Abstract
Objective
Osteoarthritis is a progressive joint disease characterized by cartilage degradation and synovial inflammation. Presence of cartilage fragments in the joint due to degradation of cartilage is thought to be associated with local inflammatory response and progressive osteoarthritic process. Understanding the mechanism by which cartilage fragments elicit this destructive process should aid in designing novel therapeutic approaches. Therefore, objective of current study is to establish an in vitro model to examine the cross-talk between chondrocytes and cartilage fragments–stimulated macrophages.
Design
Cartilage fragments were prepared from femoral head cartilages of mice and analyzed using a scanning electron microscope and particle size analyzer. Bone marrow–derived macrophages were co-cultured with cartilage fragments and chondrocytes using transwell co-culture system. Macrophage inflammatory mediators in supernatant of cultures were determined by ELISA and gene expression of macrophages and chondrocyte were quantified by qRT-PCR.
Results
Shapes of cartilage fragments were irregular with sizes ranged between 0.54 and 55 μm. Macrophages cultured with cartilage fragments released significantly higher concentrations of TNF-α, IL-6, and NO than those of mock and control. Consistently, gene expressions of TNF-α, IL-6, and MMP-9 were significantly increased in stimulated macrophages. The elevation in production of pro-inflammatory molecules in stimulated macrophages cultures were coincident with an increase in gene expression of chondrocyte MMP-13, iNOS, and IL-6.
Conclusion
We developed an in vitro co-culture model to study the impact of stimulation of macrophage by cartilage fragments on the expression of chondrocyte carbolic factors. Our results revealed that cartilage fragments triggered macrophages inflammatory response that enhanced the production of chondrocyte catabolic factors.
Introduction
Osteoarthritis (OA) is one of major degenerative joint diseases causing disability of aging population. The disease constitutes a significant economic burden on patients and public health services over the world as estimated that 10% of the world population suffer from this significant clinical problem. OA is characterized by progressive degeneration of cartilage, menisci and ligaments, and synovial inflammation. The development of OA occurs in response to inappropriate mechanical stress, eventually leading to greater catabolic activity in the joint associated with destruction of cartilage, synovial membrane and subchondral bone, and inflammation of the synovium.1,2 Chondrocytes are the major cellular components of articular cartilage and are essential for regulating cartilage homeostasis. On the other hand, in pathological condition, they produce an array of carbolic factors, including matrix metalloproteinases (MMP) and aggrecanases (ADAMTS) after exposing to mechanical forces and intrinsic and extrinsic inflammatory mediators. Synovial inflammation (synovitis) is another crucial factor that contribute to sustained catabolic activity in the joint and the progression of OA.3-5 Despite of advances in understanding the pathogenesis of OA, the precise mechanism by which chondrocytes promote the catabolic activity in the joints is not fully understood. A better understanding of the molecular pathways and signals responsible for this pathological condition should lead to development of more effective therapeutic intervention.
Cartilage wear “cartilage fragments” in joint space due the degradation of cartilage is considered as the principal feature of OA and has been targeted as indicator for diagnosis of OA.6,7 Histological findings of human OA tissues revealed that the presence of cartilage fragments in the synovium is accompanied with local cellular reaction and inflammatory response.8,9 In line with these findings, intra-articular injection of cartilage fragments induces synovitis and osteoarthritic changes in animal models.10,11 They can activate immune system through danger-associated molecular patterns (DAMPs) after interaction with pattern recognition receptors (PRRs). Therefore, it is pertinent to investigate the potential role of cartilage fragments in the etiology and progression of osteoarthritic disorders.
Given the fact that macrophages are the main immune cells in the synovial lining of joints, we believe that these cells are the first cells encountering cartilage fragments and causing excessive synovial inflammation associated with progressive OA.12-14 Once macrophages recognize cartilage fragments by their receptors, they produce endosomal enzymes to digest fragments after phagocytosis resulting in endosomal damage and persistent inflammasome activation. Consequently, activated macrophages release inflammatory mediators that facilitate the recruitment of other cells, such as lymphocytes, leading to chronic synovitis, joint pain, swelling and functional incapacity. Moreover, there is an increasing body of evidence that activated macrophages secrete enzymes that alter chondrocyte function leading to cartilage degeneration.15,16 We speculate that the presence of cartilage fragments in the synovial fluid triggers macrophage inflammatory response that leads to cartilage degeneration. Therefore, the purpose of the current study is to establish an in vitro model to examine the impact of cartilage fragments-stimulated macrophages on the expression of chondrocyte catabolic factors. Our model allowed to study the cross-talk between chondrocytes and activated macrophages by cartilage fragments.
Materials and Methods
Ethics Statement
All procedures for animal experiments were performed based on the ethical guidelines approved by the Institute of Animal Care and Use Committee of the Hokkaido University Graduate School of Medicine (approval ID: 17-0085).
Preparation of Cartilage Fragments
Cartilage fragments were prepared from femoral head cartilages of 4-week-old specific pathogen–free (SPF) C57BL/6 male mice (n = 40; Clea, Tokyo, Japan) as earlier discrbed. 17 Isolated cartilages were rinsed by ice-cold phosphate buffered saline (PBS) and then crushed by Multi Beads Shocker (Yasui Kikai, Osaka, Japan) for 1 minute at 2500 rpm. The condition was optimized to obtain cartilage fragments with sizes comparable to these isolated from OA synovial fluid.6,7 The resulting cartilage fragments were washed twice by ice-cold PBS and then examined by Toxin Sensor Single Test Kit (Genscript, Piscataway, NJ, USA) for endotoxins detection. Levels of endotoxins were below the detection limit of kit (0.015 EU/mL). The shape and surface topography of cartilage fragments were assessed using a scanning electron microscope (S-4800 Hitachi High-Technologies Corporation, Tokyo, Japan). Distribution of fragments based on their sizes (equivalent circle diameter [ECD]) was analyzed by a particle image analyzer Morphologi G3 (Malvern Instruments, Worcester, UK). Thereafter, samples with sizes ranged between 0.5 and 55 μm were kept at −80°C for further use.
Co-culture of Macrophages Stimulated by Cartilage Fragments and Chondrocytes
Bone marrow cells (BMCs) were isolated from the long bone of the same mice sacrificed for cartilage fragments. 18 BMCs were cultivated in medium (GIBCO DMEM/F12 GlutaMAX-I, Invitrogen, LifeTech Austria, Vienna, Austria) with antibiotics (penicillin 200 U/mL, streptomycin 0.2 mg/mL, and amphotericin B 2.5 μg/mL; Sigma-Aldrich, St Louis, MO, USA) supplemented with 10% heat-inactivated fetal bovine serum (FBS; Sigma-Aldrich), 25 mg/L penicillin/streptomycin and incubated in a humidified atmosphere containing 5% CO2 for 24 hours. Thereafter, nonadherent cells were washed twice with ice-cold PBS buffer (Nacalai Tesque Inc., Kyoto, Japan) and differentiated into macrophages by culturing in the fresh medium supplemented with 50 ng/mL mouse recombinant macrophage colony-stimulating factor (MCSF; Peprotech, Tokyo, Japan) for 7 days. Cultures were regularly replenished with fresh medium every 3 days. Our protocol routinely resulted in >95% CD11b+ as verified by flow cytometric analysis (data not shown). Differentiated macrophages were detached on day 7 of culture by treatment with 1% trypsin–ethylenediaminetetraacetic acid (EDTA) solution (GE Healthcare, Chicago, IL, USA) for 5 minutes, washed with ice-cold PBS, and seeded onto 24-well plates at ratio 2 × 105. Macrophages were stimulated by cartilage fragments at ratio of 1:5 for 24 and 48 hours ( Fig. 1 ). Stimulation with ratios 1:1 and 1:2 showed only minor response with no significant difference as compared to control cultures (data not shown). Control cultures were cultivated with 3-µm latex beads (Sigma-Aldrich, St Louis, MO, USA) at same ratio (1:5) and mock cultures with medium alone. In parallel, freshly isolated chondrocyte 19 were seeded (2 × 105) onto cell culture transwell insert (Falcon cell culture inserts, BD, Franklin Lakes, NJ, USA) and added to a 24-well plate of macrophage cultures. An additional culture was prepared for cartilage fragments and chondrocytes without macrophages in the transwell culture system to assess the effect of cartilage fragments on chondrocytes with no direct contact. Furthermore, the supernatant of cell cultures and cells were harvested for cytokine quantification and gene expression analysis after 24 and 48 hours. All experiments were repeated at least 2 times to obtain reproducible data.
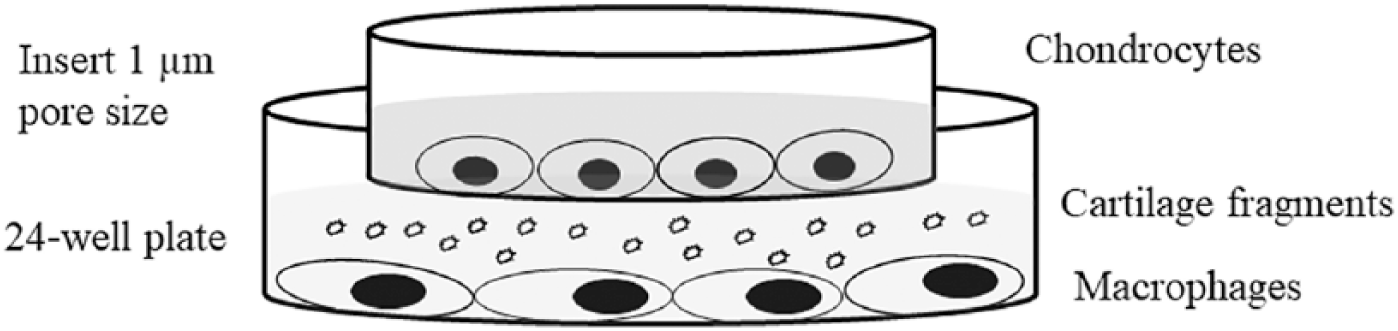
In vitro model for co-culturing cartilage fragments–stimulated macrophages and chondrocytes using transwell system.
Electron Microscopic Examination of Stimulated Macrophage
Differentiated macrophage from C57BL/6 mice (1 × 105) in culture medium were transferred into Eppendorf 1.5 mL tube and exposed to cartilage fragments at ratio of (1:5) for 10 hours using a rotating mixer. Thereafter, cells were fixed in 1.5% formaldehyde and 1% glutaraldehyde in cacodylate buffer at pH 7.4 for 15 minutes. The cells were subsequently rinsed in 3 series buffers of cacodylate buffer, 1% osmium tetroxide buffer, and cadodylate buffer. The cells were then dried in a graded ethanol series followed by embedding in epoxy resin. Thin sections were stained with 2% uranyl acetate for 10 minutes at room temperature, rinsed in d-H2O, and stained for 6 minutes with Reynolds lead citrate stain (Sigma-Aldrich). The cells were visualized using a Hitachi H-7100 transmission electron microscope (JEM-1400 Plus Nihon Denshi, Tokyo, Japan) at 80 kV.
Quantification of TNF-α and NO
Detection of tumor necrosis factor–α (TNF-α; R&D System, Minneapolis, MN, USA), interleukin-1β (IL-1β), and IL-6 (Thermo Scientific, Waltham, MA, USA) in the supernatant of cultures was performed using commercial ELISA (enzyme-linked immunosorbent assay) kits according to each manufacturer’s instructions. On the other hand, detection of nitric oxide (NO) was performed by Griess reagent (Promega, Madison, WI, USA) according to the manufacturer’s instructions.
RNA Isolation and Quantitative Real-Time Polymerase Chain Reaction
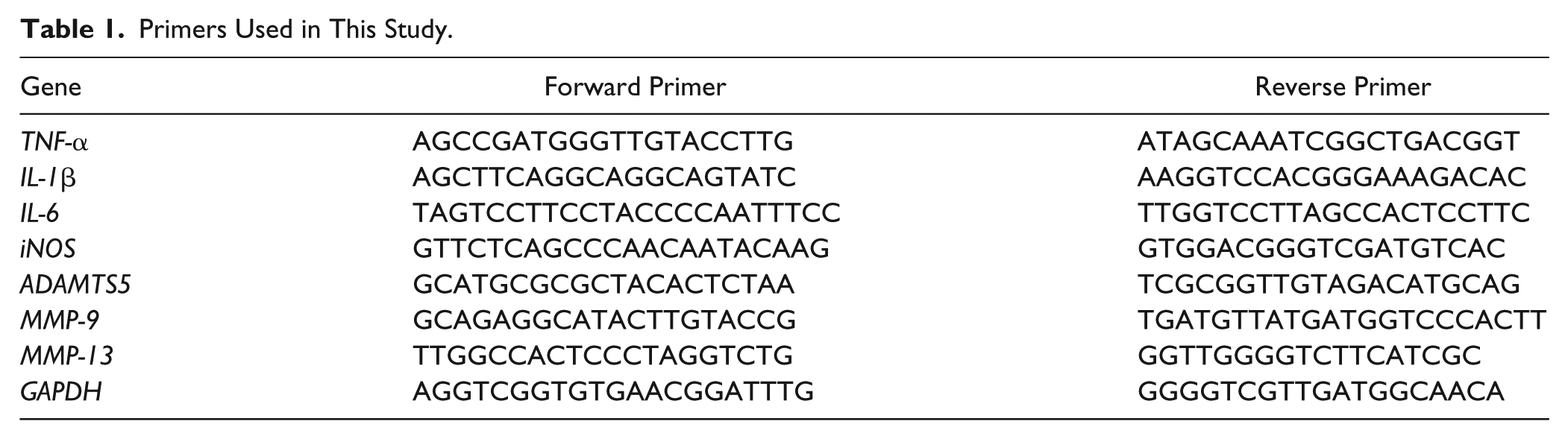
Cultured macrophages or chondrocytes were harvested and lysed with TRIzol Reagent (Invitrogen Carlsbad, CA, USA) for RNA purification. RNA was purified using RNeasy Mini kit columns (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. Next, purified 0.5 µg DNA-free RNA was reverse transcripted using GoScript Reverse Transcriptase and random primer (Promega), according to manufacturer’s instructions. Quantitative real-time polymerase chain reaction (qRT-PCR) was performed using SYBR Premix Ex Taq II (Takara, Shiga, Japan) with specific primers ( Table 1 ) on a Thermal Cycler Dice System 2 (Takara). Gene expression of target gene was calculated after normalizing to the expression of GAPDH housekeeping gene using the 2−ΔΔCt method.
Primers Used in This Study.
Statistical Analysis
Statistical analysis for differences between control and test groups were determined by Student t tests (GraphPad Software Inc., La Jolla, CA, USA) and were considered statistically significant when P < 0.05.
Results
Characterization of Cartilage Fragments
To obtain cartilage fragments with equivalent sizes and shapes to these isolated from OA synovial fluids, isolated cartilages were crashed, and the resulting wears were analyzed by scanning electron microscopy and particle image analyzer. Of note, the resulting wears had irregular shapes with sponge-like surface topography ( Fig. 2A ), and exhibited sizes ranged between 0.54 and 55 μm with a mean of 3.11 μm ( Fig. 2B ). These results demonstrate a successful preparation of cartilage fragments with comparable sizes and shapes to these isolated from OA synovial fluids.
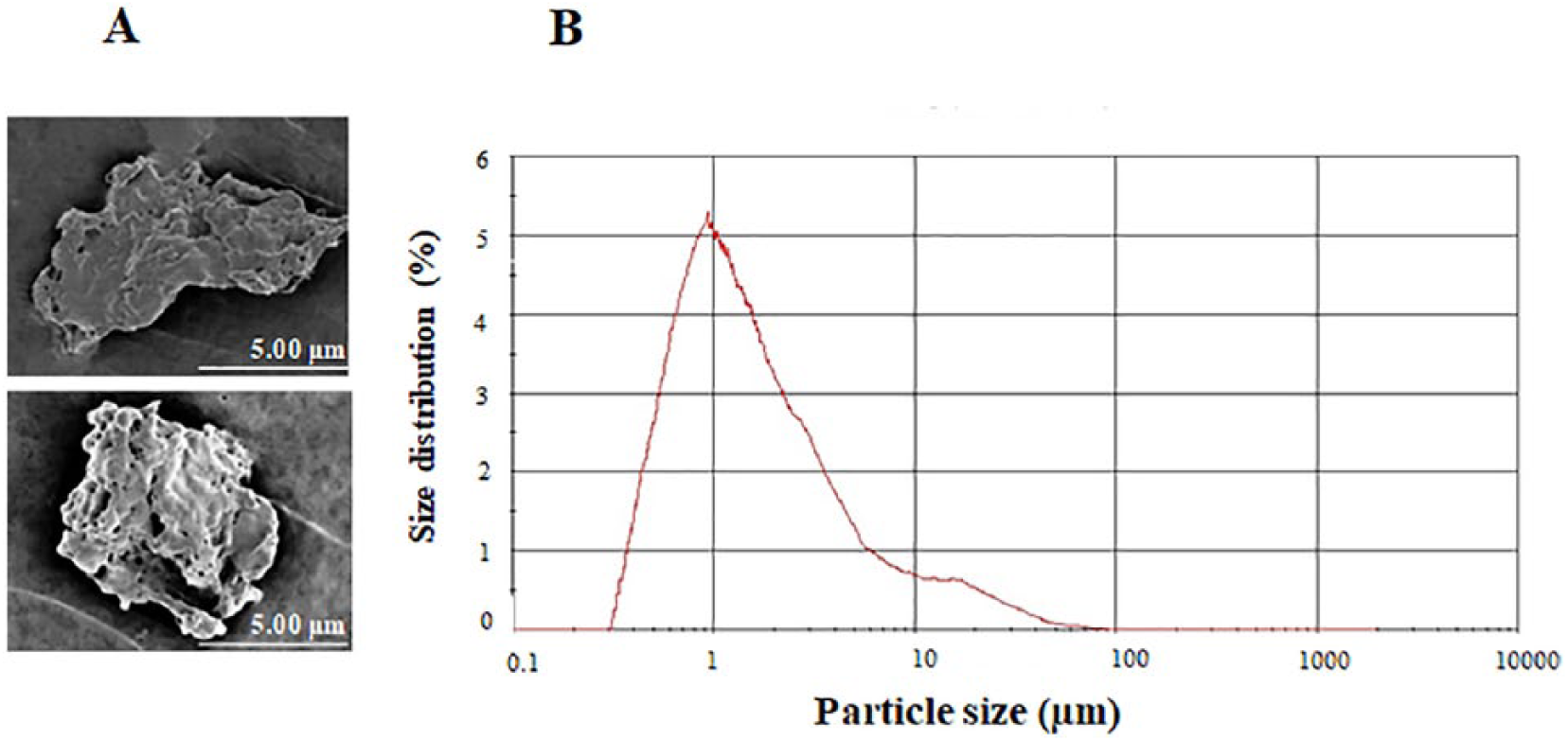
Characterization of cartilage fragments. (A) Scanning electron microscopic analysis of cartilage fragments showing the shape and sizes. (B) Size distribution of cartilage fragments as determined by a particle image analyzer. Scale bar is 5 µm.
Activation of Macrophages by Cartilage Fragments
To evaluate the activation state of macrophages after exposure to cartilage fragments in vitro, differentiated macrophages from mouse BMCs were co-cultured with cartilage fragments derived from same animals and their responses were analyzed using transmission electron microscopy for cell morphological, ELISA for inflammatory mediator production, and qRT-PCR for gene expression analyses. Strikingly, activated macrophages due to fragments uptake exhibited distinctive morphologies of active phagocytes including irregular shapes and enlarged phagolysosome ( Fig. 3 ). Moreover, co-cultured macrophages with cartilage fragments for 24 and 48 hours produced substantial amount of TNF-α, IL-6, and NO, but not IL-1β as compared with mock and control cultures ( Fig. 4A and data not shown). Consistently, gene expressions of pro-inflammatory markers, including TNF-α, IL-6, and MMP-9, but not IL-1β were significantly elevated in macrophages cultured with cartilage fragments after 48-hour culture ( Fig. 4B and data not shown). Together, these results reveal that macrophages are activated in the presence of cartilage fragments and produce common pro-inflammatory mediators known to be associated with progression of OA.
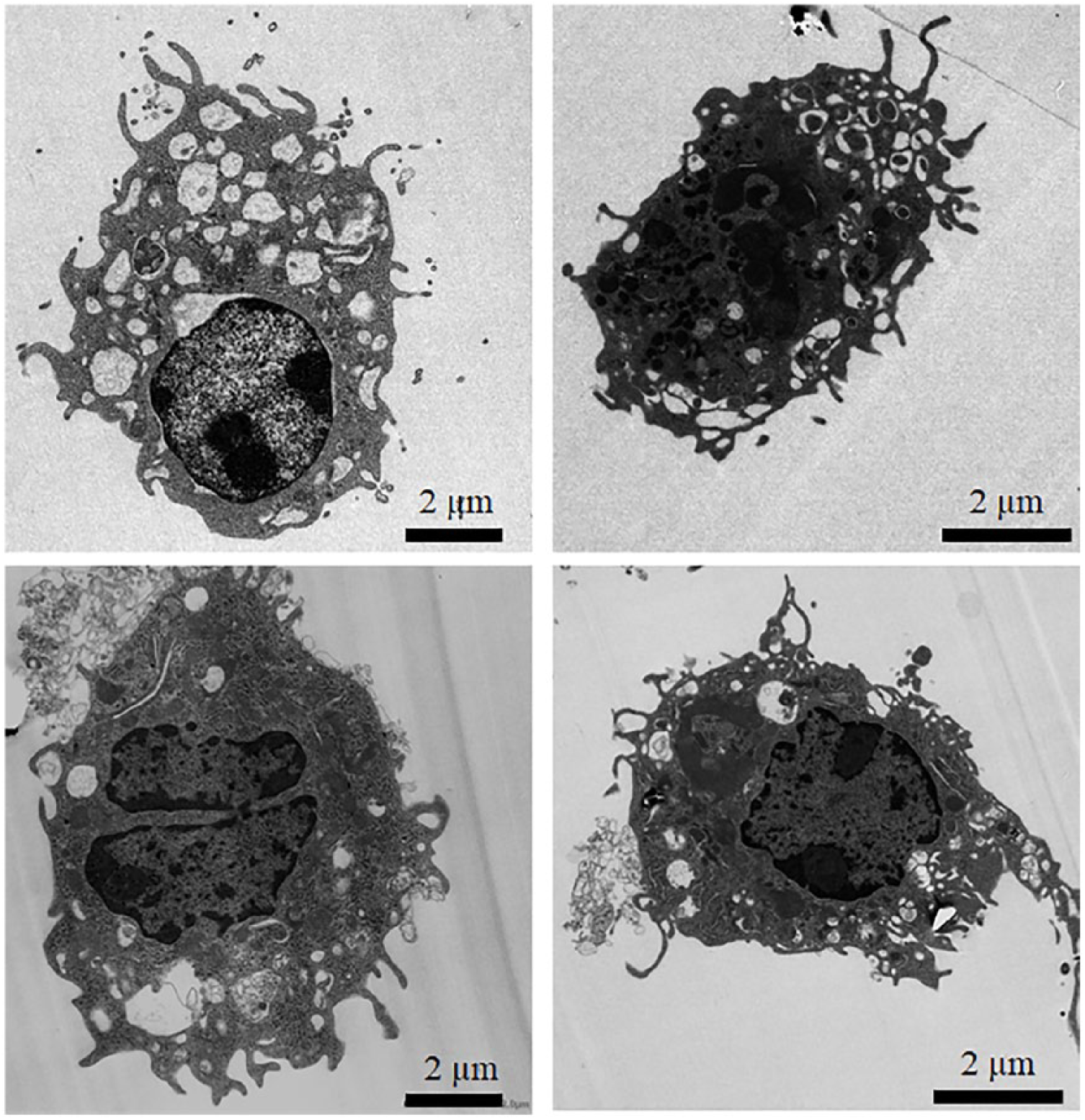
Morphological characterizations of macrophages exposed to cartilage fragments in vitro analyzed by transmission electron microscopy. Representative images for activated macrophages by cartilage fragments that exhibited distinctive morphologies of activated phagocytes, including irregular shapes and enlarged phagolysosome. Scale bar is 2 µm.
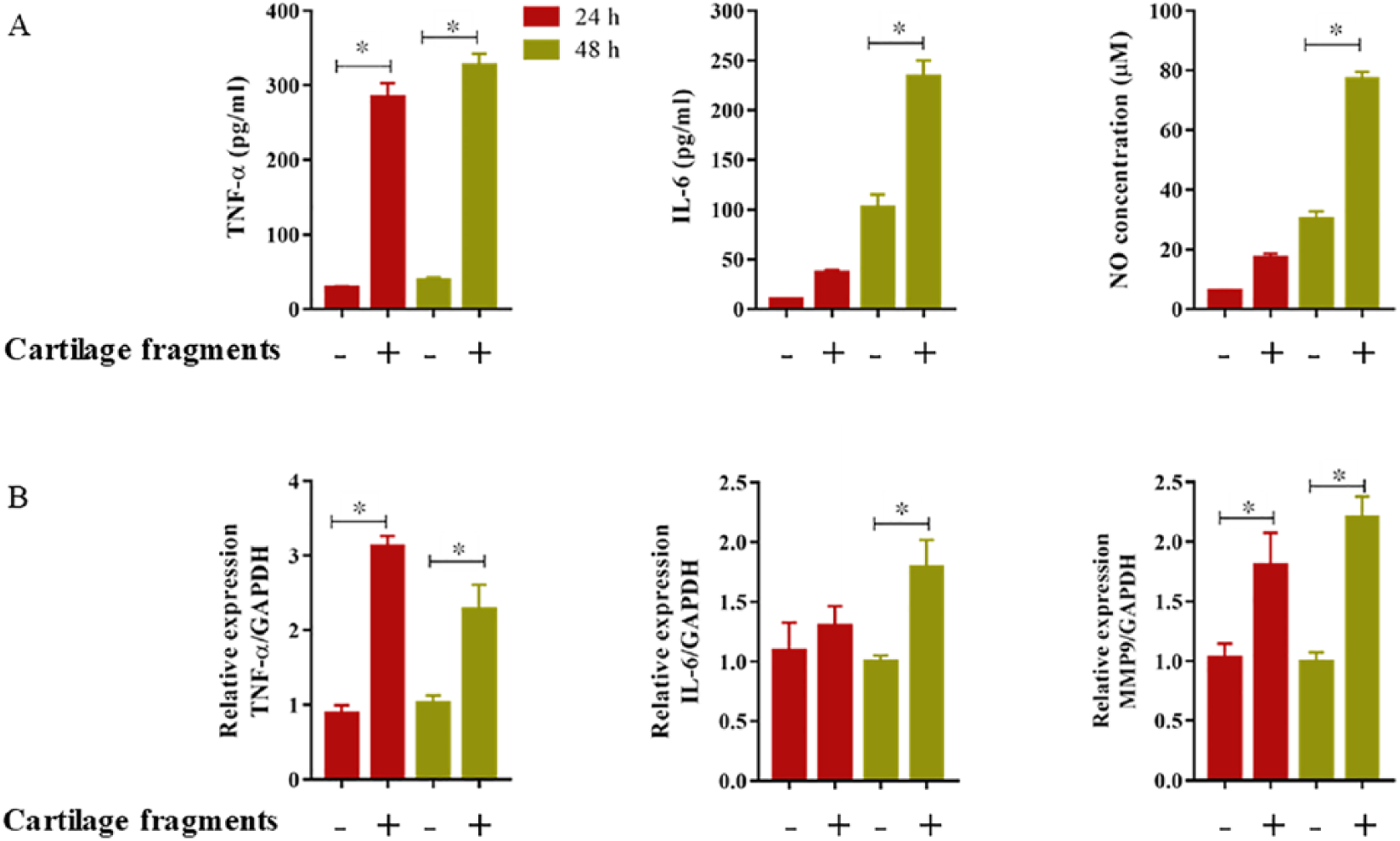
Detection of macrophage pro-inflammatory mediators, including tumor necrosis factor–α (TNF-α), interleukin-6 (IL-6), and nitric oxide (NO) after exposure to cartilage fragments for 24 and 48 hours. (A) Protein detection in the supernatant of macrophages cultures. (B) Gene expression in macrophages as analyzed by quantitative real-time polymerase chain reaction (qRT-PCR). The values are shown as the mean ± standard error of the mean (mean ± SEM) of 4 measurements (n = 4). (+) indicates presence of cartilage fragments and (−) indicates absence of cartilage fragments. Statistical significances were determined using Student t test and were considered statistically significant when P < 0.05. *Indicates significant difference.
Elevation of Chondrocyte Catabolic Factors in Co-culture Model with Stimulated Macrophage by Cartilage Fragments
To further assess the impact of stimulated macrophages on chondrocytes in the co-culture model, catabolic factors gene expression in the co-cultured chondrocytes was analyzed by qRT-PCR. Interestingly, elevation in production of pro-inflammatory molecules in macrophages cultures were coincident with an increase in expression of catabolic factors of chondrocytes, including MMP-13, inducible nitric oxide synthase (iNOS), and IL-6 but not ADAMTS5 ( Fig. 5 and data not shown). These increases were significantly different in co-cultured chondrocytes for 48 hours. It is worth noting that expressions of catabolic factors of co-cultured chondrocytes with cartilage fragments in the transwell culture system (no direct contact) and these co-cultured with macrophages stimulated with latex beads were not significantly increased after 24 or 48-hour culture (data not shown). These data show the significant impact of stimulated macrophage by cartilage fragments on the behavior of chondrocytes and suggest their role in progression of OA.
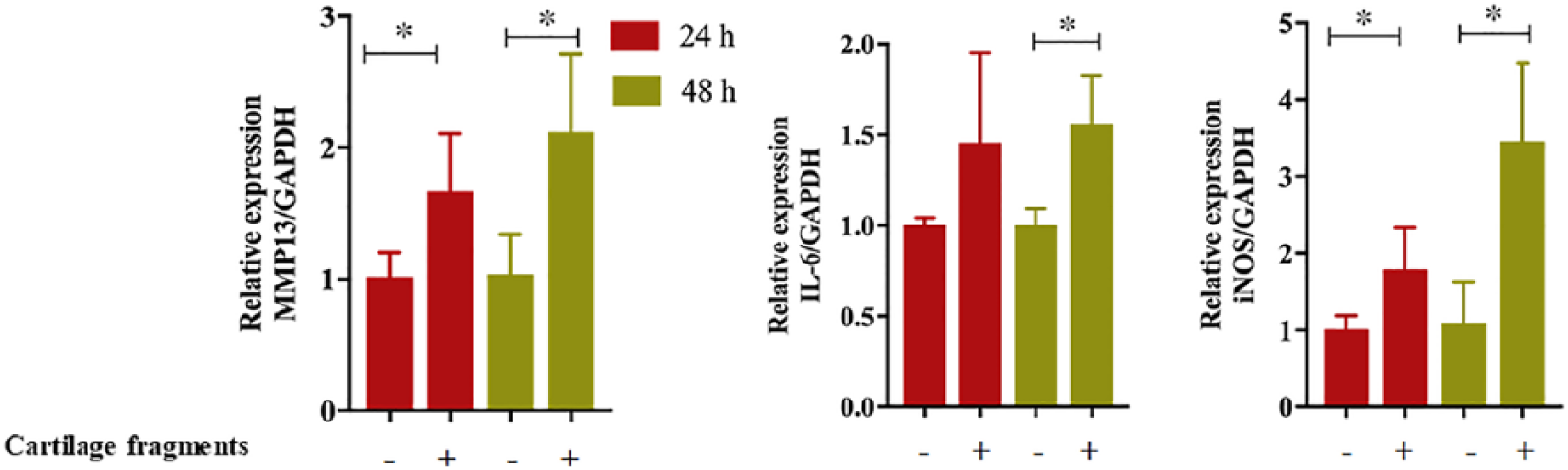
Expression of chondrocyte catabolic factors in co-culture system with macrophages stimulated with cartilage fragments. Gene expression was detected in cultured chondrocytes by quantitative real-time polymerase chain reaction (qRT-PCR). (+) indicates presence of cartilage fragments and (−) indicates absence of cartilage fragments. The values are shown as the mean ± standard error of the mean (mean ± SEM) of 4 measurements (n = 4). Statistical significances were determined using Student t test and were statistically significant when P < 0.05. *Indicates significant difference.
Discussion
Osteoarthritis has been traditionally characterized as wear-and-tear disease; however, recent research has highlighted it as an immunopathological disease.20,21 The increases of chondrolytic mediators and the activation of innate immune system are recognized as the central features of disease.20-22 Continuous exposure of synovial macrophages to cartilage fragments eventually leads to an excessive inflammatory response with adverse effects on chondrocyte function. Therefore, we believe that a better knowledge defining the cross-talk between chondrocytes and activated macrophages in the presence of cartilage fragments should provide important clues for development of therapeutic intervention. To this end, we developed a co-culture model mimicking articular environment with stimulation by cartilage fragments to study of the impact of stimulated macrophage by cartilage fragments on the expression of chondrocyte carbolic factors associated degradation of cartilage.
Our co-culture model allowed us to study the contribution of activated macrophages by cartilage fragments to chondrocyte inflammation, disfunction and degeneration. Cartilage fragments triggered macrophages to produce inflammation mediators, including TNF-α and IL-6 but not IL-1β. This eventually led to an elevation in chondrolytic factors such as MMP-9, MMP-13, and iNOs, which are known as major markers of catabolism of OA cartilage. 23 These results can be explained based on the fact that TNF-α and IL-6 act as negative regulators for chondrocyte proliferation and articular cartilage metabolism. Likewise, IL-6 neutralization reduces cartilage damage in experimental murine OA.24,25 These findings are consistent with the concept that damaged tissues activate innate immune system and trigger inflammatory responses through DAMPs. DAMPs are identified by pattern recognition receptors such as Toll-like receptors and Nod-like receptors. In fact, damaged joint tissues, including extracellular matrices such fibronectin, aggrecan, biglycan, tnenascin C, and intracellular proteins necrotic cells are implicated in the activation of innate immune response and the chronic inflammation in OA.26,27
Another interesting finding in our study is that in co-culture model, catabolic factors of chondrocytes were not upregulated in the absence of macrophages. This highlights the importance of macrophages in the pathogenesis and progression of OA. Such results suggest that inhibition of activation of inflammatory macrophages by cartilage fragments may offer a novel strategy for controlling the progression of OA and cartilage degeneration. Although there is a consensus that macrophage-mediated inflammation is involved in catabolic processes of synovium, there was no study showing the contribution of cartilage fragments to these processes.3,13-16 Generally, intraarticular fragments isolated from knee have irregular shapes with micrometer sizes ranging between 1.4 and 234 μm.6,7 Response of macrophages is essentially driven by the size and shape of fragments. Macrophages can phagocytize foreign particles with sizes <10 μm, while larger sizes cannot be phagocytized but can initiate foreign body giant cell formation.28,29 Of significance, small and elongated fragments are expected to elicit higher and severe inflammatory responses.30,31 In line with this view, cartilage fragments used in our model were prepared to fell within a range of 0.54 to 55 μm that covers phagocytosable- and nonphagocytosable-sized fragments for mimicking actual in vivo condition of OA patients.
The major limitations in the current study include inability to use human samples in this model and small number of catabolic markers detected in chondrocytes. In fact, we were not able to prepare cartilage fragments from patients undergoing knee arthroplasty due to the damaged or inflamed condition of cartilage tissue in these patients. Moreover, we have detected the most common chondrocyte markers associated with joint catabolism to assess cross-talk between macrophages and chondrocytes in the presence of cartilage fragments. In our future study, we are planning to apply RNA-Seq technology to fully understand the molecular responses of the tested cells in our model.
Taken together, our co-culture model provides a useful tool mimicking in vivo condition of synovium to explore the mechanism of progressive OA and to identify potential molecular targets for therapeutic intervention. The presence of cartilage fragments in the joint seemed to contribute to progression of OA through triggering macrophage inflammatory responses. We believe that inhibiting the interaction between macrophages and cartilage fragments might reduce the progression of OA and cartilage degeneration.
Conclusion
We established an in vitro model to study the cross-talk between macrophages and chondrocytes in the presence of cartilage fragments. Cartilage fragments triggered inflammatory response of macrophages that enhanced catabolic factors of chondrocytes. Thus, inhibiting the activation of macrophages inflammatory response triggered by cartilage fragments may offer a novel strategy for controlling the progression of OA and cartilage degeneration. Our further studies aim at identifying novel therapeutic agents for blocking the adverse effects of stimulated macrophages on chondrocytes by the use of our current co-culture model.
Footnotes
Acknowledgments and Funding
The authors thank Dr. Zenta Joutoku, Dr. Shinji Matsubara, Dr. Ryosuke Hishimura, Mrs. Kayo Nakamura, Mrs. Eriko Tagishi, and Mrs. Mayumi Suzuki for their kind help and support in our laboratory. The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded by the Grant-in-Aid for Challenging Exploratory Research (17K19707) from Japan Society for the Promotion of Science.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Ethical Approval
All procedures for animal experiments were performed based on the ethical guidelines approved by the Institute of Animal Care and Use Committee of the Hokkaido University Graduate School of Medicine (approval ID: 17-0085).
