Abstract
Objective
This study tested the hypothesis that presolidified chitosan-blood implants are retained in subchondral bone channels perforated in critical-size sheep cartilage defects, and promote bone repair and hyaline-like cartilage resurfacing versus blood implant.
Design
Cartilage defects (10 × 10 mm) with 3 bone channels (1 drill, 2 Jamshidi biopsy, 2 mm diameter), and 6 small microfracture holes were created bilaterally in n = 11 sheep knee medial condyles. In one knee, 10 kDa chitosan–NaCl/blood implant (presolidified using recombinant factor VIIa or tissue factor), was inserted into each drill and Jamshidi hole. Contralateral knee defects received presolidified whole blood clot. Repair tissues were assessed histologically, biochemically, biomechanically, and by micro–computed tomography after 1 day (n = 1) and 6 months (n = 10).
Results
Day 1 defects showed a 60% loss of subchondral bone plate volume fraction along with extensive subchondral hematoma. Chitosan implant was resident at day 1, but had no effect on any subsequent repair parameter compared with blood implant controls. At 6 months, bone defects exhibited remodeling and hypomineralized bone repair and were partly resurfaced with tissues containing collagen type II and scant collagen type I, 2-fold lower glycosaminoglycan and fibril modulus, and 4.5-fold higher permeability compared with intact cartilage. Microdrill holes elicited higher histological ICRS-II overall assessment scores than Jamshidi holes (50% vs. 30%, P = 0.041). Jamshidi biopsy holes provoked sporadic osteonecrosis in n = 3 debrided condyles.
Conclusions
Ten kilodalton chitosan was insufficient to improve repair. Microdrilling is a feasible subchondral marrow stimulation surgical approach with the potential to elicit poroelastic tissues with at least half the compressive modulus as intact articular cartilage.
Keywords
Introduction
The microfracture technique developed by Steadman et al., 1 is routinely used in the clinic to repair full-thickness cartilage lesions. While microfracture therapy and augmented microfracture therapy can lead to improved function and reduced pain,2-4 the resulting cartilage repair tissues are heterogeneous and can vary from no repair (bone), to fibrous repair, fibrocartilage, to stalled hyaline or hyaline-like repair.3-6 Human cartilage repair biopsies taken 13 months postoperative demonstrated the potential for microfracture or augmented microfracture to elicit full-thickness hyaline-like cartilage repair tissues5,6; therefore, research to further optimize the marrow stimulation technique is amply justified.
This study investigated a novel surgical resurfacing approach in a large animal model that we term subchondral marrow stimulation (SMS). SMS consists in the creation of a subchondral bone channel in a debrided lesion that is then treated with a subchondral implant.7-10 The SMS approach is distinguished from most augmented marrow stimulation approaches where an implant is delivered or fixed to the exposed bone in the base of a microfractured cartilage lesion.11-14 An SMS implant is more intimately associated with the bone marrow stromal cells that contribute to the repair process, and is also protected from biomechanical stresses that could affect implant retention. Because marrow stimulation holes of different depth and diameter generated with different tools can heal with distinct rates and produce different tissues,15,16 the SMS implant must be tailored to the particular surgical marrow stimulation approach.
A subchondral, rapidly degrading 10 kDa chitosan-NaCl/blood implant was previously shown to effectively stimulate hyaline cartilage repair compared to drilling alone in an aged rabbit SMS model.7,8 Chitosan is a biodegradable and biocompatible polysaccharide containing glucosamine and N-acetyl glucosamine, with accelerated clearance rates for lower molecular weight polymers. 17 Presolidified chitosan/blood implants generated with chitosans of different molecular weights were successfully delivered and retained in sheep subchondral bone defects created with Jamshidi biopsy needles, and positively influenced the osteochondral repair response. 9 To date, presolidified chitosan/blood implants were tested for their capacity to heal 1.5- to 2-mm osteochondral defects in rabbit and sheep models.7-9 It is currently uncertain whether these implants can be used to stimulate cartilage resurfacing of a debrided critical-size cartilage lesion through an SMS approach. Because implants with slow subchondral clearance rates can inhibit subchondral bone regeneration and delay cartilage resurfacing,10,18 this study explored the feasibility of using a rapidly degrading chitosan/blood implant to resurface cartilage lesions in a sheep SMS model.
In this study, a sheep model was chosen because of the relatively thick, dense subchondal bone plate in this species, which mimics sclerosis in osteoarthritic patients. 19 Because microfracture can produce bone compaction of dense subchondral bone plate,20,21 we created three 2-mm diameter channels with a microdrill bit and 11G Jamshidi needles as tools to cleanly remove bone and improve recruitment and migration of marrow elements to defects. Procoagulants were used to presolidify the chitosan-NaCl/blood implants, including recombinant human factor VIIa (rhFVIIa) which has chemotactic and mitogenic properties, and tissue factor (TF), which reliably clots blood of different species.22-24 To control for the use of coagulation factors to generate presolidified implants and uncertain impact on repair responses, control contralateral bone channel defects were treated with blood clot implant presolidified with the same coagulation factor. Repair tissues were analyzed at day 1 for bone structure and implant retention, and at 6 months for repair cartilage macroscopic appearance, histological quality, biochemistry, biomechanics, and subchondral bone structure.
Materials and Methods
Chitosan-Blood Implants
An autoclave-sterile solution of chitosan-HCl pH 5.4, 50 mOsm containing chitosan with 81.9% degree of deacetylation (DDA), number-average molecular weight (Mn) 10.5 kDa and polydispersity (PDI, Mw/Mn) 3.37 was generated in-house from chitosan sourced from Marinard (Gaspé, QC, Canada) as previously described. 7 Rhodamine isothiocyanate (RITC)-chitosan (81.9% DDA, Mn = 10.3 kDa, PDI = 2.21, 1% mol/mol RITC/chitosan) was prepared in-house, 25 dissolved at 5 mg/mL in dilute HCl at 90% protonation and filter-sterilized. Chitosan was stored in frozen aliquots at −80°C. Sterile clinical-grade recombinant human factor VIIa (rhFVIIa, NiaStase, Novo Nordisk, Copenhagen, Denmark) was prepared at 500 µg/mL in sterile water, stored at −80°C as frozen aliquots, and on the day of surgery diluted to 100 µg/mL with Ringer’s lactated saline. Tissue factor (TF) (Innovin, Dade-Behring, Cedarlane, Mississauga, Ontario, Canada), supplied as lyophilized preparation with phospholipids, was reconstituted with 2 mL sterile water to form a 5× concentrated solution. 200 µL 10 kDa chitosan-HCl and 50 µL 750 mM filter-sterile 750 mM NaCl were combined with 1.5 mL of fresh autologous sheep blood to generate a liquid mixture at a 6:1 blood:chitosan ratio. To create rhFVIIa implants, 35 µL of rhFVIIa was added to 1.5 mL uncoagulated blood (controls) or to chitosan-NaCl/blood mixture (implants) in a cryovial containing 3 surgical steel beads, shaken, and then drawn into a 2-mm inner diameter sterile glass tube, which was placed aseptically at 37°C for 10 minutes and then extruded onto a sterile Petri dish. Because the blood implant failed to coagulate with rhFVIIa in the fourth sheep in the study, implants for the 7 remaining sheep were generated with TF. Fifteen microliters of TF was pipetted in a line in a sterile 100 mm tissue culture dish and 100 µL of uncoagulated blood or chitosan-NaCl/blood was pipetted on top of the line of tissue factor, and allowed to coagulate in the Petri dosh held on the palm of the hand (for warmth) 1 minute. Control tissue factor implants were generated using the same approach but using whole blood. Solid tube-shaped implants were trimmed into ~3-mm long pieces with a scalpel prior to inserting in the defects. Day 1 chitosan implants were generated with 25 µL of RITC-chitosan added to the 10 kDa chitosan-NaCl/blood mixture and TF solidification.
Animals and Surgical Procedures
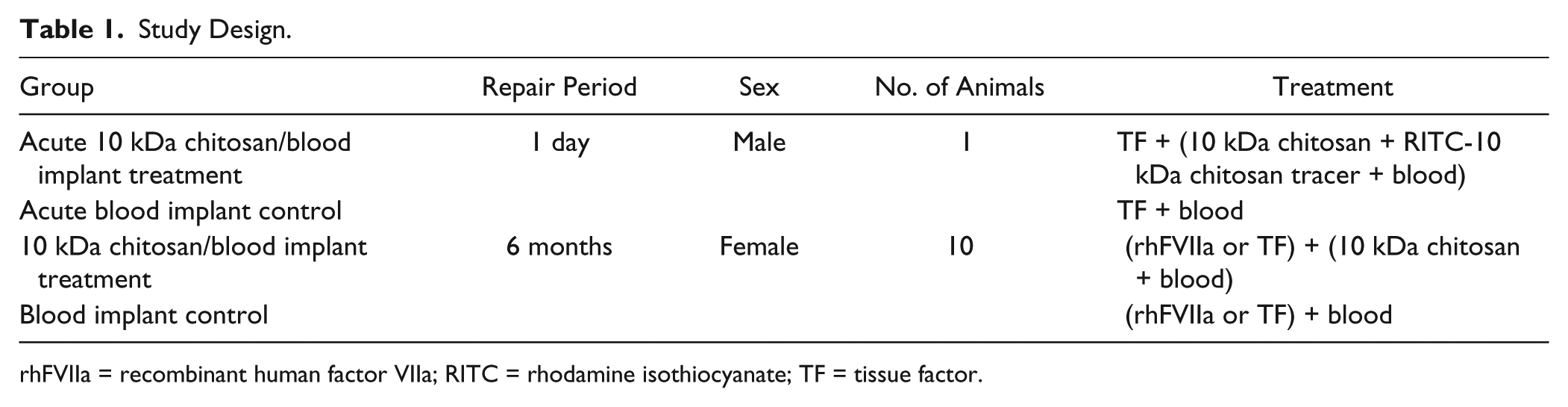
Approval for use of animals in research was given by University of Guelph Animal Care Committee (Protocol #3113), which is compliant with the Canadian Council for Animal Care guidelines. Skeletally mature Arcott cross sheep (n = 10 females, n = 1 male, 2-4 years, 65-93 kg) from specific pathogen-free herds were chosen for this study. The male sheep was used for acute day 1 repair assessments and the female sheep were randomized according to weight for distribution into treatment groups to evaluate 6-month repair (Table 1). Sheep were anaesthetized with intravenous ketamine and diazepam and then maintained on isoflurane gas. All animals received intravenous prophylactic antibiotics (20,000 IU/kg penicillin and 2.2 mg/kg gentamicin) and nonsteriodal anti-inflammatory pain medication (1.1 mg/kg flunixin) during surgery. Using aseptic technique, a medial infrapatellar arthrotomy was created to expose the medial femoral condyle (MFC) of each leg. A 10 × 10 mm2 full-thickness cartilage defect debrided of the calcified layer was made in the middle of the central weightbearing area of the MFC with a curette, then using a sterile aluminum foil template to precisely position all holes, 2 holes were generated inside the defect with a 11G Jamshidi needle (2 mm inner diameter, Cardinal Health, Dublin, OH, USA, using sterile silastic sleeves fitted outside the Jamshidi needle to achieve a 4mm target hole depth), and 1 hole was created with a high-speed handheld drill and 2.1 mm diameter microdrill burr with a round head (target 3 mm deep, Fine Science Tools, North Vancouver, British Columbia, Canada) followed by 6 holes with a microfracture awl (~3 mm deep, Sontec, Centennial, CO, USA) ( Fig. 1A ). In treated defects, presolidified chitosan-blood implant was delivered to the 2 Jamshidi holes and drill hole then 200 µL of the uncoagulated chitosan-NaCl/blood-rhFVIIa or chitosan-NaCl/blood mixture was pipetted on top of the defect. In contralateral control defects, Jamshidi and drill holes received whole blood clot implant presolidified with rhFVIIa or TF and 200 µL of the uncoagulated whole blood-rhFVIIa or whole blood was pipetted on top of the defect ( Table 1 ). Three animals were treated with rhFVIIa and 7 animals were treated with TF with alternating left and right defects receiving chitosan-blood implant ( Table 1 ). The arthrotomy site was closed in 3 separate layers with absorbable suture material and skin staples. All animals received postoperative nonsteroidal anti-inflammatory pain medication (1.1 mg/kg flunixin IV, 100 µg transdermal fentanyl patch) for 3 days, followed by flunixin as required. Sheep were euthanized by IV injection of pentobarbital. During necropsy, all joints were humidified with lactated Ringer’s solution. Defect sites were photographed with a digital camera and harvested with a band saw. In a blinded fashion, repair tissue macroscopic appearance was evaluated for the presence of osteophytes (0 = none, 2 = severe) and with the International Cartilage Repair Society (ICRS) scoring system that assesses Degree of defect repair (4 = level with surrounding cartilage, 3 = 75% repair depth, 2 = 50% repair depth, 2 = 25% repair depth, 0 = 0% repair depth; Integration with the border (4 = 100%, 3 = <1-mm fissure, 2 = 75%, 1 = 50%, 0 = <50%), and macroscopic appearance (4 = intact smooth surface, 3 = slight fibrillation, 2=small scattered fissures or cracks, 1 = severe small or few large fissures, 0 = severely abnormal or lesions below bone plate), to provide an Overall repair assessment (12 ICRS grade I normal, 8-11 ICRS grade II near normal, 4-7 ICRS grade III abnormal, 0-3, ICRS grade IV severely abnormal).26,27
Study Design.
rhFVIIa = recombinant human factor VIIa; RITC = rhodamine isothiocyanate; TF = tissue factor.
Acute defect characterization (
Biochemical Assays
Full-thickness repair cartilage samples from the distal defect and control cartilage from outside the defect of the same joint sample was collected with a 3 mm biopsy punch and scalpel, snap-frozen and stored at −80°C. Sample wet mass varied from 1 to 20 mg. Papain digests were generated and assayed by dimethyl methylene blue (DMMB) assay for glycosaminoglycan (GAG) content using a standard curve of shark cartilage chondroitin sulfate sodium salt, as well as by hydroxyproline assay with L-hydroxyproline standard. 28 Collagen content was estimated assuming a conversion factor of 13.15% w/w hydroxyproline content.
Biomechanical Tests
Osteochondral cylinders were cored with 11G Jamshidi biopsy needles from the distal defect and from the lateral femoral condyle from the same joint and placed in humidified chambers. Thickness measures were made from calibrated images of the biopsies taken with a dissection microscope. A mechanical tester (Mach-1 v500c, Biomomentum, Laval, Quebec, Canada) was used to perform unconfined compression tests (directly on articular surface of the osteochondral core) with 10% precompression offset, and five 2% stress-relation ramps. Em (MPa), Ef (MPa) and permeability (k, 10−12 m4/N·s) were extracted from the analysis of the fifth 2% stress relaxation ramp using a fibril-reinforced poroeleastic model. 29
Micro–Computed Tomography Analyses
Excised medial femoral condyles were fixed in a 4% (w/v) paraformaldehyde (P-6148, Sigma, Oakville, Ontario, Canada) 0.1 M sodium cacodylate (C-0250, Sigma, Oakville, Ontario, Canada) solution, pH 7.3 at room temperature for 24 hours, chilled to 4°C, and scanned using a micro–computed tomography (µCT) scanner (eXplore Locus, GE Healthcare Pre-Clinical Imaging, London, Ontario, Canada) at a resolution of 21 μm (defects), or 45 µm (intact condyles from age-matched normal female sheep from another study). Depthwise data analyses coordinated the different resolution. All scans were calibrated with an SB3 phantom (1.82 g/cm3, Gammex, Middleton, WI, USA) and reconstructed using GE Medical Systems eXplore Reconstruction Utility. A standardized volume of interest (VOI) encompassing the entire subchondral bone plate within the defect repair area was generated using GE Medical Systems eXplore MicroView version 2.2ABA, to measure bone mineral density (BMD), tissue mineral density (TMD), and bone volume fraction (BVF). To capture data from the subchondral bone plate in this species, the VOI was 9 mm wide × 5 mm long × 0.75 mm deep 30 with an adapted surface that followed the convex curvature of the condyle. 31 Subchondral bone cysts/residual holes in the MFC were defined as bone voids at least twice the volume of surrounding trabecular spaces, and the incidence scored by visual scanning through each 3-dimensional (3D) μCT image. Volume of subchondral bone overgrowth into the cartilage repair tissue area was captured by an adapted surface VOI above the projected tidemark. To investigate the gradient of bone morphometry parameters between the articular surface through the subchondral bone plate to trabecular bone, a depthwise analysis of bone volume fraction (BVF) 32 was performed with two 3 mm × 3 mm × 8 mm deep VOIs. The VOIs were placed in the central defect area covering the 2 microfracture and mid-defect Jamshidi hole with the inner border of the VOIs along the centerline of the defect, or a corresponding area in n = 4 intact condyles from age-matched normal female sheep. The VOIs began 1 mm above the subchondral bone plate and extended into the subchondral bone. The bone threshold was calculated using Ng’s method 33 and the BVF was calculated for each 45 µm (normal condyles) or 21 µm (defects) thick slice oriented parallel to the joint surface. BVF data within each slice were averaged and presented as a single value with respect to distance perpendicular to the articular surface. Data points were interpolated to generate a profile of BVF by depth for each VOI. Jamshidi hole depth in acute defects was determined by the bone length of the recovered Jamshidi biopsies using calibrated digital dissection microscope images.
Histological Processing, Staining, and Immunohistochemistry
Paraformaldehyde-fixed samples were decalcified in 0.5N HCl/0.1% glutaraldehyde at 4°C, cut transversely into 3 blocks, embedded in paraffin, and 6 µm thick serial sections collected through 2 distinct levels in the defect, at the proximal drill hole, and middle Jamshidi hole. As recommended for histological endpoints in cartilage repair studies, 34 a systematic preplanned staining order was established prior to sectioning. Two distinct sections per level were stained with Safranin O/fast green/iron hematoxylin, hematoxylin and eosin, or immunostained for collagen type I and collagen type II as previously described, 5 using 1 mg/mL pronase and 25 mg/mL hyaluronidase (Sigma-Aldrich, Oakville, Ontario, Canada) for antigen retrieval, 10 μg/mL collagen type I monoclonal antibody I-8H5 (MP Biomedicals, Solon, OH, USA) or 1:10 diluted hybridoma supernatant of collagen type II monoclonal antibody II-II6B3 (Developmental Studies Hybridoma Bank [DSHB], Iowa City, IA, USA), 12 µg/mL biotinylated goat anti-mouse, avidin-AP, and red substrate detection with levamisole to block endogenous phosphatases (Vector Laboratories Inc., Burlington, Ontario, Canada) and light iron hematoxylin counterstaining for collagen type II-stained sections. Stained sections were digitally scanned at 40× magnification using a Nanozoomer RS (Hamamatsu, Olympus) and calibration pinwheel, for histological scoring.
ICRS-II Histological Scoring
Histological scoring was carried out by 2 blinded and trained readers (AB and MH) using ICRS-II criteria, 35 including: (1) tissue morphology, (2) matrix staining, (3) cell morphology, (4) chondrocyte clustering, (5) surface architecture, (6) basal integration, (7) formation of a tidemark, (8) subchondral bone abnormalities/marrow fibrosis, (9) inflammation, (10) abnormal calcification/ossification, (11) vascularization within the repair tissue, (12) surface/superficial assessment, (13) mid/deep zone assessment, and (14) overall assessment, and 2 additional parameters including repair tissue integration and adjacent cartilage health, with left and right edges of each repaired defect scored separately and then averaged. Each parameter was scored by visual analog scale (VAS) in which a hash mark is placed on a 100 mm line, and the distance measured with a ruler to produce a value between 0 (poorest repair) and 100 (optimal repair). Since the defect width was large (10 mm), scoring was systematically carried out left and right halves of the defect, and then the scores for each defect half and each defect level were averaged to create one average score per category for each defect. Scores between readers were averaged, except when there was a difference between reader scores of 30% or more, in which case the blinded section was reevaluated by both readers to reach a consensus score.
Inverted Epifluorescence Microscopy
Chitosan implant residency after 1 day was assessed by inverted epifluorescence microscopy at 1.25x magnification to visualize RITC-chitosan tracer (only present in day 1 implants) macroscopically, while unstained and unmounted cryosections were viewed by epifluorescence combined with low bright-field to add bone structure to the fluorescent image.
Safety Outcome Measures
Lameness was evaluated after surgery on a scale of 0 (no lameness) to 5 (severe lameness). Sheep were monitored for incisional infection and general health throughout the study.
Statistical Analyses
The General Linear Model (GLM, Statistica, V6.1, Statsoft, Tulsa, OK, USA) was used to evaluate significant effects due to clot factor, chitosan, and hole type, with Fisher’s least squares difference (LSD) post hoc analysis to test for univariate effects (n = 10). Hole type (drill, section 1 vs. Jamshidi, section 2, n = 20 control and treated holes) was tested as a single categorical predictor of histological scores. Differences in the average subchondral bone properties (BMD, BVF, and TMD) used defect as the statistical unit, and condition as a predictor with 3 different conditions: treated 6-month defects (n = 8), control 6-month defects (n = 9), and intact normal age-matched female condyles (n = 4). Student t test was used to analyze differences in biochemical content and biomechanical parameters between repair tissue and cartilage tissue outside the defect.
Results
Macroscopic Appearance and Implant Delivery and Retention
Presolidified implants were easily inserted into all 3 bone channels ( Fig. 1A and B ) by press fitting and apparent capillary action (unpublished observations). µCT assessments of the day 1 defects revealed some variability in initial defects despite care taken to debride only the calcified cartilage layer with a curette, and the use of silastic sheaths to control Jamshidi needle penetration into the bone. Isolated patches completely debrided of the ~750 µm bone plate were observed (arrows, Fig. 1C ), and Jamshidi holes ranged from 2.5 to 10 mm deep (4.6 ± 1.2 mm, Fig. 1C ). µCT 2-dimensional images showed that Jamshidi holes and drill holes communicated freely with adjacent trabecular bone marrow compared with shallow microfracture holes with low marrow access ( Fig. 1C ). All day 1 condyles showed extensive subchondral hematoma ( Fig. 1D and E ). Marrow stimulation hole bleeding at surgery and subsequent subchondral hematoma was previously seen in sheep models, at 1 day after generating 6 Jamshidi needle perforations in intact sheep medial condyles 9 ( Fig. 1G-I ) and at 4 hours postmicrofracture of full-thickness cartilage lesions 11 ( Fig. 1J - L ). At 1 day postoperative, fluorescently labeled presolidified chitosan/blood implant was resident and localized to the top of the bone hole ( Fig. 1E and F ). Fluorescent chitosan was also detected along the bone bed of the defect, due to liquid implant pipetted over the defect (data not shown).
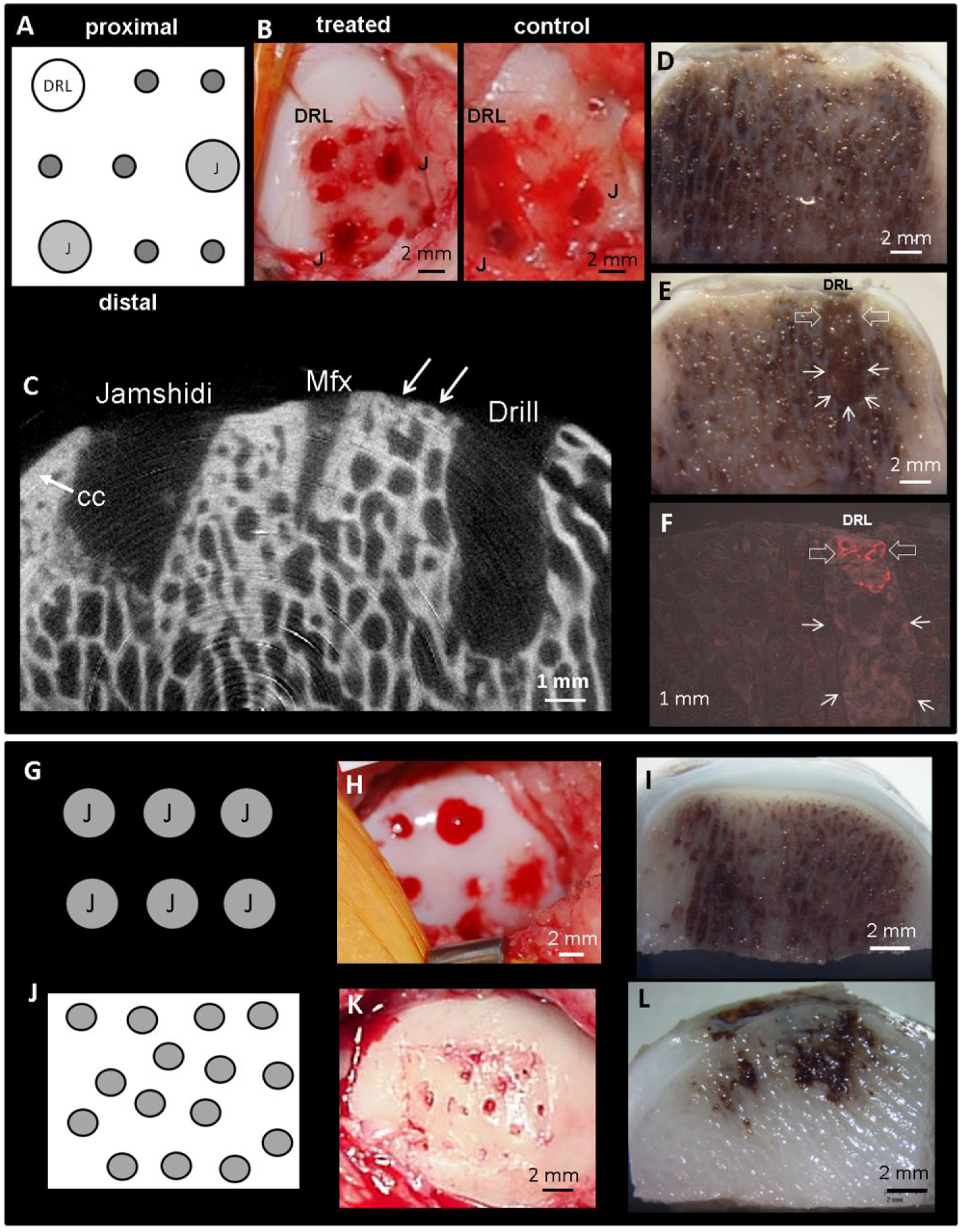
Subchondral bruising at day 1 along the condyle periphery was followed by large osteophyte formation at 6 months postoperative in all medial condyles (white arrows, Fig. 2A and B ). Some osteophytes encroached on the repair site. More specifically, in 9 condyles (control and treated), osteophyte tissue had developed up to the defect edge, and in 3 treated defects osteophyte tissue appeared to encroach into the defect. After 6 months of repair, most of the Jamshidi and drill holes were resurfaced with a white and firm repair tissue; however, resurfacing was sometimes inhomogeneous and incomplete, especially over microfractured areas ( Fig. 2C and D ). Sporadic osteonecrosis was observed around the central Jamshidi hole in 1 sheep knee treated with TF-chitosan-blood implant, and bilaterally around the Jamshidi hole in another sheep treated with TF-blood clot implant or TF-chitosan-blood implant ( Fig. 2E ). The average ICRS macroscopic score was 4 ± 2 for both treated and control defects, where 12 indicates normal cartilage and 4 to 7 indicates abnormal repair.
Macroscopic appearance of a defect after 1 day (
Subchondral Bone Responses
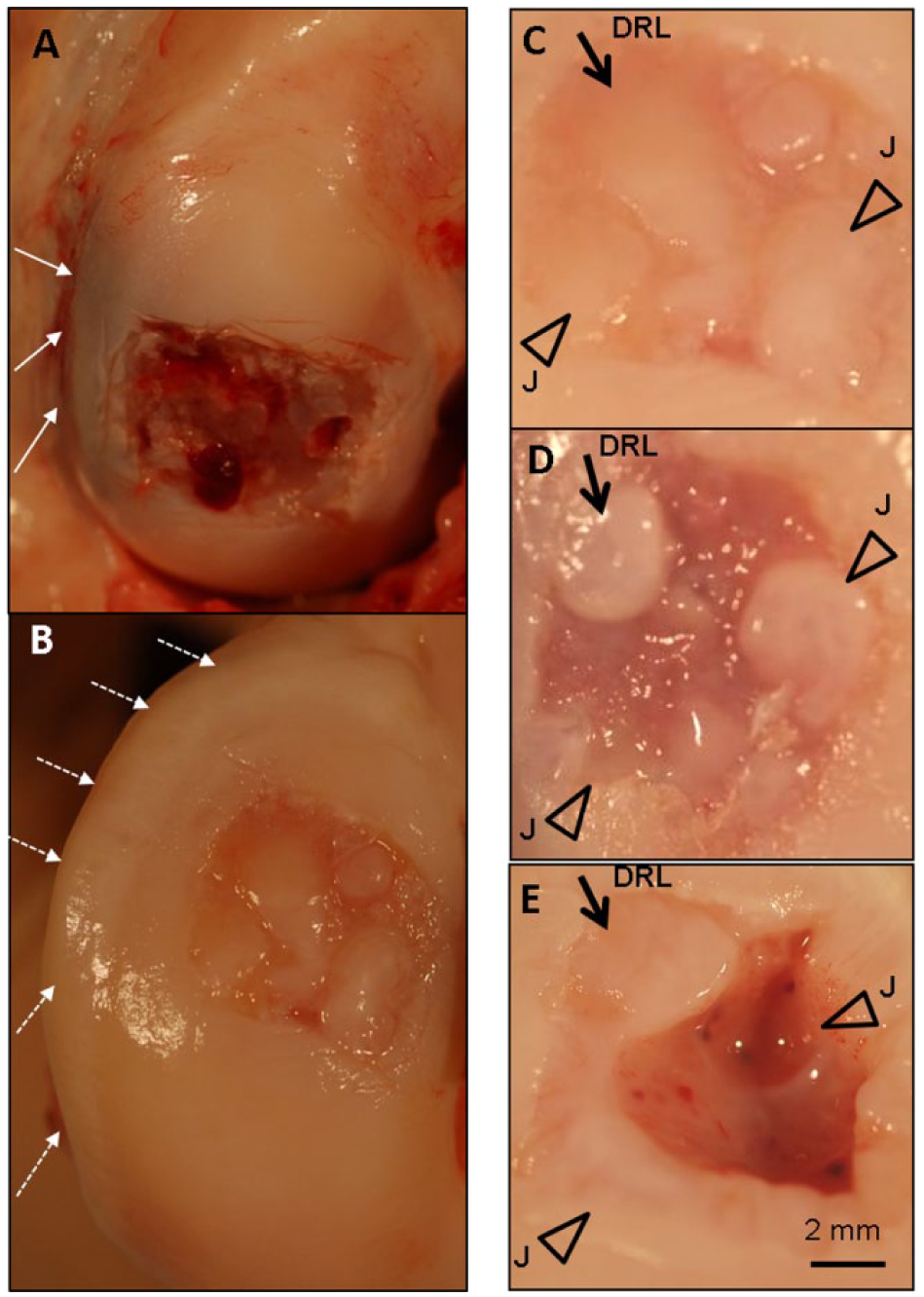
Specific changes in the subchondral bone architecture were detected between 1 day and 6 months postoperative. In a 3D VOI encompassing the entire defect subchondral bone plate ( Fig. 3A1 and A2 ), acute defects showed 50% lower BMD, a 60% lower BVF, and the same TMD when compared with intact condyles ( Fig. 3A3 ). TMD describes mineral density within bone tissue. After 6 months of repair, bone plate BVF, BMD, and TMD were all significantly lower than intact condyles (P ≤ 0.01, Fig. 3A3 ). The relative loss of TMD in the bone plate of 6 month defects compared with intact condyles and day 1 defects suggested that subchondral bone remodeling and the formation of a hypomineralized repair bone tissue had taken place.
Micro–computed tomography (μCT) measures in sheep condyles. (
Intact condyles and day 1 defects had no subchondral cysts; however, all the condyles in the 6-month groups had at least 1 cyst, and on average 2 to 3 per condyle ( Fig. 3B ). Defects treated with chitosan had an average lower number of cysts than their corresponding controls, but the difference was not significant ( Fig. 3B ). Bone overgrowth into the cartilage defect area was seen in 3 MFCs (0.3-9.1 mm3), of different treatment groups.
A depthwise µCT analysis was carried out in 2, 8-mm-deep 3D volumes of interest (VOI) in the central area of the defect treated with chitosan/blood versus blood implant ( Fig. 3C1 and C2 ). Three defects with sporadic osteonecrosis were treated separately as outliers. Normal condyles and day 1 defects showed a characteristic peak and linearly declining BVF as the measure passed through the calcified cartilage and bone plate to the cancellous bone below ( Fig. 3C3 ). Note that the peak BVF in this depthwise analysis reflects the maximally dense calcified tissue at the cartilage-bone interface. At 6 months postoperative, peak BVF was ~40% lower in both treated and control defects compared with intact and day 1 condyles (P = 0.015 treated vs. intact, P = 0.00013 control vs. intact, Fig. 3C3 ). The averaged depthwise repair bone measures showed a smooth decline in BVF; however, individual repair bone depthwise profiles revealed an irregular BVF mainly in TF-treated defects, where dips in the BVF profile revealed sporadic surface resorption, subchondral cysts, and a generally irregular trabecular bone structure in the deeper zones ( Fig. 4 ). The 3 defects with osteonecrosis (6-month outliers) showed subchondral bone loss up to 6 mm below the defect surface ( Figs. 3C and 4 ). To summarize, a significant amount of mineralized bone plate tissue was removed by debridement, drill and Jamshidi holes. After 6 months of repair subchondral bone plate showed evidence of remodeling and partial repair by a hypomineralized bone tissue.
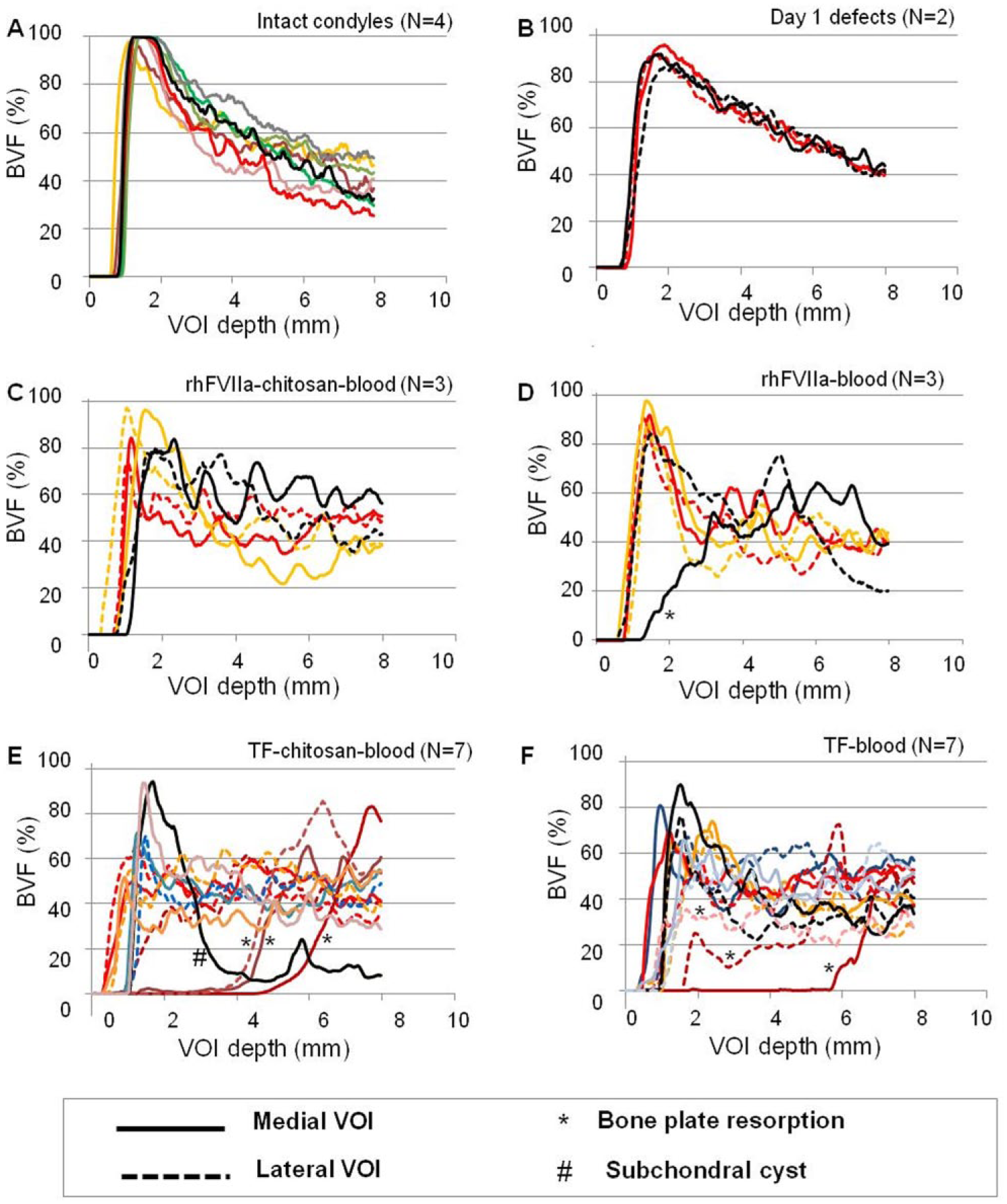
Individual depthwise micro–computed tomography (μCT) profiles for intact age-matched female condyles using the 3 mm deep volumes of interest (VOIs) presented in
Figure 3C
(1-2) for intact condyles (
Histological Repair Assessments
In all the 16 histological parameters scored, no significant differences were detected due to chitosan-blood implant versus matching control blood implants ( Fig. 5 ). By comparison, the 3 outlier defects with resorbed bone had significantly worse surface architecture (parameter V, P = 0.029), subchondral bone abnormalities (parameter VIII, P = 0.0095), inflammation (parameter IX, P < 0.0001, and surface/superficial assessment (parameter XI, P < 0.0001, Fig. 5 ). The average ICRS-II overall scores were 43 ± 21 for implant-treated, 47 ± 29 for blood clot treated, and 24 ± 35 for outliers with osteonecrosis/resorbed bone plate. When the data were stratified according to clot factor and implant, ICRS-II Overall assessment scores were 45% ± 26% for rhFVIIa chitosan/blood, 61% ± 34% for rhFVIIa-blood control, 36% ± 27% for TF-chitosan/blood, and 37% ± 27% for TF-blood control. When all 20 defects were analyzed together, drill holes showed improved repair features compared to Jamshidi holes, including better tissue morphology (P = 0.044), surface architecture (P = 0.019) and overall histological assessment (P = 0.041, 50% ± 26% vs. 34% ± 28%) scores. All defects irrespective of treatment developed repair tissue with minimal chondrocyte clustering, good basal integration, and little abnormal calcification/ossification. The average lateral integration scores ranged from 48% to 56%, where 100% is full integration, and the adjacent cartilage health ranged from 36% to 47% ( Fig. 5 ), with no significant differences due to treatment or clotting factor.
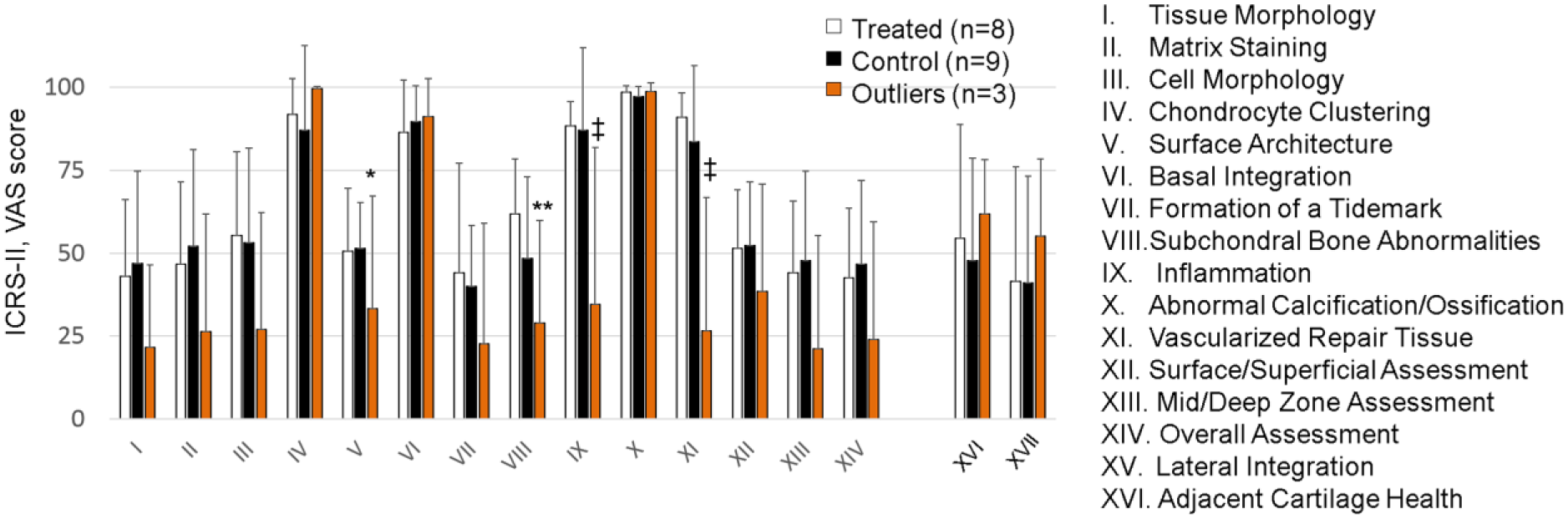
Histological visual analog scale (VAS) scoring results of groups after 6-month repair for 14 different International Cartilage Repair Society–II (ICRS-II) scoring parameters as well as lateral integration (XV) and adjacent cartilage health (XVI). Data show the mean ± standard deviation. *P = 0.029, **P = 0.0095, ‡P < 0.0001 versus treated and control.
Nearly all 6-month condylar cartilage repair tissue was uniformly stained for collagen type II with negligible collagen type I, except for areas around resorbed subchondral bone that were covered with fibrous tissue devoid of collagen type II ( Fig. 6 ). Cartilage repair tissues with full-thickness collagen type II also had uniform Safranin O staining for GAG, partly regenerating tidemark, and chondrocyte cell morphology ( Fig. 6A4-5 and C4-5 ). Cartilage repair tissues around areas of bone resorption were depleted of GAG ( Fig. 6 B1-4 and D1-4 ). In the best-case repair, a uniform resurfacing was seen with hyaline-like cartilage over well-structured bone ( Fig. 6A ).
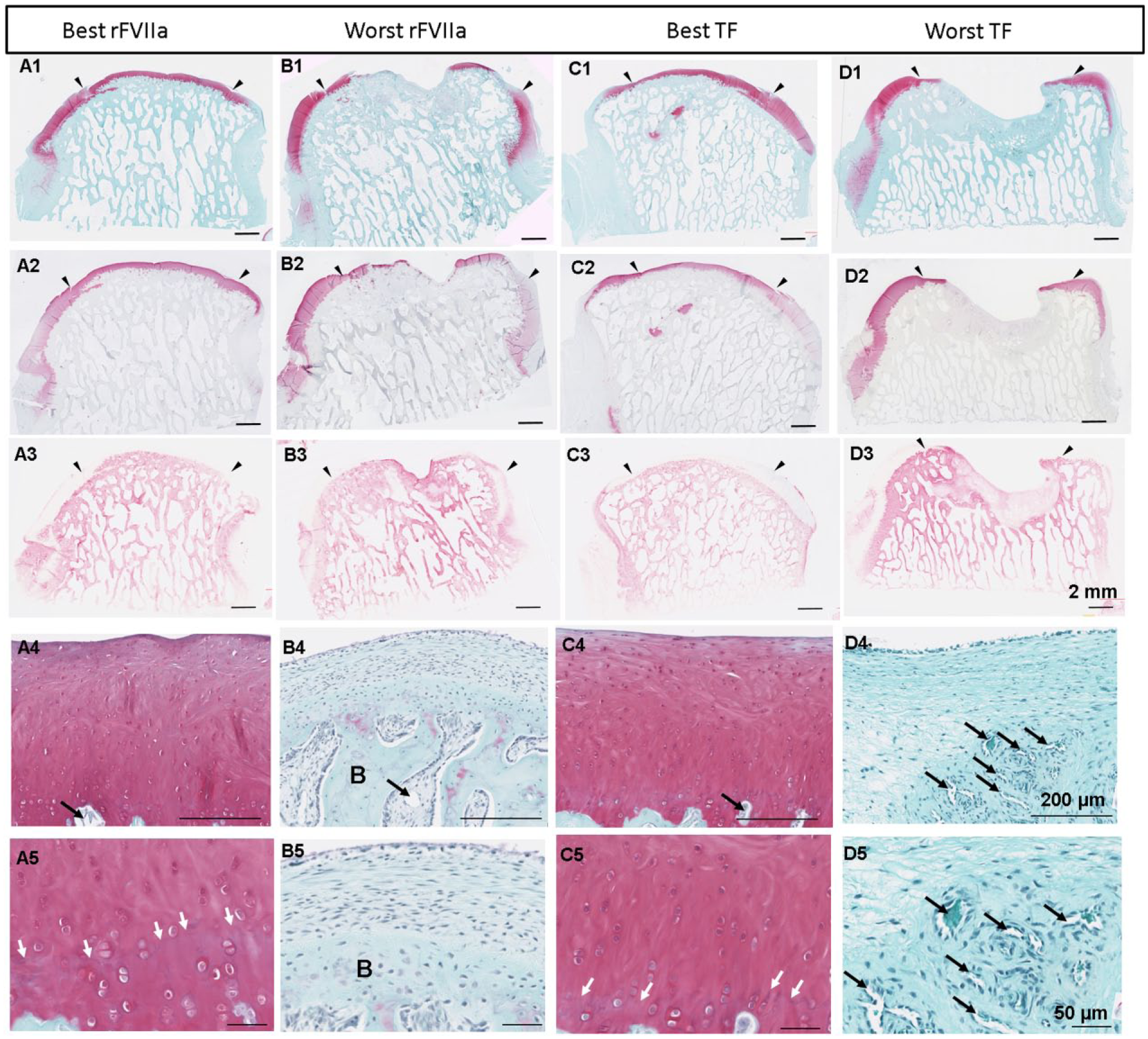
Repair cartilage/bone tissue histological appearance after 6 months. Serial sections were stained for Safranin O/fast green (
Biomechanical Properties and Biochemical Composition
Individual osteochondral repair biopsies with more intense Safranin O staining showed higher biomechanical compressive strength in unconfined compression ( Fig. 7A ). Repair tissues were on average thinner with lower GAG levels and lower compressive strength than intact cartilage outside the defect in the lateral femoral condyle. Cartilage repair tissues were slightly fibrotic with higher average collagen levels (18.9% vs. 14.7% mg/mg wet weight, Fig. 7B ) and 2-fold lower GAG (2.4% vs. 4.3% intact, P < 0.005, Fig. 7C ). Given the lack of effect of chitosan/blood implant on histological or biochemical repair tissue features, biomechanical results were grouped according to repair versus intact cartilage. The average MFC repair tissue thickness was 460 µm versus 722 µm for intact cartilage ( Fig. 7D ). Repair osteochondral tissue had a lower equilibrium modulus (Em 0.17 MPa vs. 0.28 MPa, Fig. 7D ), ~2-fold lower fibril modulus (Ef, 2.6 MPa vs 4.9 MPa, Fig. 7E ), and ~4.5-fold higher permeability than intact tissue ( Fig. 7E ).
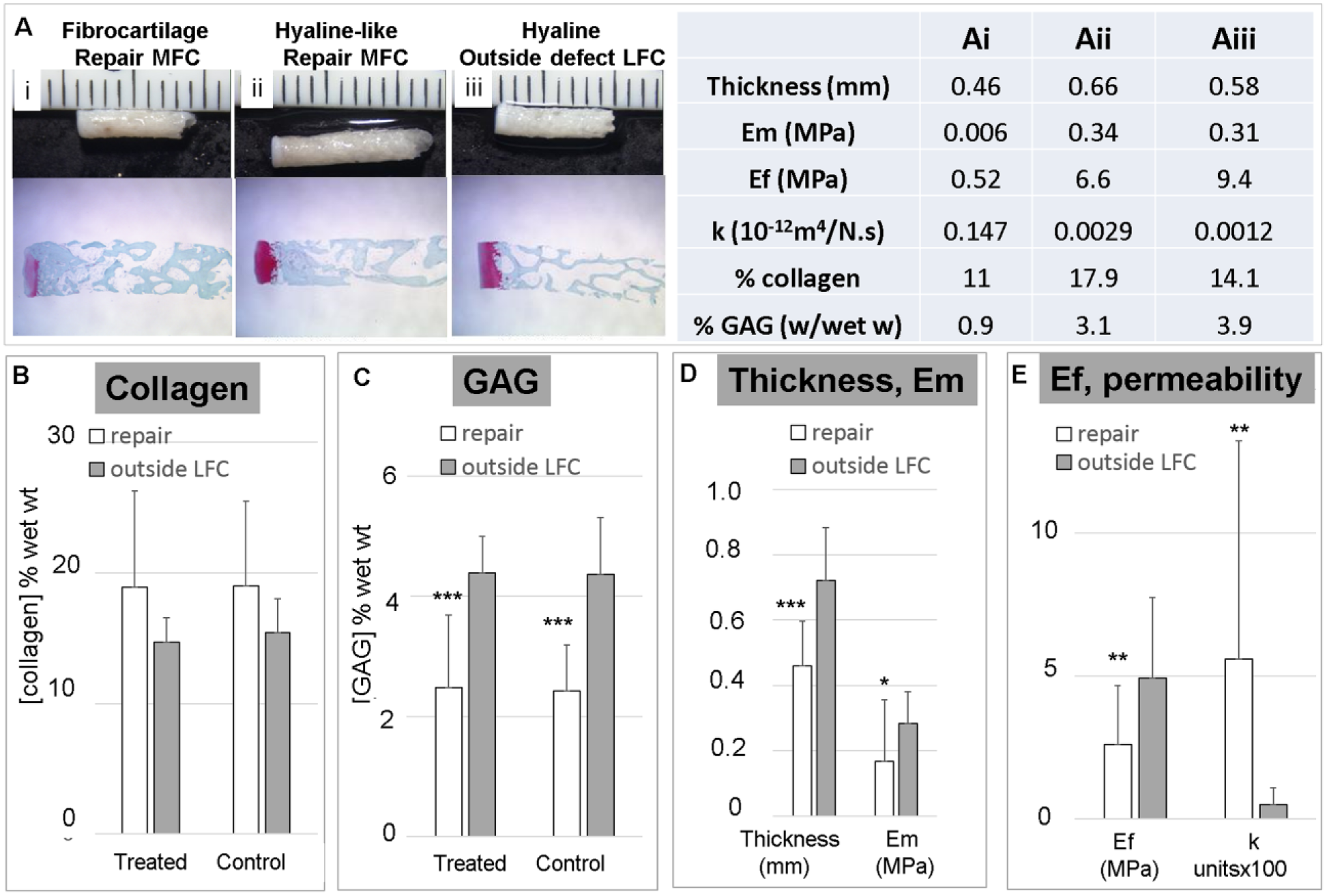
Biomechanical and biochemical analyses of cartilage repair tissues in the distal defect after 6 months. (
Safety Outcome Measures
Three knees developed superficial incisional infections that were treated and resolved with hot compresses and saline irrigation with no apparent adverse effects on bone or cartilage repair either macroscopically or histologically. Two of the biochemical samples from these 3 defects showed lower GAG levels (9 and 14 µg/mg). No sheep showed signs of knee infection or were lame at 6 months postoperatively.
Discussion
The SMS technique relies on a combination of surgical technique and implant to create access channels and a repair environment that draws mesenchymal stem cells from the bone marrow toward the cartilage defect. Chitosan/blood implants were well-retained in 2 mm diameter holes formed in the medial femoral condyle of acute defects, despite their vertical orientation at surgery, and despite the effects of gravity and motion during and after surgery. Therefore, the process of creating and retaining a presolidified chitosan-blood implant within the subchondral bone of a debrided cartilage defect is feasible, and could be potentially adapted to arthroscopic surgery. Subchondral bleeding may have pushed the implant toward the top of the hole of sheep bone channel. In a previous rabbit study, chitosan-blood implant was also resident at the top of the osteochondral defect. 7 A similar level of subchondral bleeding or bruising was observed in a previous study at 1 day after generating 6 Jamshidi needle perforations of intact sheep medial condyles. 9 By contrast only mild subchondral bruising was previously observed at 4 hours postmicrofracture. 11 These data suggest that the extensive hematoma in this study arose from Jamshidi needle perforation, although additional studies are needed to determine the extent of subchondral bruising induced by microdrilling or microfracture alone at 1 day postoperative.
In this study, chitosan-treated condyles showed fewer bone cysts than contralateral control defects—an effect previously observed in microfracture defects treated with in situ solidifying chitosan–glycerol phosphate/blood implant. 11 The effect could be explained by chitosan’s angiogenic properties,7,36 which could help restore the blood vessels ruptured by marrow stimulation. By contrast, chitosan implant had no effect on cartilage repair tissue histological features compared to contralateral controls. This may be due to the fact that chitosan implants did not completely fill the marrow stimulation holes, or that the implant was cleared too rapidly and was no longer able to influence bone-induced chondroinduction that is starting in sheep subchondral defects at 3 months postoperative. 9 Future studies will determine whether chitosan implants designed with a higher dose or slower clearance rate can elicit a better cartilage repair response. One important conclusion from this study is that subchondral implants previously optimized to influence repair in rabbit defects that heal completely after 8 to 10 weeks7,8 require further optimization to translate to an effective treatment in a more slowly-repairing large animal cartilage repair model.
In this study, larger 2 mm diameter marrow holes tended to reproducibly elicit a collagen type II+ and GAG-rich repair tissue while a less consistent resurfacing was obtained over microfracture holes, which compact the bone 20 and have less initial access to cell-rich marrow cavities ( Fig. 1C ). Drill holes produced a significantly higher overall cartilage histological quality than the central Jamshidi holes, suggesting that future developments in this area may benefit from drilling instead of biopsy needle channels.
Subchondral bone cleanly removed by debridement ( Fig. 1 ), 37 and by Jamshidi needle biopsy, drilling ( Fig. 3 ), 16 and trans-osseous drilling using a Kirchner wire, 38 is slow to restore mineral density. Ideally, it would be possible to debride just the calcified cartilage layer without damaging the bone plate, but Mika et al 39 showed that conservative debridement with a curette leaves a layer of calcified cartilage, which is known to block repair from marrow-stimulation defects.11,40 Current data show that despite great care, the bone plate (which can be quite thin in normal condyles 32 ) is frequently breached using a curette or a burr.41,42 Orth et al 37 recently showed that debrided sheep condyle defects, with or without microfracture, have significantly reduced bone plate BMD and BV/TF at 6 months compared to adjacent bone in a sheep model. Our results support and extend these data to show that surgical disruption of the subchondral bone plate integrity leads to sustained resorption and delayed new bone formation. This is manifested by a relatively stable loss of BMD and BVF and lower TMD at 6 months compared with day 1 and intact condyles. Lower BVF and BMD levels in the incompletely repaired bone plate may lead to reduced mechanical properties.43-45 The comprehensive analyses in this study allow new insights leading us to propose that a softer cartilage-repair bone interface could have a potential protective effect on maturing cartilage repair tissues with 2-fold lower GAG, slightly higher collagen content, and 2-fold lower fibril modulus compared with intact tissues.
In this study, the average repair tissue ICRS-II histological overall assessment scores of ~45% (excluding 3 outlier defects) are consistent with ~42% or ~46% overall scores previously reported for repair biopsies from human clinical trials at 12 to 13 months postmicrofracture.4,6 In 3 defects treated with rhFVIIa-blood implant, the average 61% ICRS-II overall assessment score is higher than the ~55% overall assessment score previously reported in human defects treated by characterized chondrocyte implantation 4 or microfracture and BST-CarGel. 6 The improved overall assessment score for rhFVIIa-blood clot control defects was accompanied by improved subchondral bone repair compared with TF-blood controls ( Fig. 4 ). One limitation of our study was that we did not have a control group where the experimental sites were allowed to bleed with no implant. We also tested only one dose level of rhFVIIa, although dose-dependent effects are seen after intravenous infusion,46-48 and on in vitro cell proliferation and endothelial migration.22,24 To demonstrate the potential for rhFVIIa in cartilage repair, additional studies are needed to compare surgery-only with rhFVIIa delivered locally at different dosages, and perhaps with a species-specific recombinant factor. Indeed, very little is known about the role of clotting factors and subchondral bleeding in osteochondral repair. Deeper drilling creates more subchondral bleeding 20 and elicits more cartilage regenerate in rabbits, but mainly in the subchondral bone area. 15 Subchondral bone bruising/hemorrhage was quite extensive in this model, compared with microfracture-only sheep defects, 11 and despite delivery of TF-implant. Chronic intra-articular bleeding is known to lead to arthrosis. 49 More experiments are needed to determine whether osteophytes seen at 6 months postoperative may develop in response to subchondral bleeding or to clot factor treatment.
Sporadic osteonecrosis around 3 TF-treated Jamshidi defects could potentially be explained by TF diffusing into the local vasculature and inducing local intravascular thrombosis and sporadic bone necrosis.50-52 However, no such adverse events were reported in the use of TF for sinus bone grafting in human patients. 53 Certain states of inflammation can induce TF expression inside of blood vessels. 54 Bleeding induced through deeper debridement and marrow stimulation does not always induce spontaneous bone regeneration, 55 and when combined with certain implants (i.e., TF-blood (±chitosan), platelet-rich plasma–collagen hydroxyapatite, collagen type II, matrix-associated autologous chondrocyte implantation/collagen types I and III), further bone resorption can ensue for reasons that remain unclear.56-58 More research is needed to understand why some implants elicit bone resorption instead of anabolic bone repair and how to coordinate implant-host interactions to elicit bone and hyaline cartilage repair during the more protracted bone healing sequence in large animals and humans.
Footnotes
Animal Welfare
The present study was carried out in compliance with the Canadian Council for Animal Care guidelines which follow international, national, and institutional guidelines for humane animal treatment and comply with relevant legislation.
Authors’ Note
Institutions where the study was performed: Ecole Polytechnique, Montreal, QC, Canada (principle institution); University of Guelph (animal experimentation); Biomomentum (mechanical testing).
Acknowledgments and Funding
We thank Viorica Lascau, Jun Sun, Genevieve Picard, Gaoping Chen, Adam Harris, T. Duncan Smith, Vincent Darras, and Michelle Bridges for valuable technical contributions. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Science and Engineering Research Council (NSERC Strategic, STPGP 365025), Canadian Institutes of Health Research (CIHR, BME 303615), Fonds de la Recherche sur la Santé du Québec (FRSQ) Groupe de Recherche sur les Sciences et Technologies (GRSTB). Salary support: ON Graduate Research Fund (ADB), FRQ-S National Researcher Career Fellowship (CDH).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This work was partly funded by an industry-partnered government grant (Natural Sciences and Research Engineering Council of Canada, and Piramal Healthcare) awarded to CDH and MBH. CDH is a member of the Board of Directors and a shareholder of ORTHO RTi. MBH is a shareholder of ORTHO RTi, and is a consultant for High Meadow Consulting Inc., Arthritis Innovation Corp, Merial, Elanco. EQ is an employee of Biomomentum, Inc.
Ethical Approval
Ethical approval for this study was obtained from the University of Guelph (Protocol #3113) that was recognized by the Direction of Research at Ecole Polytechnique (Certificates # ANI-0405/02 and BIO-0809-02).
