consequence of decellularization.
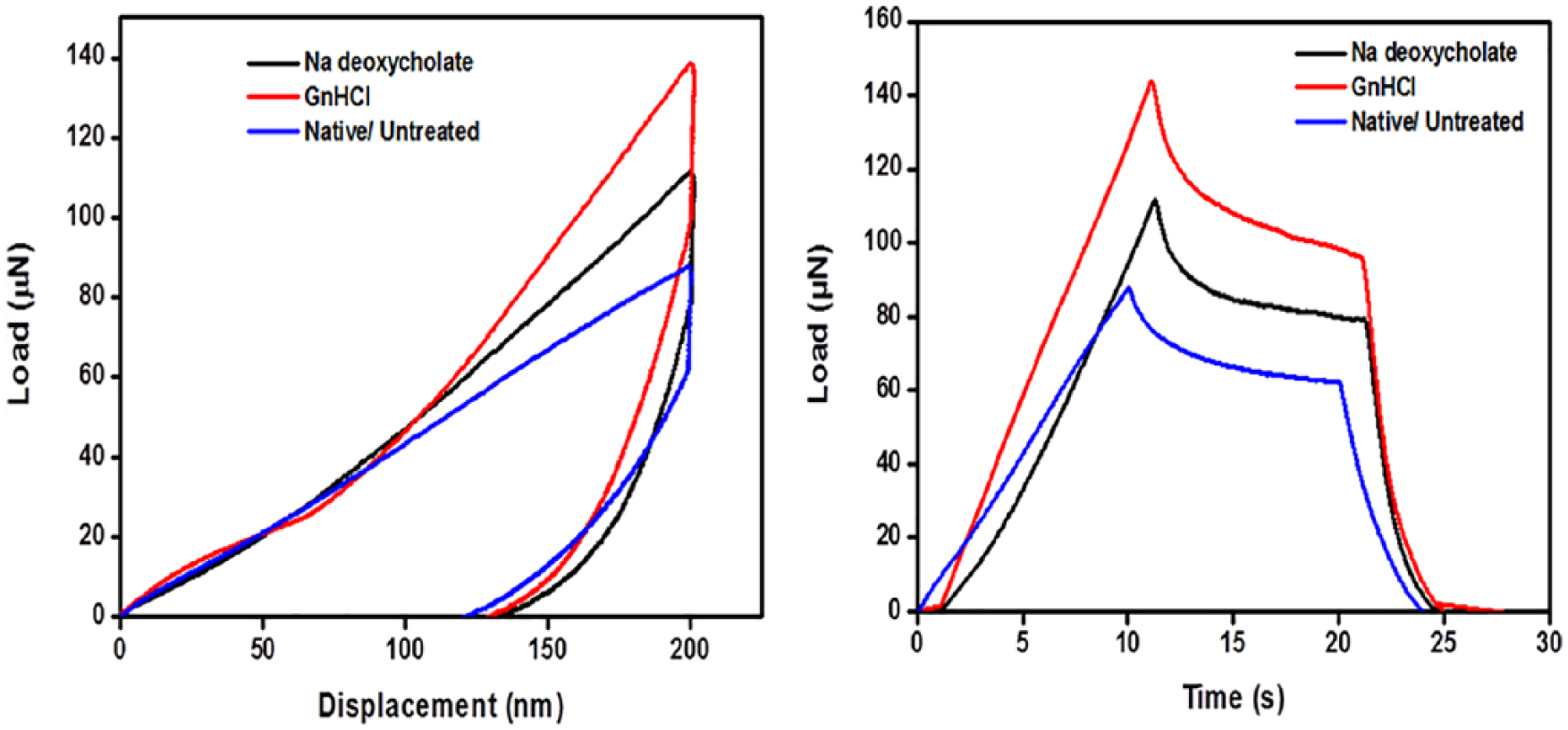
Images revealed a typical load-displacement and stress relaxation (load vs. time) curves obtained through nanoindentation of decellularized (Na-deoxycholate and GndHCl) and native/ fresh cartilage samples (wet condition).
Elastic Modulus (E), Hardness (H), and Stress Relaxation Values of Decellularized and Native Cartilages.

Swelling Behavior
Regardless of the chemical treatments, all the samples showed high swelling property in a quite similar way and gained saturation within 3 hours. All the samples exhibited maximum swelling within first 15 to 30 minutes starting with a ~2.09-, 4.52-, and 1.64-fold increases in wet weight of Na-deoxycholate-treated, GndHCl-treated, and native cartilage samples, respectively ( Fig. 4d ). Although, the initial behavior of decellularized groups showed higher water absorption capacity than native group, but post equilibrium, changes were insignificant amongst the groups.
In Vitro Hemocompatilibity
In this study, the degree of hemolysis is calculated by the amount of hemoglobin present in the supernatant. Herein, regardless of human and animal blood samples, it was noticed that Na-deoxycholate- and GndHCl-treated samples were in the range of slightly hemolytic (i.e., 2.87% and 2.92%) and nonhemolytic (i.e., 1.30% and 1.33%) range, respectively, whereas, native cartilage was found to be highly hemolytic ( Fig. 4e ).
In Vitro Cell Viability and Cytotoxicity Assay by MTT
The percentage of cellular viability ( Fig. 6A ) and cell cytotoxicity ( Fig. 6B ) were monitored in effects with decellularized and native cartilage extracts on rabbit PBMC at specified time intervals (24, 48, and 72 hours) and the data were also compared with positive and negative controls. Considering the optical density values, it was observed that the cellular viability of decellularized cartilage treated groups (Na-deoxycholate and GndHCl) and TCP remained nonsignificant at every time points. The percentage cellular viability was found to be ~87.27% and ~90% (24 hours), ~83.33% and ~104% (48 hours), ~108.10% and ~120% (72 hours) for Na-deoxycholate- and GndHCl-treated groups, respectively. Contrarily, the cellular viability of native cartilage significantly decreased after 72 hours compared with its treated counterparts. However, the percentage viability of native cartilage group, 2% and 4% DMSO was found in decreasing order after 72 hours, that is, ~54% (24 hours), ~29.66% (48 hours), and ~31.36% (72 hours) for native group, ~51.51% (24 hours), ~33.33% (48 hours), and ~27.02% (72 hours) for 2% DMSO and ~36.66% (24 hours), ~23.33% (48 hours), and ~3.6% (72 hours) for 4% DMSO.
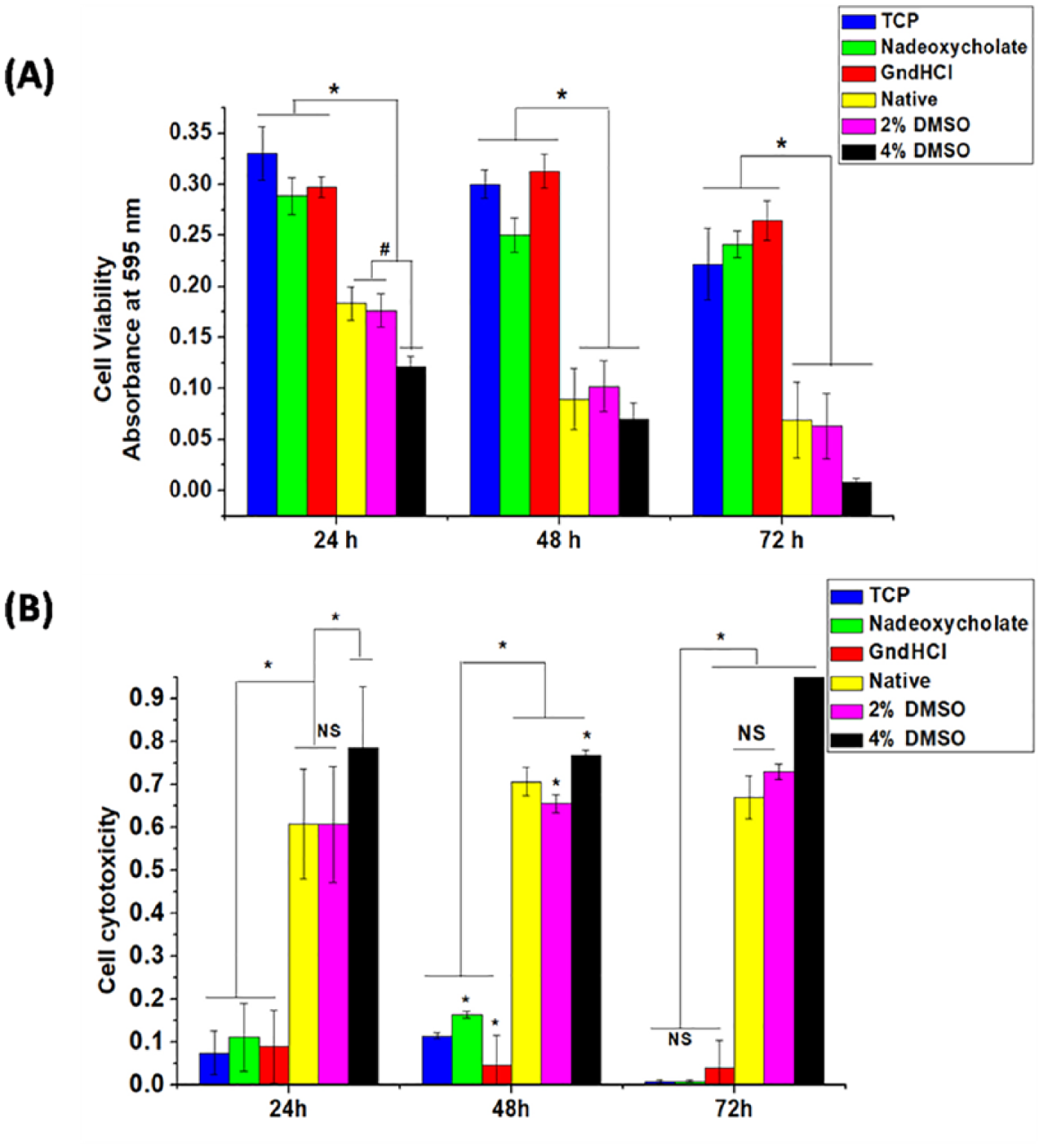
Cell viability (
From the above data, cellular toxicity ( Fig. 6B ) was also calculated. In a quite similar way, cell cytotoxicity in effect to treated cartilage groups were found to be nonsignificant compared with TCP (positive control) and also toxicity was monitored to decrease after 72 hours. Whereas, cell cytotoxicity of native group and 2% and 4% DMSO groups were noticed to be increasing by the end of 72 hours ( Fig. 6 ).
In Vivo Results
The gross observation of the operative site was studied post 1M and 3M and tissue biopsies were taken for histological and biochemical analysis. An interesting view was revealed which showed that decellularized cartilage materials were well organized maintaining their proper orientation ( Fig. 7.II ). Any sign of tissue necrosis and inflammation surrounding the site were undetectable. Conversely, native cartilage engrafted area revealed inflammation and accumulation of tissue necrosis surrounding the site ( Fig. 7.II ).
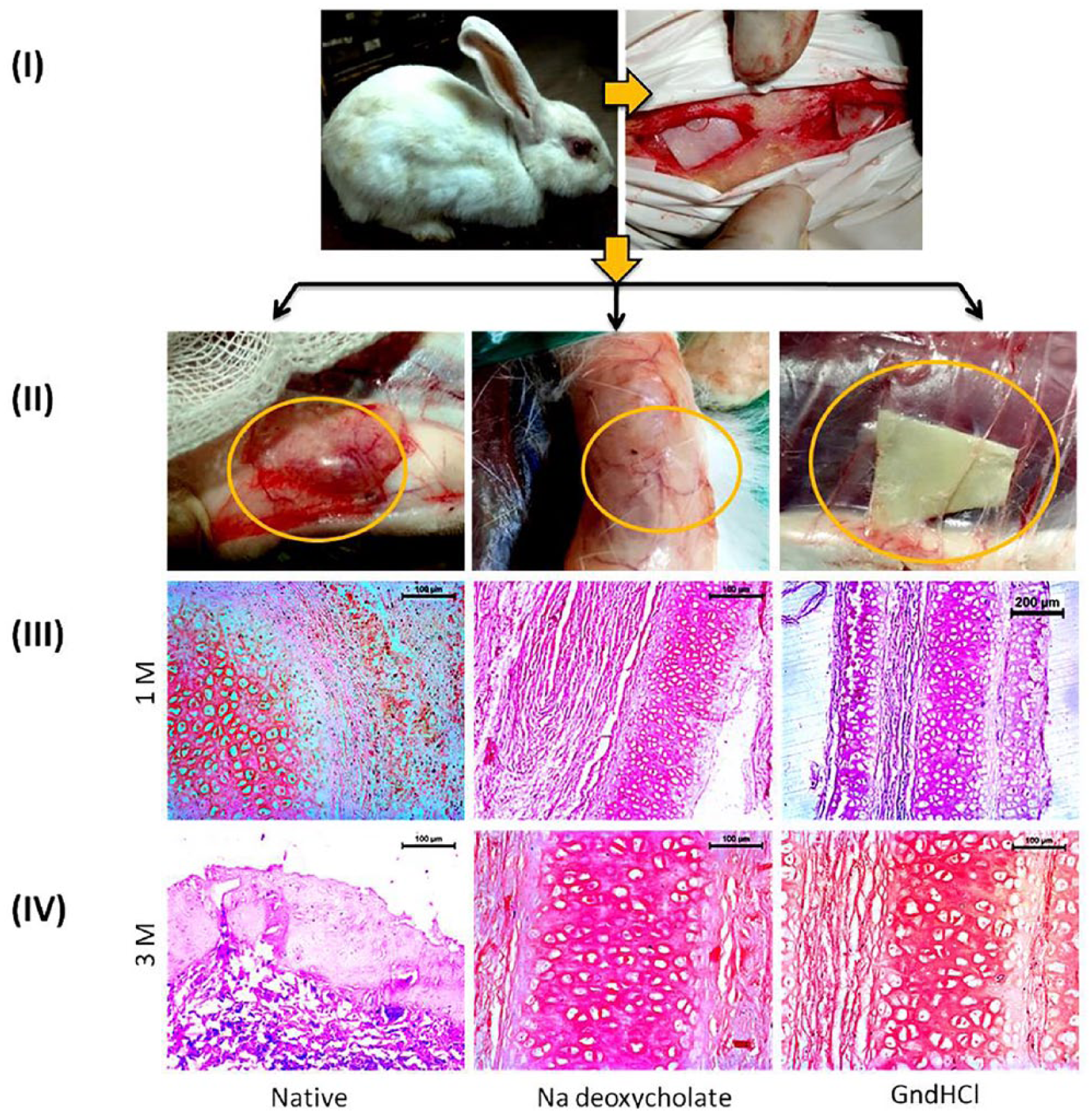
In vivo experiment on rabbit, subcutaneous implantation (
In order to monitor the host-implant biocompatibility, tissue sections (1M) were stained by H&E and microscopic observation revealed presence cellular cavity and trabeculae with no sign of angioinvasion ( Fig. 7.III ). A few numbers of immune cells and macrophages were noticed at the cartilage-tissue interface. The 3M sections depicted better attachment to the dermal and subdermal tissues. The cellular and structural orientation was appropriately coordinated with each other. No existence of macrophages, lymphocytes, plasma cells, and immature fibroblasts was spotted ( Fig. 7.IV ). On the other hand, untreated cartilage explants (both 1M and 3M) were found to be surrounded with mononuclear cells, mostly macrophages. Presence of lymphocytes, vacuolated cells and fibroblast cells was also noticed ( Fig. 7.III ). Cellular infiltration was also monitored to be limited in the peripheral area of the explants. Tissue necrosis and degeneration of cells was observed on the 3M explants quite prominently ( Fig. 7.IV ).
In Vivo ECM Study
The images of tissue sections (1M and 3M) stained by Sirius red for collagen fibers ( Fig. 8 ) represented compact arrangement of collagen firmly packed between cells and surrounding supporting matrix. The in situ structural alignment of the collagen fibers was maintained properly along with normal angiogenesis. In order to understand the orientation of elastic fibers, the tissue sections (1M and 3M) were stained with Weigert’s resorcin fuschin stains and the images ( Fig. 8 ) revealed presence of an extrusive elastin mass surrounding the area. The proliferating elastic fibers were detected all over the matrix and also at the periphery. Alcian blue staining was performed (1M and 3M) for the analysis of sGAG, one of the important ingredients of ECM. The microscopic evaluation depicted presence of sGAG in the matrix, suggesting proper accumulation of ECM without any abnormality ( Fig. 8 ). The ECM consisting of collagen, elastin, and sGAG maintained their structure fairly well. In contrast, the staining images (1M and 3M) of native cartilage implanted group showed deformed orientation of collagen and elastic fibers ( Fig. 8 ). The intensity of stains was limited as a result of loose and scattered alignment of ECM. The sGAG staining by Alcian blue was found to be very faintly visible ( Fig. 8 ).
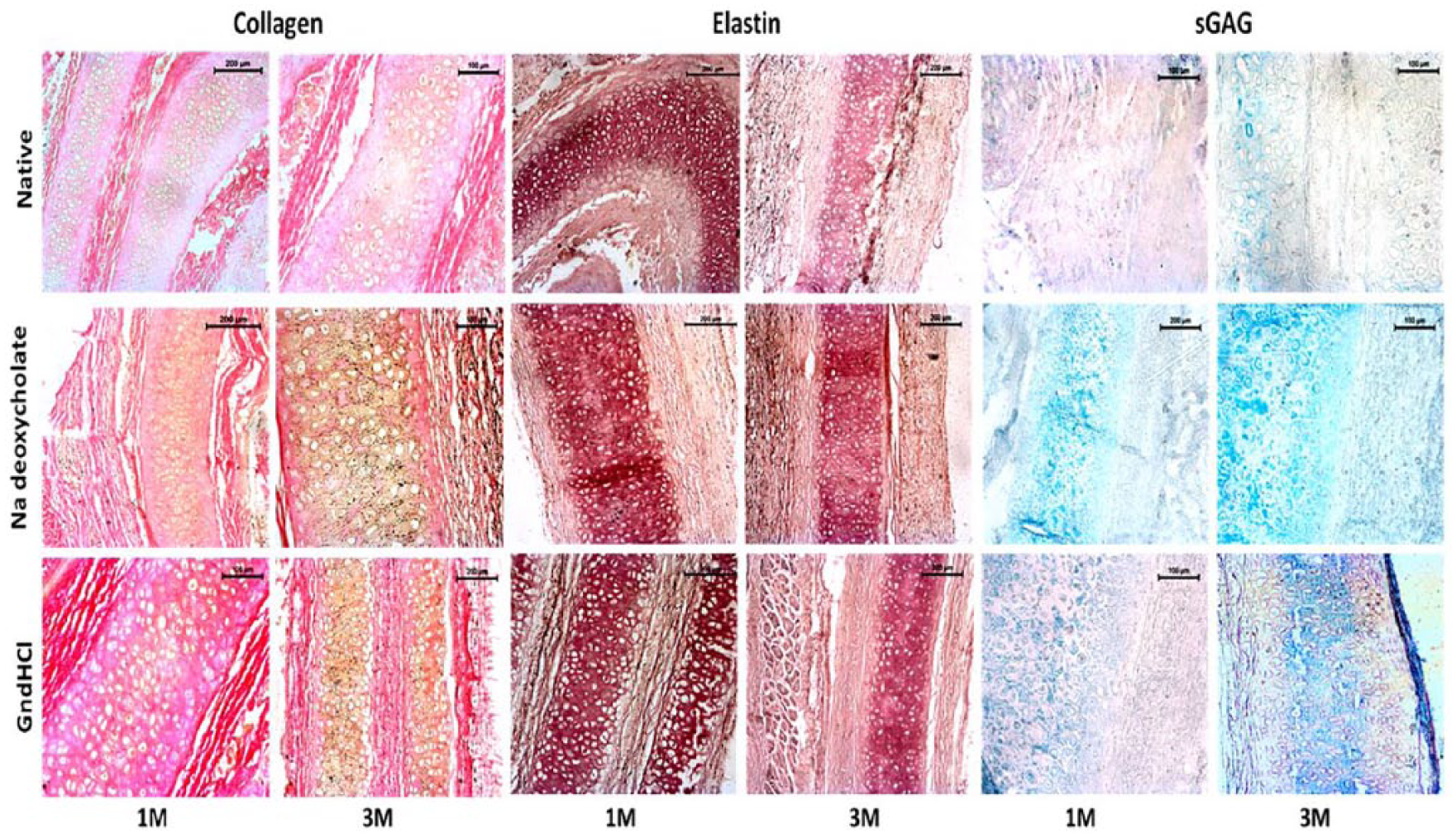
Extracellular matrix (ECM) study of decellularized and native cartilage explants post 1 month (1M) and 3 months (3M). Images of collagen, elastin, and sulfated glycosaminoglycan (sGAG) staining. Scale bar: 50 µm, 100 µm, 200 µm.
Biochemical Study
Biochemical estimation of DNA, collagen, and sGAG was carried out from the tissue biopsies at the predefined time points (1M and 3M) along with tissues from normal animal (without implant). This relative study was performed to distinguish the difference between normal and engrafted tissues. The quantity of DNA was estimated at specific time points and found that there was no significant difference in DNA concentration among the treated groups post 1M, that is, 13.71 ± 0.963, 10.76 ± 1.6, and 12.73 ± 1.331 µg/mL for Na-deoxycholate-treated, GnDHCl-treated, and native groups, respectively ( Fig. 9a ). However, compared with the healthy animal (i.e., 44.4 ± 1.44 µg/mL), the data were quite distinguishable. On contrary, concentration of DNA in treated groups (Na-deoxycholate and GnDHCl) was remarkably increased (44.5 ± 2.246 and 40.23 ± 1.16 µg/mL, respectively) after 3M and surprisingly showed similar result as of healthy group (i.e., 44.4 ± 1.44 µg/mL). Conversely, the concentration in the native group was decreased (8.22 ± 0.64 µg/mL). The results of total collagen and sGAG concentration revealed that after 1M of material implantation, the amount of sGAG ( Fig. 9b ) and collagen ( Fig. 9c ) within the treated groups remained similar with no drastic changes, although much less than normal healthy group. But at the end of 3M, there was a dynamic increase of these components in the Na-deoxycholate- and GndHCl-treated cartilage implanted groups, quite likely to the normal group. On contrary, native cartilage treated group gave decreasing concentration of collagen and sGAG content ( Fig. 9b and c ).
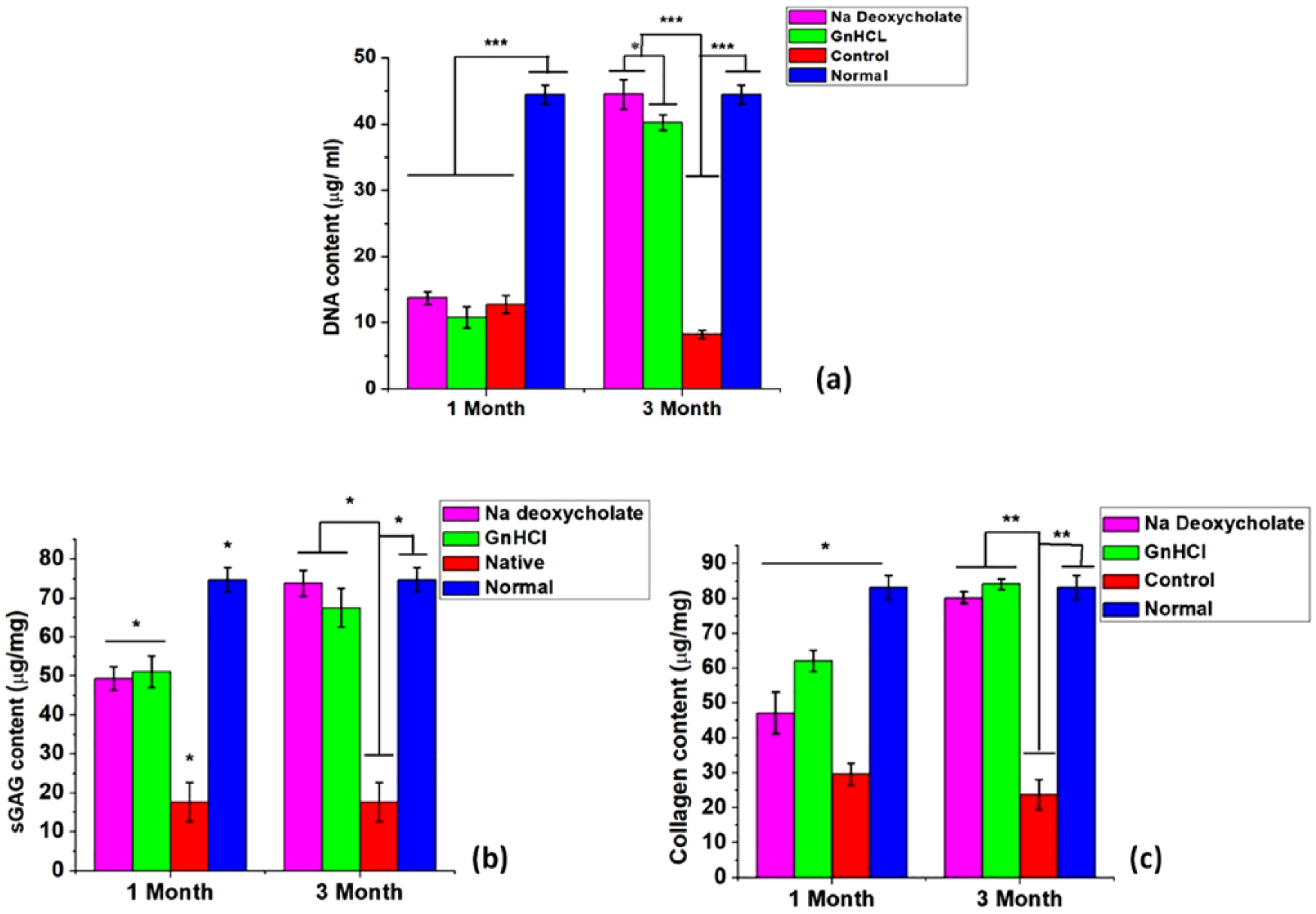
Biochemical assay of decellularized, control and normal tissue explants post 1 month (1M) and 3 months (3M) showing (
Discussion
Despite being rigid and flexible, cartilage lacks an intrinsic regenerative capacity unlike other self-repairing tissues, like bone. Hence, healing of cartilage postinjury is inevitably difficult. As a result, a scarcity for sufficient substitute matrices still persists. 1 In this regard, tissue engineering plays the most promising role, and the utilization of biomaterials, scaffolds has come with exceptional outcomes.28,29 The definition of an appropriate scaffold or biomaterial comprises on certain parameters, which include, biocompatibility, mechanical stability, and availability. 30 Besides, biochemical stability and integrity of the materials and native collagen network containing fibers and fibrils should not be altered. Furthermore, the developed matrices should also provide a niche for proliferation, adhesion, and migration of cells into the matrix as well as generating newly synthesized ECM components. Thus, a proper procedure is required that will possibly eliminate immunogenic components, rejection reactions, and risk of disease transmission 14 while at the same time, keeping the matrix integrity intact. The aim of this research was to evaluate the efficacy of different decellularization techniques on caprine ear cartilage and to observe changes in biomechanical, structural, and biocompatibility properties post decellularization. Two different chemical agents were used; one being Na-deoxycholate and the other being guanidine hydrochloride (GndHCl). The first group of decellularized cartilage materials by Na-deoxycholate followed by SDS treatment resulted in removal of nuclear bodies, although keeping the ECM unaffected.19,20 Similarly, in the second group, a combined effect of NaOH, GnDHCl, and H2O2 led to elimination of cellular components, DNA/ RNA and hydrolysis of protein and sGAG content. 18 Histological techniques are one of the primary and basic steps to study cellular structure and orientation microscopically. H&E staining confirmed absence of cellular bodies in both the decellularized groups. This cellular loss was further ascertained quantitatively by measuring the DNA content of the decellularized and native cartilage tissues and observed a significant loss of DNA among the former groups. A decrease of ~88% was noticed among the decellularized groups (i.e., Na-deoxycholate and GndHCl) compared with the native cartilage. Therefore, these techniques meet the criteria for complete decelllularization of the ear cartilage by reducing the DNA content to less than 50 ng/mg tissue, 15 which on the other hand, minimizes the chances of immune responses as long as in vivo implantations are concerned. Besides, maintenance of structural integrity of matrix is equally important despite of cellular loss. Collagen, one of the leading and richly available proteins of ECM, provides mechanical strength and controls chondrogenic differentiations. 31 The intention of retaining the collagen and elastic fibers unaffected post decellularization was one of the major challenges to look into. Having said that, the collagen and elastic fibers were found to be compactly arranged together as well as maintaining their proper orientation without any drastic changes after decellularization with Na-deoxycholate and GndHCl as assessed histologically by Sirius red and Weigert’s elastin staining. The SEM images also supported the outcome showing unaltered alignment of collagen and elastic fibers among the decellularized groups. The quantification of collagen biochemically depicted little changes in the decellularized groups compared with native matrix. The difference between the concentrations of collagen was insignificant ( Table 1 ). However, proteoglycans, another vital organic component of cartilage matrix, were decreased post decellularization. Removal of sGAGs is one of the important requirements as this phenomenon promotes the cellular loss by controlling the water flow as well as diffusing the decellularizing agents into the matrix. 18 The staining result by Alcian blue and chemical assay by DMMB demonstrated qualitative and quantitative loss of sGAG, respectively. In the first group, SDS treatment possibly caused the reduction7,32 whereas in the second group, NaOH treatment led to the destruction of sGAG along with cellular loss. 18 Although the average loss of sGAG was monitored to be ~73% (P ≤ 0.05) in both the decellularized groups, the remaining content showed no toxic effects. On contrary, numerous properties of sGAGs, like reacting with cytokines, engaging growth factors, enhancing water retention capacity of the matrix and also improving ECM gel properties could be the reason of enhanced biocompatibility of the matrix after decellularization.33,34 Hence these data altogether led to conclusion that the chosen decellularization techniques of combined usage of Na-deoxycholate plus SDS and GndHCl cocktail were successfully removed the cellular part while keeping the ECM unaffected.
The TGFβ superfamily plays crucial roles in various stages of cartilage development and maintenance. 35 In this study, on decellularization, concentrations of TGFβ and TGFβ1 were found to be significantly decreased in Na-deoxycholate-treated cartilage compared with native cartilage, whereas GndHCl-treated cartilage group showed insignificant loss. This may happen as a result of chemical treatments along with rigorous washing. It has been observed that TGFβ superfamily has been vastly used to promote MSC chondrogenesis in various in vitro culture and in vivo articular cartilage repair.35,36 Therefore, these matrices may also be utilized as a vehicle to provide spatial and temporal control TGFβ to induce in vitro chondrogenesis of stem cells as well as in vivo cartilage defects repair. This may lead to another area of research that needs further elaboration, discussion, and development.
Most biological tissues, including different organ tissues, ocular tissues, and cartilage tissues, exhibit time-dependent mechanical behavior due to their viscoelastic nature of tissues. In order to determine the physiological functionality of these tissues, such as energy dissipation and load bearing, it is necessary to understand the viscoelastic nature of these tissues. Nanoindentation is the novel technique to investigate the stress relaxation behavior of viscoelastic materials at nanoscale, by measuring the load-time history on a constant input displacement on samples. The mechanical property of the decellularized materials was performed to monitor whether the chemical agents disrupted the structural property and mechanical strength of the cartilages. In the present study, decellularized cartilages, treated with Na deoxycholate and GndHCl showed ~38.21% and ~84.15% improvement in stress relaxation, respectively, as compared with untreated sample and also the cartilage samples that were treated with GndHCl show significant improvement in hardness and elastic modulus, compared with other samples. Moreover, this can be due to exhibiting more viscoelastic nature of the cartilage when it was treated with GndHCl. Surprisingly, the increased property indicated better stiffness and more active energy dissipation mechanism most likely as a consequence of decellularization. Therefore, from this novel nanoindentation study, it can be concluded that the mechanical functionality of the cartilage materials was not only maintained post decellularization but also increased (GndHCl > Na-deoxycholate) significantly.
The capacity to uptake water as well as retaining it within the matrix is one of the most important and basic characteristics of a scaffold or biomaterial for successful tissue engineering applications. Since, the decellularizing agents discard the cellular parts of any material, it is important to monitor whether this causes any changes in the swelling behavior of the samples. Additionally, the swelling ability plays pivotal role in in vitro cellular proliferation 37 and in vivo implantation by preserving its shape yet protecting nutrient and media loss from the matrices respectively. In our study, the swelling behavior of the decellularized cartilage matrices showed a similar increment as native cartilage. The initial increment was higher in GndHCl (~4.52-fold) and Na-deoxycholate (~2.09-fold) compared with native group (~1.64-fold). But the changes were unnoticeable post equilibrium. Therefore, despite the importance of the swelling ability of any biomaterials, this particular study on decellularized cartilage matrix has not been ventured yet. Hence, this phenomenon also assured that the developed decellularized matrices maintained their basic structure post chemical treatments and they could be used as a vehicle for many important researches in future such as for local delivery of drugs and so on.
Biocompatibility can be defined as a harmless relationship between the host tissue and materials without any unfavorable or adverse reactions as well as lysis of RBCs. 38 Thrombogenesis occurs due to presence of some procoagulant factors in the circulation as a result of hemolyzed RBCs. 39 As a result of osmotic pressure induced by the matrices, red blood cells of the animal and human blood are burst, causing an influx in the optical density of the incubated solution. Therefore, the more RBCs broken during the experiment are directly proportional to higher hemolytic index. According to the ASTM F756-00 (2000), if the material hemolytic index data are below 2%, within 2% and 5%, and >5%, then the sample can be considered as nonhemolytic, slightly hemolytic, and hemolytic, respectively. As already mentioned, the prime intention of our study was to successfully implement these decellularized matrices as acceptable xenografts in in vivo system. Having said that, such clinical experiments are mostly associated with blood contact with the host, which, if overlooked, will result in hemolysis and finally necrosis or graft rejection. Surprisingly, as the literature revealed, researches with acellular biomaterials, precisely acellular cartilage xenografts have not dealt with hemocompatibility study much clearly, hence, whether these samples are implantable or not, was one of the important and novel observations. However, our experiment depicted that decellularized cartilages lay within the range of nonhemolytic (GndHCl) to slightly hemolytic (Na-deoxycholate) whereas, native cartilage showed high hemolysis activity. Therefore, the results confirmed that these newly developed decellularized matrices could be successfully implanted in future clinical practices.
The initial results obtained after 24 hours showed that rabbit PBMC was least affected by the treatment of decellularized cartilage extracts (50 µg/mL), which was maintained throughout until 72 hours. There was no significant difference in viability with positive control whereas native cartilage extract and 2% and 4% DMSO–treated cells were found to be reduced significantly by the end of 72 hours. The toxicity analysis also showed similar results where 4% and 2% DMSO–treated groups showed maximum toxicity to the cells followed by native cartilage extract. In contrast, toxicity was found minimum in treated groups and nonsignificant with TCP.
In vitro analysis plays important role to monitor the effects of acellular cartilage extracts with native extracts. Cellular antigens and proteins (cellular and structural, viz., sGAG) are mostly involved in raising immunologic reactions which affect the cellular viability and cell cytotoxicity, causing eventual cell death and necrosis. In addition, application of chemical agents to obtain decellularization sometimes causes toxicity to the host. Therefore, it was a vital step to monitor the effects of decellularized cartilage extracts on rabbit PBMC. The comparison was obtained with native cartilage extracts and DMSO, which is a potent toxic agent to cells. 40 The rabbit PBMC was cultured for 72 hours and cell viability and cytotoxicity was measured by MTT assay. Cellular viability was maintained throughout the experiment when cultured with decellularized cartilage as compared with TCP (TCP containing only cells without any treatment). Whereas, the viability was significantly lost in case of native cartilage, 2% and 4% DMSO–treated group. Similarly, cell cytotoxicity results indicated reduced or very less toxic effects of decellularized cartilages, whereas native cartilage extracts showed higher toxicity level quite similar to DMSO (2% and 4%, respectively). This contrasting outcome was well expected quite similar to our previous work 41 as after cellular loss, the structural part of cartilage becomes unresponsive and inert to the surrounding atmosphere.
There have been numerous instances of using variety of xenografts in cartilage tissue engineering.42-44 Also, acellular cartilages are being parallelly employed9,43,45 but mostly limited to in vitro analysis. Despite the importance of in vitro experiments, results of in vivo analysis play an influential role for any successful clinical applications as there is a noticeable difference between the two environments. Promising results of in vitro analysis under controlled laboratory conditions may work differently under in vivo atmosphere in contact to host. Therefore, considering this fact, our experiments particularly concentrated on the in vivo applications to monitor what exactly occurs in real situation under direct contact with host cells as these data will provide a clear view of host-graft relationship that is what finally counts for prolonged clinical trials. Fresh and decellularized cartilages were placed subcutaneously on rabbit and left in situ for 1 and 3 months. On gross observation of native cartilage implanted group, initiation of tissue necrosis was observed after 1 month, which strongly developed after 3 months where a typical foreign body response was noticed causing inflammation and deformed structure of the xenograft. This was further confirmed microscopically by H&E staining where the specimen showed presence of vacuolation, macrophages, and degenerated stages of surrounding host tissue. In contrast, the decellularized cartilage explants were monitored to maintain their basic structure without accumulation of any immunogenic cells as assessed by H&E staining. Although, initial results of 1M explants depicted few infiltrated macrophages surrounding the graft but this occurrence might be explained as a normal event due to host’s early immune response against foreign body material. 46 This may happen because of decellularization, which not only has abandoned the functional cellular part of the materials but also has indirectly suppressed the immune response triggered by the host cells post grafting. Therefore, a typical morphology of the sections exhibited compatibility of the decellularized materials with the host tissue, irrespective of the chemical agents, that is, Na-deoxycholate and GndHCl ( Fig. 7.IV ). Meanwhile, the collagen, elastin, and sGAG staining of decellularized explants post 1 and 3 months revealed systematic orientation of collagen and elastic fibers, proteoglycan was also moderately available. In contrast, the untreated cartilage tissue explants showed uneven arrangement of collagen and elastic matrix, limited appearance of proteoglycan and tissue degeneration as a consequence of necrosis and graft rejection.
The corresponding biochemical assessment of sGAG, collagen content depicted similar data that supported the former ECM study. A correlated study was conducted with a group of normal healthy animal, and from the quantitative analysis of DNA, collagen, and sGAG content, it was clearly seen that despite of having lesser concentration of DNA, sGAG, and collagen, possibly described as an effect of host’s early immune response in contact to cartilage material, which later on faded away, eventually all of these increased to give a final value similar to normal group unlike untreated explanted tissue which gave decreased values possibly because of graft rejection or tissue necrosis.
In conclusion, this investigation has led to the development of a biocompatible decellularized cartilage matrix that influenced the proliferation and migration of surrounding host cell into the matrix resulting in rigorous matrix synthesis and enhanced biomechanical properties. 47 In the field of cartilage tissue engineering, the utilization of xenografts, unlike autografts and allografts is limited. We intended to develop non immunogenic, biocompatible acellular caprine cartilage matrices to be utilized in osteochondral defects and transplantation applications. Having said that, more elaborate future studies are required to explore the regenerative capacity of the decellularized matrices in reconstruction of cartilage defects as well as its biomechanical alterations in such stressed environment.
Footnotes
Acknowledgments and Funding
Authors wish to express their sincere thanks to both Honorable Vice Chancellors of Jadavpur University and West Bengal University of Animal and Fishery Sciences, Kolkata; the directors of IIT Roorkee, IIT Kharagpur, and CSIR-CGCRI, Kolkata for their support for carrying out this work. Thanks are also due to the personnel who helped during all characterizations reported in the article. The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The total in vivo animal trial was conducted in accordance with the “Principles of Laboratory Animal Care” from the Institutional Animal Ethical Committee (IAEC), West Bengal University of Animal and Fishery Sciences (WBUAFS), West Bengal, India (Permit No. IAEC/59(i) dated July 27, 2018).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.