Abstract
Background
Full-depth cartilage lesions do not heal and the long-term clinical outcome is uncertain. In the symptomatic middle-aged (35-60 years) patient, treatment with metal implants has been proposed. However, the cartilage health surrounding these implants has not been thoroughly studied. Our objective was to evaluate the health of cartilage opposing and adjacent to metal resurfacing implants.
Methods
The medial femoral condyle was operated in 9 sheep bilaterally. A metallic resurfacing metallic implant was immediately inserted into an artificially created 7.5 mm defect while on the contralateral knee the defect was left untreated. Euthanasia was performed at 6 months. Six animals, of similar age and study duration, from a previous study were used for comparison in the evaluation of cartilage health adjacent to the implant. Cartilage damage to joint surfaces within the knee, cartilage repair of the defect, and cartilage adjacent to the implant was evaluated macroscopically and microscopically.
Results
Six animals available for evaluation of cartilage health within the knee showed a varying degree of cartilage damage with no statistical difference between defects treated with implants or left untreated (P = 0.51; 95% CI −3.7 to 6.5). The cartilage adjacent to the implant (score 0-14; where 14 indicates no damage) remained healthy in these 6 animals showing promising results (averaged 10.5; range 9-11.5, SD 0.95). Cartilage defects did not heal in any case.
Conclusion
Treatment of a critical size focal lesion with a metal implant is a viable alternative treatment.
Introduction
Articular cartilage is a remarkably strong tissue capable of enduring great loads over time but has a limited healing capacity intrinsic to its avascular nature.1-7 It has been suggested that full-depth cartilage lesions (International Cartilage Research Society, ICRS grade 3-4), often associated to sports or other intense activities, do not heal.1,2,7-9 While many of these lesions cause pain and disability, others remain asymptomatic but might still progress to osteoarthritis (OA). 10 Some researchers therefore advocate that treatment is warranted both for symptomatic and prophylactic reasons. 11 It has been suggested that even after various kinds of treatment these lesions might progress to OA.12-14 However, because not all untreated focal lesion evolve to symptomatic knee OA, the treatment algorithm for these lesions remains controversial. 15
Modern treatment of focal condylar full thickness cartilage lesions include biological repair by autologous cartilage reimplantation, a technique that is technically demanding, requires sophisticated laboratory resources and comprises a meticulous rehabilitation programme.16-18 Another treatment pathway, so called focal knee resurfacing with metallic (FKRM) implants, is gaining popularity but still lacks conclusive scientific evidence to support its biological safety and efficacy for the whole patient group suffering full depth cartilage lesions. 19 Based on our previous preclinical reports, we suggest FKRM as a safe treatment strategy for the symptomatic middle-aged active patient (35-60 years), where biological treatments have failed or are shown to be less effective.19-21
We have previously evaluated FKRM in terms of osseointegration and positioning of femoral condylar implants and the resulting local wear on the opposing tibial cartilage, as well as the impact on general cartilage health of the sheep knee20,21 (joint cartilage homeostasis 22 ). The first aim of the present study was to compare short-term cartilage alterations in knees with lesions immediately treated with FKRM implants with those where full depth cartilage lesions were left untreated. The second aim was to follow the natural history of the cartilage adjacent to the implants by evaluating the macroscopic and microscopic reaction of the cartilage to the implant. To the best of our knowledge, the cartilage health surrounding FKRM implants has yet not been evaluated.
Materials and Methods
Animals
Nine healthy female sheep (Swedish landrace) from 2 breeders entered the present experiment. The mean age and weight of the sheep were 2.8 years and 76.5 kg, respectively. Three animals were lost to follow-up (as described below). For comparison of cartilage health adjacent to the implant, data from a prior experiment not previously analyzed, were used. Thus, all sheep of similar age (mean 2.8 years), sex, race, and study duration (6 months) using identical implant and evaluation protocol were analyzed (n = 6/13) for cartilage health adjacent to the implant. 23 Older sheep or sheep with longer follow-up were excluded from this comparison. Hence, a total of 12 animals were descriptively analyzed for health of cartilage surrounding the implant both macroscopically and microscopically (see below). The rationale was, for ethical reasons, to minimize the number of animals used.
The animals were housed at the Department of Clinical Sciences, Swedish University of Agriculture Sciences (SLU) in Uppsala, Sweden They were observed daily to monitor general condition, signs of pain and lameness (where grade 0 was normal gait and 1-4 was mild, moderate, major or severe lameness, respectively). 24 Euthanasia was performed at 6 months using an overdose pentobarbital (100 mg/mL) after securing blood samples. The knees were removed from the body and prepared further as described below. The Animal Ethics Committee, Uppsala Sweden, approved the protocol.
Implant
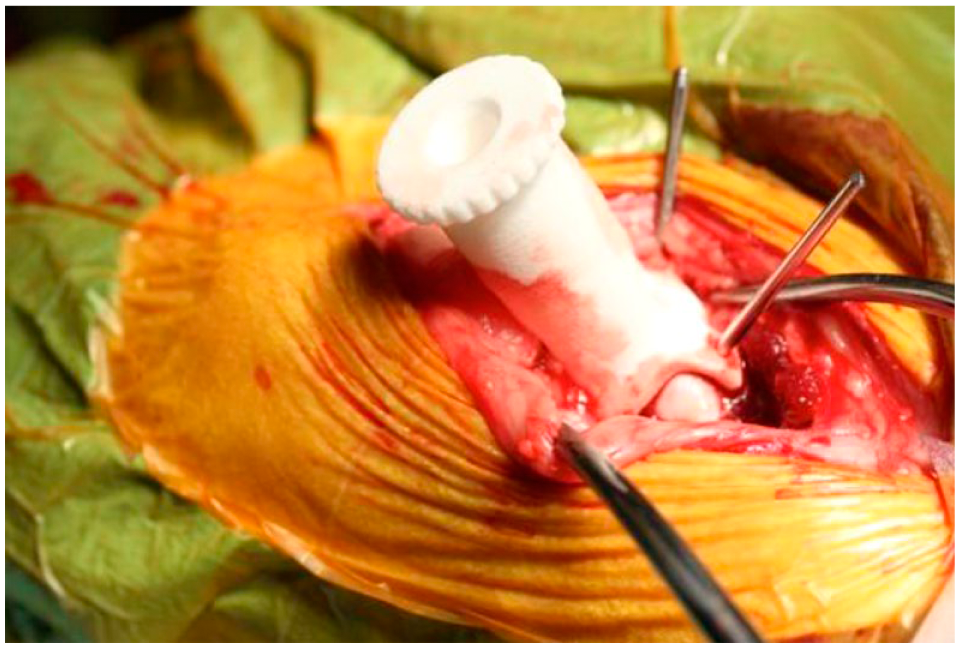
The implant (diameter 7.5 mm) had a double-curved (radii 19 and 12 mm) articulating surface modeled after computed tomography (CT) scans of a “standard” sheep knee, and was manufactured from implant-grade cobalt-chrome by a computer-aided design and manufacturing (CAD/CAM) process. The implants were coated with commercially pure titanium (60 µm) on which a layer of hydroxyapatite (HA; 60 µm) was plasma sprayed (Plasma Biotal Ltd, Buxton, UK). The articulating surface was then polished to a roughness (Ra) < 0.03 µm. The monobloc implant ( Fig. 1 ) had a 10-mm peg (diameter 2 mm) introduced into an undersized (diameter 1.8 mm) drill hole in the bone for primary interference fit. The implants were manufactured and provided by Episurf AB (Stockholm, Sweden).

Episealer second-generation double-curved and double-coated focal knee resurfacing implant (Episurf AB, Stockholm, Sweden).
Anesthesia
The animals were anesthetized according to the same protocol as used in previous experiments.20,21,23 They were operated in dorsal recumbency and the surgical field was aseptically prepared.
Surgery
Surgery was performed on both knees by the same surgeons (HNS, NMC, and LR), where the created defect on one side was immediately treated and on the other knee the defect was left untreated; randomly assigned. The medial femoral condyle was exposed through a medial parapatellar 5- to 6-cm incision and the operation was carried out using a set of specially designed instruments: First, a centralizing aiming guide with a built-in guiding tube, adapted to the contour of the weightbearing condylar surface was applied and fixed to the condyle by means of 3 pins engaging the metaphysis outside the articulating cartilage ( Fig. 2 ). Through the guiding tube, sitting perpendicular to all tangents of the articulating surface, a specially designed drill was used to cut the cartilage and the underlying bone in a way to exactly correspond to the shape of the implant. According to previous studies, 20 we aimed to position the implant at a level 0.5 mm recessed below the surrounding cartilage
Individualized guiding device showing the guiding tube that follows the curvature of the condyle, securing drilling perpendicular to the condyle in all planes.
On the other knee, a critical size defect of 7.5 mm was created to induce femoral condyle osteoarthritis in an otherwise stable joint as major lesions (14 mm) tend to cause significant degenerative changes at the opposing tibia. 8 A curette was used to cut the cartilage with special care taken not to damage the subchondral bone. Finally, the joint capsule was sutured in a continuous pattern using polydioxanone (PDS, Ethicon) and the subcutaneous tissue and skin were closed in a similar pattern using polyglicaprone 25 (Monocryl, Ethicon). No surgical complications occurred during the operations. The sheep were extubated in their stables and under continuous observation and regained consciousness within 1 hour postsurgery.
Laser Measurements of Implant Position
The medial femoral condyle with the implant was used for analysis. A negative print of the medial femoral condyle was subsequently scanned using a high precision (<1 µm) laser-scanning device (www.nikonmetrology.com; LK, Scandinavia, Stockholm, Sweden). The contour of the femoral condyle including the implant was digitized using a specific software program (Metris Focus Inspection 9.2) and the radius of the condyle curvature was determined in both the sagittal and coronal planes. The surface of the implant was then marked with 5 different reference points (center, anterior, posterior, medial, and lateral). From these landmarks, the implant height (mm) relative to the surrounding cartilage surface was calculated ( Fig. 3 ). Using the relative height (h) and the interdistance (d) of the anteroposterior or mediolateral data points, respectively, the angulation (tilt) of the implant relative to the surrounding condylar surface could be calculated by trigonometry as arctan (h/d).

Laser scanning of the implant in transversal (left) and antero-posterior (right) plane. Reconstructed 3-dimensional image shows one implant with excessive tilting leading to protrusion.
Macroscopic Cartilage Evaluation
The joints were inspected macroscopically according to Outerbridge (0-4) and a modified O’Driscoll score (0-6 points instead of 0-10 as the parameter restoration of contour and cartilage erosion of the graft was not possible to evaluate).25-27 High-resolution photographs (Canon EOS 450D, EF-S 17-55mm f/2.8 IS USM lens fixated at a distance of 0.3 m, using 35 mm focal length) were taken of the medial and lateral femoral and tibial condyles. Two blinded independent observers (NMC and HB) evaluated the photographs of each tibia plateau separately. Articular cartilage lesions were classified according to a scale of 0 to 4, where grade 0 is normal, grade 1 is fibrillation (softening not possible to evaluate on photograph), grade 2 is superficial fissures (not reaching the subchondral bone), grade 3 fissures to the subchondral bone, and grade 4 exposed subchondral bone. 28
Microscopic Cartilage Evaluation
After removal of soft tissues and photography, the articular cartilage of the tibia was dissected and placed in 2% glutaraldehyde + 1% paraformaldehyde in 0.1 M sodium cacodylate buffer, pH 7.4 and stored in a refrigerator. Segments chosen to represent areas of cartilage facing the implants and from the anterior and posterior third of the joint including the control medial tibial plateau, lateral tibial condyles and from the lateral femoral condyle were cut. Semithin sections were stained with Safranin O and used for light microscopic analysis. These specimens were blinded and scored at random by 2 observers (NMC and HB). Damage to the articular cartilages was evaluated according to a modified Mankin score as recommended by Osteoarthritis Research Society International (OARSI) for histological assessment of osteoarthritis in sheep. 29 The inner, middle, and outer section of each segment was analyzed and the most severe lesion was used for scoring. The scores of the observers were averaged, where outliers with a difference of more than three points were scored again until consensus was reached. 30
Health of Cartilage Surrounding the Implant
To evaluate the health of the cartilage bordering the implant a score was created using both macroscopic and microscopic parameters. These parameters were modified from existing classifications evaluating cartilage repair.31-34 Macroscopically, cartilage-implant contact area, general appearance and color of the cartilage abutting the implant and cartilage flow were evaluated (score 0-7). Microscopically, the cartilage quality, integration with the implant (or demarcation lines), and also the implant level in relation to surrounding cartilage were evaluated (score 0-7). These parameters are combined with the intent to help researchers and clinicians to evaluate the health of the cartilage adjacent to the implant. A maximum score of 14 points suggests optimal cartilage health, whereas 0 points is the worst-case scenario. We propose that a score of 14 to 10 suggests satisfactory results, a score of 9 to 5 moderate results, and a score of 4 to 0 denotes unsatisfactory chondrointegration (see Table 1).
A Proposed Score for Macroscopic and Microscopic Evaluation of Cartilage Health Surrounding an Implant.

Macroscopic score 0 to 7, microscopic score 0 to 7, total score 0 to 14 points.
Evaluation of Cartilage Defect
The defects were evaluated macroscopically using high-resolution photographs according to ICRS cartilage repair score (0-12) and microscopically according to O’Driscoll cartilage repair score (0-24).32,33
Histomorphometric Analyses of Osseointegration
For methods on the evaluation of osseointegration and data in detail, see Martinez-Carranza et al. 21 In summary, the medial femoral condyle containing the implant was prepared for light microscopy, according to the ground sectioning technique by Donath and Breuner. 35 The sections were stained with Sanderson’s RBS stain and counterstained with acid fuchsin (both Dorn & Hart, Villa Park, ILUSA). The specimens were examined with a Zeiss Supra VPN-40 field cathode scanning electron microscope using the backscatter detector. The resulting images were evaluated using ImageAccess (Imagic, Glattbrugg, Switzerland) software. The amount of the bone-to-implant contact was measured and expressed in percentage.
Statistical Methods
Data are presented as means with their range, standard deviation (SD), or estimated 95% confidence intervals, or shown in a box-plot. A 2-factor analysis of variance (ANOVA) model was used to compare cartilage damage related to operative treatment or joint location and interactions between those factors. Independent-samples t test was used to compare means between groups. P values were set at 0.05. For nonparametric values, the Wilcoxon signed ranked test was used to compare results for matched pairs, where a test statistic (Z-score) exceeding 1.96 indicates statistical significance. Calculations were performed using the SPSS 15.0 for Windows package.
Results
General and Joint Health of the Animals
The general health of the animals was good prior to operations. One sheep died immediately after extubation and 2 sheep were sacrificed at 2 months, due to septic arthritis and lameness, respectively. The wounds healed without complications. Postoperatively, all sheep (n = 6) showed various degree of limp that decreased gradually, and at the time of euthanasia no sheep showed lameness. Joint health as indicated by the modified O’Driscoll score showed no changes in range of motion, fibrosis, or cartilage appearance (average 0.0 out of maximum 6 points).
Implant Height and Tilt
Height of implants (n = 6) as assessed by the mean of 3 transversal and 3 anteroposterior data points in the implant measured from laser scans, averaged 0.60 mm recessed (range 0.16-0.82) with a standard deviation (SD) of 0.23 mm for the whole group (aimed at 0.5 mm recessed). Furthermore, the mean frontal (transversal) and sagittal (anteroposterior) tilt was 3.7° (range 0.4°-7.3°) and 2.6° (range 0.8°-4.6°), respectively ( Fig. 4 ).
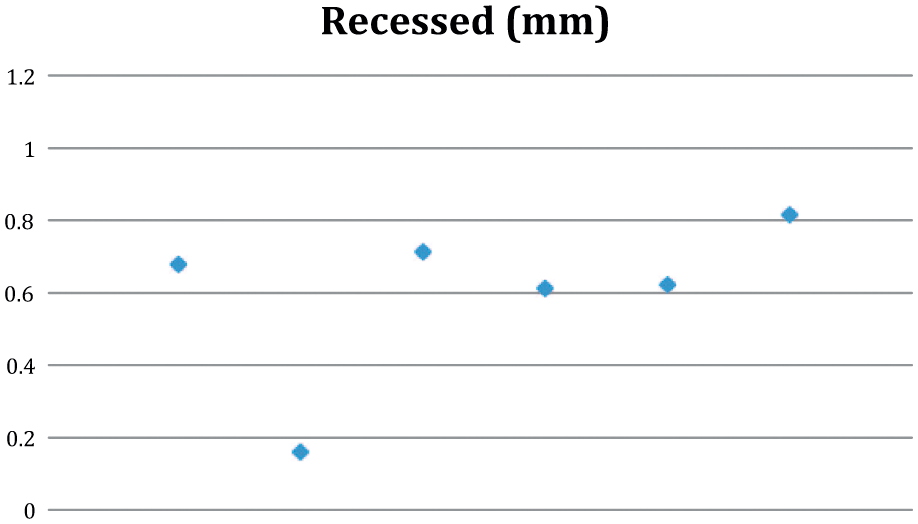
Raw data of the implant position in terms of averaged height (in millimeters; zero denotes the cartilage level). Observe that the implants are aimed at an ideal position of 0.5 mm recessed. Individual values are scattered along the x-axis. Observe one superficial implant.
Macroscopic Cartilage Evaluation
The macroscopic cartilage evaluation (Outerbridge 0-4) (n = 6) of the medial tibial cartilage surface showed modest cartilage damage both in the surface opposing the implants 1.6 (range 0.5-4.0) and in tibia opposing the untreated defect 0.9 (range 0.0-1.5) with no statistically significant difference between sides (Z = 0.707, P = 0.480). Lateral tibia and femoral surface showed no or minor damage macroscopically ( Fig. 5 ).
High-resolution photograph used to evaluate macroscopic cartilage damage of tibial condyles. To the left observe tibial cartilage damage on the medial side consequent to a tilting and protruding implant ( Fig. 3 ). To the right is a representative specimen of pristine cartilage of both tibial plateaus.
Microscopic Cartilage Evaluation
The opposing tibial plateaus of both the treated and untreated defect showed a varying degree (range 1-15) of articular cartilage damage evaluated according to the modified Mankin score as recommended by OARSI (0-27 units). Average wear, including implanted and control knees from a previous study, is shown (n = 12) in a box-plot ( Fig. 6 ).
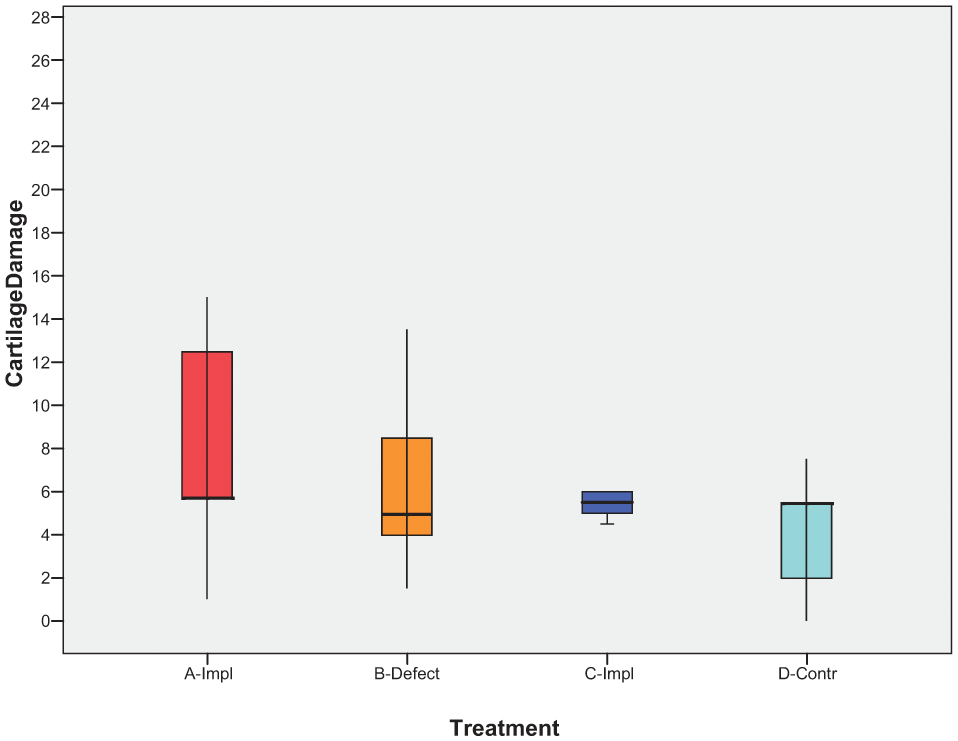
Box-plot showing all data denoting cartilage damage according to modified Mankin as recommended by OARSI (0-27). Box denotes first and third quartiles and whiskers show the minimum and maximum value, respectively. The band inside the box denotes the median. Red and orange show group of ewes (n = 6) operated with implant or defect, respectively. Dark blue and light blue show group of ewes (n = 6) operated with implant or nonoperated control knee, respectively (matched from previous experiment).
Cartilage damage of the medial tibial plateau opposing the implant showed no statistically significant difference (P = 0.51; 95% CI −3.7 to 6.5) compared with the medial tibial plateau opposing the nontreated defect of the contralateral knee (n = 6). Using repeated-measures ANOVA we showed a statistically significant difference in cartilage damage between different joint compartments. Medial femoral compartment was significantly less damaged compared with medial (P = 0.02; 95% CI 0.45-8.14) or lateral tibial compartments (P = 0.004, 95% CI 1.59-5.00), respectively ( Fig. 7 ).
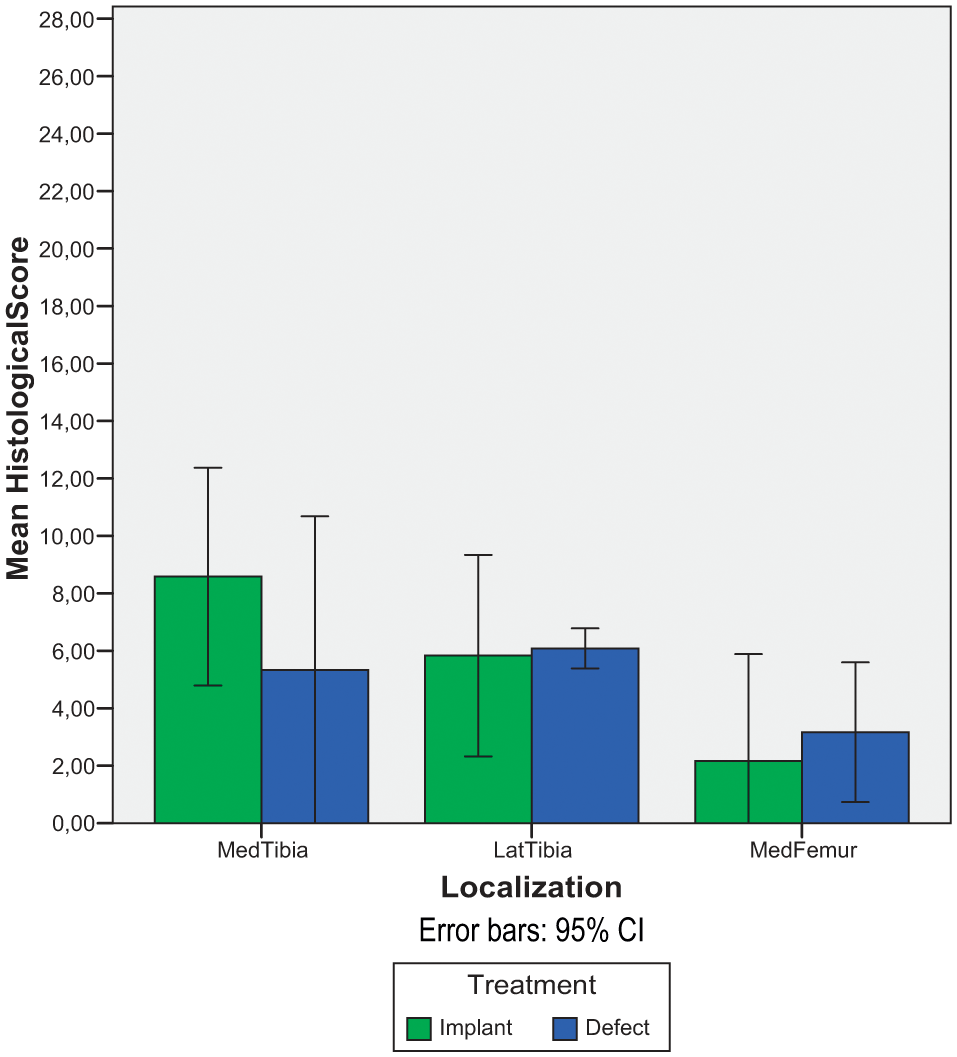
Cluster bar that shows the mean histological damage (Mankin 0-27) of medial tibial, lateral tibial, and medial femoral compartments, respectively. Green bars indicate knees operated with implant and blue bars indicate knees with an untreated defect. Error bars denote 95% confidence intervals (CI).
The tibial cartilage of the lateral compartment showed no statistically significant difference compared to the medial compartment (P = 0.51, 95% CI −4.61 to 2.61). Differences between compartments were not related to treatment with implant or left untreated (P = 0.38) (n = 6).
Cartilage Health Adjacent to the Implant
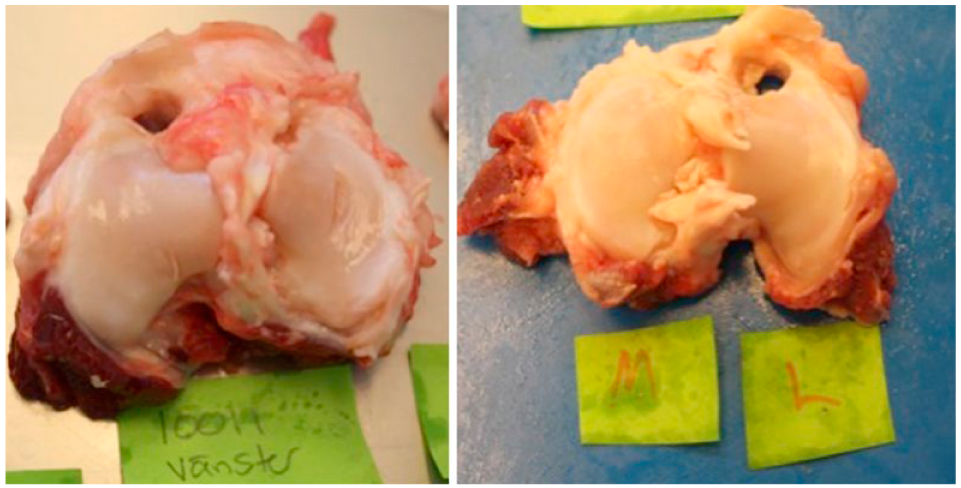
Cartilage health adjacent to the implant ( Fig. 8 ) scored macroscopically (n = 6) averaged 5.75 (range 5.0-6.5), SD 0.52, and microscopically (n = 6) 4.75 (range 4.0-5.5), SD 0.52. Total score averaged 10.5 (range 9.0-11.5, SD 0.95). Six comparable animals from our prior experiment showed similar scores; macroscopically (n = 6) on average 6.9 (range 6.5-7.0, SD 0.20), microscopically (n = 4) 5.125 (range 3-6, SD 1.44), and total score (n = 4) averaged 12.125 (range 10-13, SD 1.44), respectively.
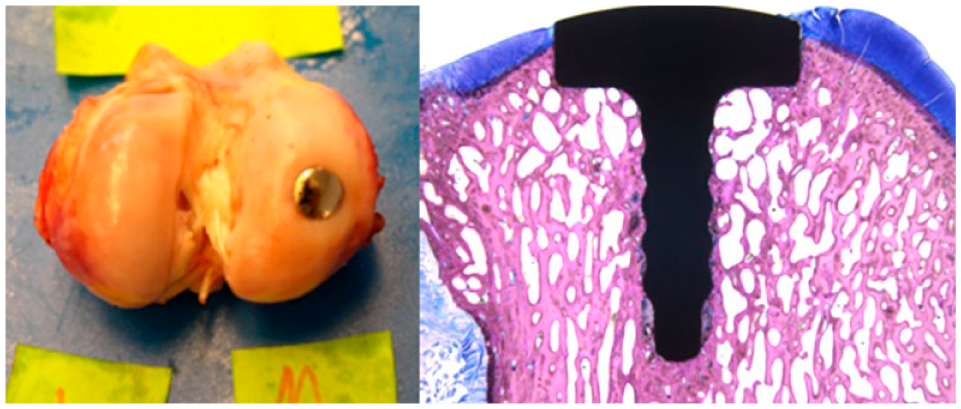
Macroscopic (left) and microscopic (right) images used to evaluate cartilage health surrounding the implant. In the shown example, the macroscopic evaluation scored 6 (scale 0-7), while the microscopic evaluation scored 5 (scale 0-7). Osseointegration (right) was satisfactory (98% bone-to-implant contact).
Cartilage Repair of the Defect
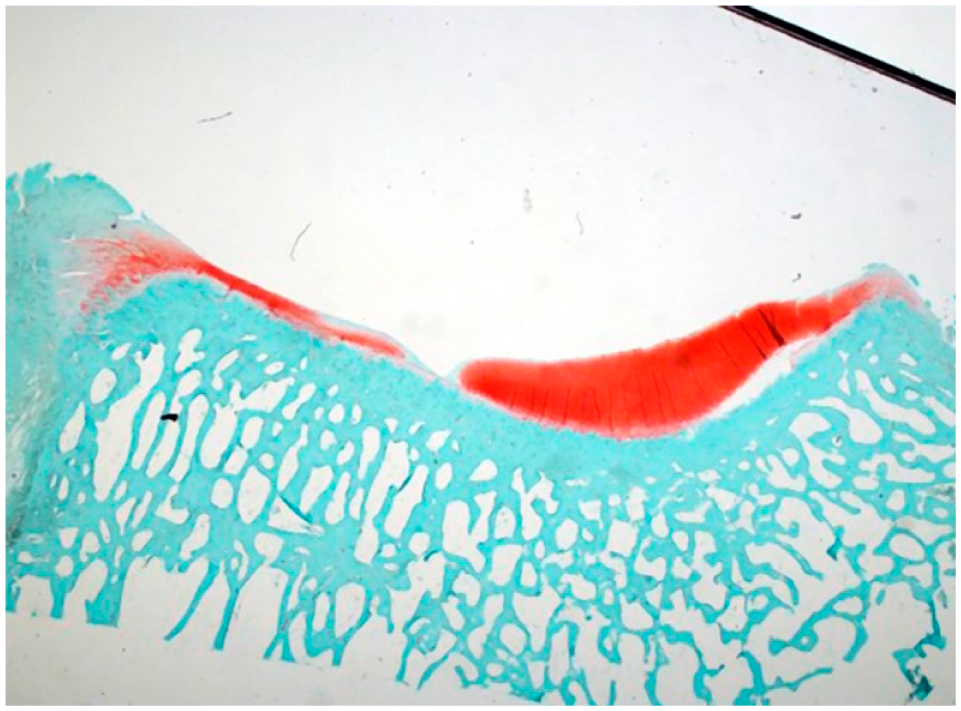
Cartilage repair of untreated defects scored macroscopically (n = 6) on average 5.0 (range 0.5-5.0) and microscopically (n = 6) on average 8.0 (range 2.0-14.0) ( Fig. 9 ).
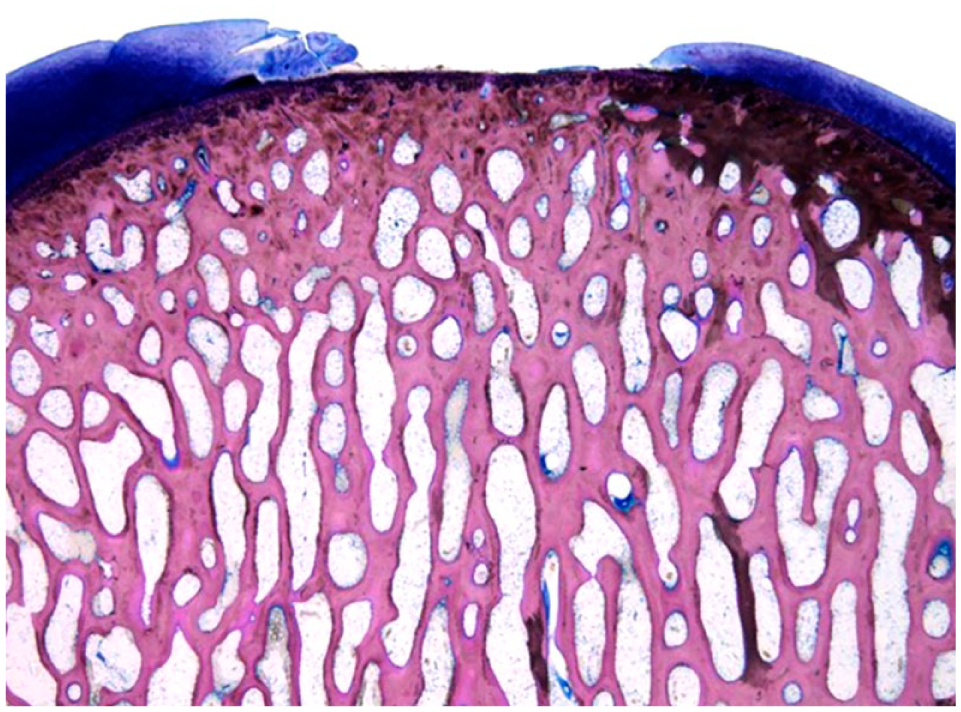
A representative microscopy image of an untreated full-depth cartilage lesion (7.5 mm) at 6 months.
Histomorphometry
Bone-to-implant contact averaged 79.3% (range 41.9%-98.8%) ( Fig. 8 , right).
Discussion
The main finding of this study was the modest cartilage damage of medial tibial plateaus, whether defects of the opposing femoral condyles were treated with metal implants (FKRM) or left untreated ( Fig. 10 ). Because no statistically significant difference related to treatment was demonstrated, it is adequate to suggest that receiving a femoral implant is not more harmful (at short term) than to leave the cartilage defect untreated.
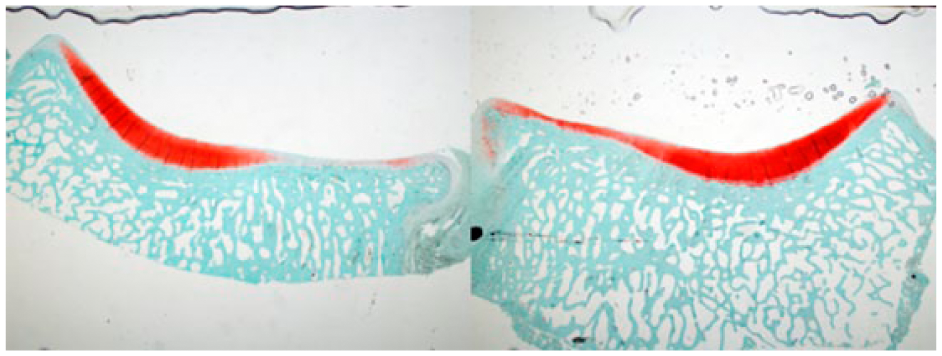
Cross-sectional histological pictures (Safranin O) of the tibial cartilage opposing the implant (left), and opposing an untreated defect (right). Observe the modest and similar cartilage damage of both knees.
In a previous study, we found cartilage damage of the medial tibia opposing defects treated with FKRM as well as in the nonoperated healthy knee. 23 We acknowledged that cartilage damage was partly age-related and consequently only young ewes (2-3 years) were considered for inclusion in the present study. Still, cartilage wear occurred in the medial compartment, regardless if a defect was treated or left untreated. The degree of cartilage degeneration was comparable to what others have found in healthy knees of goats of equivalent age,27,30 and similar to our nonoperated knees from previous experiments ( Fig. 6 ). It could be speculated that even healthy joint cartilage exhibits moderate lesions, often minor fissures that demonstrates the metabolic capability of cartilage repair in the young ewe. Analyzing the cartilage health of the knee joint in different compartments, we did not find any statistically significant difference in cartilage damage between medial and lateral compartments either when defects of the medial femoral condyle were treated or left untreated. This is contradicting the view of others who suggested that the homeostasis of the whole knee is altered by focal lesions, FKRM implants, or microfracturing, and mediated by metalloproteases from the damaged cartilage reaching distant compartments with the synovial fluid.22,27,30,36 In contrast, our current data indicate that an inserted implant in the medial compartment or an untreated defect does not predispose for general cartilage deterioration. This is in line with our previous data supporting the impression of only local effects secondary to FKRM implants.
The second main finding of this study was that cartilage bordering the implant remained healthy. We recognized that the integrity of the implant-cartilage interface has to be maintained not to risk deleterious effects on the joints general health. We have previously reported on the role of hydroxyapatite covering the circumference of the implant hat and suggest that the histochemical properties of this material enhance integration between FKRM implants and the surrounding cartilage ( Fig. 11 ). 21 Because previous studies have evaluated cartilage lesions or repair tissue only, we could not rely on those validated classifications.31-34 In an attempt to evaluate the implant-cartilage interface in a standardized manner, we created a method (Table 1) based on previous applicable aspects on cartilage repair. Our 2 groups of animals (6 animals from a previous experiment and 6 animals from the present experiment) about 3 years of age, with unilateral implants, yielded satisfactory results in terms of cartilage health abutting the implant and its chondrointegration.
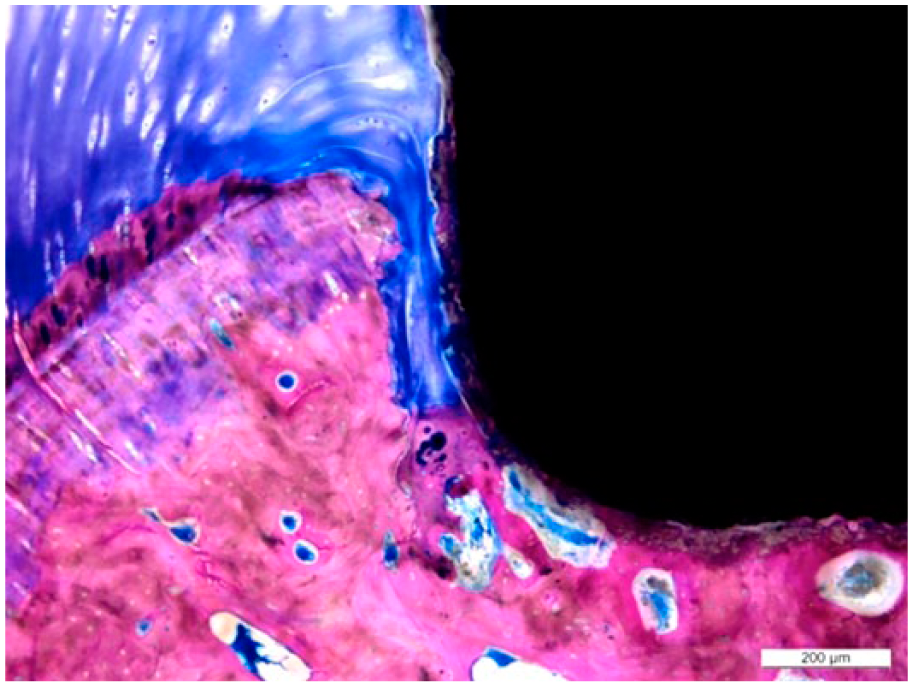
An excellent integration of cartilage to the hydroxyapatite-covered areas of the hat without intervening soft tissue (chondrointegration).
In our group of 6 ewes, we had an osseointegration of about 80%, which is high when compared with experimental data reported by others but less than in our previous experiments which was about 90%.22,36-38 We think the reason for this somewhat inferior result is a design change of the fixating peg, using an irregular shape resembling the threaded design used by others.27,38 Theoretically, this would increase the bone-implant contact area, but since all implants are press-fitted, bone might rather be pushed away leaving voids, resulting in less bone to implant contact. We have therefore reverted to our primary design with straight pegs and would rather advice some caution when using threadlike designs.
We have previously demonstrated a correlation between implant position and cartilage damage and recommended that the implant should be seated somewhat recessed (0.5 mm). 37 In our present group of 6 animals, 1 implant was both shallow and tilted and consequently protruded (0.4 mm), which could explain the devastating damage to opposing tibia ( Figs. 3 - 5 and 12 ). An implant should never protrude, and we believe that the most accurate and consistent method to achieve optimal position is using a custom-made implanting device or other advanced instrumentation.
Cross-sectional histological picture (Safranin O) of the tibal cartilage surface opposing a protruding implant (secondary to tilting).
The spontaneous healing of the untreated defects was minor as shown in other studies, and in most cases even progressed in terms of erosion at the border of the defect at short-term (6 months).4,8
A major limitation of this study is the small sample of animals left for histological analysis due to the relatively large number of dropouts (3/9). Also limiting the interpretation of the results is the wide range of cartilage damage of both treated and untreated knees complicating statistical evaluation. Hence our results should only be used to create hypotheses that should be tested in future experiments. The inclusion of animals from a previous study could be criticized but was for ethical reason in order to minimize the number of animals used. Because those animals were of similar age and general joint health, and the tissue samples from the implant-cartilage interface had not been previously analyzed, we believe the use of the 2 data sets was justified.
Conclusion
Based on our results it is adequate to postulate that these implants constitute a reasonable alternative treatment for symptomatic localized cartilage defects of the femoral condyle. The implant positioning is accurate and consistent, osseointegration is reliable and cartilage health adjacent to the implant is satisfactory. Nevertheless, we advise caution when treating asymptomatic localized defects with metal implants until supported by further research.
Footnotes
Acknowledgments and Funding
Dr. Ryd was involved with study design and performance of surgeries. Dr. Ryd reports financial support as a board member from Episurf Medical AB, Stockholm. For this reason he did not participate in the analysis of data and writing of the manuscript. HN-S performed all surgeries and was responsible for care of animals. We thank Episurf Medical AB. Stockholm for kindly providing implants and instruments and financial support for animal studies.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Martinez-Carranza reports grants from Stockholm County Council, Karolinska Institutet, Karolinska University Hospital, and from Episurf Medical AB. Stockholm, Sweden, during the conduct of the study. Dr. Berg reports grants from Stockholm County Council, Karolinska Institutet, and grants from Episurf Medical AB. Stockholm, during the conduct of the study. Dr. Lagerstedt and Dr. Nurmi-Sandh report nonfinancial support for laboratory facilities from Episurf Medical AB. Stockholm, during the conduct of the study. Dr. Schupbach reports nonfinancial support for laboratory facilities from Episurf Medical AB, Stockholm during the conduct of the study.
Ethical Approval
Ethical approval for this study was obtained from the Animal Experiments Ethical Board; Uppsala Sweden; C271/8 and C135/10.
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
