Abstract
Objective
Despite the mechanical and biological roles of subchondral bone (SCB) in articular cartilage health, there remains no consensus on the postoperative morphological status of SCB following bone marrow stimulation (BMS). The purpose of this systematic review was to clarify the morphology of SCB following BMS in preclinical, translational animal models.
Design
The MEDLINE and EMBASE databases were systematically reviewed using specific search terms on April 19, 2016 based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. The morphology of the SCB was assessed using of microcomputed tomography (bone density) and histology (microscopic architecture).
Results
Seventeen animal studies with 520 chondral lesions were included. The morphology of SCB did not recover following BMS. Compared with untreated chondral defects, BMS resulted in superior morphology of superficial SCB and cartilage but inferior morphology (specifically bone density, P < 0.05) of the deep SCB. Overall, the use of biological adjuvants during BMS resulted in the superior postoperative morphology of SCB.
Conclusions
Alterations in the SCB following BMS were confirmed. Biologics adjuvants may improve the postoperative morphology of both SCB and articular cartilage. Refinements of BMS techniques should incorporate consideration of SCB damage and restoration. Investigations to optimize BMS techniques incorporating both minimally invasive approaches and biologically augmented platforms are further warranted.
Introduction
The treatment of symptomatic cartilage defects remains challenging despite intense focus from the orthopaedic and scientific communities. Bone marrow stimulation (BMS) is considered the first-line treatment for symptomatic cartilage defects because of its minimally invasive approach, cost-effectiveness, and limited surgical morbidity 1 ; it is also considered a gold standard for comparison of emerging cartilage repair technologies. 2 BMS techniques (microfracture and/or drilling) stimulate cartilage healing by directly accessing the regenerative elements of bone marrow by penetrating the subchondral bone (SCB) directly from within the defect. 3 The resultant regenerative mesenchymal clot contains stem cells and growth factors that support the formation of fibrocartilage within the defect. Patient-reported outcomes have shown significant improvement following marrow stimulation in the short to medium term with a progressive decline in the performance of the repair tissue in the medium to long term. Inferior biomechanical properties of fibrocartilage and postinjury joint homeostasis have been implicated.3-6
The SCB consists of a superficial plate and deep spongy bone that performs important mechanical and biological roles to support and maintain the overlying cartilage.7-11 The SCB is known to absorb 70% of compressive loads mechanically and have cross-talk with the articular cartilage, communicating biochemical signals through transport pathways between them. 7 Contextually, it is currently unclear if the surgical trauma required during BMS procedures (removal of the calcified cartilage layer and penetration of the SCB) is recoverable. Given the importance of SCB in cartilage health,7-11 persistent deficits in its structure may affect the quality and sustainability of the repair tissue. 12
The purpose of this systematic review was to clarify the morphology of SCB following BMS in preclinical, translational animal models.
Methods
Search Strategy
Two independent reviewers performed a systematic review of the MEDLINE and EMBASE databases in April 19, 2016 based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 13
The search terms were: (subchondral bone plate OR subchondral bone plate over growth OR subchondral bone plate remodeling OR osteocartilage regeneration OR osteocartilage repair OR tidemark) AND (bone marrow stimulation OR microfracture OR drilling OR abrasion). References of all searched articles were also screened for possible inclusion. The inclusion and exclusion criteria are shown in Table 1 .
Inclusion and Exclusion Criteria.

Two independent reviewers screened the titles, abstracts and full-text articles of all searched studies by applying the aforementioned criteria. Any disagreements were resolved by consensus and if disagreement persisted, a senior author was consulted ( Fig. 1 ).
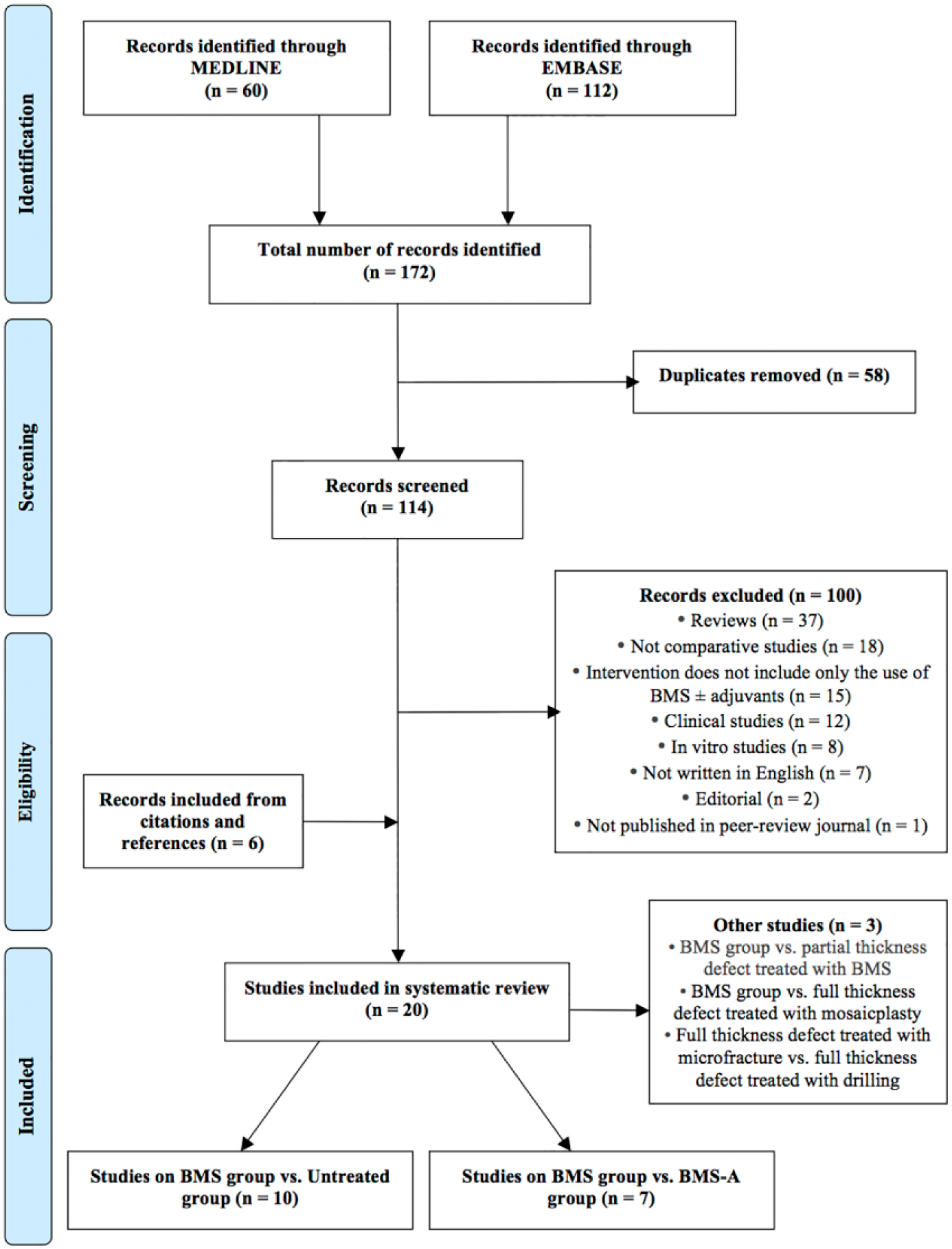
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram.
Data Evaluation
A standardized sheet was used to extract predetermined variables. The methodological quality of evidence (QoE) was evaluated using the Modified Horn Scale ( Table 2 ). 14 This is an 8-point scale designed to evaluate the QoE in animal studies. Each criterion was worth 0 or 1 point; 1 point was awarded when the study reports the criteria being mentioned. Studies scoring <4 points were graded as having poor QoE and studies with ≥4 points were graded as having good QoE.
Modified Horn Scale.

BMS, bone mallow stimulation; µCT, microcomputed tomography.
The included studies were categorized into 2 groups based on the character of the chondral lesion: (1) full-thickness chondral defect treated with BMS (BMS group) versus full-thickness chondral defect untreated (untreated group) and (2) BMS group versus full-thickness chondral defect treated with BMS + adjuvants (BMS-A group). Studies were then divided according to their time(s) until evaluation into 3 time phases: acute (0 to 2 weeks), subacute (2 weeks to 3 months) and late (≥6 months). Individual morphological assessment was then made according to the various anatomical parts of SCB: deep SCB and superficial SCB. This included microcomputed tomography (µCT) to assess the bone densities and histology to assess microscopic architectures.
SCB and Cartilage Assessment
µCT assessments were made only for the SCB and was divided into deep and superficial components according to previously published criteria. 15 The deep SCB was assessed using “bone volume over total volume” (BV/TV; %) and the superficial SCB was assessed using “bone surface area over bone volume” (BS/BV; mm−1). Both these are measurements of bone densities of the deep SCB and superficial SCB, respectively. Assessments of superior/inferior µCT results were made within the study and generalized across the included studies.
Histological assessments were made for both SCB and cartilage according to various reported grading systems within individual studies (American Association of Equine Practitioners lameness scale, histomorphometry, immunohistochemistry, International Cartilage Repair Society II score, O’Driscoll score, Modified O’Driscoll score and Sanz score). Assessments of superior/inferior histological assessments were made within the study first; trends were then generalized across the included studies. Histological assessments of the deep SCB and superficial SCB were defined anatomically. 7 Histological assessment of the deep SCB was noted when microscopic architectural assessment of the spongy bone was made and histological assessment of the superficial SCB was noted when microscopic architectural assessment of the bone plate was made.
Definition of Morphology
Morphology of the SCB referred to its structural components assessed by µCT and/or histology. In histological analysis, superior morphology of cartilage referred to increased hyaline-like repair with inferior morphology of cartilage referred to the contrary. In µCT analysis, superior morphology could not be made in reference to increased or decreased bone density as this is currently unknown for the SCB specifically and therefore, the term was avoided in reference to bone density.
Results
BMS Group versus Untreated Group
The BMS group versus untreated group studies are presented in Table 3 .
BMS Group versus Untreated Group Studies.
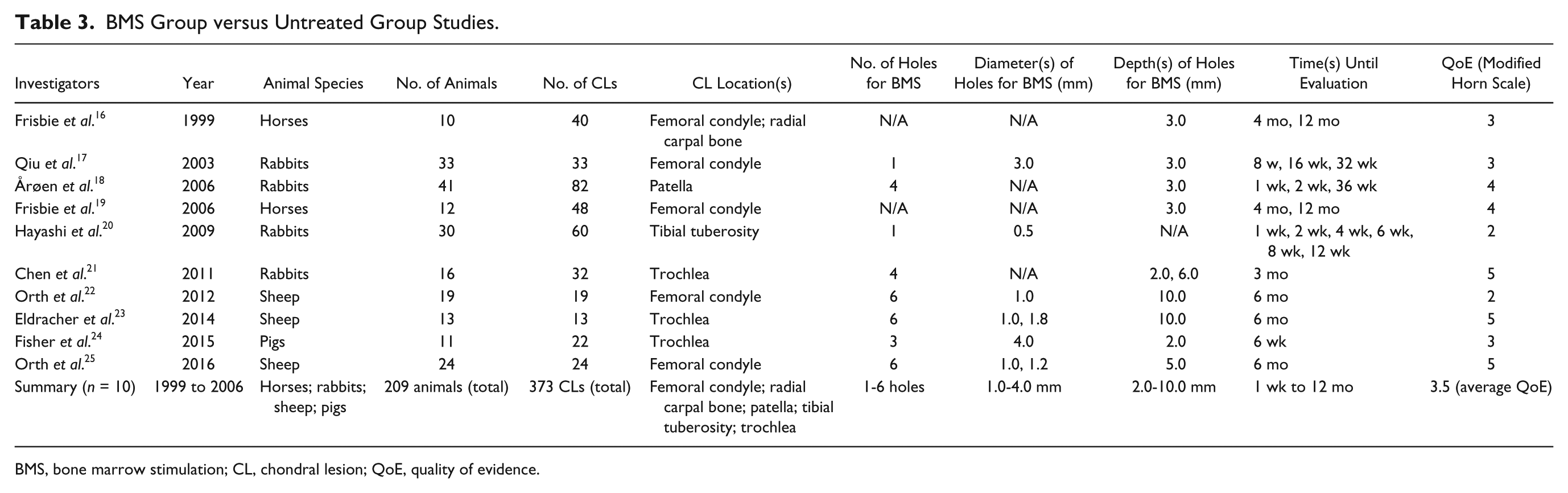
BMS, bone marrow stimulation; CL, chondral lesion; QoE, quality of evidence.
Deep SCB
µCT assessment of the deep SCB included 5studies ( Table 4 ). A subacute phase study by Fisher et al. 24 indicated in both µCT and histology that neither the BMS or untreated group restored the bone density or histological architecture of the deep SCB back to normal (P < 0.05). Bone density comparisons between the BMS and untreated group revealed conflicting findings in the subacute phase. Chen et al. 21 indicated significantly increased bone density in the untreated group compared with the BMS group whereas Fisher et al. 24 indicated the opposite of these findings. However, 3 late phase studies further supported the findings of Chen et al. 21 indicating significantly increased bone densities in the untreated group than the BMS group (P < 0.05).22,23,25
BMS Group versus Untreated Group Summary of Results.
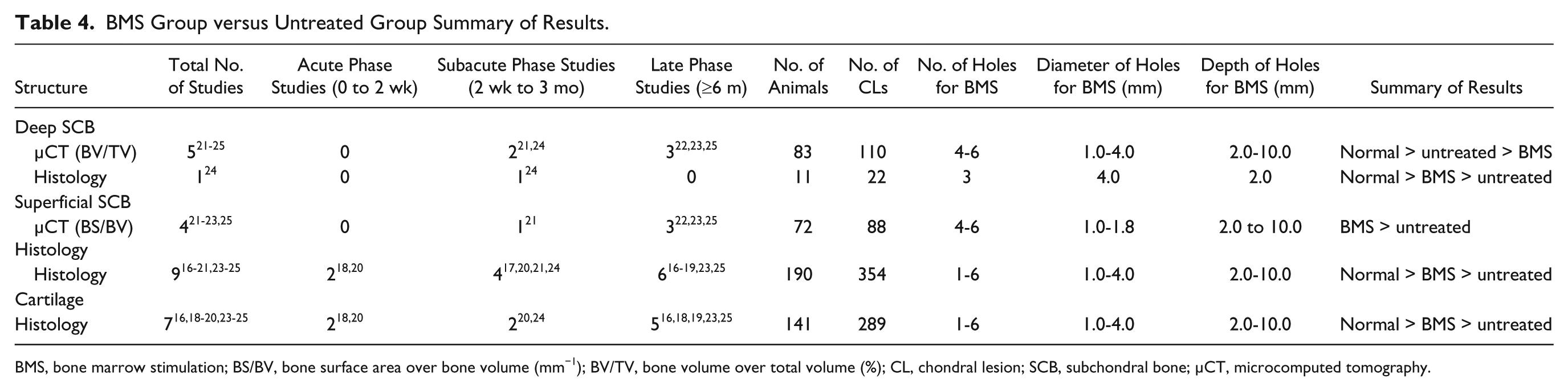
BMS, bone marrow stimulation; BS/BV, bone surface area over bone volume (mm−1); BV/TV, bone volume over total volume (%); CL, chondral lesion; SCB, subchondral bone; µCT, microcomputed tomography.
Histological assessment of the deep SCB included 1 study in the subacute phase ( Table 4 ). 24 Inferior histological architectures were noted in both the BMS and untreated groups compared with normal. Additionally, superior histological architecture was indicated in the BMS group compared with the untreated group.
Superficial SCB
µCT assessment of the superficial SCB included 4 studies ( Table 4 ). µCT assessments did not compare either the BMS or untreated group to normal.21-23,25 However, all studies indicated significantly increased bone densities in the BMS group compared with the untreated group (P < 0.05).
Histological assessment of the superficial SCB included 9 studies ( Table 4 ). In 6 studies that were collectively included in all time phases, inferior histological architectures were indicated in both the BMS and untreated groups compared with normal.17,19-21,23,24 Four studies that were also collectively included in all time phases indicated superior histological architectures in the BMS group compared with the untreated group.16,18,24,25 Both µCT and histological assessments of the superficial SCB were similar, except for the lack of comparison to normal by µCT in both the BMS and untreated groups. Interestingly, Qiu et al. 17 using drilling demonstrated in the only study of an osteochondral model (n = 33; osteochondral lesions = 33) that the superficial SCB had in fact overgrown beyond its native tidemark.
Cartilage
Histological assessment of the cartilage included 7 studies ( Table 4 ). In 5 studies that were collectively involved in all the time phases, inferior histological architectures were noted in both the BMS and untreated group compared with normal.18-20,23,24 Three studies compared histological architectures between the BMS and untreated group in the acute and late phases revealed that the BMS group was significantly superior to the untreated group (P < 0.05).16,18,25 However, a subacute phase study by Fisher et al. 24 instead indicated similar histological architecture between the BMS and untreated group.
Relationship Between Morphology of SCB and Cartilage Repair
Six studies of all time phases compared the morphological relationship of the SCB and cartilage in the BMS to untreated groups; increased bone density and superior histology of the superficial SCB was indicated to be related with increased hyaline-like cartilage repair.16,18-20,23,25 Two of these studies also assessed the deep SCB in the late phase, indicated that worsening morphology (specifically bone density) of the deep SCB related with increased hyaline-like cartilage repair.23,25 However, 1 study conflicted both these findings in the subacute phase, indicating superior morphology of the deep SCB relating to increased hyaline-like cartilage repair and that there was no morphological relationship between the superficial SCB and hyaline-like cartilage repair. 24
BMS Group versus BMS-A Group
The BMS group versus BMS-A group studies are presented in Table 5 .
BMS Group versus BMS-A Group Studies.
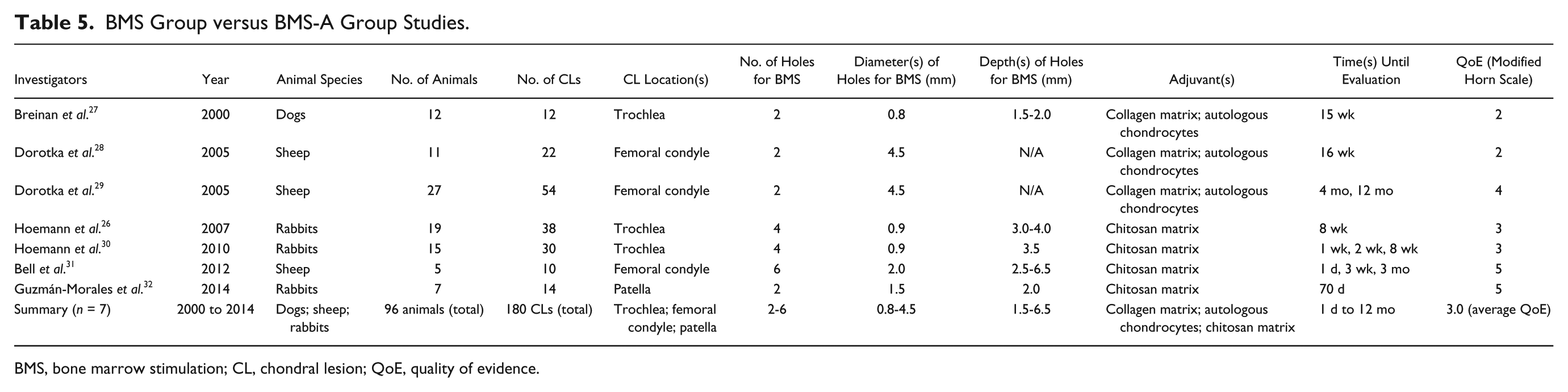
BMS, bone marrow stimulation; CL, chondral lesion; QoE, quality of evidence.
Deep SCB
µCT assessment of the deep SCB included 1 study in the subacute phase using chitosan matrix as the adjuvant. 26 µCT assessments did not compare either the BMS or BMS-A groups with normal. However, significantly increased bone density was indicated in the BMS-A group compared with the BMS group (P < 0.0005). Histological assessment of the deep SCB was not made in this study.
Superficial SCB
µCT assessment of the superficial SCB also included 1 study in the subacute phase using chitosan matrix as the adjuvant. 26 Similarly, no µCT assessments compared either the BMS or BMS-A groups to normal but significantly increased bone density was indicated in the BMS-A group compared with the BMS group (P < 0.0005).
Histological assessment of the superficial SCB included all 7 studies of this category ( Table 6 ). In all 7 studies that were collectively included in all time phases, no histological assessments in relation to normal for either the BMS or BMS-A groups were made.26-32 Histological assessments made between the BMS and BMS-A groups revealed contrasting results depending on the adjuvant used. Four studies in the acute and subacute phases using chitosan matrix adjuvants indicated superior histological architecture in the BMS-A group compared with the BMS group.26,30-32 A range of results was presented when collagen matrix and autologous chondrocytes adjuvants was compared with the BMS group. Breinan et al. 27 indicated superior histological architecture in both the collagen matrix and autologous chondrocytes groups compared with the BMS group. However, 2 other studies by the same first author (Dorotka),28,29 compared collagen matrix and autologous chondrocytes adjuvants to the BMS group alone at 4 months revealed inferior histological architecture in the collagen matrix adjuvant but superior histological architecture in the autologous chondrocytes adjuvant. However, one of these studies also evaluated the outcomes at 12 months and indicated superior histology in both the collagen matrix and the autologous chondrocytes groups at that time point. 29
BMS Group versus BMS-A Group Summary of Results.
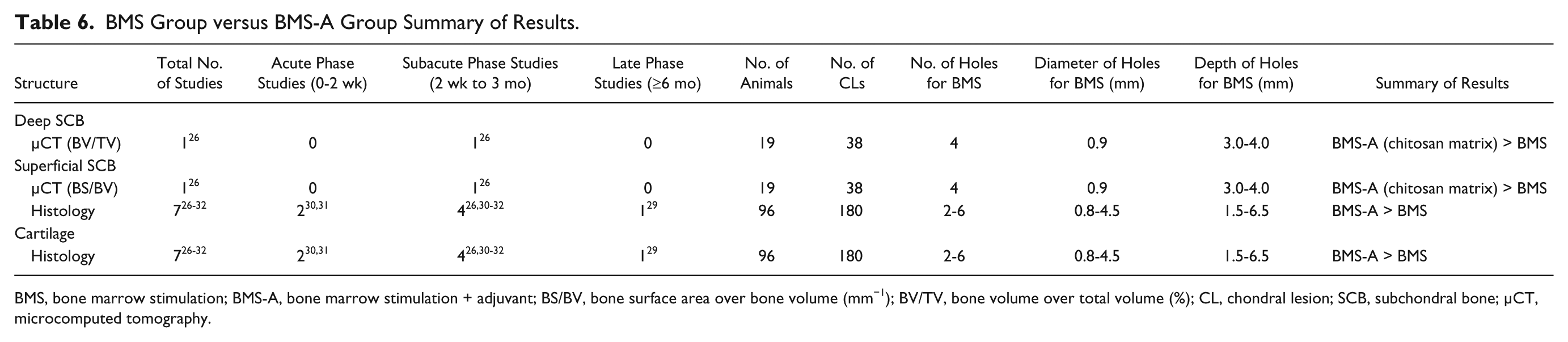
BMS, bone marrow stimulation; BMS-A, bone marrow stimulation + adjuvant; BS/BV, bone surface area over bone volume (mm−1); BV/TV, bone volume over total volume (%); CL, chondral lesion; SCB, subchondral bone; µCT, microcomputed tomography.
Cartilage
Histological assessment of the cartilage also included all 7 studies of this category ( Table 6 ). The findings here were similar to that of the histological assessment of the superficial SCB except in the study by Breinan et al., 27 where similar results were revealed in both the collagen matrix and the autologous chondrocyte groups compared with the BMS group. However, this result was statistically insignificant in the study.
Relationship Between Morphology of SCB and Cartilage Repair
Seven studies of all time phases compared the morphological relationship of the SCB and cartilage in the BMS and BMS-A groups indicated that increased bone density and superior histology of the SCB related with increased hyaline-like cartilage repair. This was observed in 4 studies of all time phases assessing the morphology of the superficial SCB28-32 and 1 study assessing the morphology of both the superficial and deep SCB. 26 One study revealed a lack of relationship between the morphology of the superficial SCB and hyaline-like cartilage repair. 26
Discussion
The consensus of postoperative morphological status of SCB following BMS and its impact on cartilage repair and clinical outcomes has not been established yet, despite the advocates of the important role of the SCB in cartilage repair. 12 This systematic review demonstrated that contemporary BMS techniques resulted in alterations of the SCB in preclinical animal models. Specifically, bone density of the deep SCB significantly decreased to the untreated defect. In addition, biological adjuvants appeared to improve the morphology of the SCB compared with BMS alone. Although no conclusive outcomes have not been achieved in available studies, increased bone density and superior histology of superficial SCB may be important for cartilage repair. In this systematic review, animal studies were different experimental designs including different BMS techniques, different animal species, different locations of defects in the joint (trochlea vs tibia vs condyles). The variations in articular cartilage and SCB biomechanics between joints should be carefully accounted for when interpreting these results. 33 Nevertheless, these outcomes may be valuable for further research because of the consistency of the findings among the studies.
Successful cartilage repair and re-loading at the defect site would be expected to increase bone density due to an innate plasticity of bone that allows it to adapt to shield greater forces in accordance with Wolff’s law. 34 However, this was not observed in the current study for the deep SCB; superficial SCB was not reported. Although the etiology of these findings is uncertain, a number of potential mechanisms may be responsible for this phenomenon and warrant consideration. At the tissue level, inferior biomechanical properties of fibrocartilage and loss of the calcified cartilage layer may alter force transduction and fluid flow within the defect and in the SCB below. 35 At the cellular level, mechanotransduction of bone cells (similar to chondrocytes) is mediated by primary cilia responding to load-mediated local shifts of interstitial fluid pressure. 36 It is currently unclear if penetration of SCB with exposure to the joint environment, loss of the calcified cartilage (and vascular watershed) and more permeable fibrocartilaginous repair tissue are capable of disrupting osteoblast mechanotransduction locally at the microfractured defect.
Given the importance of SCB in cartilage health,7-11 the relationship between the affected SCB and cartilage repair tissue quality and quantity was important concern in this systematic review. Although no conclusive outcomes have not been achieved in current available studies, increased bone density and superior histology of superficial SCB may be important for cartilage repair.16,18-20,23,24 The current study also demonstrated that bone density of the deep SCB is significantly reduced in the BMS group when compared with the untreated group. These characterizations provide a basis to investigate less detrimental approaches to improve the quality and quantity of the reparative fibrocartilage. In accordance, Gianakos et al. 12 reported superior SCB morphology for 1.0 mm diameter awls compared with the 2.0 mm in the talar cadaver model. Eldracher et al. 23 reported superior cartilage repair and SCB morphology for 1.0-mm diameter drilling compared with 1.8 mm at 6 months in the sheep trochlear defect model. Orth et al. 25 also reported superior cartilage repair and SCB morphology but for 1.0 mm diameter awls compared with 1.2 mm at 6 months in the medial femoral condyle of the sheep model. While the smaller awl resulted in reduced morphological disruption of the SCB, SCB cysts and intralesional osteophytes were observed at similar frequencies. In contrast, Marchland et al. 37 compared 0.5- and 0.9-mm diameter drill holes in the rabbit model observed no difference in the cartilage tissue repair or SCB morphology at 6 months. Kok et al. 38 similarly observed no difference between 0.45- and 1.1-mm diameter awls in the talar osteochondral defects of a goat model. With regard to the relationship between morphology of deep SCB and quality and quantity of reparative cartilage tissue, the current study showed the conflict findings.23-25 Despite these findings, as similar to superficial SCB, deep SCB is supposed to be important structure for cartilage repair, based on the function of SCB for the articular cartilage. Chen et al. 39 reported that 6-mm depth holes produced statistically superior cartilage repair compared with 2-mm depth holes at 3 months in the rabbit trochlear defect model. Further investigations should focus on the relationship between morphology of SCB and quality and quantity of reparative cartilage tissue as well as less impassive subchondral penetration approaches.
The optimal approach to improve the quality of cartilage repair is to enhance regeneration of the cartilage whilst also promoting the restoration of the SCB affected by BMS. Currently, various biologics have been used in cartilage repair, including platelet-rich plasma, concentrated bone marrow aspirate, scaffold-based therapy and chondrocyte-based therapy. Successful short-term functional outcomes following these biological adjuvant therapies in bone marrow stimulation have been reported.6,40 However, little information remains available for their capabilities for the health of the SCB in BMS. Outcomes from the current systematic review suggest that biological adjuvants (collagen matrix, autologous chondrocytes and chitosan matrix) may improve the morphology of the SCB than that of BMS alone. Postulations can include innate osteochondroconductive and/or -inductive properties of the biological adjuvants. Future studies that incorporate both the morphology of the SCB and cartilage health as measures of BMS success are needed to confirm this finding.
Several inherent limitations could have hindered the accuracy of findings in this study. The QoE was assessed according to Modified Horn Scale whereby the validity remains questionable, as no studies have validated this scale to date. Secondly, the translation of the findings from animal to human models remains a challenge as the potential differ in joint loading and innate structural stability in animals can mislead the understanding in humans. Finally, the marked heterogeneity from the vast range of animals, surgical techniques and outcome measures exhausted may affect the clinical translation of the results. Nevertheless, as these findings were consistent, the outcomes from current study is valuable for future studies investigating the SCB.
Conclusion
In this systematic review, animal studies were different experimental designs, including different BMS techniques, different animal species, different locations of defects in the joint (trochlea vs. tibia vs. condyles). Nevertheless, obtained outcomes were the consistency among the studies. The current study demonstrated that BMS techniques resulted in alterations of the SCB and that biological adjuvants appeared to improve the morphology of the SCB compared with BMS alone. Refinements of BMS techniques should incorporate consideration of SCB damage and restoration. Investigations to optimize BMS techniques incorporating both minimally invasive approaches and biologically augmented platforms are further warranted.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: John G. Kennedy is a consultant for Arteriocyte, Inc; received research support from the Ohnell Family Foundation, Mr. and Mrs. Michael J. Levitt, Arteriocyte Inc; is a board member for the European Society of Sports Traumatology, Knee Surgery, and Arthroscopy (ESSKA), International Society for Cartilage Repair of the Ankle (ISCRA), American Orthopaedic Foot & Ankle Society (AOFAS) Awards and Scholarships Committee, International Cartilage Repair Society (ICRS) finance board.
Ethics approval
Ethical approval was not sought for the present study because this study is a systematic review of published animal studies.
Animal welfare
Guidelines for humane animal treatment did not apply to the present study because this study is a systematic review of published animal studies.
