Abstract
Objective
To describe histological and metabolic characteristics of glenohumeral joint (GHJ) articular cartilage and compare to knee and ankle joints.
Design
Macroscopically healthy human humeral head, glenoid, knee, and ankle articular cartilage were obtained from tissue donors (N = 16, 9 males, 7 females; age 45-78 years), within 24 hours of death. Gross morphology of each joint was assessed using Collins grading. Cartilage explants were removed from the entire surface of each joint, cultured for 48 hours with or without interleukin-1β and processed for histology with Safranin O, proteoglycan (PG) synthesis/content, and polymerase chain reaction for collagen II, aggrecan, and SOX9. Results were compared between uncultured and cultured controls and across all 3 joints.
Results
Structural differences were seen on histology between GHJ cartilage and knee and ankle cartilage of the same Collins grade, specifically, depletion of Safranin O staining in the extracellular matrix. Treatment of glenoid and humerus specimens with IL-1β demonstrated a trend toward decreased PG synthesis in each explant but this decrease did not reach significance. There was no significant difference in PG synthesis between humerus, glenoid, knee, and ankle samples at baseline, day-0 control, 48-hour control, and 48 hours after treatment with 0.1 ng or 10 ng of IL-1β. There were no significant increases in collagen II, SOX9, and aggrecan expression in glenoid and humeral head cartilage samples treated with IL-1β compared to baseline controls.
Conclusions
GHJ articular cartilage did not significantly differ from ankle or knee cartilage with regard to PG synthesis and gene expression. However, it did differ in its histological appearance in normal state.
Introduction
Articular cartilage has limited regenerative capacity as it is an avascular tissue with restricted supply of oxygen, blood, and nutrients. 1 The most common cause of cartilage degeneration is a loss of balance between anabolic and catabolic processes.2,3 Osteoarthritis (OA) is a degenerative process affecting articular cartilage, synovium, joint capsule, and bone in part mediated by interleukin-1β (IL-1β).4,5 IL-1β serves as both an autocrine and paracrine signaling factor to disrupt cartilage homeostasis by inhibiting the synthesis of aggrecan, promoting release of inflammatory mediators, and stimulating matrix degrading enzymes such as matrix metalloproteinases (MMPs) that serve to breakdown proteoglycans (PGs), a key component of articular cartilage extracellular matrix (ECM).3,4,6-8
While certain joints more frequently develop symptomatic OA such as the hip, metatarsophalangeal joints of the hand, and knee, others including the ankle and shoulder experience symptomatic osteoarthritis less often.5,9-11 Although the incidence of articular cartilage injury cannot be directly equated to symptomatic OA, it is accepted that cartilage degeneration precedes OA. To date, few studies have compared the biochemical composition and response to inflammatory mediators commonly seen in the development of OA between different human joints. Epidemiological, radiological, and pathoanatomical studies have clearly demonstrated a significantly lower incidence of OA in the ankle joint compared to the tibiofemoral joint.5,12-14 As authors have noted, anatomical and biomechanical differences exist between the knee and ankle joint with the ankle joint being more congruent and stable than the knee under load, which may in part contribute to the disparity in OA. 5 A previous study in cadaveric human tissue demonstrated that chondrocytes within ankle cartilage have a decreased response to catabolic factors including IL-1 when compared to knee cartilage. 12 Furthermore, ankle chondrocytes synthesize PGs at a higher rate than that found in knee cartilage, suggesting a greater capacity for repair in the ankle. 5 However, no studies have extended this work to glenohumeral joint, a joint that has historically inconsistent outcomes of cartilage reparative and restoration procedures leading to increasing frequency of arthroplasty.15-20 While many cartilage surgical techniques that have been studied in the shoulder have been shown to be effective in the knee and ankle, the glenohumeral joint possess unique challenges given that the etiologies of disease are often different (i.e., frequently following instability events 21 ), and the thickness of articular cartilage is notably different as compared to the knee and ankle.20,22-27 In addition, our surgical experience is that the glenohumeral joint appears to be disproportionately more vulnerable to surgical insults such as simple shoulder arthroscopy compared to other joints. A greater understanding of the biochemical and morphological nature of glenohumeral joint articular cartilage is an important and necessary process to help improve treatments and direct further research.
The purpose of this study was to describe histological, morphological, and metabolic characteristics of glenohumeral joint articular cartilage and, whenever possible, compare to knee and ankle joints from the same donors. A secondary objective was to compare the response of articular cartilage to IL-1β between the 3 joints in human specimens with regard to gene expression, PG content, histology, and immunohistochemistry.
Materials and Methods
Reagents
Dulbecco’s modified Eagle medium (DMEM), Hams F-12 medium (F12), fetal bovine serum (FBS), penicillin/streptomycin/fungizone (PSF) composed of 10,000 units/mL penicillin, 10,000 µg/mL of streptomycin, and 25 µg/mL of fungizone, gentamicin concentration 100 µg/mL, and Trizol reagent were purchased from Invitrogen (Carlsbad, CA). QScript cDNA SuperMix was purchased from Quanta Biosciences (Beverly, MA). RT 2 qPCR Primer Assay (330001) for SOX9 (PPH02125A, Accession No. NM_000346.3), and housekeeping gene GAPDH (PPH00150F, Accession No. NM_001256799) were purchased from QIAGEN (Germantown, MD). Collagen type II (Accession No. NM_001844.4) and aggrecan (Accession No. NM_001135.3) were purchased from Integrated DNA Technologies (San Diego, CA). SYBR Green was purchased from Biorad (Hercules, CA). IL-1β (Cat# 201-LB) was purchased from R&D systems (Minneapolis, MN). 35 S-sulfate was obtained from Perkin-Elmer (Boston, MA), and alcian blue (8GX) dye was purchased from Biorad (Hercules, CA). Hoechst 33258 dye (Cat# 09460) and 1,9-dimethylmethylene blue dye (Cat# 03610) were purchased from Polysciences, Inc. (Warrington, PA). Antibodies for Immunohistochemistry—Aggrecan, monoclonal (Cat# AB186414); Collagen II, polyclonal (Cat# AB34712); Collagen type X, monoclonal (Cat #182563); and SOX9, monoclonal (Cat# AB195550)—were purchased from Abcam (Cambridge, MA). Ultra-Sensitive ABC Peroxidase Rabbit IgG Staining Kit (Cat# 32054) was purchased from Fisher Scientific (Pittsburgh, PA). All other used chemicals were molecular biology grade and purchased either from Sigma (St. Louis, MO) or Fisher Scientific (Pittsburgh, PA).
Tissue Culture
Macroscopically healthy human humeral head, glenoid, knee, and ankle cartilage were obtained from tissue donors (N = 16, 9 males, 7 female between the age of 45 and 78 years), through the Gift of Hope Organ & Tissue Donor Network (Itasca, IL) within 24 hours of death ( Table 1 ).
Donor Demographics and Tissue Explant Information a .

PCR = polymerase chain reaction.
Most, but not all, donors had bilateral joints available.
Donors had no known history of joint disease and only macroscopically normal joints were used for the study. Of note, one patient had a small, isolated grade II femoral chondral defect which was included in polymerase chain reaction (PCR) for comparison. As all samples were cadaveric the Deceased Subject Rule was applied, and no institutional review board (IRB) review and approval were necessary from our institution as stated in the IRB exempt letter. Gross morphology of each joint was assessed using Collins grading.28,29 Full-thickness human articular cartilage was dissected from the articular surface and 4 mm explants were made using a biopsy punch. Baseline was defined as the day the tissue was received. All cultures were carried out in serum. The explants were kept for 5 days in steady state in DMEM/F12 in a 1:1 ratio supplemented with 10% FBS, 1% PSF, and 0.2% gentamicin (complete medium). The medium was changed every other day for 5 consecutive days to reach steady state conditions. 4 The cultures were maintained at 37°C in a humidified atmosphere of 5% CO2. After 5 days in complete medium, the medium was replaced with fresh complete medium with or without interleukin-1β (IL-1β; 0.1 ng/mL or 10 ng/mL). 3 The treatment duration was for 48 hours.
Proteoglycan Synthesis
After 48 hours of IL-1β treatment, the cultured explants were pulse-labeled with 10 uCi/mL 35 S-sulfate for 4 hours, after which the culture media and explants were collected separately and stored at −20°C. 35 S-sulfate PGs in the media and papain digested explants were quantified by liquid scintillation counting following a rapid filtration assay, as previously described. 30 The concentration of 35 S-sulfate PGs synthesized was normalized to the cellular DNA content measured by the DNA assay using Hoechst 33258 dye. 31 This method utilizes digested aliquots of cartilage mixed with dye solution and fluorescence emission that has been shown to be sensitive to as little as 6 ng of DNA. PG synthesis is quantified in counts per minute normalized to microgram of DNA.
Proteoglycan Content
After 48 hours of IL-1β treatment, the cultured media and explants were collected separately. Media was not digested and used as is. The explants were digested with papain using the protocol of Masuda et al. 30 To assay PG content in both culture media and papain-digested explant samples, the DMMB assay was utilized. 32 Values of PG content in media and explant were normalized to the DNA content measured in the corresponding explant sample. 31 Proteoglycan content is quantified in micrograms and normalized to microgram of DNA.
Histologic Analysis
Explants were fixed in 4% paraformaldehyde, dehydrated, embedded in paraffin, sectioned in 6-µm sections with a microtome, and processed for histology and immunohistochemistry. For histologic study, the tissue sections were stained with Safranin O for PGs and fast green for proteins. 33 Integrity of the cartilage surface and its underlying matrix were analyzed with a Nikon Eclipse E800 (Nikon Inc., Garden City, NY) microscope for surface changes, abnormalities in cellularity, and patterns of Safranin O staining reflective of PG distribution.
Immunohistochemistry
The same serial sections used for histologic analysis were used for immunohistochemistry. Prior to primary antibody, slides were incubated at 60°C in a water bath in a 10 mM sodium citrate buffer, pH 6.0, for 20 minutes to unmask the antigen. Sections were incubated overnight at 4°C with primary antibody: anti-aggrecan (2 µg/mL), anti-collagen II (5 µg/mL), and anti-SOX9 (5 µg/mL). Biotinylated secondary antibody was used from the ABC kit for 1 hour followed by the ABC reagent for 30 minutes and DAB substrate for no more than 10 minutes (visual control for staining). The slides were washed, dehydrated, and mounted. Evaluation and documentation of the results was done with a Nikon Eclipse E800 microscope with a Spot 2 camera; the pictures were taken with the MetaMorph software program (Universal Imaging Corporation, Downingtown, PA).
RNA Isolation
Total RNA was isolated using Trizol reagent, as previously described. 34 Thermal cycling process with SYBR Green I was performed using CFX Connect Real-Time System (Bio-Rad). RNA isolation was performed with the following steps (2 step Amp+Melt):
95°C—2 minutes
95°C—0.05 seconds
60°C—30 seconds
Go to Step 2—39 times
95°C—0.05 seconds
Melt curve 65°C to 95°C, increments 0.5°C for 0.05 seconds
4°C forever in the end-hold
Four to 5 disks of 4 mm of cartilage were used to obtain 1 µg of RNA from which to make complementary DNA. Tissue was flash frozen in liquid nitrogen and stored at −80°C until RNA isolation was ready to be performed. Livak and Schmittgen’s method was used to analyze the relative changes in gene expression from real-time quantitative polymerase chain reaction experiments. 35 The housekeeping gene used for normalization was glyceraldehyde-3-phosphate dehydrogenase (GAPDH).
Statistical Analysis
Kolmogorov-Smirnov testing for normality of distribution was used to determine if variables were parametric or nonparametric. Independent sample t test was used to determine mean difference between 2 independent parametric variables and Mann-Whitney U testing was used for nonparametric variables. Furthermore, paired-samples t test was used to determine mean difference within each sample group at different time points, and Kruskal-Wallis H testing for comparisons between the different types of cartilage explants for nonparametric quantitative variables at multiple time points. Post hoc pairwise analysis was used if Kruskal-Wallis H analysis demonstrated a significant difference between 3 or more variables. All reported P values are 2-tailed, with an alpha level set at 0.05 for statistical significance (SPSS version 23.0; IBM Corp., Armonk, NY).
Results
Among collected joints, there were fewer normal shoulder joints than knee or ankle joints based on Collins grade. Even when normal based on modified Mankin score 36 and Collins grade, there were changes in the histological appearance of cartilage from humerus and glenoid. At grade 0 there was evidence of depletion of Safranin O staining in the ECM with apparent increased metabolic activity within territorial matrix evidenced by the intense red staining around chondrocytes unlike normal appearance of the cartilage from the knee and ankle ( Table 2 ).
Histologic Images of Safranin O Staining of Extracellular Matrix for Glenoid, Humeral Head, Knee, and Ankle Articular Cartilage for Collins Grades 0 and 2 Samples a .
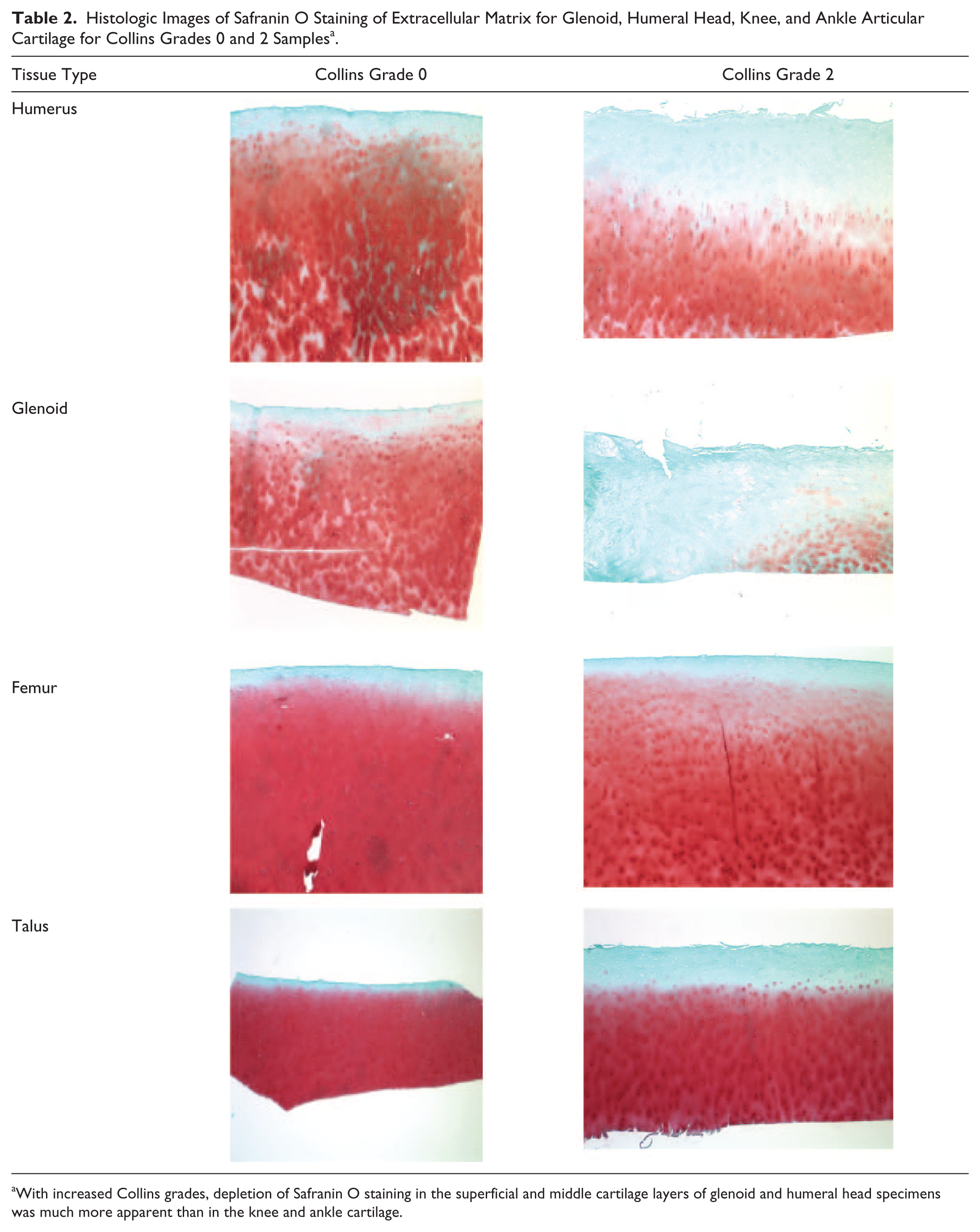
With increased Collins grades, depletion of Safranin O staining in the superficial and middle cartilage layers of glenoid and humeral head specimens was much more apparent than in the knee and ankle cartilage.
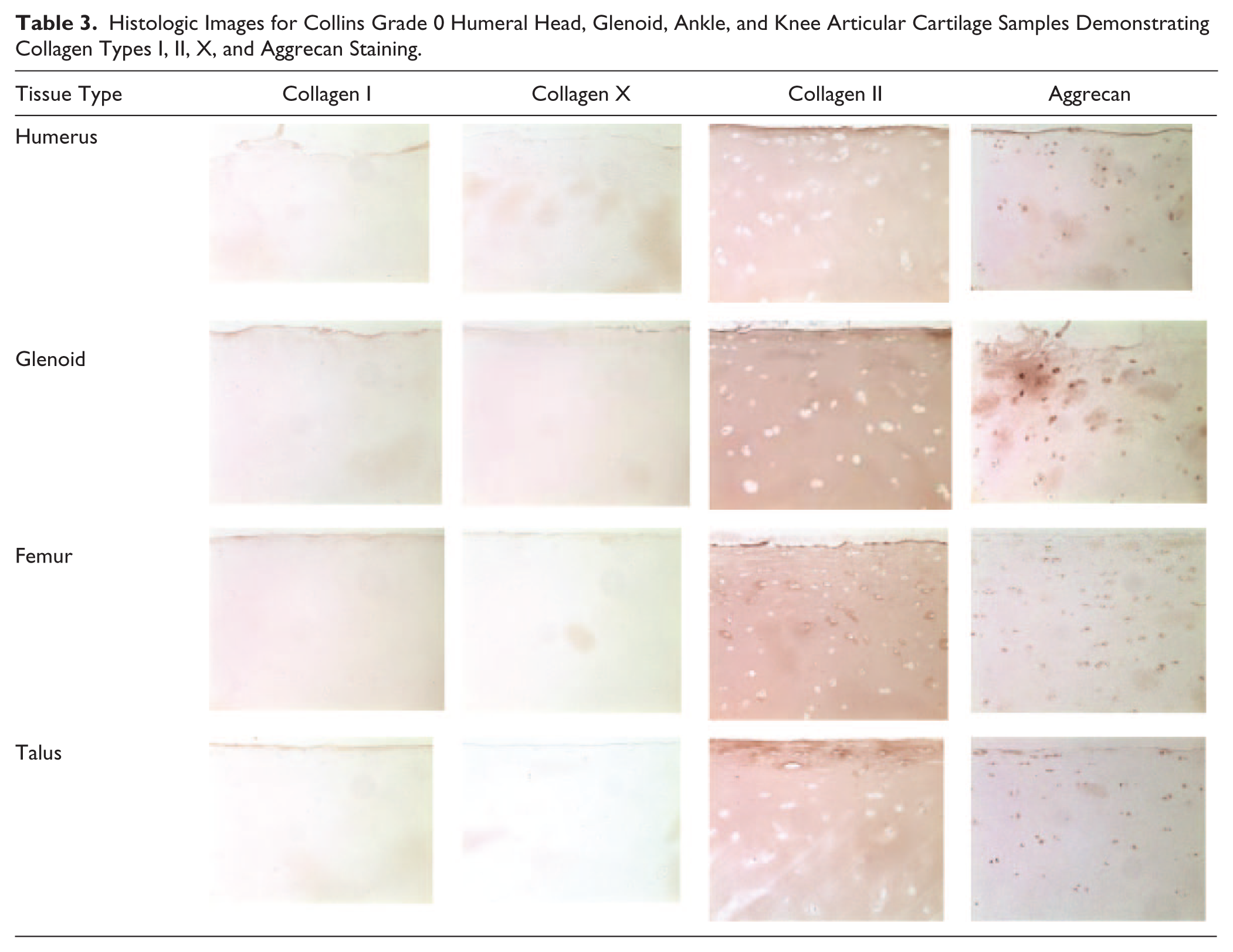
Immunohistochemistry (IHC) staining for collagen types I and X were negative in humeral head, glenoid, ankle, and knee specimens. Staining was positive in all joints for collagen type II and aggrecan in Collins grade 0 samples ( Table 3 ).
Histologic Images for Collins Grade 0 Humeral Head, Glenoid, Ankle, and Knee Articular Cartilage Samples Demonstrating Collagen Types I, II, X, and Aggrecan Staining.
The treatment of glenoid and humerus specimens (Collins grades 0-1; N = 4) with IL-1β showed a trend toward an approximately 2-fold decrease in PG synthesis when normalized to µg of DNA of each explant as compared to 48-hour control specimens (N = 4); however, this decrease in comparison to 48-hour control specimens did not reach significance in humerus samples at 0.1 ng/mL IL-1β (P = 0.294) or 10 ng/mL IL-1β (P = 0.226). Likewise, glenoid samples approached but did not reach statistically significant decreases in PG synthesis when treated with 0.1 ng/mL IL-1β (P = 0.071) or 10 ng/mL IL-1β (P = 0.065) as compared to 48-hour control glenoid specimens. There was no difference in PG synthesis rates between humerus and glenoid samples (Collins grades 0-1) at baseline, day 0 control, 48-hour control, and 48 hours after treatment with 0.1 ng/mL or 10 ng/mL of IL-1β (P > 0.05 for all). When humerus and glenoid samples were compared to femur and ankle samples from the same donors (Collins grades 0-1) at the same treatment stages, no significant differences in total proteoglycan synthesis were identified using Kruskal-Wallis H testing (Baseline, P = 0.124; Day 0, P = 0.897; 48-hour control, P = 0.384; 48-hours of IL-1β 0.1 ng/mL, P = 0.431; 48 hours of IL-1β 10 ng/mL, P = 0.172). Despite the lack of differences in statistical significance, metabolism in humerus and glenoid showed a tendency to be higher than of that in the knee or ankle based on the level of PG synthesis ( Fig. 1 ).
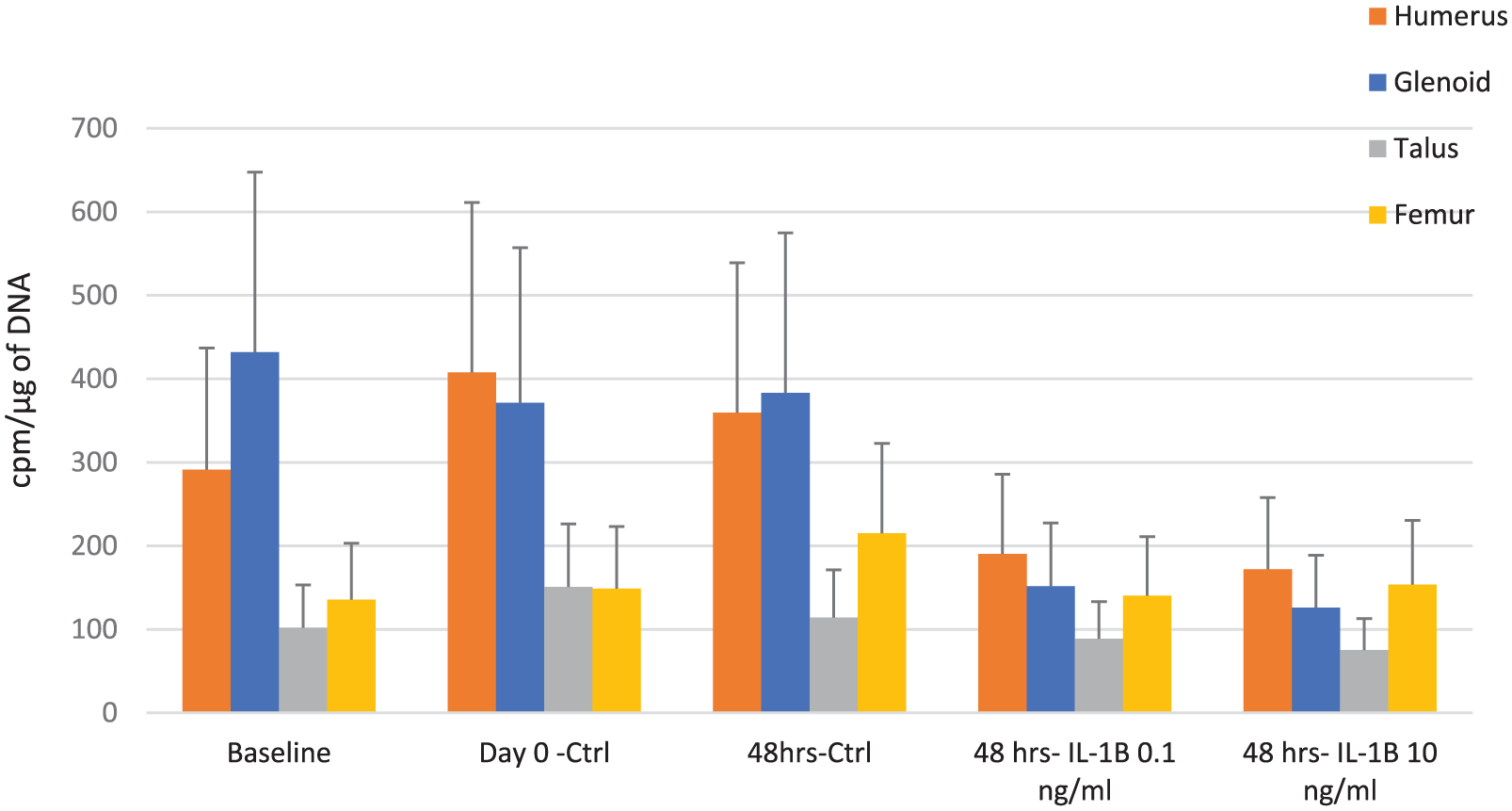
Proteoglycan synthesis per microgram of DNA between glenoid, humeral head, ankle, and femur explants for bilateral joints from 4 donors.
When PG content was evaluated separately in media and matrix, no significant differences at any point between control samples for both the humerus and glenoid specimens and those treated with either concentration of IL-1β either in media or in matrix (P > 0.05 for all) were identified. When compared to ankle and femur specimens (Collins grades 0-1) using one-way Kruskal-Wallis H analysis, there was no significant difference in PG content in media at any point ( Table 4 ). Similarly, when comparing Collins grades 0-1 samples for differences in PG content in matrix only at the same end points, no significant differences were found between the 4 joints ( Table 4 ).
Proteoglycan Content Quantified in Micrograms in the Media and Matrix Separately Normalized per Microgram of DNA of Humeral Head, Glenoid, Femur, and Talus Cartilage Explants from the Same 4 Tissue Donors at 4 Different Treatment Points a .
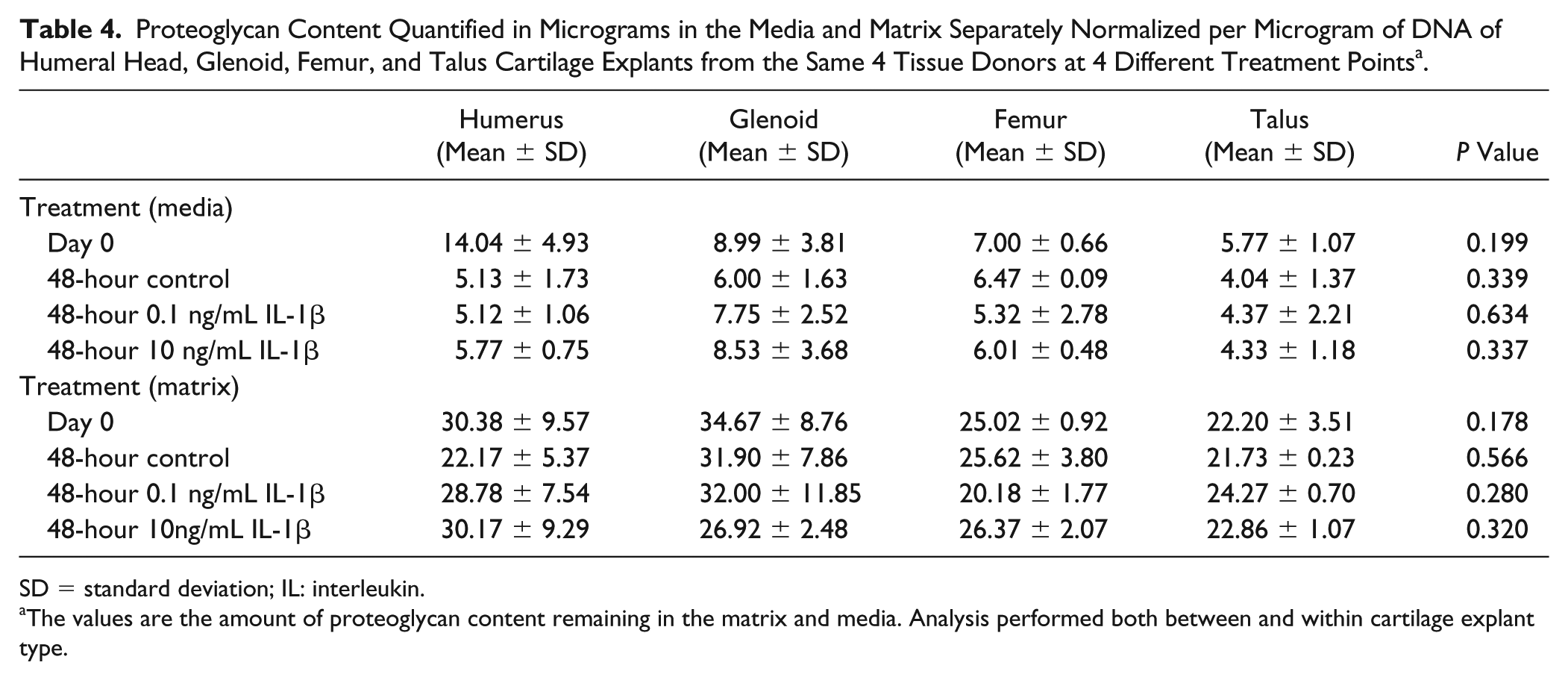
SD = standard deviation; IL: interleukin.
The values are the amount of proteoglycan content remaining in the matrix and media. Analysis performed both between and within cartilage explant type.
PCR gene expression at baseline for collagen type II, SOX-9, and aggrecan was compared between knee and ankle cartilage explants from the same donors (N = 3 knee; N = 4 ankle) to PCR gene expression relative to the humeral head explants from the same donors (N = 10). Of note, humeral head samples ranged between Collins grades 0-1, ankle samples ranged from Collins grades 0-1, and knee samples ranged from Collins grades 1-2. There were no significant differences in gene expression for collagen type II, aggrecan, and SOX9 between knee and ankle relative to the humeral head specimens at baseline (i.e., the day the tissue was received); however, there was a trend toward a higher aggrecan expression in the ankle and grade 2 knee cartilage (Fig. 2A-C).
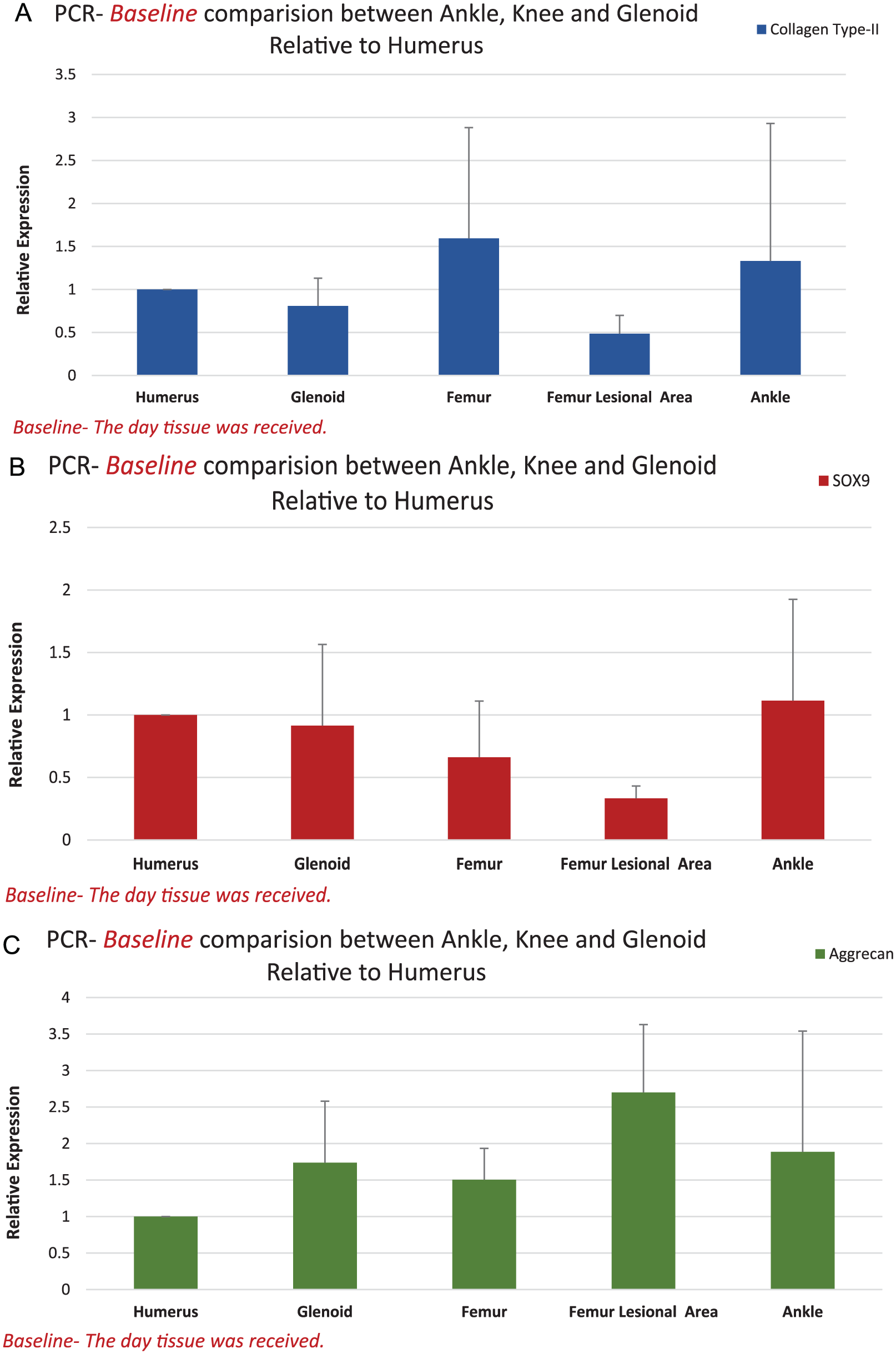
Baseline comparison of gene expression using polymerase chain reaction between Collins grades 0-1 ankle explants, humeral head Collins grades 0-1 explants, femur Collins grades 0-1, damaged femoral lesion area Collins grade 2, and glenoid Collins grades 0-1 explants for (
With culture alone, expression of all 3 genes did not demonstrate significant changes in expression when compared to baseline control within both glenoid or humerus explants. IL-1β treatment of glenoid and humerus cartilage did not significantly increase expression of genes of interest (P > 0.05 for all) ( Figure 3A-F ), though some modulation in gene expression was noticed.
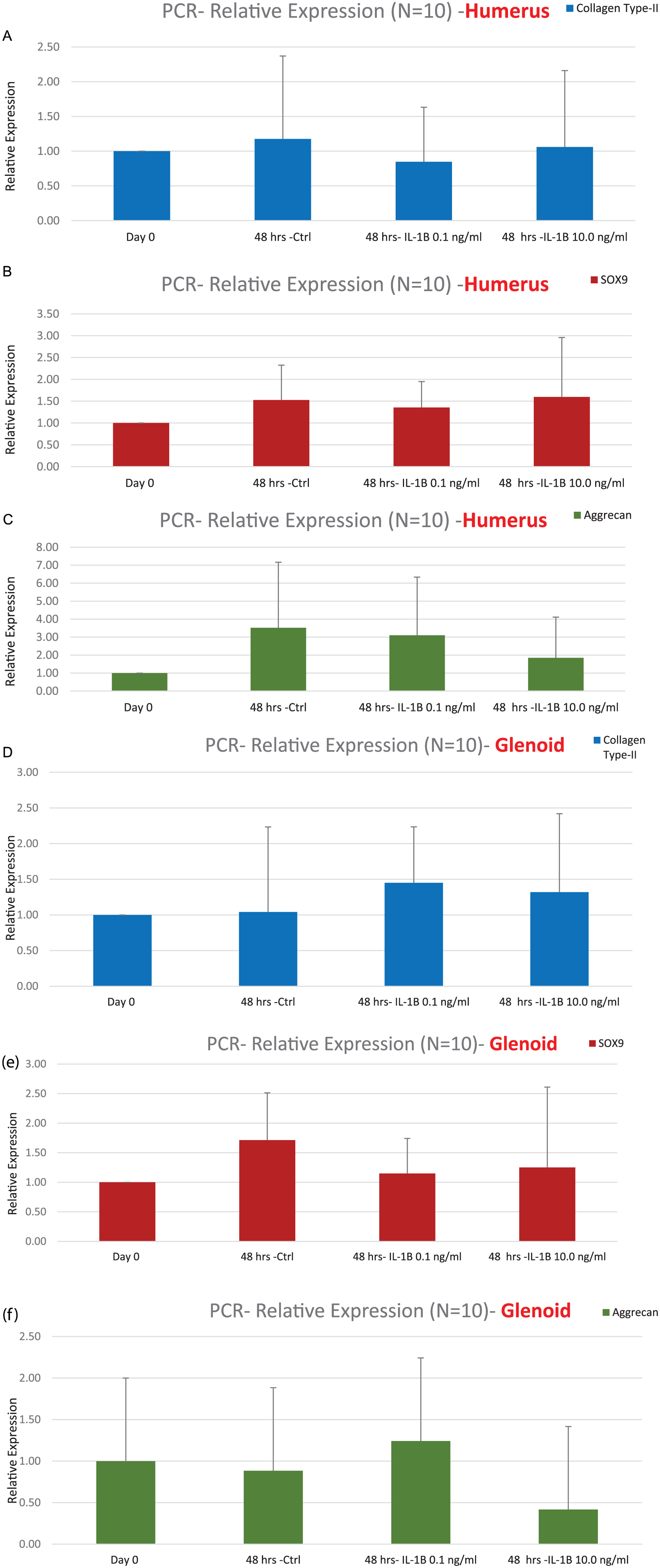
(
Discussion
This is the first attempt to compare morphological appearance and responses of human articular cartilage from 3 anatomic locations: shoulder, knee and ankle. The principle findings of this study demonstrate subtle and distinct structural differences between human glenohumeral joint articular cartilage and articular cartilage from knee and ankle joints. Specifically, humeral head and glenoid cartilage demonstrated early degenerative changes in the extracellular matrix appearance already at normal morphological and histological state. In addition, the more intense Safranin O staining was found around chondrocytes and early evidence of matrix depletion in the interterritorial matrix of the humeral head and glenoid cartilage than in the knee and ankle samples suggesting potential remodeling processes characterized by deposition of PGs in the territorial matrix. However, glenohumeral joint cartilage did not demonstrate a significant difference in PG synthesis when compared to similar Collins grade cartilage samples taken from the knee and ankle joints of the same human donor at all observational time points. Taken together, these findings indicate that normal shoulder cartilage might be distinct in the level of its metabolism even at morphologically normal state.
The extracellular matrix, comprised largely of sulfated glycosaminoglycans, afford articular cartilage compressive stiffness under loads that is critical to preventing chondral and subchondral damage. With regard to ankle and knee cartilage matrix, authors have previously shown the dynamic stiffness to be higher in the ankle than the knee due to higher sulfated glycosaminoglycan content and lower water content in the ankle. 37 This higher sulfated glycosaminoglycan content has also been shown to correlate with a higher rate of PG synthesis by ankle chondrocytes as compared to knee chondrocytes. 38 These findings have not previously been compared to glenohumeral joint cartilage, which is a more unstable and non–weight-bearing joint. While the higher rate of PG synthesis was not shown to be statistically significant in the present study when compared to knee and ankle cartilage, this study may serve as a pilot study for which future investigations may build upon to evaluate in more detail and with the use of a larger sample size the metabolic and biomechanical differences of the cartilage from glenohumeral joint that might be responsible for poor healing ability of the joint.
The treatment of glenoid and humerus specimens with 0.1 ng/mL and 10 ng/mL of IL-1β did lead to a decrease in overall PG synthesis; however, these findings approached but did not reach statistical significance. This may be due to a wide variation in morphology of cartilage specimens, even when the specimens were of the same gross morphology grade, and a relatively low number of observations. Nevertheless, this study serves as a launch point from which future investigations may target and direct research to better elucidate the response of shoulder articular cartilage to injury and/or catabolic mediators.
On PCR analysis, IL-1β treatment of glenoid and humerus cartilage did not demonstrate significantly elevated expression of key matrix genes involved in chondral repair, specifically collagen type II, SOX9, and aggrecan. The limited increase in expression of key chondral matrix genes may suggest limited intrinsic reparative capacity of the glenohumeral joint when subjected to inflammatory stressors, which may in part explain inconsistent results with reparative procedures such as microfracture. 17 Furthermore, the outcomes of cartilage palliative, reparative, and restorative procedures of the glenohumeral joint have yielded inconsistent results with some authors noting significant improvements at short-term and mid-term outcomes while others demonstrate less promising results.15-20 While this study is unique and contains the largest number of samples to date, future studies with larger sample sizes would be beneficial to try and elucidate what effect, if any, IL-1β or the combination of various pro-inflammatory mediators have on the expression of key matrix genes to gain a better understanding of how glenohumeral joint articular cartilage responds to catabolic environment.
Authors have previously demonstrated differences between articular cartilage in the knee and ankle, and glenohumeral joints, namely, anatomical differences with regard to cartilage thickness. Specifically, the cartilage thickness of the humeral head has been reported to be 0.28 to 2.2 mm and the glenoid to be 1.4 to 2.4 mm,22,23 in contrast to the 3.3 to 4 mm thickness of the distal femur 24 and 1 to 2 mm of the talus.25-27 These anatomical differences along with differences in joint biomechanics may play a role in the etiology of focal and diffuse chondral diseases in each respective joint. Furthermore, as it pertains to the glenohumeral joint, the relatively thin articular surface is important to understand the pathogenesis of iatrogenic injury, for example, during thermal capsullorhaphy and for use of osteochondral grafts. 22
This study is unique in its scope trying to extend previous work comparing knee and ankle cartilage to that of the glenohumeral joint. In addition, these findings may serve to spawn future research into better understanding the mechanisms of glenohumeral joint degeneration and restoration to improve treatment algorithms.
There are several limitations of the present study. First, while a concerted effort was taken to compare cartilage samples from the joints taken from the same donor, this was not always possible. Second, there was less tissue available from the glenoid than from humerus or knee and ankle, which limited the number of treatment groups and analyses performed on glenoid cartilage, thus affecting the comparison between the joints. Third, the cartilage explants were harvested from the entire surface of each joint with no specific attention to the location within joint. This definitely could influence the outcomes; however, the need of multiple explants for each metabolic analysis dictates chosen experimental design. Finally, this study is limited most significantly by the number of donor specimens available for inclusion, which is commensurate with previous studies but is nevertheless a limiting factor to a greater power of analysis due to a wide range of “normal” state for glenohumeral joint cartilage.
Conclusion
Glenohumeral joint articular cartilage did not significantly differ from that of ankle or knee with regards to PG synthesis and gene expression. However, it did differ in its histological appearance in normal state. These data suggest that glenohumeral joints can show signs of early osteoarthritis in older, deceased individuals with histologically and macroscopically normal-appearing knee and ankle cartilage. More work is needed with larger numbers of samples to further understand the biology of glenohumeral joint articular cartilage and its responses to catabolic cytokines.
Footnotes
Authors’ Note
This work was performed at the Rush University Medical Center.
Acknowledgments and Funding
The authors would like to acknowledge the Gift of Hope Organ and Tissue Donor Network, Itasca, Illinois, and the donor’s family. We also thank Dr. Arkady Margulis for assisting in tissue procurement and Dr. Lev Rappoport for assistance with histological studies. The study was supported in part by the Klaus Kuettner chair for osteoarthritis research (SC).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not sought for the present study because all samples were cadaveric.
Informed Consent
Informed consent was not sought for the present study because all samples were cadaveric.
