Abstract
Objective
This study aimed to compare standard saline lavage to combination saline and high-pressure carbon dioxide (CO2) lavage in removing marrow elements from osteochondral allografts.
Design
Six fresh hemicondyles were obtained. Three osteochondral allograft plugs (15-mm diameter, 6-mm depth) were harvested from each hemicondyle and randomized to 1 of 3 treatment arms: A, no lavage; B, 1 L standard saline lavage; C, simultaneous saline (1 L) and 1-minute high-pressure CO2 lavage. After hematoxylin and eosin staining, a “percentage fill” of remaining marrow elements was calculated for each overall sample and then repeated in 3 distinct compartments for each sample based on depth from surface: 1, deepest third; 2, middle third; and 3, most superficial third. Trial arms B and C were compared with 1-tailed Student t tests.
Results
Group A had an overall percentage fill of 51.2% ± 8.8%. While both lavage techniques decreased overall remaining marrow elements, group B yielded significantly higher percentages of remaining marrow elements than group C (28.6% ± 16.5%, 14.6% ± 8.7%, P = 0.045). On depth analysis, group A exhibited homogenous filling of trabecular space (63.0% ± 15.5%, 67.6% ± 13.7%, and 55.2% ± 10.1% in zones 1, 2, and 3, respectively). Both lavage arms equally removed marrow elements from superficial zone 3 (B, 17.4% ± 9.2%; C, 15.6% ± 12.4%, P = 0.41) and middle zone 2 (B, 30.2% ± 17.7%; C, 21.4% ± 15.5%, P = 0.18). However, group C lavage removed significantly more marrow elements in deep zone 1 than group B (29.7% ± 10.9%, 58.5% ± 25.2%, P = 0.01).
Conclusion
Combination saline and high-pressure CO2 lavage more effectively clears marrow elements from osteochondral allografts than saline alone.
Introduction
Osteochondral allograft transplantation (OCA) has become a dominant treatment strategy for symptomatic osteochondral defects.1,2 Common indications for OCA include posttraumatic chondral defects and osteochondral lesions, such as osteochondritis dessicans and avascular necrosis. 3 Long-term success rates of OCA for focal chondral and osteochondral defects have ranged between 50% and 90% in the literature.4-6 While graft failure can be attributed to several factors, the role of the host immune response to antigenic cellular content and major histocompatibility complexes of graft marrow cells and bone lining cells remains debated within the orthopedic community.7,8 Intact cartilage is immunologically privileged due to its rigid extracellular matrix. Allograft bone and remaining marrow elements, however, have been shown to stimulate an immune response in the host that may affect graft survival. 8
Given that postoperative antibody development may impact graft integrity and longevity, there is tremendous interest in decreasing the amount of antigenic cellular material present in osteochondral allografts. 9 Current practice guidelines recommend a thorough lavage of the donor graft with sterile saline before implantation. 10 However, no study to date has quantitatively assessed the efficacy of this lavage on removing marrow elements. The application of high-pressure carbon dioxide bone cleaning may offer an improvement from standard saline lavage. 11 The purpose of this study was to quantify the impact of standard saline lavage alone versus saline lavage with simultaneous high-pressure carbon dioxide (CO2) lavage on removing bone marrow elements from donor osteochondral allograft tissue. The authors hypothesized that osteochondral allografts receiving standard lavage with sterile saline would show decreased bone marrow cellular material compared with grafts receiving no treatment at all, and that the addition of high-pressure CO2 lavage would further assist in removing marrow elements.
Methods
After obtaining institutional review board exemption for this cadaveric tissue study, 6 fresh hemicondyles were obtained from an osteochondral allograft supplier (JRF Ortho, Centennial, CO). All grafts were centrifuged by the tissue bank as part of standard tissue processing and were stored in a nutrient media containing antibiotics, Eagle’s minimal essential medium (EMEM), and fetal bovine serum. To mimic clinical conditions but not interfere with patient care, hemicondyles were donated immediately after the supplier’s self-imposed 28-day expiration date.12,13 Samples arrived on the day of the experiment and were placed in a basin of sterile saline while graft harvest instruments were prepared. Three osteochondral allograft plugs were harvested from each hemicondyle (Arthrex OATS System, Naples, FL) ( Fig. 1 ). To mimic clinical practice, osteochondral plug diameter was determined using a 15-mm harvesting reamer, while plug depth was measured with a ruler to a depth of 6 mm. Once harvested, osteochondral plugs were randomly assigned to 1 of 3 treatment arms: A, no lavage; B, 1 L standard pulse saline lavage (Pulsavac System, Zimmer/Biomet, Warsaw, IN); C, simultaneous saline lavage (1 L) and 1-minute high-pressure CO2 lavage (CarboJet, Kinamed, Camarillo, CA) ( Fig. 2 ).
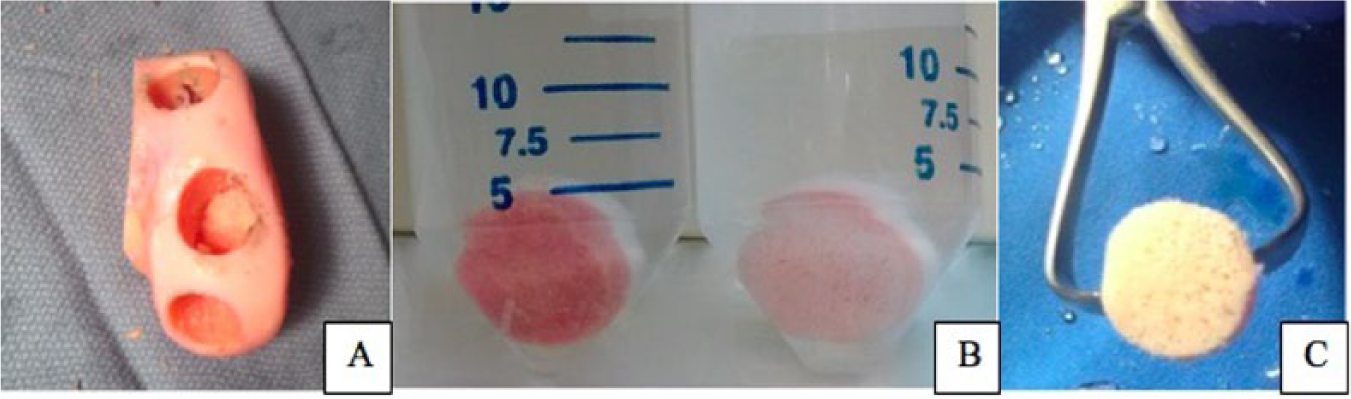
Human femoral hemicondyle after harvest of 3 osteochondral allograft plugs (
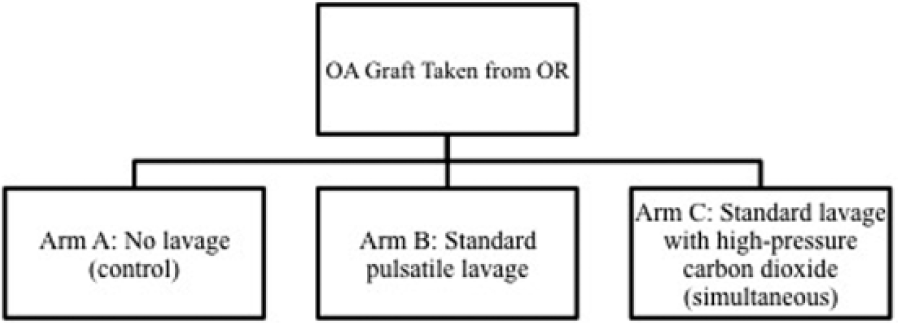
Three trial arms of the osteochondral allograft lavage study.
In groups B and C, 1 L of saline took 1 minute to dispense, so both groups received an equal duration of treatment. During both saline and carbon dioxide lavage, a towel clamp was used to grasp the osteochondral plug along the subchondral plate in order to avoid damage to cartilage. The towel clamp was rotated regularly throughout both cycles to allow for equal lavage distribution across the entirety of the plug. In all cases, the lavage instrument was held 1 cm away from the plug and directed perpendicular to the bony surface.
After lavage treatment, all samples were fixed in 10% neutral buffered formalin for 3 days in a temperature-controlled environment. After fixation, all samples underwent decalcification in aqueous 60% formic acid:40% sodium citrate solution. Once decalcified, samples were embedded in paraffin, sectioned, and subjected to staining with hematoxylin and eosin to identify remaining bone marrow elements residing in the trabecular space. Sections were viewed by light microscopy at 4× and 20× views to evaluate the presence of remaining cellular material. Percentage of remaining cellular material was calculated using a “percent fill” technique that has been utilized in prior studies.14,15 The purpose of the percent-fill technique is to show the 2-dimensional area occupied by marrow elements as a percentage of all available free trabecular space. All calculations of area were carried out using ImageJ software (National Institutes of Health, Bethesda, MD).
To assess depth of penetration of both lavage modalities, calculations were repeated in 3 distinct compartments for each sample based on depth from surface: 1, deepest third, adjacent to cartilage; 2, middle third; and 3, most superficial third ( Fig. 3 ). Since cartilage thickness varies depending on location of graft harvest on the hemicondyle, each 6-mm depth plug contained a slightly different depth of available subchondral bone. This minor variability was accepted to mimic clinical practice. As such, the 3 zones of depth for subgroup analysis were identified on each sample by viewing the entire section on ImageJ and dividing available depth of subchondral trabecular space into thirds. Trial arms B and C were compared using 1-tailed t tests with significance set at P < 0.05 (SPSS Software, Armonk, NY).
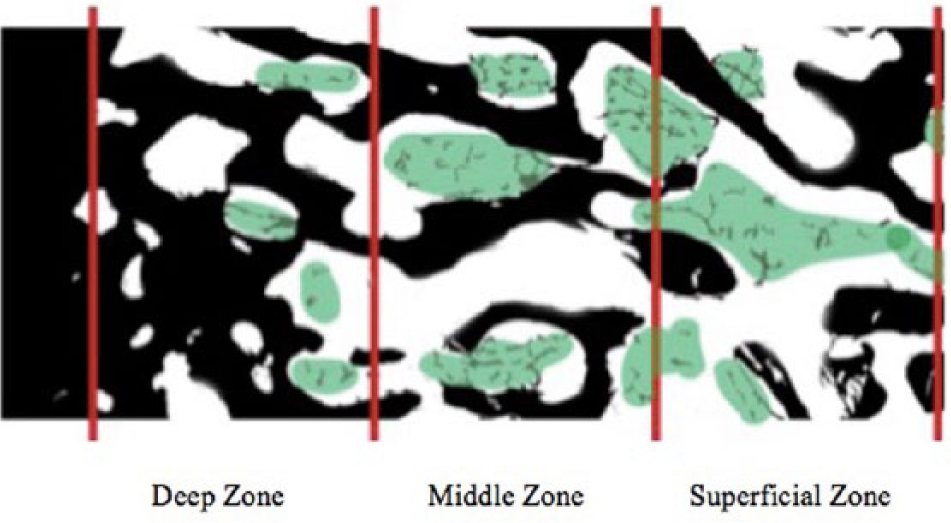
Illustration of decalcified osteochondral allograft histology showing bone (black), trabecular space still containing marrow elements (green), and trabecular space that has been washed free of marrow elements (white). Using this terminology, “percent fill” equals: [Green area / (Green area + white area)] × 100.
Results
In total, 4 samples were taken from the left lateral hemicondyle, 1 from the right lateral hemicondyle, and 1 from the left medial hemicondyle. The average age of the grafts at the time of lavage was 37.0 days since donor death. All efforts were made to harvest to a depth of 6 mm. Cartilage depth ranged from 2 to 3 mm, leaving 3 to 4 mm trabecular space to be analyzed according to zones of depth.
Analysis of the percentage of remaining bone marrow elements after various lavage modalities revealed that both standard saline lavage and saline lavage in combination with high-pressure CO2 substantially decreased the percentage fill of osteochondral allografts. Group A had an overall percentage fill of 51.2% ± 8.8%. While both lavage techniques decreased overall remaining marrow elements versus the control group, group B yielded significantly higher percentages of remaining marrow elements than group C (28.6% ± 16.5%, 14.6% ± 8.7%, P = 0.045) ( Fig. 4 ). On subgroup depth analysis, control samples exhibited homogenous filling of trabecular space (63.0% ± 15.5%, 67.6% ± 13.7%, and 55.2% ± 10.1% in zones 1, 2, and 3, respectively). Both lavage arms equally removed marrow elements from superficial zone 3 (B, 17.4% ± 9.2%; C, 15.6% ± 12.4%, P = 0.41) and middle zone 2 (B, 30.2% ± 17.7%; C, 21.4% ± 15.5%, P = 0.18). However, group C lavage removed significantly more marrow elements in deep zone 1 than group B (29.7% ± 10.9%, 58.5% ± 25.2%, P = 0.01) ( Fig. 5 ).
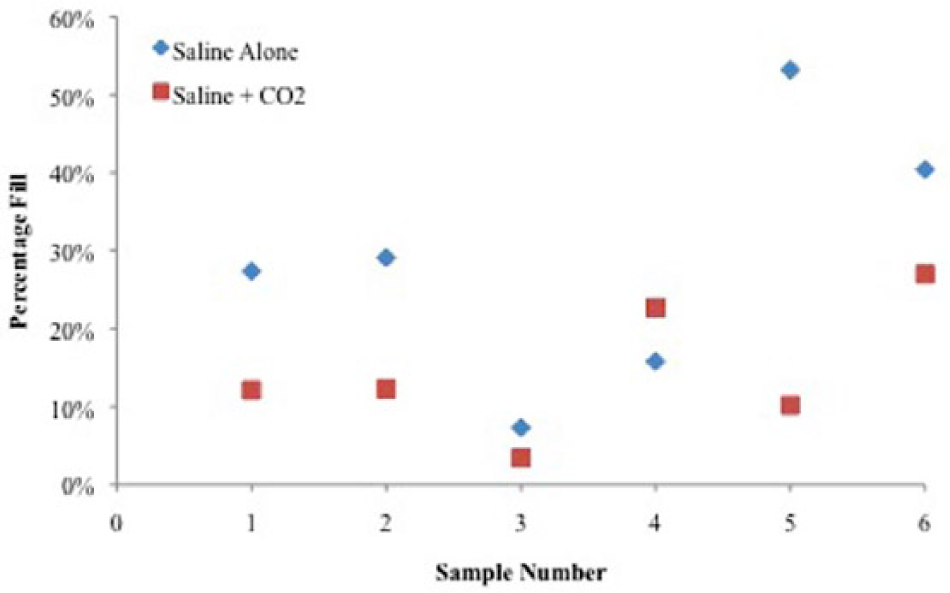
Comparison of percentages of marrow elements remaining for all samples after lavage with both saline alone and combination saline and carbon dioxide.
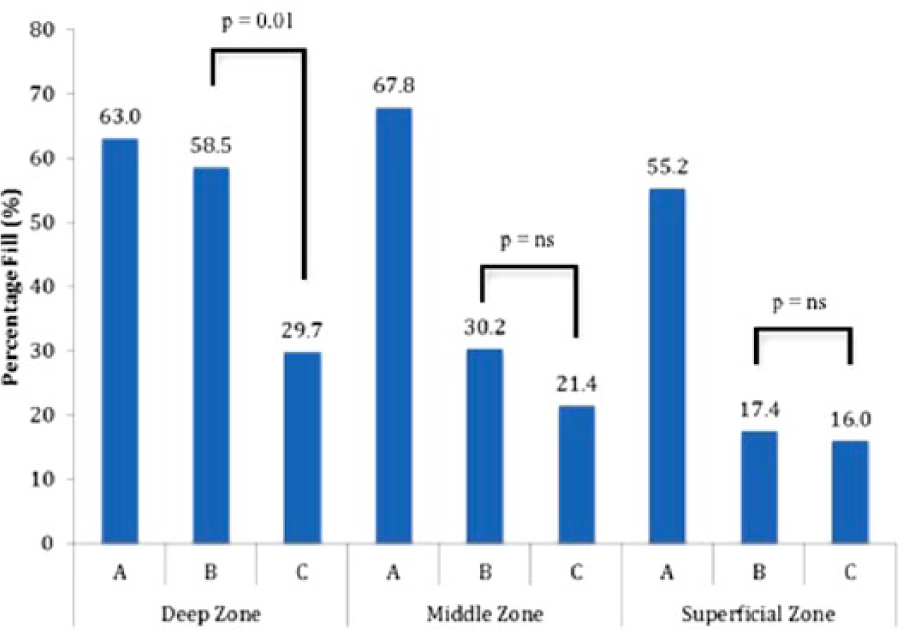
Marrow elements remaining did not differ between treatment groups in the superficial or middle zones (
Discussion
While the role of the immune response associated with OCA remains unclear, studies of failed osteochondral allografts have demonstrated both histological and radiographic evidence of immune-mediated graft rejection.9,16 OCA remains unique among allogeneic transplantation procedures in that it does not involve human leukocyte antigen (HLA) or ABO blood group matching to prevent an immune response; nor do recipient patients receive any form of postoperative immunosuppression. While cytotoxic treatment and freezing of grafts have both been shown to eliminate the host immune response,17-21 their downstream negative impact on chondrocyte viability precludes their use in OCA. 22 In the search for a treatment modality that can best remove immunogenic marrow elements without subjecting chondrocytes to thermal or cytotoxic insult, our results suggest that improved lavage therapy may be the next step forward. Combination saline and high-pressure CO2 lavage halves the amount of remaining material when compared with saline lavage alone. On subgroup analysis, improvement appears most pronounced in the deepest third of the OCA plug. This likely occurs with both lavage modalities working in synergistic fashion: carbon dioxide gas penetrating more deeply into the trabecular space, with saline flushing the liberated elements away.
Both animal and human studies have suggested that antibody responses to OCA are elicited by membrane-associated histocompatibility complexes found in blood and marrow cells occupying the trabecular space. 8 In a rat model, Stevenson et al. 23 found that major histocompatibility complex (MHC) class I–mismatched fresh grafts had the highest antibody titers and subsequently exhibited rapid pedicle occlusion with diminished internal remodeling of cortical bone when compared with frozen mismatched grafts. In humans, Chu et al. 4 partially attributed higher failure rates in bipolar allograft procedures (50% vs. 7% with unipolar allografts) to increased surface area in bipolar implants, as anti-donor antibody titer frequency and magnitude correlated with graft size. Similarly, Hunt et al. 7 found that large grafts (>10 cm2) were 36 times more likely to elicit an antibody response than small graft (<5 cm2). While the antibody-positive group in this study had a higher failure rate (36% vs. 21%), this failed to reach statistical significance. In regards to imaging outcomes, Sirlin et al. 9 compared magnetic resonance imaging findings with development of post-OCA antibody responses and found that those who developed serum anti-HLA antibodies demonstrated greater edema, thicker graft-host interface, more abnormal graft marrow, and a higher proportion of surface collapse. These findings collectively suggest that the immune response elicited in the host may affect long-term outcomes after OCA.
Despite this evidence, multiple studies have raised doubts about the clinical ramifications of postoperative antibody development and its impact on graft survival. Two separate histological studies of failed OCA explants by Kandel et al. 24 and Oakeshott et al. 25 failed to identify endothelial cell swelling, vasculitis, or other evidence of an immune reaction against the grafts. While Williams et al. 26 found 1 case of mononuclear lymphocyte infiltration and four cases of pannus formation in an analysis of 26 failed OCAs, the authors concluded due to lack of vasculitis and inflammatory infiltrate that these were not immune-mediated reactions. Ultimately, failure after OCA is likely multifactorial, with the immune response playing a variable role based on sensitization of the host. Findings by Fraitzl et al. 27 support this notion, as the authors found that OCAs transplanted to sensitized hosts exhibited much more rapid graft deteriorization, T-lymphocyte and macrophage invasion, and marrow cavity fibrosis than OCAs transplanted to nonsensitized hosts. Since allogenic chondrocytes remain protected by a dense, avascular matrix, sensitization and subsequent cellular and humoral immunity likely stems from exposure to elements within the trabeculae.8,16 More specifically, marrow elements, such as collagen molecules and their degraded fragments, represent potential immunogenic proteins within bone allografts, and their removal may alleviate immunological reactivity and improve graft outcome.28,29 On the basis of our findings, combination CO2 and saline lavage accomplishes this goal most effectively.
Our study has several limitations. Our small sample size (n = 6) was due to limited graft availability, as all grafts within the 28-day postmortem period were prioritized toward patient care. As a direct consequence of this, our study utilized grafts that were recently expired (average age of 38 days postmortem) and may not be entirely representative of the cellular milieu residing in the trabecular space. However, while prolonged graft storage has been shown to affect chondrocyte survival, there is no evidence to suggest that it affects the quantity of marrow elements contained within the trabecular space. This is supported by the abundant amount of marrow elements found in our control group. Second, our histological analysis focused more on quantity of marrow elements, rather than the specific immunogenic components within the trabeculae. Given that the cellular components driving immunogenicity remain unclear, we reasoned that absolute reduction in total marrow elements would likely parallel a reduction in those components responsible for the immune response. Similarly, there may be an immunogenic component within graft bone that is acellular that would not be addressed by this technique. There may also have been minor differences in bony anatomy between donor grafts, such as bone density, which could affect the ability of lavage to remove bone marrow elements. To control for these subtle differences in underlying bony anatomy, all effort was made to harvest all 3 OCA plugs from the same portions of each hemicondyle. Finally, given that this was a controlled laboratory study, we were unable to assess the clinical ramifications of improved lavage. However, given the dramatic decrease in the percentage of marrow elements, particularly in the deep zone of our samples, we feel this technique can be immediately translational to use in clinical care. Future in vivo studies are needed to determine whether decreases in remaining marrow elements affect graft survival and clinical outcomes.
Footnotes
Acknowledgments and Funding
This was an internally funded study. However, the CarboJet device was donated by Kinamed (Camarillo, CA, USA) and the hemicondyles were donated by JRF Ortho (Centennial, CO, USA).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ABY reports research support from Arthrex, Inc. and NuTech Medical. BJC reports royalties from Arthrex, DJO Global, and Elsevier; serves as a paid consultant to Arthrex, Carticept Medical, Regentis Biomaterials, and Zimmer; has stock or stock options held in Carticept Medical and Regentis Biomaterials; has received research or institutional support from Arthrex, Medipost, the National Institutes of Health (NIAMS and NICHD), and Zimmer; has received nonincome support (such as equipment or services), commercially derived honoraria, or other non–research-related funding (such as paid travel) from Athletico, Össur, Smith & Nephew, and Tornier.
Ethical Approval
Ethical approval for this study was waived by the Rush University Medical Center Institutional Review Board because of use of de-identified cadaveric specimens.
