Abstract
Background
The repair of osteochondral lesions remains a challenge due to its poor vascularity and limited healing potential. Micronized cartilage matrix (MCM) is dehydrated, decellularized, micronized allogeneic cartilage matrix that contains the components of native articular tissue and is hypothesized to serve as a scaffold for the formation of hyaline-like tissue. Our objective was to demonstrate in vitro that the use of MCM combined with mesenchymal stem cells (MSCs) can lead to the formation of hyaline-like cartilage tissue in a single-stage treatment model.
Design
In group 1 (no wash), 250 µL MCM was reconstituted in 150 µL Dulbecco’s phosphate-buffered saline (DPBS) for 5 minutes. Group 2 (saline wash) included 250 µL MCM washed in 20 mL DPBS for 30 minutes, then aspirated to remove all DPBS and reconstituted in 150 µL DPBS. Group 3 (serum wash): 250µL MCM washed in 20 mL DPBS for 30 minutes, then aspirated and reconstituted in 150 µL fetal bovine serum. Each group was then added to 50 µL solution of MSC suspended in DPBS at a concentration of 1.2 × 106 cells/350 µL. After 3 weeks, the defects were extracted and sectioned to perform viability and histologic analyses.
Results
Stem cells without rehydration of the MCM showed almost no viability whereas near complete cell viability was seen after rehydration with serum or saline solution, ultimately leading to chondrogenic differentiation and adhesion to the MCM particles.
Conclusion
We have shown in this proof-of-concept in vitro study that MCM can serve as a scaffold for the growth of cartilage tissue for the treatment of osteochondral lesions.
Introduction
Articular cartilage, classified as hyaline cartilage, is made of chondrocytes, type II collagen, and proteoglycans to produce a smooth, low-friction gliding surface capable of withstanding compressive and shearing loads. Because of its lack of vascular supply and encasement of chondrocytes in matrix limiting migration, articular cartilage has limited healing potential. Microfracture surgery was introduced as a method to address full-thickness cartilaginous defects of the knee and has subsequently been used to treat osteochondral lesions of the talus (OLT).1,2 This technique involves penetration of the subchondral bone to stimulate extrusion of bone marrow to the defect site. However, microfracture alone results in the synthesis of both fibrocartilage and hyaline cartilage that fails to mimic the biomechanical properties and longevity of native hyaline cartilage.3,4 Though encouraging short-term data have been published, long-term results remain variable and prompts the search for superior techniques for the treatment of these lesions.5,6
More recent advances in surgical and laboratory techniques have resulted in the development of treatments, such as autologous chondrocyte implantation (ACI) and matrix-induced autologous chondrocyte implantation (MACI), aimed at restoring the native hyaline cartilage structure and function. These treatments, however, require multiple procedures for final implantation, excessive cost, and may require a suitable donor site with associated morbidity. Therefore, there remains a need for a cost-effective, one-step implantation system with limited patient morbidity to treat OLTs.
Biologic adjuncts, such as platelet-rich plasma, hyaluronic acid, and bone marrow aspirate concentrate (BMAC) can augment cartilage repair. 7 Mesenchymal stem cells (MSCs) found in BMAC have the ability to differentiate into chondrocytes and osteoblasts. 8 Cartilage repair treated with BMAC in animal models has been shown to more closely mimic the composition of native articular cartilage with improved integration into the defect.9,10 Early clinical data demonstrate the effectiveness of BMAC used in conjunction with other substrates to treat osteochondral lesions though high quality comparative studies remain lacking. 11
Numerous biologic and synthetic scaffolds have been proposed to provide the framework for cartilage ingrowth and repair. These options include bone graft substitute, hydroxyapatite-based scaffolds, and collagen-based scaffolds.11-13 Micronized cartilage matrix (MCM) (BioCartilage, Arthrex, Naples, FL) is a composed of dehydrated, decellularized, micronized allogeneic cartilage that is hypothesized to serve as a scaffold composed of native articular tissue components for the formation of hyaline-like tissue. Early results of MCM have shown promising results in the treatment of OLTs ( Fig. 1a ). 14 The objective of this study is to investigate the single-stage treatment of OLTs with a combination of MSC and MCM.
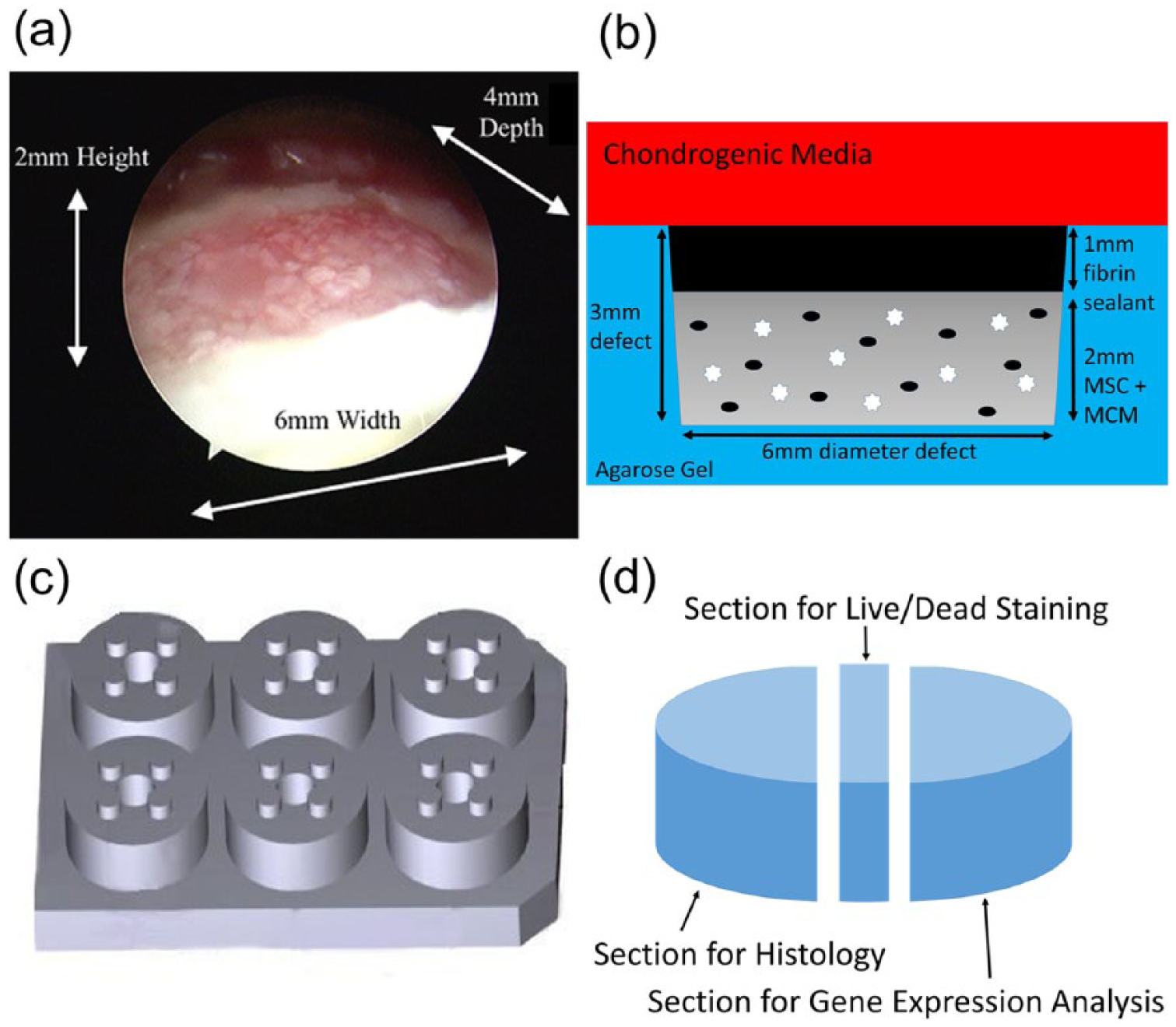
(
Materials and Methods
Cells/Viability
Human bone marrow–derived stem cells were obtained from a single donor at an associated stem cell bank within our institution and expanded in monolayer culture using standard protocols to a maximum passage of 4. There were 2 experimental time points at day 0 and 3 weeks. The time point at day 0 was to establish viability and baseline conditions of MSC mixed with MCM. Three experimental groups with matched control groups were tested for their biocompatibility using LIVE/DEAD Cell Viability Assay (Thermo Fisher Scientific, Waltham, MA) at time points 30 and 60 minutes. Experimental group 1 (no wash): 250 µL MCM reconstituted in 150 µL Dulbecco’s phosphate-buffered saline (DPBS) for 5 minutes was added to a 50 µL solution of MSC suspended in DPBS at a concentration of 1.2 × 106 cells/350 µL. Experimental group 2 (saline wash): 250 µL MCM was first prewashed in 20 mL DPBS for 30 minutes by combining the MCM and DPBS in 50 mL polypropylene centrifuge tubes and manually agitating for 30 minutes in order to hydrate the initially anhydrous MCM. The mixture underwent centrifugation for 2 minutes to settle the MCM out of solution and the DPBS was aspirated out to yield prewashed MCM. This was then reconstituted in 150 µL DPBS and finally combined with a 50 µL solution of MSC that had been suspended in DPBS at a concentration of 1.2 × 106 cells/350 µL. Experimental group 3 (serum wash): 250 µL MCM prewashed in 20 mL DPBS for 30 minutes was aspirated to remove all DPBS and then reconstituted in 150 µL fetal bovine serum (FBS) and added to a 50 µL solution of MSC that had been suspended in FBS at a concentration of 1.2 × 106 cells/350 µL. Each experimental group was matched with a control group identical to the experimental group but without MCM. Imaging was performed on a Nikon TE2000 inverted fluorescent microscope with 10× magnification and viability was quantified using ImageJ software as live cell counts normalized to live cell counts in controls.
Chondrogenic Culture
Once viability was established, we studied chondrogenesis after 3 weeks. To approximate the defect dimensions seen in OLT in vivo, we used a custom-designed and manufactured polysulfone mold to create circular (6 mm diameter by 3 mm depth) indentations in agarose gel (0.8%) (
Fig. 1b
). Each defect sat above an underlying 3 mm layer of agarose. There were 4 such defects per well of a 6-well plate, and each plate contained 2 wells of each of the experimental groups and the control group (
Fig. 1c
). Based on the viability assays, the MCM for all groups was first washed for 30 minutes in DPBS and aspirated. Experimental group defects were filled with 55 µL of (1) MSC (200,000 cells) in FBS and (2) MSC (200,000 cells) in FBS and MCM (0.8:1 FBS to MCM). The control group defects were filled with 55 µL of FBS and MCM (0.8:1 FBS to MCM). A layer of TISSEEL Fibrin sealant was placed to a height of 1 mm on top of each defect and allowed to set for 5 minutes, to simulate surgical practice. Finally, 6 mL of chondrogenic media (α-modified Eagle’s medium [αMEM] with dexamethasone [0.1 µM], transforming growth factor–β [TGFβ; 10 ng/mL],
Analysis
At the end of the 3-week time point, defects were extracted and divided into sections, each directed to a separated analysis ( Fig. 1d ). A 300 µm slice of each defect was set aside for live-dead staining, while the larger section was dedicated to histology analysis, as described by the protocols below.
Live-Dead Staining
The 300 µm slice of each defect was stained using the Live/Dead kit and Hoechst 33342, according to the manufacturer’s protocol. Images were taken using an inverted fluorescence microscope (Nikon TE 2000 or Zeiss XXX) with 10× objectives.
Histology
On harvesting, tissue samples were immersed in 4 mL 4% paraformaldehyde for 24 to 40 hours at room temperature for fixation. Following this, the fixative was removed; the samples were washed in deionized water, and then immersed in 5 mL 70% ethanol and stored at 4°C until embedding. Tissue samples were then sent out for embedding in paraffin and prepared for histological analysis. Histological analysis was performed using hematoxylin and eosin stain for cellularity and safranin-O stain for proteoglycans. We evaluated the fate of the micronized cartilage and whether it retained its proteoglycan and collagen content over time, whether it became repopulated by MSC and how well it integrated with the matrix secreted by the MSC.
Statistical Analysis
Statistical comparisons were made to control groups using analysis of variance with Dunnett’s test, in JMP Pro 12.1 software.
Results
After 3 weeks of culture in chondrogenic media, we observed that the combination of MSC and MCM grossly produced cohesive structures that were easily handled ( Fig. 2a ). Furthermore, histological analysis showed the presence of differentiated chondrocytes staining for proteoglycans which also adhered to MCM fragments ( Fig. 3 ). Without the MCM, the stem cells did not form viable constructs (not shown). In constructs that contained both MSC and MCM, the 3-week cell viability was more than 98%, with no dead cells visible in many constructs ( Fig. 2b and c ). MSC were almost immediately killed when added directly to the dry MCM ( Fig. 4a ). Rehydrating the MCM prior to the addition of cells was required to maintain the viability of the added MSC. There was no statistically significant difference between rehydration using serum or saline ( Fig. 4b and c ). The MCM itself generated a “dead cell” signal initially, therefore the normalized total number of live cells in each condition was used for statistical comparisons ( Fig. 4a ).
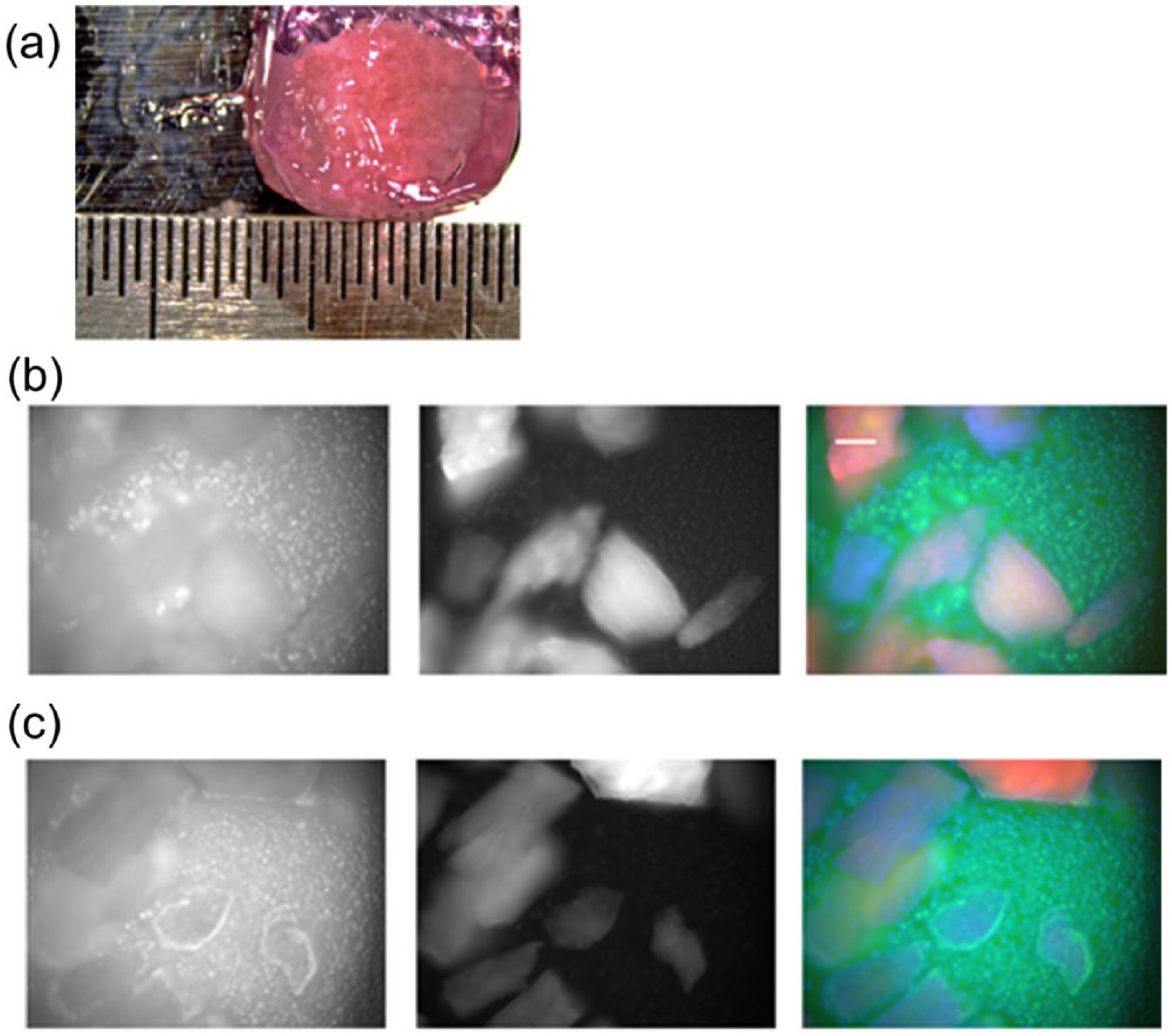
(
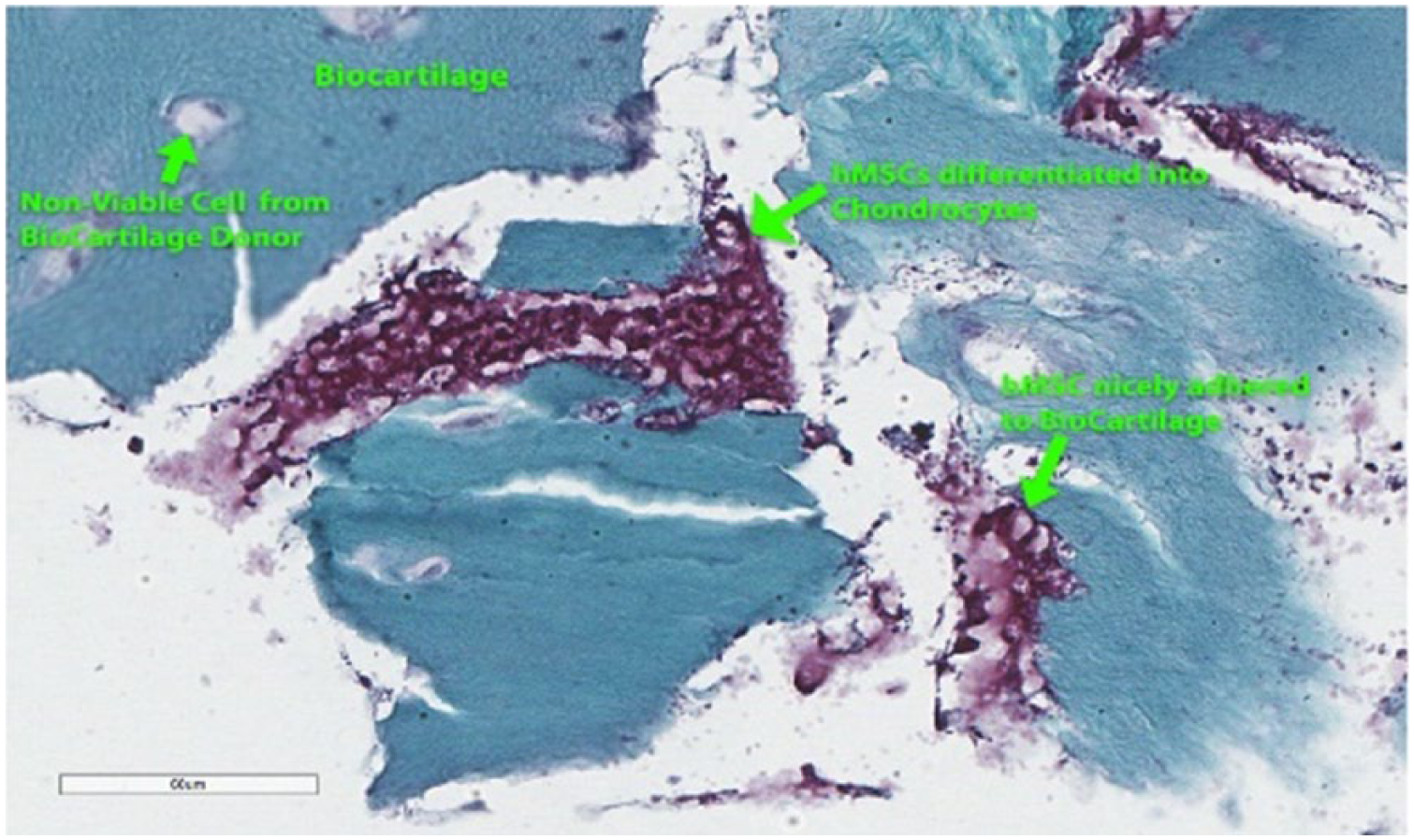
Harvested defects show chondrocyte differentiation adherent to micronized cartilage matrix (MCM) and have secreted a cartilage matrix. Staining is with hematoxylin and eosin for cellularity and safranin-O for proteoglycan content.
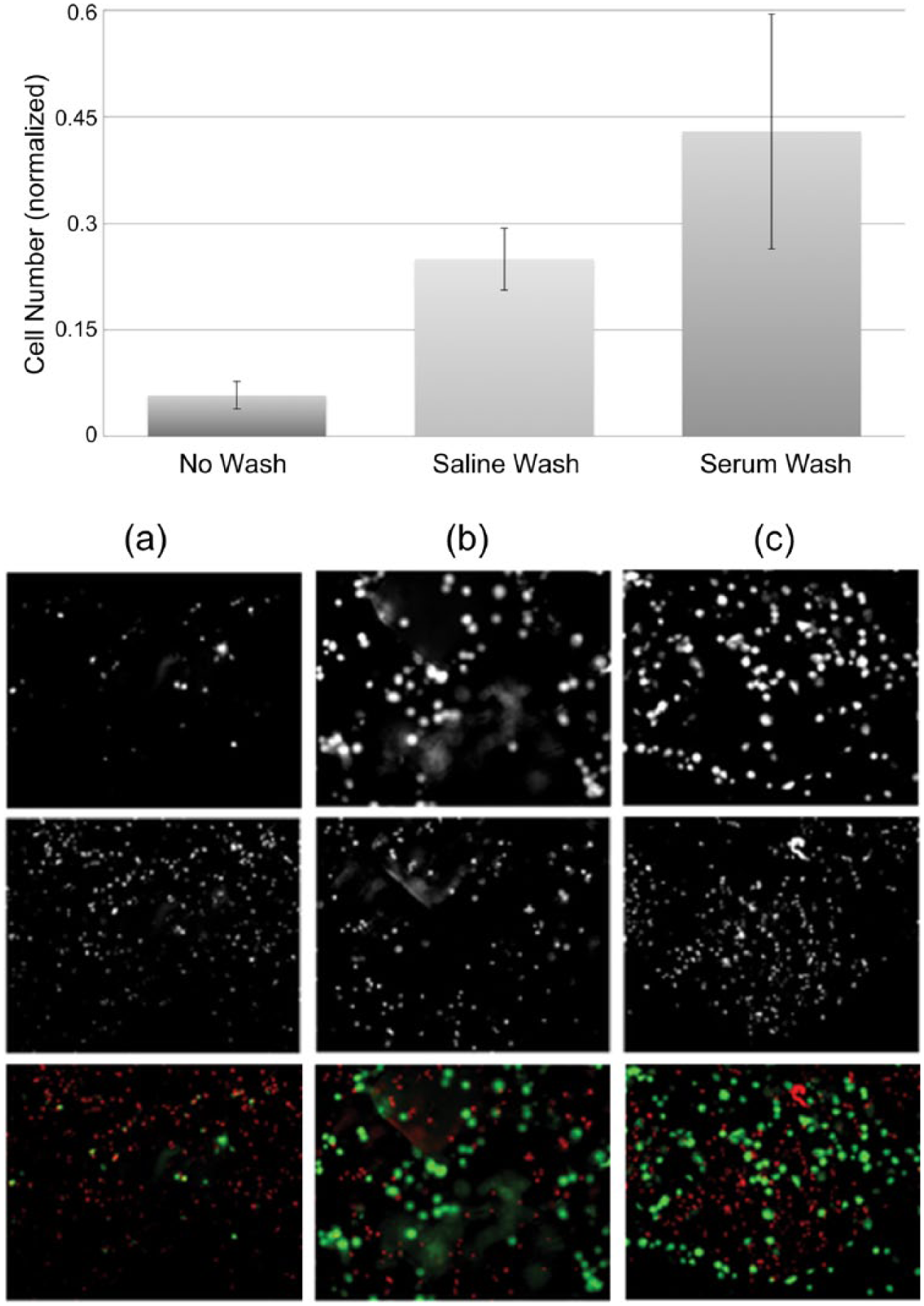
Washing micronized cartilage matrix significantly increases cell survival. Cell number has been normalized by calculating the ratio of each viability experiment to its corresponding control group average. (
Discussion
The goal of any cartilage repair procedure is the restoration of hyaline articular cartilage to fill the defect and the integration with adjacent native cartilage. Microfracture, based on its ease of use and safety, became the most widely used repair technique to induce formation of cartilage-like tissue within chondral defects, but the results have been variable. Short- to mid-term results of the microfracture technique yielded significant clinical improvement in up to 80% of patients but outcomes were noted to deteriorate over time.3,15 As noted in the systematic review by Mithoefer et al., 16 secondary arthroscopic procedures observed limited repair with fibrillated tissue. Histologically, most of these repairs resulted in either fibrocartilage or mixed fibrocartilage and hyaline cartilage, with limited type II collagen and proteoglycan content. Though subsequent promising techniques such as ACI and MACI are now popular, these are limited by their significant cost and surgical exposure required for implantation.
The advent of newer biologics has spurred interest in the development of a cost-effective single-stage procedure to treat chondral defects. Our study demonstrates that dehydrated micronized cartilage matrix can be a suitable scaffold for the chondrogenic differentiation of bone marrow–derived stem cells. After 3 weeks of culturing under chondrogenic media conditions, we observed robust cell survival with nearly 100% viability. Macroscopically, the final cultures resulted in structures resembling those used in current 2-stage techniques such as in ACI or MACI procedures, suggesting chondrogenic differentiation of the stem cells. Microscopic evaluation demonstrated cartilage matrix deposition occurred surrounding the cells after 3 weeks of chondrogenic culture with adherence of the cells to the allogenic MCM, further supporting our conclusion. These results provide a foundation for subsequent laboratory studies on the use of MCM.
Few studies thus far have investigated the use of MCM for the treatment of osteochondral lesions, and the few clinical series reporting on its use are limited to short-term follow-up. Lu et al. 17 initially showed that minced cartilage had the potential to be a suitable scaffold for the formation of hyaline-like tissue in animal model chondral defects. Malinen et al. 18 later reported on the use of particulate cartilage for the treatment of femoral condyle defects in baboons, showing that freeze-dried preparations led to both macroscopic and microscopic evidence of cartilage regeneration. Fortier et al. 19 more recently described the use of MCM in equine knee models and found that MCM combined with platelet-rich plasma resulted in improved histologic and imaging outcomes compared with microfracture alone after 13 months, though no difference in integration with host cartilage was observed. In early human clinical use, no complications have been encountered thus far, and clinical trials are ongoing. 20 Desai 14 has recently published a surgical technique of using the MCM for osteochondral lesions of the talus, which includes microfracture to encourage native mesenchymal stem cell extravasation to the lesion followed by placement of MCM through a percutaneous needle combined with autologous blood solution beneath a fibrin sealant. One primary advantage with this surgical technique is that it may be performed arthroscopically without the need for open visualization or repair. The use of MCM has further been reported on chondral defects of the glenoid, capitellum, and tibial plateau.21-24 However, there is limited data to guide the optimal clinical treatment protocol for the use of MCM, and additional laboratory studies could further enhance the current technique.
Here we have demonstrated an in vitro method to ensure cell viability on implantation of MCM with MSC. From this proof of concept study, the goal is to evaluate cell viability at additional time points. Also, reverse transcription-polymerase chain reaction should be carried out to evaluate mRNA expression levels of the following chondrocyte-specific genes: collagen type II, aggrecan, link protease, collagen type I, and versican. This study also did not look at the ability for this chondral repair technique to integrate with the adjacent native cartilage and this would be an additional investigation worth pursuing. In the laboratory setting, we found that a rehydration step of 30 minutes was optimal. This was an unexpected, yet understandable, finding. Without the initial step of rehydrating the MCM, mostly dead cells were observed. This may have clinical implications in determining the appropriate surgical technique to encourage chondrocyte survival and differentiation. Further evaluation of this in vitro finding is required and correlation to a clinical setting is being evaluated.
Conclusion
In conclusion, we have shown that the combination of mesenchymal stem cells with rehydrated MCM in either saline or serum in vitro results in nearly 100% cell viability and supports chondrogenic differentiation. This study serves as the first step in developing a single-stage surgical treatment for osteochondral lesions. Further studies are warranted to determine the gene expression levels of key components of hyaline cartilage and to refine the growth technique to enable in vivo osteochondral defect repair.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Eric Giza and Christopher Kreulen are paid consultants for Arthrex Inc.
Ethical Approval
Ethical approval was not sought for the present study because no human or animal subjects were used.
Informed Consent
Informed consent was not sought for the present study because no human subjects were utilized.
Trial Registration
Not applicable.
