Abstract
Objective
Articular cartilage is an avascular tissue with limited ability of self-regeneration and the current clinical treatments have restricted capacity to restore damages induced by trauma or diseases. Therefore, new techniques are being tested for cartilage repair, using scaffolds and/or stem cells. Although type II collagen hydrogel, fibrin sealant, and adipose-derived stem cells (ASCs) represent suitable alternatives for cartilage formation, their combination has not yet been investigated in vivo for focal articular cartilage defects. We performed a simple experimental procedure using the combination of these 3 compounds on cartilage lesions of rabbit knees.
Design
The hydrogel was developed in house and was first tested in vitro for chondrogenic differentiation. Next, implants were performed in chondral defects with or without ASCs and the degree of regeneration was macroscopically and microscopically evaluated.
Results
Production of proteoglycans and the increased expression of collagen type II (COL2α1), aggrecan (ACAN), and sex-determining region Y-box 9 (SOX9) confirmed the chondrogenic character of ASCs in the hydrogel in vitro. Importantly, the addition of ASC induced a higher overall repair of the chondral lesions and a better cellular organization and collagen fiber alignment compared with the same treatment without ASCs. This regenerating tissue also presented the expression of cartilage glycosaminoglycan and type II collagen.
Conclusions
Our results indicate that the combination of the 3 compounds is effective for articular cartilage repair and may be of future clinical interest.
Keywords
Introduction
Damages to the joint cartilage represent a significant issue of public health. 1 Articular cartilage presents reduced ability of self-repair and the current treatments for large degeneration are still limited. 1 In this scenario, tissue engineering has been proposing new solutions for cartilage formation, based on the conjunction of different sorts of scaffolds and cells. 2 Although several preclinical studies have been conducted using type II collagen hydrogels, fibrin sealant, and adipose-derived stem cells (ASCs), 3 their combination has not yet been investigated for focal defects. In this study, we tested the conjunction of these 3 compounds on cartilage repair of rabbit knees. Our results indicate that this combination is effective for articular cartilage repair and may be of interest for future clinical applications.
Methods
ASCs were isolated from human adipose tissue samples obtained from healthy female donors (age range 25-50 years), during abdominal liposuction procedure for esthetic purposes at the Hospital Medical School, University of Campinas, Brazil. The study was conducted in accordance with the Declaration of Helsinki and was approved by the local Ethical Committee Board. Culture procedures were performed as previously described 4 and the cells were used after confirmation of purity and capacity of differentiation into chondrogenic, adipogenic, and osteogenic lineages (Suppl. Fig. S1). Hydrogel was prepared in house using type II collagen derived from chicken sternum cartilage, as detailed in the Supplementary Material. Live/dead staining showed high cell viability and density in the scaffold (Suppl. Fig. S2).
The ability of the hydrogel to sustain ASC chondrogenic differentiation was first tested in vitro. The protocol was executed as previously described 4 and the experiments were performed 4 times independently, using ASC from 4 different donors. Expression of collagen type I (COL1α2), collagen type II (COL2α1), aggrecan (ACAN), and sex-determining region Y-box 9 (SOX9) was analyzed by quantitative polymerase chain reaction. Primer sequences are described in the Supplementary Table S1. Next, in vivo cartilage repair was evaluated using surgical defects (3 mm) performed in the knees of 4 New Zealand White rabbits (~12 weeks old). The animal procedures were approved by the Committee on Animal Research and Ethics of University of Campinas and are detailed in the Supplementary Material. The defects in the right knees were filled with hydrogel containing 106 cells (hydrogel with ASCs), whereas the defects in the left knees were filled with cell-free hydrogel as matched controls (hydrogel-only). All the implants were attached with fibrin sealant. After 12 weeks, the rabbits were sacrificed and the defect areas were analyzed by means of gross morphology and histological examination. Hematoxylin and eosin staining and alcian blue staining were performed according to standard procedures. Second harmonic generation (SHG) imaging was carried out as previously described. 5
Results
During in vitro differentiation, the initially viscous and transparent material suffered retraction and appeared as a small whitish microsphere, firm in consistency and easily handled, about the size of 1-mm diameter ( Fig. 1A ). At the end of 3 weeks, the material was stained with toluidine blue and cells were collected for gene expression analysis. Toluidine blue staining indicated the production of abundant extracellular matrix proteoglycans, especially at the periphery of the scaffold. The cells showed adherence and were well distributed throughout the hydrogel ( Fig. 1B ). A pronounced upregulation of COL2α1 was observed by the induction of chondrogenic differentiation (P < 0.05, Mann-Whitney test). The expression of SOX9 and ACAN also increased compared with control ( Fig. 1C ), indicating an effective chondrogenic character of the ASC. COL1α2 was also upregulated, which is expected after 3 weeks of ASC chondrogenic differentiation. 6
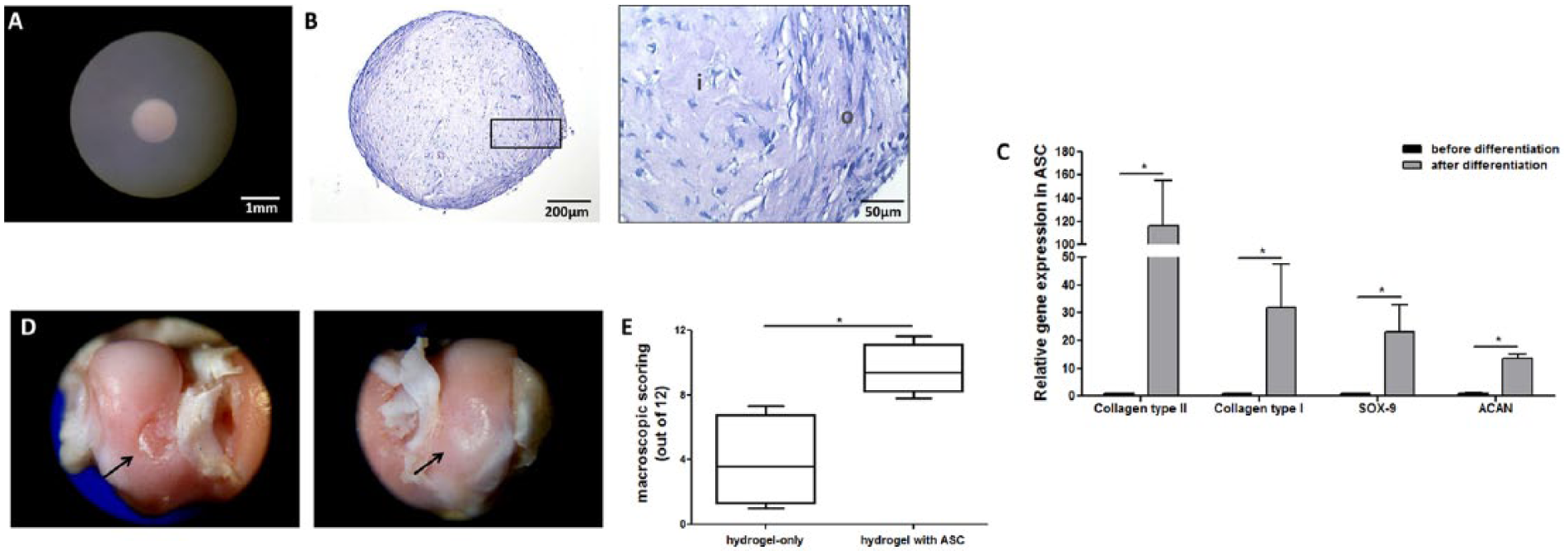
In vitro chondrogenic differentiation of adipose-derived stem cells (ASCs) in type II collagen hydrogel and macroscopic analysis of in vivo cartilage repair. (
The implants caused no postsurgical inflammation and the animals exhibited normal behavior and gait. After 12 weeks of the surgical procedure, macroscopic analysis revealed great differences between the defects treated with hydrogel-only and with hydrogel in combination with ASCs. The control group displayed a depressed and irregular tissue covering the defects, with limited integration with surrounding area. In contrast, the hydrogel with ASC implants appeared with neo-tissue graft, presenting a smooth and flush surface with the surrounding native cartilage and a moderately flat regularity ( Fig. 1D ). Macroscopic cartilage repair was scored by 5 blinded observers according to the International Cartilage Repair Society (ICRS) scoring system (0-12 points). 7 Control implants received a score of 3.58 [1.00-7.33]; whereas a score of 9.41 [7.83-11.67] was given to the hydrogel with ASC treated implants ( Fig. 1E ). P < 0.05; Mann-Whitney test (data are shown as median [min-max]).
Tissue organization was microscopically analyzed by hematoxylin and eosin staining and SHG analysis was used to evaluate collagen fibers formation and orientation. Contrary to the native cartilage ( Fig. 2A-D ), the lesions treated with hydrogel-only were occupied by an unstructured tissue ( Fig. 2E and F ). SHG microscopy showed disorganized collagen fibers, lacking defined orientation ( Fig. 2G and H ). In contrast, all the defects treated with the hydrogel in combination with ASC appeared hypercellular and presented intact surrounding native cartilage with no apparent signs of degeneration, but 2 different patterns of cellular organization were observed. One of them presented a high number of uniformly distributed cells in the defect area ( Fig. 2I and J ) and a better organization of collagen fibers compared with hydrogel-only treated lesions ( Fig. 2L and M ), supporting a remodeling process. The other pattern of organization was characterized by the presence of clusters of chondrocyte-like cells, especially in the deeper zones, suggesting an even more advanced healing process. The territorial and interterritorial matrices were well defined ( Fig. 2N and O ). SHG showed increased intensity of fluorescence around the cell clusters and appeared more similar to the normal cartilage ( Fig. 2P and Q ). Fast Fourier transform (FFT)–based analysis ratio from these defects was increased in comparison with hydrogel-only treated lesions (3.26 ± 0.30 vs. 2.63 ± 0.19, respectively) and was similar to the native tissue (3.93 ± 0.4).
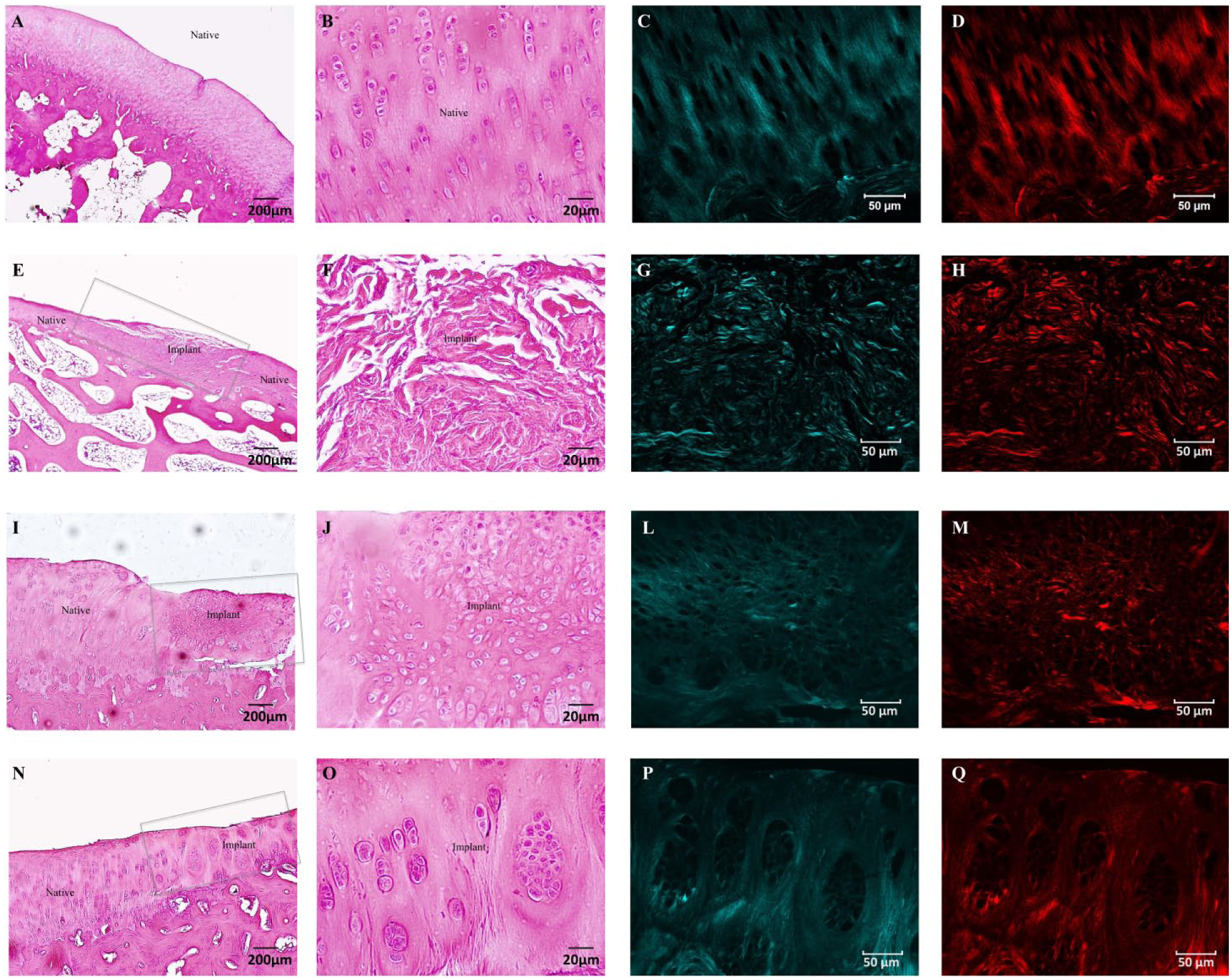
Microscopic tissue organization of the implant areas and surrounding native tissue. (
Some sections of the defects treated with ASCs plus hydrogel were chosen for alcian blue staining and immunohistochemical analysis of type I and II collagen. We observed the presence of glycosaminoglycans and expression of type II collagen, indicating the formation of hyaline cartilage in the regenerating area (Suppl. Fig. S3). Type I collagen was mainly detected in the central area and in a thin layer on the surface of the adjacent native cartilage, which may be explained by a systematic effect of focal articular cartilage injury, as previously observed. 8 Immunostaining for type I collagen was decreased in the border zones of the defect, which contained cells in a more advanced stage of maturation (Suppl. Fig. S3).
Quantitative histologic evaluation was performed by 4 blinded individuals using the Pineda scoring system (0-14 points), 9 in which the lowest score indicates a better cartilage repair. The average histologic score in the hydrogel-only group was 7.25 ± 2.0, while hydrogel with ASC group received a score of 3.3 ± 1.1 (P < 0.05; Mann-Whitney test).
Discussion
In this short communication, we report the efficacy of the combination of type II collagen hydrogel, ASCs, and fibrin sealant on cartilage repair. Since the application of only hydrogel and fibrin sealant led to inferior tissue regeneration, our data confirm previous studies that showed a central role of stem cells in obtaining a neo-tissue with similar morphology of the native cartilage. 10 Although the study of ASC was important in the past, it is now widely accepted that they present good results in terms of cartilage formation and regeneration. 11 However, the majority of the studies were performed after in vitro chondrogenic induction of ASCs. Our data indicate that expanded ASCs are capable of inducing cartilage repair without prior differentiation.
It is well known that the use of xenogeneic cells may induce rejection. However, there are increasing evidences that ASCs present immunosuppressive effects in many circumstances. 12 This property has been used as an advantage for xenogeneic transplantation and several studies have been conducted using the injection of human ASC in animal models. 13 In accordance with this, we observed no inflammation or lymphocytic/macrophagic infiltrates after 12 weeks of the injection of human ASCs in our rabbit model, suggesting that the human ASCs were well accepted by the host organism.
Fibrin sealant was used to add the hydrogel the property of fixing to the substrate. The integration of injured areas with adjacent native cartilage and the anchorage to the underlying subchondral bone indicated that the hydrogel was successfully attached to the defects. Moreover, we believe that fibrin prochondrogenic characteristics 14 may have reinforced cartilage repair. Biosynthesis of collagen type I may indicate the formation of fibrous tissue and may be refined in further studies. However, a certain degree of type I collagen during in vivo cartilage repair has been frequently reported.8,15
In conclusion, the use of ASC without prior differentiation, combined with an in-house developed hydrogel, and fibrin sealant, turned out to form an easily handle material, which succeeded in promoting cartilage repair. Although a long-term analysis should be performed, these results are promising and may be of scientific interest for additional tests.
Footnotes
Authors’ Note
Authors Mariana Lazarini and Pedro Bordeaux-Rego contributed equally to this work.
Acknowledgments and Funding
The authors would like to thank Prof. Benedicto de Campos Vidal from University of Campinas for providing collagen type II, and Tereza Salles and Paulo Latuf Filho for their valuable technical assistance. We also thank Raquel Foglio for English review and Michel Moraes for figure formatting and design. This work was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq/MCT), Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) and Fundação de Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for the use of human cells was obtained from the Research Ethics Committee of the University of Campinas (approval number: 838/2008).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation. The study was approved by the Ethics Committee on Experimental Animal Research at the University of Campinas (approval number: 1714-1).
Supplementary Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
