Abstract
Background
Many in vitro damage models investigate progression of cartilage degradation after a supraphysiologic, compressive impact at the surface and do not model shear-induced damage processes. Models also neglect the response to uninterrupted tribological stress after damage. It was hypothesized that shear-induced removal of the superficial zone would accelerate matrix degradation when damage was followed by continued load and articulation.
Methods
Bovine cartilage underwent a 5-day test. Shear-damaged samples experienced 2 days of damage induction with articulation against polyethylene and then continued articulation against cartilage (CoC), articulation against metal (MoC), or rest as free-swelling control (FSC). Surface-intact samples were randomized to CoC, MoC, or FSC for the entire 5-day test. Samples were evaluated for chondrocyte viability, GAG (glycosaminoglycan) release (matrix wear surrogate), and histological integrity.
Results
Shear induction wore away the superficial zone. Damaged samples began continued articulation with collagen matrix disruption and increased cell death compared to intact samples. In spite of the damaged surface, these samples did not exhibit higher GAG release than intact samples articulating against the same counterface (P = 0.782), contrary to our hypothesis. Differences in GAG release were found to be due to tribological testing against metal (P = 0.003).
Conclusion
Shear-induced damage lowers chondrocyte viability and affects extracellular matrix integrity. Continued motion of either cartilage or metal against damaged surfaces did not increase wear compared with intact samples. We conjecture that favorable reorganization of the surface collagen fibers during articulation protected the underlying matrix. This finding suggests a potential window for clinical interventions to slow matrix degradation after traumatic incidents.
Introduction
A common risk factor for posttraumatic osteoarthritis (PTOA) is joint trauma. While there are multiple clinical events leading to PTOA, including cartilage damage, 1 compressive injuries, 2 anterior cruciate ligament injuries, 3 and bone fractures,1,4 the underlying mechanisms regulating the response to the acute injury by the whole joint are yet to be fully understood.1,5 Given that cartilage damage can increase the risk of PTOA 20-fold, 1 understanding the progression from initial cartilage injury to PTOA is necessary in determining critical time points for intervention in order to delay or prevent arthritic disease. 6
To that end, effective in vitro models should provide controlled mechanical conditions to evaluate the temporal relationship between injury and response. 7 When developing studies to understand this relationship, models should account for the idea that 2 different insult types may lead to PTOA: (1) single, acute stress events and (2) chronic stress abnormalities accumulated over time from small mechanical insults. 5 For acute events, most cartilage injury models apply a single supraphysiologic impact to create the initial insult.8,9 For chronic stress, few in vitro models exist, usually through high loading cycles of dynamic compression.10-12 Modeling shear damage to the surface, which could be classified as a form of chronic stress, has not been well evaluated in cartilage explant studies. This is despite the clinical association between shear damage and the development of osteoarthritis (OA). Physical insults, such as those due to meniscal injury 13 or due to the presence of uric acid crystals from gout, 14 as well as biological diseases, such as the genetic loss of superficial zone protein, 15 have been linked. For the latter, the loss starts a cascade of effects including increased friction, increased shear stress across the surface, and ultimately increased cartilage wear and joint degradation. 16 Furthermore, shear stress has been documented with in vitro studies as a negative factor in cartilage failure as a response to injury and extracellular matrix wear.17,18
Development of a superficial zone damage model through excessive shear would highlight the role of the superficial zone in the progression from healthy cartilage to PTOA. Superficial zone integrity is important for healthy cartilage function, as it provides resistance to shear forces due to the orientation of the collagen fibers. Additionally, it serves as a protective barrier to fluid flow and loss of matrix proteins, because under mechanical loading, the collagen fibers at the surface collapse, resulting in a low permeability state.19,20 It is postulated that once the superficial zone is damaged, matrix degradation and a loss of cartilage function follow, leading to the development of PTOA. 6
Here, we created an in vitro model of shear-induced cartilage surface damage. Tribological testing was divided into 2 periods: the damage induction period which utilized the shear damage model to create a superficial zone injury; and the continued articulation period which evaluated the effect of articulation on cartilage with damaged and intact surfaces. For damage induction, the shear damage was created through articulation against polyethylene (PE), a material previously shown to damage articular cartilage,21-23 resulting in a loss of the superficial zone. For the continued articulation period, models of the natural joint with a cartilage-on-cartilage interface and of the partially reconstructed joint with a cartilage-on-metal interface both simulated the complex motion of an articular joint. The overarching hypothesis was that excessive shear damage to the superficial zone of cartilage would accelerate wear when the damage was followed by continued articulation against cartilage or against metal.
Materials and Methods
Cartilage Specimens
Cartilage samples were removed from 4 stifle (knee) joints of 6- to 8-month-old steers that were obtained from a local abattoir within 48 hours of slaughter and kept intact and refrigerated. Joints were dissected to expose the femoral trochlear groove with visual joint integrity confirmed for a lack of bruising of the cartilage surface and for the absence of blood-tinted synovial fluid. Representative samples of each animal were evaluated to observe an intact cartilage surface and minimal cell death throughout the sample depth.
Using a custom oval punch (20 mm length × 14 mm width), full-thickness cartilage discs were dissected from the trochlear groove and trimmed to size (~3 mm height). Samples were secured in porous PE wafers under semiconfined compression, which were then placed in polyether ether ketone specimen cups. To create a cartilage counterface for articulating motion against cartilage, strips (45 mm length × 8 mm width × 3 mm height) were removed from the trochlear rim with a scalpel, trimmed to exact size, and secured to a Delrin ball with size 0 silk surgical ties (Ethicon Inc., Somerville, NJ). Initial wet weights of all samples were collected. Tissue was cultured in DMEM–Ham’s F-12 (1:1) media containing 1% Mini-ITS (50 mM insulin, 2 µg/mL transferrin, 2 ng/mL selenous acid, 25 µg ascorbic acid, and bovine serum albumin/linoleic acid at 420/2.1 µg/mL) and antibiotic solution (penicillin, streptomycin, amphotericin B, and gentamicin) at 37°C, 5% CO2, 95% humidity. The preculture period was 5 days with media replenished daily.
Tribological Apparatus
The tribological test simulator was initially designed for articulation against chondrocyte-seeded scaffolds, as published in Wimmer et al. 24 From the perspective of an articulating joint, such as the knee, the motion is characterized by rolling and sliding with a gliding contact point that is smaller than the overall wear path. 24 To create this motion, a pin-on-ball concept was used where a cell-seeded scaffold (original design) or cartilage tissue (current design) acts as the pin, which is pressed onto a conforming ball. Complex shear force patterns are created on the scaffold/cartilage pin surface through both the phase-shifted rotating motion trajectories of both bodies and the cyclic compressive loading. The loading pressure is akin to contact forces seen in vivo during walking.25-27 The creation of a migrating contact point in the model further allows for improved lubrication 28 and for greater cell viability. 29
For tribological testing, strips and balls were mounted onto the joint motion simulator with their corresponding discs. The 4-station simulator sits in another incubator with the same conditions as the culture periods. Balls were lowered and loaded to an approximate contact pressure of 2 MPa (~45 N loading in the simulator). Balls rotated at a frequency of 0.5 Hz and a stroke of 30°. Explants rotated at a frequency of 0.1 Hz and a stroke of 15°. The resulting contact area was 20 mm2 with a curvilinear path of about 5.2 mm, which was similar for both the material balls and the cartilage strip. The wear area was about 40% of the surface of the cartilage. Testing occurred for 3 hours per day (5,400 cycles) over the entire 5-day testing period (27,000 total cycles). Culture medium was collected and freshly replenished after each 3-hour test and after the overnight incubation period between tests (rest period). Media was stored at −80°. Media was analyzed at individual test or rest time points. Final wet weights were collected after the last testing period.
Superficial Zone Damage Induction Period
After tissue harvest and the preculture, cartilage discs were divided into 2 groups: intact surface or shear induced-damage surface (damage). During days 6 to 7, samples randomized into the damage group articulated against PE for 3 hours per day, resulting in a loss of the superficial zone over the articulated wear path. PE was chosen as the material is unsuitable for hemi-prosthetic joint replacements as seen in animal 21 and clinical 22 studies. In addition, preliminary short-term experiments showed that cartilage articulation against PE caused removal of the superficial matrix and limited cell death in the superficial zone, suggesting that a cell response to damage is still possible. 23
During this 2-day period, intact surface samples underwent tribological testing in their assigned subgroup: unloaded free-swelling control, model of the natural joint with articulation against cartilage, or model of the partially reconstructed joint with articulation against metal. The cartilage-on-cartilage articulating model (CoC) serves as the natural joint model, replicating both the sliding and rolling motion and the articulating interface seen in vivo. The metal-on-cartilage articulating model (MoC) represents a partially reconstructed joint as the metal (cobalt chromium molybdenum alloy) is commonly used in joint hemiarthroplasty as a cartilage counterface. 30 The groups are outlined in Figure 1 .
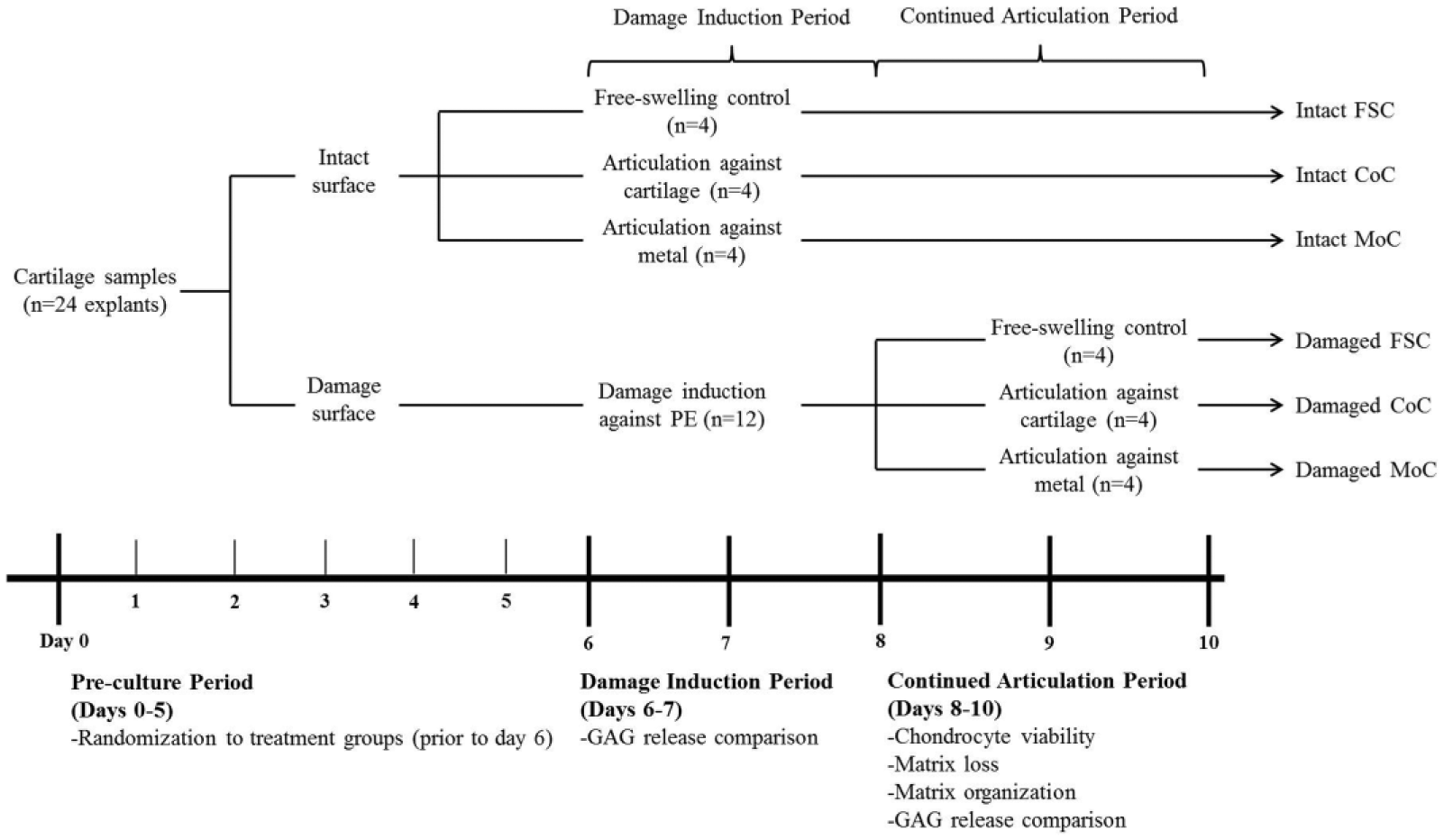
Schema describing damage induction period and continued articulation period with the resulting 6 testing groups for the study. Timeline of study design displaying the randomization into the groups for intact versus damaged surface and for continued tribological testing. Testing periods occurred for 3 hours per day. The rest period is defined as the 21 hours between testing periods. Media was collected after every test and rest period and was individually assessed for GAG release before being summed for major time points at (1) postdamage induction, (2) after continued articulation, and (3) during the rest periods.
Continued Articulation Period
For days 8 to 10, intact and damage samples underwent additional tribological testing. Intact samples remained in their respective groups. Damaged surface samples were randomized to the same subgroups: unloaded free-swelling control (FSC), articulation against cartilage (CoC), or articulation against metal (MoC). Figure 1 summarizes the timeline of events and the analysis collection points.
Media Analysis: Sulfated-Glycosaminoglycan (GAG) Release
Measuring the release of GAGs into the culture media is a proxy method of evaluating cartilage extracellular matrix wear. 31 To analyze the effect of surface damage and articulation, the dimethylmethylene blue dye reaction32,33 was used to measure GAG concentration in the collected media. Using a spectrophotomorphic plate reader, absorbances were read at 530 nm and 595 nm with the standard determined from bovine nasal septum (BCO-3015; Axxora Platform, San Diego, CA). GAG release was analyzed for 3 significant comparison points (as shown in Fig. 1 ): after the shear-induced damage period; after the continued articulation period; and after the 21-hour “rest” periods immediately following testing.
Tissue Analysis: Tissue Loss, Histology, and Viability
Tissue loss was evaluated as the differences in wet weight from the end of testing after the continued articulation period and from the initial wet weight after tissue procurement.
At the completion of tribological testing after day 10, samples were dissected through the longitudinal axis of the wear scar for histological analysis. Half of the dissected sample was fixed in formalin, processed for paraffin embedding, sliced to a thickness of 5 µm, and exposed to deparaffinization washes in xylene and decreasing concentrations of alcohol. Slides were stained with Safranin-O/fast green to evaluate the presence of matrix proteoglycans 34 and imaged with 4× and 10× objectives. Morphology was assessed using previously reported modified scoring criteria, 35 originally established by Mankin and Lippiello. 36 Samples were also stained with picrosirius red to evaluate the effect of surface damage on the collagen matrix. 37 Using a light microscope with a polarizing filter (Model BX40, Olympus, Melville, NY), birefringence of collagen within the extracellular matrix was captured with special attention to surface changes. Samples were stained at the same time and viewed with identical microscope settings for lighting.
The remaining half of the dissected sample was analyzed for cell viability. With a commercially available live/dead staining kit, samples were incubated in phosphate-buffered saline containing calcein AM and ethidium homodimer-1 for 45 minutes at 37°C (Invitrogen Molecular Probes, Eugene, OR). 34 Samples were imaged using a fluorescence-light microscope (Eclipse TE2000-S; Nikon Instruments Inc., New York, NY), charge-coupled device (CCD) camera (SPOT RT-KE Color 3-Shot, Model 7.3x; Diagnostic Instruments, Sterling Heights, MI) and 5× objective.
Statistics
Data were presented as mean ± standard error with a sample size of 4 explant discs for each group. Data were normalized to the number of contacting cartilage surfaces, that is, CoC values were divided by 2. The assumption is that the cartilage disc and strip for CoC models contribute equally to the GAG release, based on the experimental design where the volume of cartilage in contact with the media is similar between the surfaces despite the differences in shape. For all comparisons, the data were statistically blocked with respect to the animals, which decreases the variation due to animal differences. Data were confirmed to be normally distributed using normality plots with the exception of Mankin scores. Wet weight changes and GAG release data were analyzed with 2-way ANOVA for the effect of the superficial zone condition (intact, damaged) and the articulation type (FSC, CoC, MoC). Post hoc pairwise comparisons were performed with Tukey’s range test. For GAG release in day 8 through day 10 of testing, the superficial zone condition was not a statistically significant factor. Therefore, damaged and intact surface samples were combined (n = 8) and analyzed by 1-way ANOVA for the effect of articulation type (FSC, CoC, MoC). Post hoc pairwise testing was then performed using Fisher’s protected least significant difference t test. For comparison between intact and damaged surfaces, Mankin scores were analyzed with a nonparametric Mann-Whitney test. Significance was set at P ≤ 0.05. Statistical analyses were conducted with GraphPad Prism 6 (GraphPad Software, La Jolla, CA) and Design-Expert (Version 9, Stat-Ease, Inc., Minneapolis, MN).
Results
Superficial Zone Damage Induction Period
Articulation against PE caused degradation of the superficial zone layer throughout the articulation period. As a result, GAG release was significantly increased during damage induction in the first 2 days of tribological testing as compared to other groups with intact surfaces (P < 0.0001 for PE against FSC, CoC, and MoC) ( Fig. 2 ).
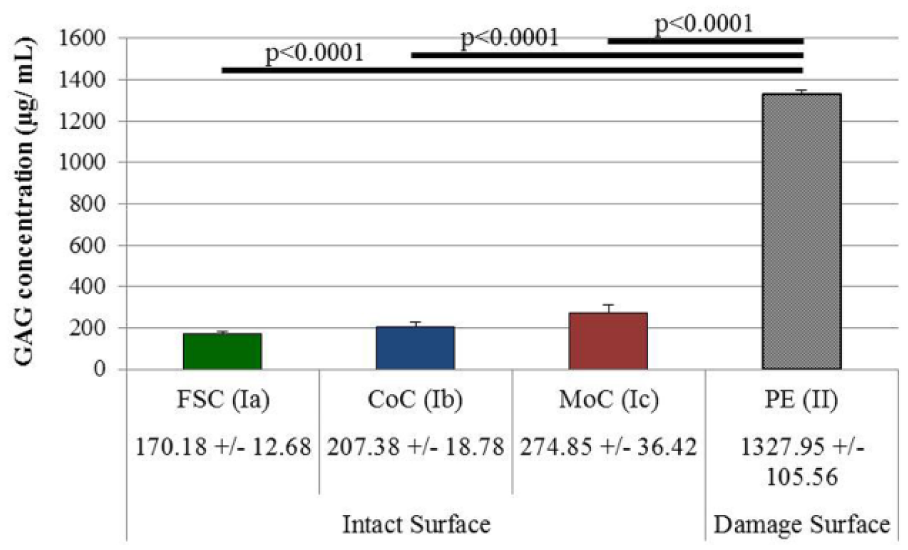
GAG release from the tribological testing period of days 6 to 7 during the damage induction. GAG release was significantly higher in the damage group (Group II) versus all intact surface samples (Ia, Ib, and Ic; P < 0.0001 for comparisons). Data presented as mean GAG release ± SEM (n = 4 for Groups Ia, Ib, and Ic; n = 12 for Group II).
Continued Articulation Period
Cartilage Wear due to Continued Articulation Period
Due to damage induction causing the loss of the superficial zone, the middle zone became the new articulating surface for the damage samples during the period of continued articulation in days 8 to 10. In this period, all intact and damage samples displayed similar trends in daily GAG release with no individual daily differences and decreasing amounts released over time ( Fig. 3a ). When analyzing the cumulative release over the 3 days, GAG release was found to be significantly influenced by the articulation type, that is, FSC, CoC, or MoC (P = 0.002) but not by the surface condition, that is, intact or damage (P = 0.762) ( Fig. 3b ). Therefore, the intact and damage surface groups were combined in the statistical analysis to increase the power to evaluate the different counterfaces for articulation. GAG release for the combined MoC group was significantly higher than the release for the combined FSC (P = 0.001) and for the combined CoC (P = 0.0004). The difference between combined FSC and combined CoC groups was not significant (P = 0.657).
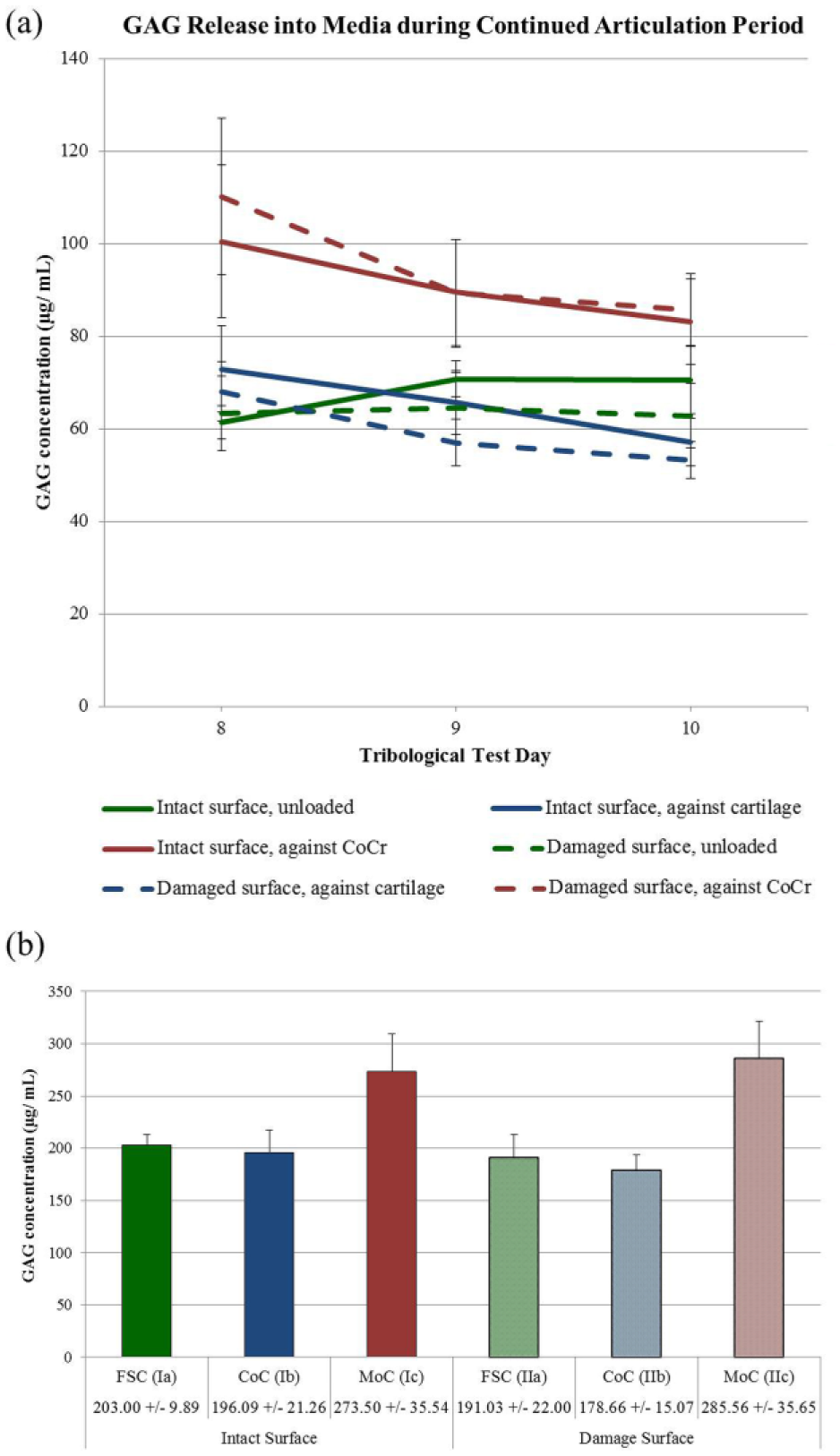
(
Cartilage Wear due to Summative Effects of Damage Induction and Continued Articulation
Cumulative GAG release over the entire 5-day period was significantly higher for damage samples versus the intact samples with surface status being a highly significant contributing factor (P < 0.0001) ( Fig. 4a ). Paired comparisons of the articulation type against damaged or intact surfaces resulted in significant differences as well (FSC: P = 0.004; CoC: P = 0.031; MoC: P = 0.013). Additionally, the individual rest time points between tests, which compare the ongoing matrix response when tribological stresses were removed, exhibited no differences in GAG content released into the media when comparing intact versus damaged surfaces and when comparing between the counterface materials ( Fig. 4b ). No differences were detected in the cumulative GAG release following the removal of mechanical stimuli ( Fig. 4c ), neither between nor within groups. Therefore, the cumulative difference in GAG loss is largely attributed to the loss during the damage induction period, as there were no differences between intact and damage groups after the continued articulation period nor during the rest times when mechanical stresses are absent.
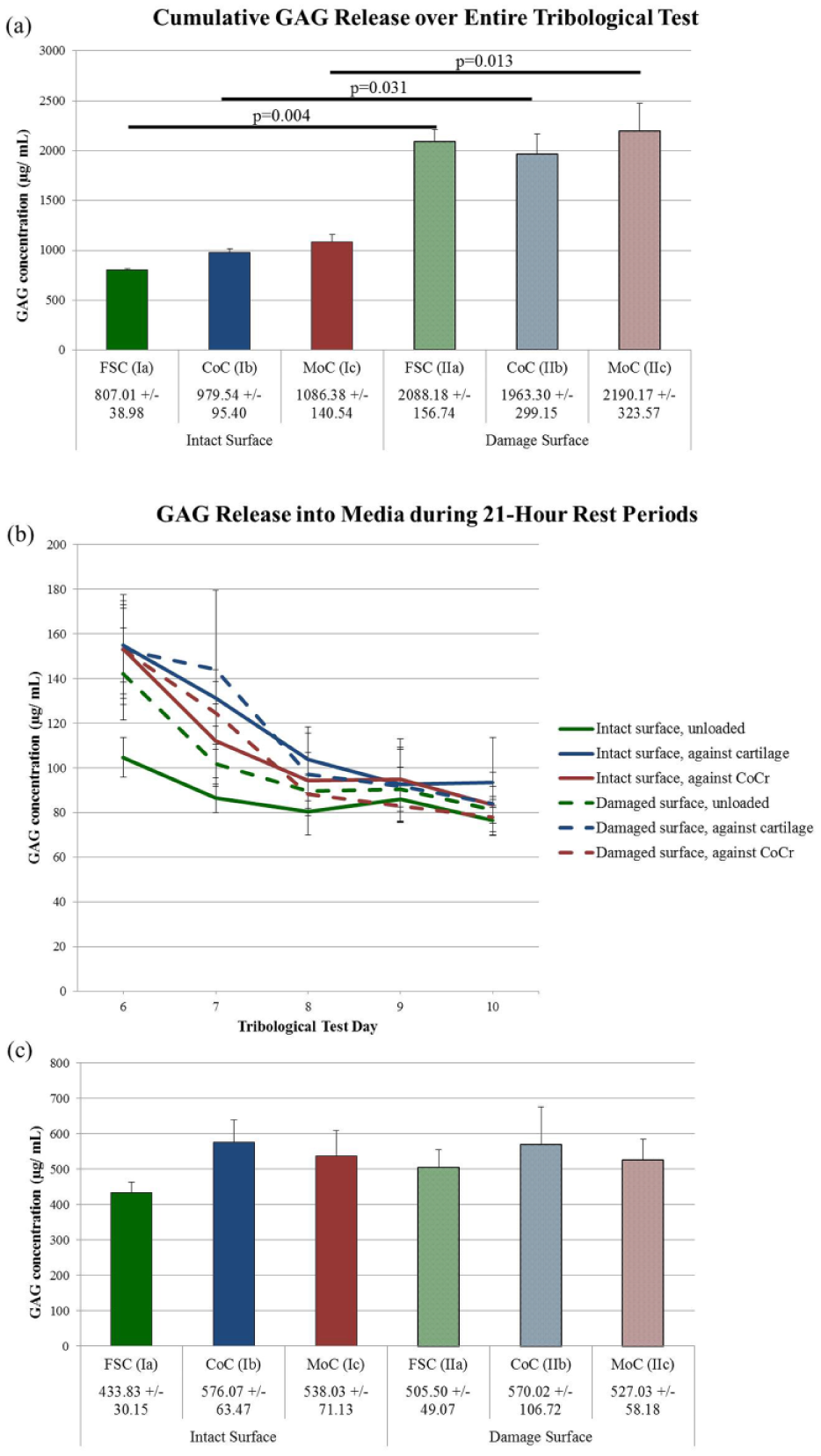
(
Increased tissue loss correlated with increased GAG released into the media
The damage induction period caused significant decreases in wet weight, resulting in about a 10% loss of tissue for damage groups. Wet weight for the damage groups was significantly lower compared to its corresponding intact group (FSC: P < 0.0001; CoC: P < 0.0001; MoC: P = 0.0002) ( Table 1 ). No differences were detected within either the intact groups or damage groups.
Change in Wet Weight from Pretest to Posttest as Mean Mass Change ± SEM. All Damaged Samples Lost Significantly More Tissue Than All Intact Samples (P < 0.001).
FSC =free-swelling control; CoC = continued articulation against cartilage; MoC = cartilage articulation against metal.
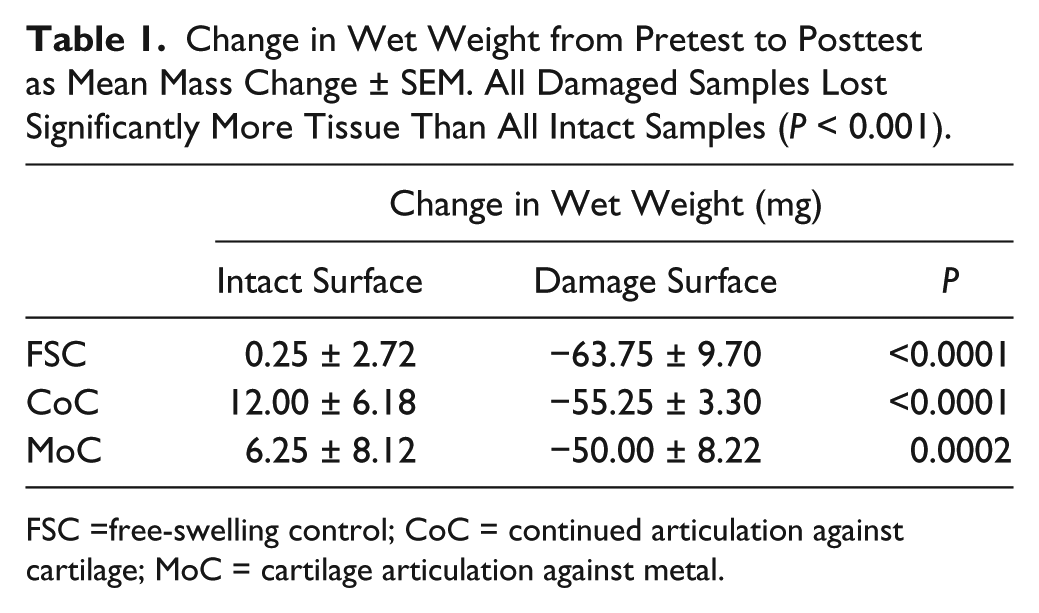
The significant tissue loss is displayed histologically in the loss of 10% to 20% of the original explant thickness from mainly the superficial zone of the wear area due to PE articulation. Evaluation of the matrix proteoglycans though Safranin-O staining reflected the surface damage as the surface status was a significant factor for differences in Mankin scores (P < 0.0001) ( Fig. 5 ). Damage surface samples displayed increased Mankin scores compared to the respective intact sample group (FSC: P = 0.035; CoC: P = 0.010; MoC: P = 0.102). Figure 5g to i magnifies the center wear area (as outlined in Fig. 5d to f with the black box) to illustrate the surface fibrillation that occurred with damage, but appeared worsened by articulation against metal. In addition to fibrillation, all damaged samples have a layer of decreased GAG at the surface (thinner blue layer), which resemble the naturally occurring layer in the intact samples. For intact surface samples, only the MoC samples displayed a minimal disruption of the superficial zone in the wear area with no differences in Mankin scores as compared to FSC or CoC.
Representative sections of histological samples stained for GAG with Safranin-O, viewed with a 4× objective. Mankin score for respective grouping located in the lower right of each image as means ± SEM. Damaged surface samples had significantly higher Mankin scores than their respective intact surface sample (P < 0.05 for all comparisons). The black box in (
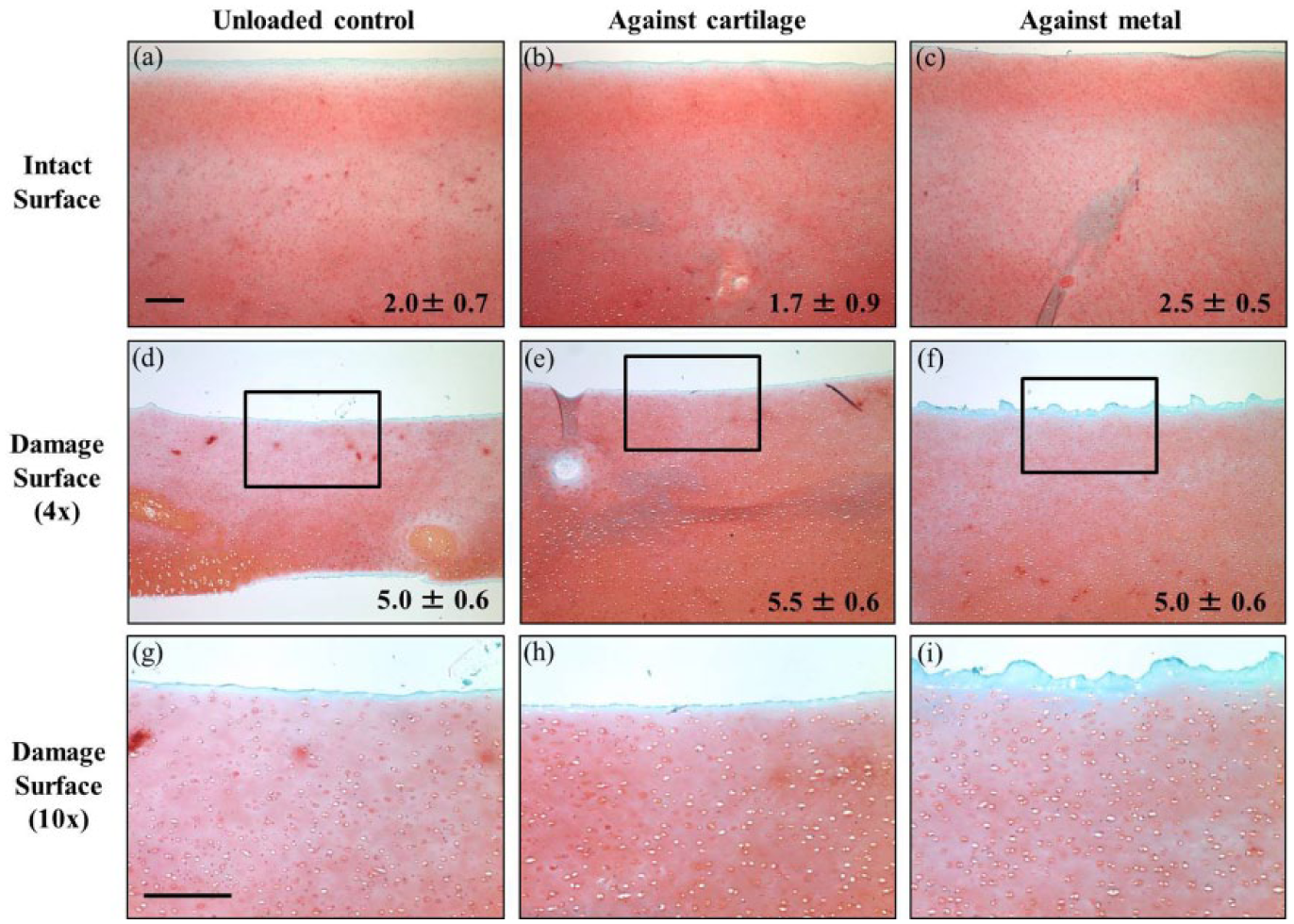
Using picrosirius red, surface disruption of collagen fibers can be seen in the intact MoC samples as compared to the FSC and the CoC ( Fig. 6a-c ). However, even more perturbation can be appreciated in the damage samples at the articular interface ( Fig. 6d-f ). There is an increase in red and orange fibers at the surface (represented by the white arrows), suggesting these thicker collagen fibers deep to the surface are in an altered orientation compared to intact samples.37,38 Figure 6g to i provides a higher magnification view of the center wear area for damage samples. The fibrillation appears to be worsened with the additional articulation against metal ( Fig. 6i ), as seen with the Safranin-O stained images as well.
Histological samples stained for collagen with picrosirius red, viewed with a 4× objective under with a polarizing filter. Increased coloration at the damaged interface, particularly seen as the red and orange colors, indicate a disruption of larger, more tightly packed fibers as the birefringence pattern changes (white arrows). The white box in (
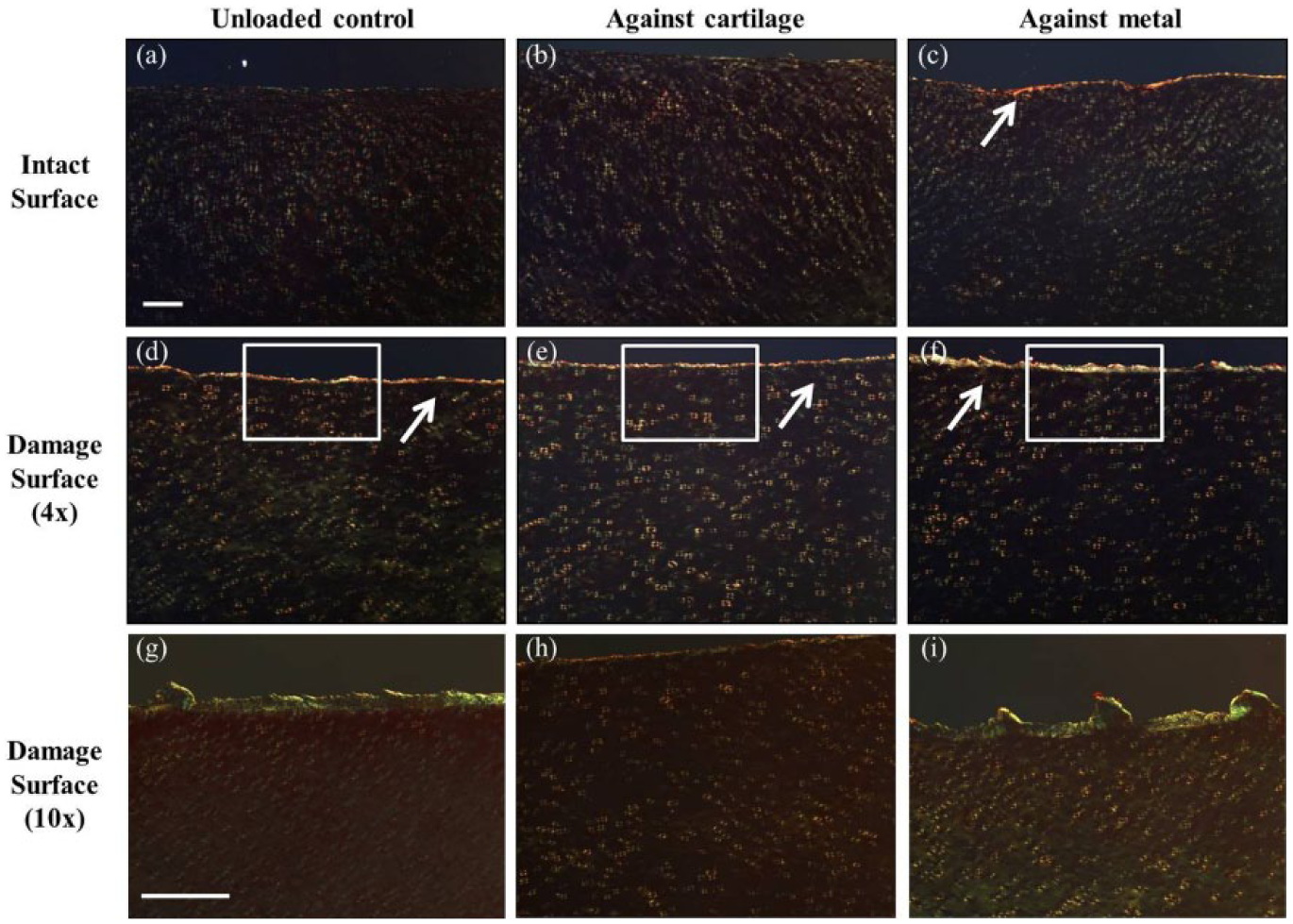
In addition to the histological matrix changes, changes were noted in cellular viability and organization. Increased cell death was caused by damage induction at the articulating surface and throughout the depth of the remaining tissue, as indicated by the red-stained cells in Figure 7b and d . In damage surface samples ( Fig. 6d-f ), displacement of the chondrocytes from the middle zone was noted, as the middle zone became the new articulating surface because of the loss of the superficial zone from damage induction.
Live/dead cross-sections from explants immediately after tribological testing from (
Cartilage Wear of the Cartilage Counterface
After articulation, cartilage strips looked grossly similar, regardless of the articulating cartilage counterface being intact or damaged. Cell viability for strips were similar ( Fig. 8a and b ), with limited cell death at the surface and increased death in the deep zone due to tissue handling and bending for conformation to the ball adapter. Histologically, strips were stained similarly with Safranin-O as no differences in Mankin scores were detected; no changes in matrix organization or cellular structure were apparent ( Fig. 8c and d ). Finally, during the rest periods between tribological tests, daily GAG release was also similar ( Fig. 8e ).
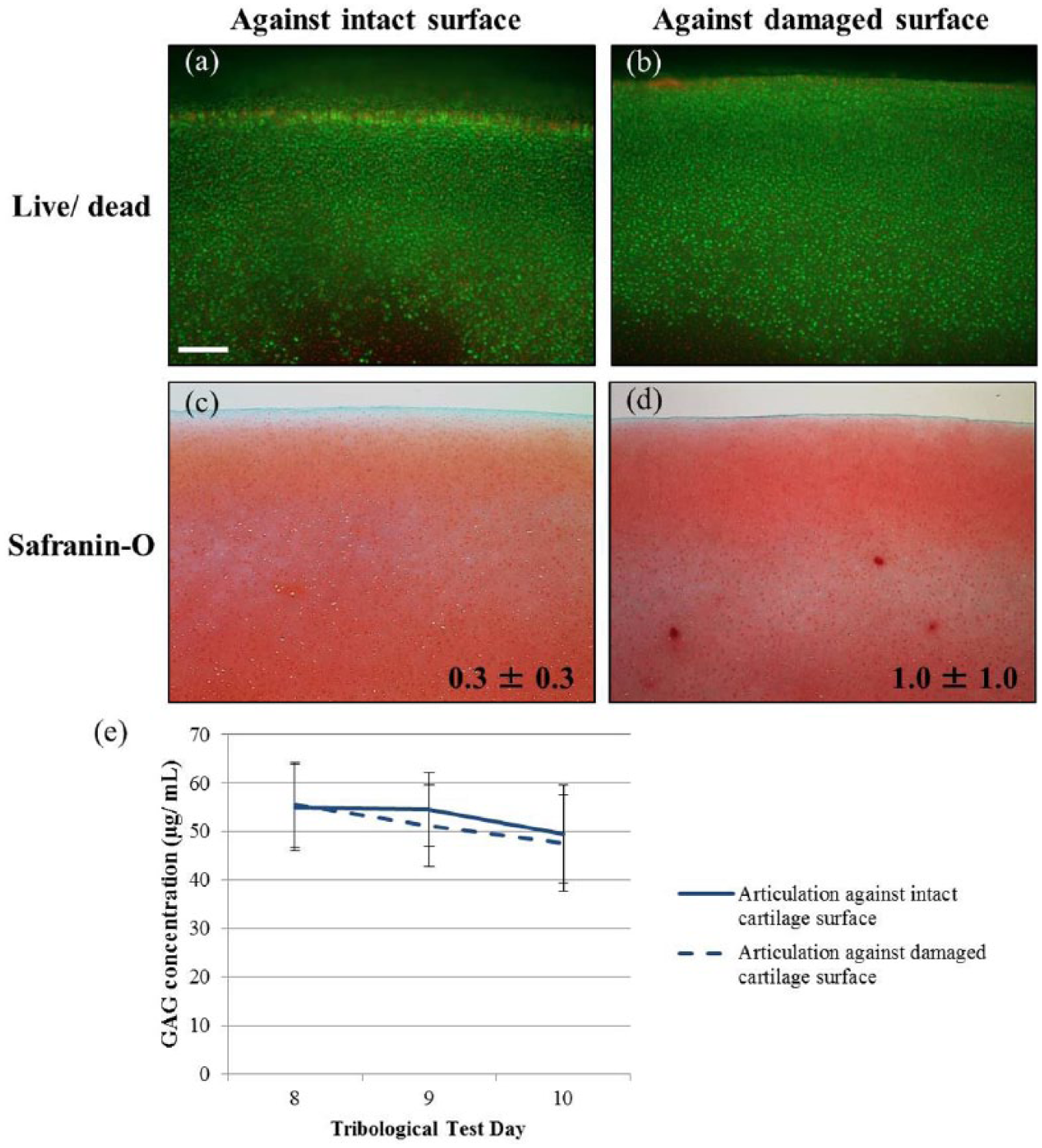
Live/dead cross-sections from cartilage strips immediately after tribological testing, which (
Discussion
This study aimed (1) to introduce a shear-induced cartilage surface damage model in which the superficial zone was removed and (2) to evaluate cartilage wear from intact and damaged surfaces in which tribological stresses were applied with articulation against cartilage and metal counterfaces. This model design is unique in that there are no other damage models that utilize shear forces to remove the superficial zone. Also, only 2 other studies applied load and motion to mechanically injured tissue.29,39 Such a model using complex articulating motion and loading against intact and damage surfaces has biological and clinical relevance. Intact models articulating against cartilage serve to further the understanding about mechanisms of normal cartilage degradation. Damage models articulating against cartilage replicates a diseased joint where one or both surfaces are potentially damaged by a shear insult. The models articulating against metal with either intact or damaged surfaces represent potential in vivo occurrences where the metal is used surgically as a cartilage replacement, such as in focal defect repairs, hemiarthroplasties, or patellar interventions in knee replacement surgery. 30
The study hypothesis that cartilage with an absent superficial zone would exhibit increased wear compared to cartilage with an intact surface was not supported. The importance of superficial zone integrity was driving the underlying hypothesis. Damage induction by articulation against PE caused injury to the cartilage surface, disruption of the collagen matrix, and increased GAG release, as best evidenced by damaged FSC samples, which remained unloaded after damage induction. However, once additional articulation against cartilage or metal was applied, any additional signs of matrix damage or differences in wear did not appear to be due to the condition of the surface but rather due to differences in articulating counterface. For instance, surface damage was not worsened by articulation against cartilage as determined by comparing FSC to CoC groups (i.e., no tissue loss, no differences in histological scoring, and no differences in GAG release). Articulation against metal caused increased GAG release and collagen disruption, supporting previous data generated in our laboratory. 40 The differences, though, were expected to be even more pronounced for MoC samples when comparing intact versus damage surface, in contrast to the actual findings. Generally, the lack of differences in GAG release in intact versus damage surfaces is contrary to multiple published hypotheses, according to which, collagen network damage causes increased fluid flow and fluid loss, leading to increased GAG loss and further matrix degradation before progressing to the ultimate development of OA.7,20,41
It was expected that GAG release for damaged samples would be higher because the integrity of the superficial zone was compromised. Also, once the new articulating surface of the damaged samples became the middle zone, differences in composition, mechanical properties, collagen fiber orientation, and gene expression patterns of the superficial zone and middle zone 42 were expected to add to the increased wear. It was beyond the scope of this study to investigate the underlying mechanisms that account for the lack of observed difference between the intact and damaged cartilage surface samples. One possibility for this lack of difference relates to a mechanism proposed in 2 different studies.7,19 The studies suggested that that in samples with an intact surface, their observed decrease in GAG release between testing and unloaded samples was due to the continuous cyclic loading that led to a decreased interstitial porosity and that changes in the type of mechanical loading would influence this release. Here, the collagen architecture at the articulating surface of damaged surfaces was disrupted (as evidenced by the increase in red/yellow/orange fibers in the plane of view). The articulation during the damage induction period may have caused reorganization of the oblique orienting collagen fibers of the middle zone, which becomes the new articulating surface for damaged samples with the loss of the superficial zone. 43 The disrupted fibers could still collapse and provide a protective barrier in order to decrease wear, similar to the role of the superficial zone in intact surface.
It is a challenge to compare these findings to those in impaction studies, the most prevalent damage model in in vitro testing. Both impaction and shear models of damage produce similar end-results with cartilage degradation, albeit through potentially different underlying mechanisms. For instance, the acute versus chronic time difference could account for contrasting results where mechanical impact studies have shown that GAG release is significantly increased as a result of the injurious compression.44,45 Interestingly, in both the acute and chronic mechanisms of PTOA, the authors attribute the GAG release to changes in the collagen network. A future study could compare shear versus impact damage in the context of the continued load and articulation to determine if the collagen matrix is disrupted differently.
The current study has several limitations related to study design. First, in utilizing the PE articulation for 2 days, there was a significant impact on the chondrocyte viability of the tissue in the worn area. This study did not evaluate the mechanism of cell death, that is, apoptosis or necrosis. This is significant in that there could be potential treatments, such as membrane stabilizing surfactants or caspase inhibitors, to promote cell survival and minimize the associated degradation. 46 Second, 3 days postdamage may have been too brief as other studies utilize longer culture periods following impaction damage.35,47 Third, the mechanical properties of the tissue samples were not measured, even though the surface collagen disruption would suggest changes in properties. Impaction studies have found decreased tensile strength with an altered collagen matrix 44 and reduced compressive stiffness and shear stiffness. 48 Last, GAG loss was the sole outcome measure of cartilage wear in response to injury, as it is a historical marker of tribological wear. 31 However, uncertainty persists regarding the underlying mechanism (e.g., is the release due to physical displacement or to proteolytic degradation), the role of metabolism and matrix protein synthesis (e.g., is this a remodeling mechanism), and the state of proteoglycans in the matrix (e.g., is there an aberrant retention affecting function). Furthermore, only evaluating these side chains limits an understanding of the relationship between the model of cartilage damage and matrix proteins, such as the aggrecan core protein, small proteoglycans, collagens, and cartilage oligomeric matrix protein (COMP). COMP, for example, has been shown clinically in patients to be influenced by mechanical factors, such as walking, 49 albeit independent of OA status.
In order to better understand the mechanism of the decreased GAG loss despite matrix surface injury, follow-up should be conducted with advanced imaging, such as immunohistochemistry, and with gene expression. For example, it is possible that lubricin generation or transfer from the cartilage strip surface plays a role in minimizing the GAG loss through creating a new superficial zone in conjunction with the changes detected in collagen fibers. Lubricin analysis could be performed via immunohistochemistry imaging of both cartilage surfaces and gene expression changes in the cartilage discs due to articulation.
In summary, we utilized a unique model of joint simulation to demonstrate that subsequent cartilage matrix wear was not impacted by damage of the superficial zone. The testing design allowed for a damage period followed by additional tribological testing in order to simulate an in vivo environment where damaged tissue can still be mechanically influenced through walking or other activities of daily living. The short-term tribological test following damage induction did not yield the expected increase in GAG release in response to the collagen disruption, which was initially caused by shear injury from articulation against PE. This result suggests a window of opportunity for clinical intervention in the immediate time after injury to repair cartilage matrix damage and prevent or slow the matrix degradation. Whether the repair should occur with biological supplementation or with bioengineered materials remains to be determined. Either of these repair strategies could be evaluated with our in vitro model of cartilage damage.
Footnotes
Acknowledgments and Funding
Special thanks to Dr. Lev Rappoport for assistance with histology and Dr. Michel Laurent for assistance with statistical analysis and interpretation. This work was supported in part by a grant of the National Institutes of Health (RLT-T32 AR052272) and by the Rush Arthritis and Orthopaedics Institute.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not sought for the present study because the bovine stifle joints utilized for cartilage harvest were obtained from a local food supplier.
