Abstract
Objective
To test whether patients with spontaneous osteonecrosis of the knee (SONK) are characterized by abnormal levels of thrombophilia-associated factors.
Design
Twenty-five patients with SONK were recruited. Inclusion criteria were (1) age >40 years, (2) acute onset knee pain not precipitated by trauma, and (3) MRI findings consistent with SONK. Exclusion criteria were (1) history of cancer and chemotherapy and (2) factors associated with secondary osteonecrosis. Blood tests included 13 thrombophilia-associated factors that were either heritable mutations or acquired factors. Descriptive statistics included medians, ranges, means, and standard deviations. Mann-Whitney test was used to compare thrombophilia-associated factor levels between the sexes. Spearman’s rank test was used to test correlations between smoking status and each thrombophilia-associated factor. Level of significance was set at 0.05.
Results
Median patient age was 62 years (range, 44-77 years). There were 16 (64%) men. Thirteen (52%) patients had thrombophilia-associated factor abnormalities of which 9 were elevated fibrinogen but this was less than 1 standard deviation above norm threshold. Other findings were 3 patients with marginally decreased antithrombin below norm threshold, low protein S Ag in only 1 patient, and factor V Leiden mutation heterozygosity in 2 patients, which was not higher than normal population prevalence. Thrombophilia-associated factors neither differed between sexes (P = nonsignificant) nor correlated with smoking status (P = nonsignificant).
Conclusion
Thrombophilia-associated factor abnormalities in patients with SONK were minimal. Therefore, clinical workup and treatment strategy in this disease should focus on addressing alternative etiologies leading to abnormal subchondral bone metabolism with focal osteopenia.
Keywords
Introduction
Knee osteonecrosis can result in significant morbidity. Three types of knee osteonecrosis have been described: (1) primary, or spontaneous osteonecrosis of the knee (SONK), most often affecting middle-aged population1,2; (2) secondary osteonecrosis of the knee, which is associated with a variety of predisposing conditions such as systemic steroid therapy, alcoholism, hemoglobinopathies, and lupus erythematosus, all of which affect younger patients and present multiple-sites involvement 3 ; and (3) postarthroscopic osteonecrosis, occurring in association with previous knee arthroscopy or after posterior cruciate ligament 4 or anterior cruciate ligament reconstruction. 5 Among the 3, SONK is considered the most prevalent type of knee osteonecrosis with a reported prevalence of 3.4% in patients older than 50 years and as high as 9.4% in those older than 65 years. 2 The pathophysiology of SONK is unclear. Multiple theories have been proposed, which include local ischemia with microvascular occlusion due to thrombotic event, which leads to subchondral necrosis and may eventually result in subchondral collapse, or alternatively, a primarily subchondral insufficiency fracture of a metabolically abnormal and osteopenic bone. 6 Although atraumatic osteonecrosis of the femoral head 7 and multifocal osteonecrosis 8 have shown both associations with thrombophilic profile, to the best of our knowledge, this has never been tested specifically in patients with SONK, despite possible common pathophysiological pathways among these medical conditions. The objective of this study was therefore to test whether patients with SONK are characterized by abnormal levels of thrombophilia-associated factors.
Patients and Methods
Twenty-five patients comprised the study group and were recruited from a tertiary sports and knee clinic. Inclusion criteria were (1) age >40 years, (2) acute onset knee pain that was not precipitated by trauma, and (3) magnetic resonance imaging (MRI) findings consistent with SONK, which included diffused prominent bone marrow signal (i.e., marrow edema) involving a knee condyle on T2-fat suppressed weighted images, and surrounding a subchondral area of low signal on T1-weighted images (i.e., SONK fragment).1,2 Exclusion criteria were (1) history of cancer and chemotherapy treatment and (2) factors associated with secondary osteonecrosis (i.e., lupus erythematosus, inflammatory bowel disease, corticosteroid therapy, and heavy alcohol consumption).
Demographics and Accompanying Conditions
There were 16 (64%) men. Median patient age was 62 years (range, 44-77 years). Smoking status was mean 8 ± 12 pack-years (range, 0-40 pack-years). Seventeen (68%) patients reported history of little smoking (≤5 pack-years), and 20 (80%) patients were nonsmokers at the time period when SONK occurred. None of the patients had previous event of deep vein thrombosis (DVT), but two patients underwent prior coronary artery bypass surgery a few years prior to the event of SONK. Two other patients with SONK who had history of cancer were excluded and did not comprise the study group.
SONK Characteristics and Accompanying Meniscal Lesions
Among the 25 patients of the study group, 21 had SONK of the medial compartment (18 of the medial femoral condyle [MFC], 3 of the medial tibial condyle [MTC]), and 4 patients had SONK of the lateral femoral condyle (LFC). In accordance with the Koshino classification system ( Table 1 ), 9 all cases involved osteonecrosis stage I or stage II ( Fig. 1 ). There were no cases of Koshino stage III or stage IV in this series. Among the 21 patients with SONK of the medial compartment, 11 cases had coexisting medial meniscus tears, of which 8 were degenerated tears of the posterior horn and 3 cases were posterior root tears. Based on knee radiographs and MRI findings of cartilage wear, 23 patients had no or only mild arthritis, consistent with Kellgren-Lawrence grade ≤2, and only 2 patients had moderate arthritis consistent with Kellgren-Lawrence grade 3. In addition, since previous studies implied possible interrelationships between medial meniscus lesions and the volume of SONK fragment,10,11 measurements of the volume of SONK fragment in each case were performed as an approximated ellipsoid as previously suggested.10,11
Koshino Classification System. 9

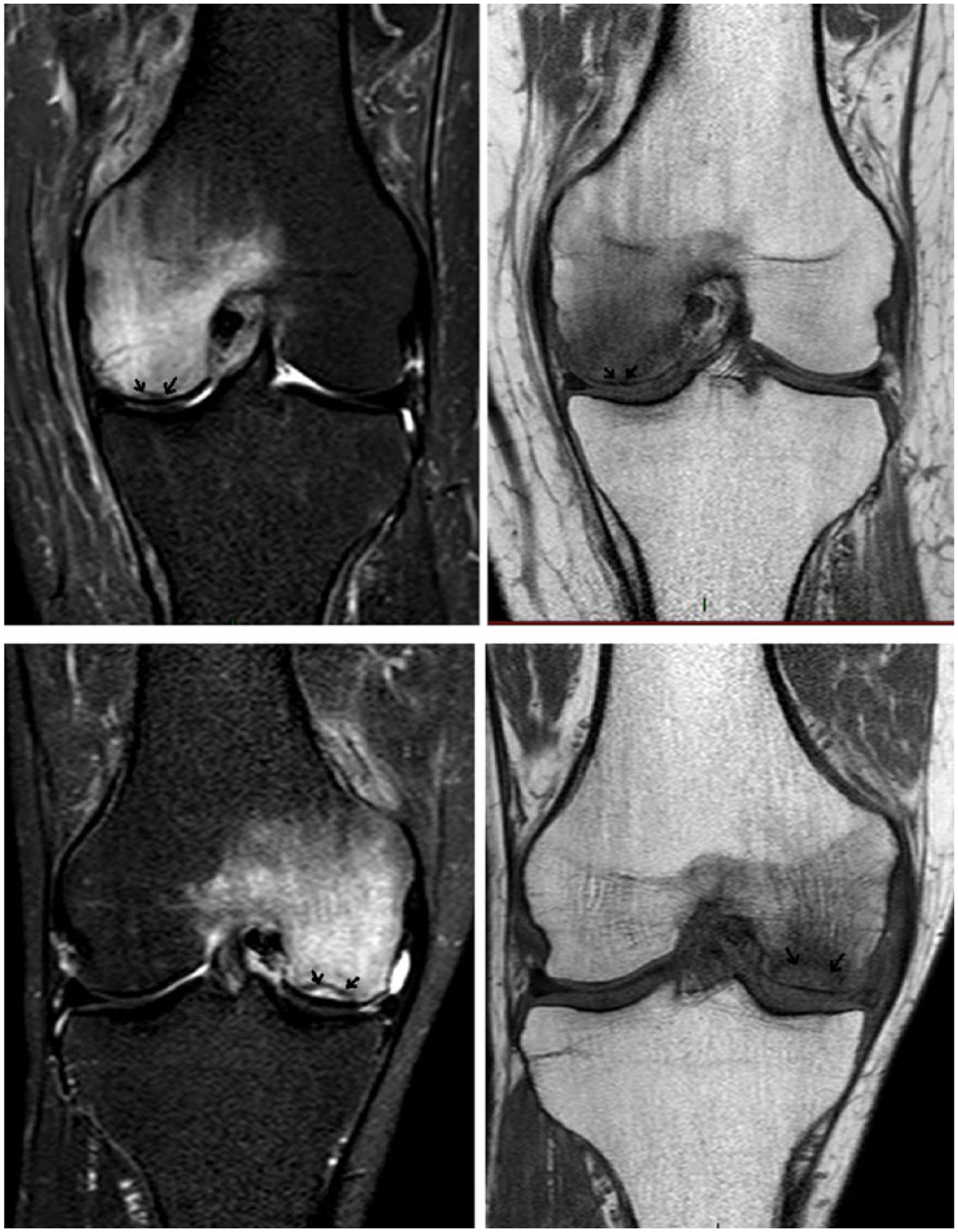
Magnetic resonance images of 2 representative patients in this series. Upper 2 images show mid-coronal cut of a left knee with subtle spontaneous osteonecrosis of the knee (SONK) fragment (arrows) surrounded by bone marrow edema of the medial femoral condyle on T2-fat-suppressed weighted image (left) and on T1-weighted image (right). This represents Koshino stage I. Lower 2 images show mid-coronal cut of a right knee with well-defined SONK fragment margins (arrows) surrounded by bone marrow edema of the medial femoral condyle on T2-fat-suppressed weighted image (left) and on T1-weighted image (right) with possible minimal flattening of the condyle but without collapse of the subchondral bone. This represents Koshino stage II.
Thrombophilic Profile Tests
Thrombophilic profile was based on published recommendations for laboratory tests screening related to thrombophilia, which included both heritable mutations as well as acquired predisposing factors. 12 Heritable thrombophilia-associated profile was evaluated by testing the following 6 factors: (1) functional antithrombin (%), (2) protein C activity [%], (3) protein S Ag free (%), (4) activated protein C resistance (ratio), (5) factor V Leiden mutation, and (6) G-20210-A mutation of the prothrombin gene. Acquired thrombophilia-associated profile was evaluated by testing factors related to antiphospholipid syndrome since it is considered the most common cause for acquired thrombophilia. All 3 components of the antiphospholipid syndrome were tested and included: (1) levels of lupus anticoagulant—Russell viper venom test (RVVT) and a silica clotting time (SCT) (ratio); (2) anticardiolipin IgG and IgM antibodies (U/mL); and (3) anti β2 glycoprotein IgG and IgM antibodies (U/mL). In addition, prothrombin time (PT) (%), international normalized ratio (INR) (ratio), activated partial thromboplastin time (APTT) (seconds), and plasma fibrin-ogen levels (mg/dL) were tested.
PT, APTT, and fibrinogen were determined in an automated BCS XP analyzer (Siemens, Marburg, Germany), using standard reagents (Dade Innovin, Dade Actin FS and Dade Thrombin) (Siemens, Marburg, Germany). Protein C activity was quantified by a chromogenic assay (Protein C, HemosIL, instrumentation laboratory). Free protein S Ag was quantified by automated latex ligand immunoassay (free protein S, HemosIL, instrumentation laboratory). Antithrombin activity was measured using chromogenic assay (liquid anti-thrombin, HemosIL, HemosIL, instrumentation laboratory). Factor V Leiden and prothrombin gene G20210A mutations were determined by real-time polymerase chain reaction (PCR) (Accustart Genotyping tough MIX, Low Rox, QUANTA biosciences). Lupus anticoagulant was detected with the diluted RVVT and SCT (HemosIL, instrumentation laboratory). Activated protein C resistance was determined using the factor V Leiden (APCTM Resistance V) kit (HemosIL, instrumentation laboratory). Anticardiolipin antibodies and β2 glycoprotein antibodies were measured by a standardized enzyme-linked immunosorbent assay (ELISA) according to the international standards of IgM (MPL) and IgG phospholipid units (GPL) (BIORAD, Hercules, CA, USA). All test analyses were supervised by qualified laboratory staff. For each factor, blood levels were interpreted as “normal” or “abnormal” in view of the normal reference range, which is the set of values 95% of the normal population falls within (i.e., 95% prediction interval). 13 Finally, after reviewing the results of blood tests, patients with abnormal fibrinogen levels were asked to consent for repeated tests, in addition to adding C-reactive protein (CRP) level test (mg/dL) in order to rule-out or confirm acute phase condition as an underlying source for elevated fibrinogen level.
Clinical data collected included medical history and comorbidities with specific attention to documented prior thromboembolic events and vascular morbidity, drug usage, smoking, malignancy treated with steroids or chemotherapy, prior surgeries, and family history of thromboembolic events. The study was approved by the institutional review board, and all participants signed informed consent.
Statistical Analysis
In the absence of published data about expected abnormal levels of prothrombotic factors in patients with SONK, a sample size of 25 patients was regarded adequate in accordance with a previous study that showed increased antiphospholipid antibodies levels in 20% of a group of 25 patients with multifocal osteonecrosis. 8 Descriptive statistics included medians with ranges and means with standard deviations. Also, because women have been shown to be at greater risk for thromboembolic events, 14 the Mann-Whitney test was used to compare thrombophilia-associated factor levels between the sexes. In addition, due to reported association between smoking, systemic inflammation, elevated fibrinogen levels, and consequently, vascular events, 15 correlations between smoking (pack-years), and each of the thrombophilia-associated factors were calculated with Spearman’s rank test. SONK volume measurements presented normal distribution using Kolmogorov-Smirnov test in the study group, and therefore the Student t test was used for comparing SONK volume between the following subgroups: (1) among the 21 patients with SONK of the medial compartment, comparisons of SONK volume were made between patients with (11 cases) versus patients without (10 cases) medial meniscus tears; (2) among the 11 patients with medial meniscus tears, comparisons of SONK volume were made between 8 cases with degenerated tear of the posterior horn versus 3 cases with posterior root tear of the medial meniscus; and (3) among all 25 patients of the study group, comparisons of SONK volume were made between patients with- (13 cases) versus patients without (12 cases) coexisting thrombophilia-associated factors. Level of significance was set at 0.05. Statistical analyses were performed using IBM SPSS-22 software package (Armonk, NY, USA).
Results
Twenty-five patients were recruited for this study. There were 16 (64%) men. Median patient age was 62 years (range, 44-77 years). Smoking status was mean 8 ± 12 (pack-years) (range, 0-40 pack-years). Seventeen (68%) patients reported history of little smoking (≤5 pack-years), and 20 (80%) patients were nonsmokers at the time period when SONK occurred. None of the patients had known risk factors for osteonecrosis such as lupus erythematosus, inflammatory bowel disease, corticosteroid therapy, heavy alcohol consumption, or systemic condition associated with hypercoagulability. None of the patients had previous event of deep vein thrombosis (DVT), but 2 patients underwent prior coronary artery bypass surgery a few years prior to the event of SONK. Two other patients with SONK who had history of cancer were excluded and did not comprise the study group. On MRI, all cases involved osteonecrosis stage I or II according to the Koshino classification system ( Fig. 1 ). 13 Coexisting knee lesions on MRI included 14 cases of meniscus tears (13 were medial meniscus posterior horn tears of which 3 were root tears, and 1 was lateral meniscus tear), and 3 cases with mild arthritic changes.
No abnormalities were observed in the three standard blood clotting tests, which were APTT (seconds), INR (ratio), and PT (%). Of the 25 patients, 13 (52%) demonstrated abnormalities supportive of thrombophilic tendency ( Table 2 ). These included 9 patients with elevated fibrinogen level (mean 437 ± 56 mg/dL, range 401-579 mg/dL), but deviations from upper norm threshold were marginal and less than 1 standard deviation in 8 of these 9 cases. Two of these 9 patients also had minimal increase in the Lupus anticoagulant SCT test, and 2 patients were heterozygotes for factor V Leiden mutation. Another 4 patients, other than the 9 patients with elevated fibrinogen, had the following thrombophilic abnormalities: 1 patient had decreased level of protein S Ag free (%), and another 3 patients had minimally decreased antithrombin (%) levels. Of the 9 patients with elevated fibrinogen levels, 5 patients consented for repeated blood tests, of whom all had elevated fibrinogen levels in the retests (mean 472 ± 62 mg/dL, range 418-576 mg/dL) while CRP levels did not indicate acute phase reaction in any of the cases (mean 1.0 ± 0.4 mg/dL, range 0.5-1.4 mg/dL).
Thrombophilia-Associated Factor Levels. a
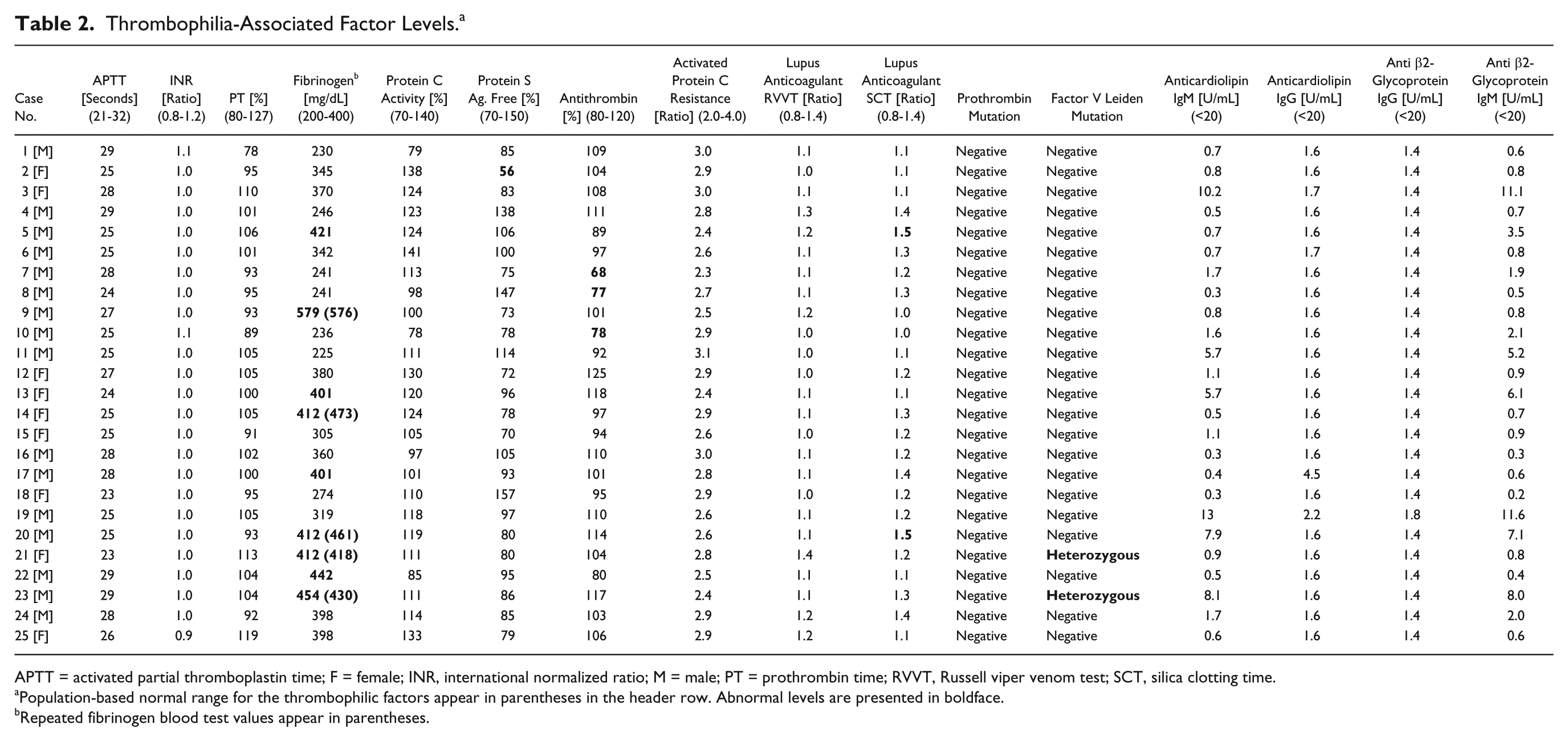
APTT = activated partial thromboplastin time; F = female; INR, international normalized ratio; M = male; PT = prothrombin time; RVVT, Russell viper venom test; SCT, silica clotting time.
Population-based normal range for the thrombophilic factors appear in parentheses in the header row. Abnormal levels are presented in boldface.
Repeated fibrinogen blood test values appear in parentheses.
Looking specifically at the 2 men who had prior coronary artery bypass surgery, one had increased fibrinogen level (579 mg/dL), and the other patient did not demonstrate any abnormality in any of the blood tests.
Female sex and smoking status (pack-years) were not associated with any of the thrombophilia-associated factors (P = nonsignificant).
Subgroup comparisons of SONK fragment volume in 21 patients with medial compartment involvement showed nonsignificant differences between 11 patients with versus 10 patients without medial meniscus tears (3853 ± 3989 vs. 1594 ± 1340 mm3, P = 0.1). Among the 11 patients with medial meniscus tears, significantly larger volume of the SONK fragment was observed in 3 patients who had medial meniscus root tear versus 8 patients who had degenerated posterior horn tear (8056 ± 5503 vs. 2277 ± 1915 mm3, P = 0.02). SONK fragment volume in the entire study group did not differ between 13 patients with versus 12 patients without thrombophilia-associated factors (2527 ± 2933 vs. 3105 ± 3283 mm3, P = 0.6).
Discussion
Based on the findings of this study, a possible linkage between thrombophilia and SONK remains questionable. While 13 of 25 (52%) patients with SONK had abnormal levels of thrombophilia-associated factors, which may be supportive of an association between these 2 conditions, the abnormalities identified were mild and of unclear clinical significance. Among the abnormalities observed, high fibrinogen level was the most consistent. Fibrinogen is a protein synthesized in the liver and it can promote blood viscosity and platelet aggregation. It is converted to fibrin by the action of thrombin. In addition, it makes clots more resistant to lysis. Accordingly, elevated levels of fibrinogen (i.e., >400 mg/dL) were associated with thrombosis in a variety of diseases of vascular origin. 16 Furthermore, while it can present high levels secondarily as an acute phase reactant, in this series fibrinogen level was likely not elevated as a result of acute phase reaction as shown by the low levels of CRP in these patients. Thus, the elevated fibrinogen levels observed may have in theory contributed to microvascular abnormalities causing ischemia as an underlying process in SONK. On the other hand, deviations from upper norm threshold in this series were almost exclusively far from “convincing” as clinically significant since these were relatively minimal and practically smaller than one standard deviation in 8 of 9 cases with elevated fibrinogen. As to factor V Leiden heterozygosity which was observed in two patients in this series, the prevalence of this abnormality is 2% to 7%, 12 implying again that our findings in patients with SONK are not exceptional relative to the general population. Protein S Ag abnormality was observed in one patient. Indeed, this was a 55-year-old woman who was not consuming hormonal replacement therapy and therefore may have been representing true thrombophilia-associated abnormality in this single patient. 17 Since the prevalence of this deficiency is less than 1%, 12 the observation of a positive patient in this small series of 25 patients may be regarded as exceptional. However, at this point, one positive patient should not be generalized to the entire group or claimed to be a characteristic exceptional finding in patients with SONK until larger populations are tested. A seemingly more convincing positive finding in this series could have been antithrombin deficiency in 3 of the 25 patients (12%) which is substantially higher than the prevalence in the general population reported as less than 1%. 12 However, looking at the individual abnormal values in these 3 cases of low levels of antithrombin showed only very marginal reduction of the values below the normal threshold, which again questions clinically significant interrelationships between thrombophilia and SONK in this series.
The literature about possible interrelationships between coagulopathy and necrosis in bone and joints is extensive although SONK was not specifically investigated in this respect until today. Thrombophilic profile was reported in patients with idiopathic and secondary osteonecrosis of the femoral head, showing increased prevalence of factor V Leiden mutation, high factor VIII levels, high levels of anticardiolipin IgG and low levels of resistance to activated protein C in these pathologies.18-21 Moreover, a recent report in patients who had multifocal osteonecrosis (MFON) showed abnormal levels of at least one prothrombotic factor in 12 (48%) out of 25 patients. 8 These included antiphospholipid syndrome abnormalities with elevated levels of anticardiolipin IgG and IgM, factor V Leiden mutation, and factor VIII, among others. 8 MFON and SONK may in theory share common underlying pathomechanisms, relating to focal ischemic events in the microvasculature. In MFON however, the infarcts are observed at the medullary canal and not necessarily at the subchondral bone, 8 while SONK affects the subchondral plate and results in injury to the hyaline cartilage-subchondral bone interface, which may appear in some cases as subchondral collapse and permanent articular chondral damage. 9 This, in addition to the multifocal involvement in MFON as opposed to SONK may explain why these populations which did not present similarly convincing data of an underlying hypercoagulable state should be viewed as 2 separate medical entities in respect to underlying pathomechanisms and treatment strategies.
Alternative pathomechanisms, other than hypercoagulability, come from a few studies who have suggested that SONK may be caused by insufficiency fracture and that the primary process in this disease is not osteonecrosis. These studies evaluated magnetic resonance imaging22,23 and histological specimens6,24,25 of patients diagnosed with SONK, and showed that necrosis was preceded by an underlying subchondral insufficiency fracture and suggested that the main histopathologic abnormality in this process may be a subchondral insufficiency fracture and not necessarily the presence of necrotic bone. The findings of the current study may be supportive in this regard (i.e., exceptional hypercoagulability was not indeed a characteristic finding in these patients).
Coexisting medial meniscus tears observed in 11 patients among 21 (52%) who had SONK of the medial compartment deserve further attention in relation to possible coexisting mechanical factors in this disease. In fact, volume of the subchondral osteonecrotic fragment in patients with SONK was previously associated with the extent of medial meniscus extrusion by some investigators, 11 while others supported high incidence of medial meniscus root tears in patients with SONK although statistically significant association between SONK volume and meniscal root tears was not identified. 10 These studies supported the coexistence of medial meniscus tears and SONK at least at the advanced stages of the disease but they did not necessarily indicated cause-and-effect relationships. In the current investigation, a trend, although without statistical significance, for larger SONK volume of the medial compartment was demonstrated in patients with medial meniscus tears versus patients without meniscus tears, while statistically significant larger volume of SONK was found in patients with medial meniscus root tears versus patients with medial meniscus degenerative tears with intact posterior root. The current series may therefore add support to the coexistence of loss of medial meniscus mechanical hoop function and larger SONK fragments, whereas degenerative medial meniscus tears with intact posterior root seemed less likely to play a role in the evolvement of larger SONK fragments in this series. Moreover, large epidemiological observations showed that incidental MRI findings of meniscus tears in the middle-aged and elderly populations may be as high as 56%, 26 and therefore the current series with more than half of the cases showing meniscus tears is not necessarily an exception in this regard.
The clinical significance of our findings deserves further research, and treatment strategies for SONK, based on a possible underlying vascular occlusion and resultant ischemia are still evolving. In fact, reports about the clinical benefit of pain reduction and bone marrow edema resolution at 4 months after the administration of a prostacyclin anti-clotting analogue in a series of patients with idiopathic bone marrow edema of the knee and similar age group as our patients deserves attention in this regard. 27 To add to the complexity of evolving pathomechanisms and suggested treatment strategies in this pathology, other investigators showed positive association between low bone mineral density and the incidence of SONK in women older than 60 years, 28 whereas treatment with low-dose intravenous bisphosphonates and high-dose oral vitamin D administration had positive effects of resolving symptoms in these cases, 1 without the addition of any anti-clotting medication.
Limitation of this study may include lack of tests of other markers of activation of the coagulation pathway such as D-dimers, thrombin-antithrombin complexes, and F1.2 levels as well as other relevant tests such as thromboelastography. This could be a basis for future research. Also, standing knee radiographs were not performed systematically, so that femorotibial angle could not be accurately measured, and therefore a potential contributing factor for the evolvement of SONK 11 was not tested.
In conclusion, thrombophilia-associated factor abnormalities in patients with SONK were minimal. Therefore, clinical workup and treatment strategy in this disease should focus on addressing alternative etiologies leading to abnormal subchondral bone metabolism with focal osteopenia.
Footnotes
Authors’ Note
This study was conducted at the Meir General Hospital, Kfar Saba, Israel
Acknowledgments and Funding
We thank Gloria Rashid, PhD, Head of the Clinical Laboratories; Tali Tohami, PhD, Head of the Hematology Laboratory; and Eran Neumark, PhD, MBA, Head of the Integrated Biochemistry Laboratory, all at the Meir General Hospital, Kfar Saba, Israel, for conducting the blood tests analyses. The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from the institutional review board (Approval number 0020-13-MMC).
Informed Consent
Written informed consent was obtained from all subjects before the study.
Trial Registration
Not applicable.
