Abstract
Objective
Dexamethasone is known to support mesenchymal stem cell (MSC) chondrogenesis, although the effects of dose and timing of exposure are not well understood. The objective of this study was to investigate these variables using a laboratory model of MSC chondrogenesis.
Design
Equine MSCs were encapsulated in agarose and cultured in chondrogenic medium with 1 or 100 nM dexamethasone, or without dexamethasone, for 15 days. Samples were analyzed for extracellular matrix (ECM) accumulation, prostaglandin E2 and alkaline phosphatase secretion, and gene expression of selected collagens and catabolic enzymes. Timing of exposure was evaluated by ECM accumulation after dexamethasone was withdrawn over the first 6 days, or withheld for up to 3 or 6 days of culture.
Results
ECM accumulation was not significantly different between 1 and 100 nM dexamethasone, but was suppressed ~40% in dexamethasone-free cultures. Prostaglandin E2 secretion, and expression of catabolic enzymes, including matrix metalloproteinase 13, and type X collagen was generally lowest in 100 nM dexamethasone and not significantly different between 1 nM and dexamethasone-free cultures. Dexamethasone could be withheld for at least 2 days without affecting ECM accumulation, while withdrawal studies suggested that dexamethasone supports ECM accumulation beyond day 6.
Conclusion
One nanomolar dexamethasone supported robust cartilage-like ECM accumulation despite not having an effect on markers of inflammation, although higher concentrations of dexamethasone may be necessary to suppress undesirable hypertrophic differentiation. While early exposure to dexamethasone was not critical, sustained exposure of at least a week appears to be necessary to maximize ECM accumulation.
Introduction
The ability to undergo chondrogenesis is a hallmark of bone marrow mesenchymal stem cells (MSCs),
1
which has generated significant enthusiasm that they may be capable of repairing articular cartilage defects. MSCs possess favorable properties for cartilage tissue engineering as they can be readily culture-expanded, and secrete robust quantities of cartilage-like extracellular matrix (ECM) after seeding into scaffolds and culture in the appropriate biochemical environment
For many years, the combination of selected growth factors and the glucocorticoid dexamethasone (Dex) has been used to induce robust MSC chondrogenesis
Effective strategies for delivering chondrogenic factors to support MSC chondrogenesis
Methods
Mesenchymal Stem Cells Isolation and Expansion
Mesenchymal stem cells were isolated from bone marrow aspirates from the iliac crest of 2- to 5-year-old horses that were euthanized for reasons unrelated to this study. Colony-forming cultures were established to isolate the MSCs from the bone marrow, 15 after which the MSCs were seeded at 2 × 103 cells/cm2 in tissue culture flasks in α-minimal essential medium, 10% fetal bovine serum, and 2 ng/mL fibroblast growth factor-basic (Peprotech, Rocky Hill, NJ) and cultured to 80% confluence over 4 days. The cells were expanded through a second passage prior to seeding in chondrogenic culture.
Agarose Encapsulation and Chondrogenic Culture
Culture-expanded MSCs were encapsulated in 2% (w/v) agarose gel at 12 × 106 cells/mL, as previously described. 15 Baseline chondrogenic medium consisted of high-glucose Dulbecco modified Eagle medium supplement with 1% ITS+ Premix (BD Biosciences, Bedford, MA), 37.5 μg/mL ascorbate-2-phosphate (Wako Chemicals, Richmond, VA), 5 ng/mL recombinant human transforming growth factor-β1 (Peprotech, Rocky Hill, NJ). 5 Cultures were maintained in 1 or 100 nM Dex (Sigma-Aldrich, Saint Louis, MO), or in Dex-free medium, for 15 or 21 days. Culture medium was changed every third day.
Quantification of Extracellular Matrix Accumulation and DNA
Following chondrogenic culture, MSCs-seeded agarose samples were weighed, and then digested in proteinase K (Sigma-Aldrich, Saint Louis, MO) in Tris–HCl solution at 60°C overnight. DNA was quantified following digestion using the Hoechst dye assay. 16 Total accumulated sulfated glycosaminoglycan (GAG) and hydroxyproline were quantified by dimethylmethylene blue 17 and dimethylamino benzaldehyde dye 18 binding assays, respectively. ECM accumulation data were normalized to the sample wet weight or DNA.
Immunohistochemistry and Histology
Samples from 15 days culture were fixed in 10% formalin for 48 hours, paraffin-embedded, sectioned, and mounted on slides. Sections were deparaffinized and rehydrated prior to staining.
Type II Collagen Immunohistochemical Staining
Samples were incubated with proteinase K (Sigma-Aldrich, Saint Louis, MO) at 37°C for 15 minutes, and then mouse anti-collagen type II IgG primary antibody using undiluted supernatant (Hybridoma Bank, Iowa City, IA) followed by donkey anti-mouse IgG secondary antibody conjugated with peroxidase at a 1:500 dilution (Jackson Immunoresearch, West Grove, PA). Antibody detection was performed using VECTOR NovaRED (Vector laboratories, Burlingame, CA). Additional sections were incubated with normal mouse serum at equal concentration to that of the primary antibody as a negative control. Equine cartilage was analyzed in parallel as a control.
Toluidine Blue
Sections were stained with 0.04% toluidine blue solution (Electron Microscopy Sciences, Fort Washington, PA) to detect the accumulation of sulfated pro-teoglycans.
Prostaglandin E2 Levels
Medium from chondrogenic cultures was collected on days 1, 3, 6, 9, 12, 15, 18, and 21, stored at −20°C, and then analyzed for PGE2 concentration using a commercially available enzyme-linked immunosorbent assay kit (Enzo Life Sciences, Farmingdale, NY). PGE2 secretion was normalized to the sample wet weight or DNA.
Alkaline Phosphatase
Medium from chondrogenic cultures was evaluated for alkaline phosphatase activity. Media samples were incubated with SIGMA
RNA Extraction and Real-Time PCR
Mesenchymal stem cells–seeded agarose samples were collected on days 3, 6, 9, and 15 in TRIzol reagent (Life Technologies, Grand Island, NY) and stored in −80°C. Samples were removed from TRIzol, frozen in liquid nitrogen, and pulverized. TRIzol was first added to the pulverized samples, followed by chloroform at a ratio of 1:5 to the TRIzol. Samples were centrifuged at 12,000
Primer and Probe Sequences.

Experimental Design
The effects of Dex concentration on MSC chondrogenesis was evaluated by culturing samples in chondrogenic medium containing a maximum of 100 nM Dex, 5 which is the most commonly used concentration for supporting bone marrow MSC chondrogenesis. From 100 nM, we evaluated the effect of reducing the concentration of Dex by comparing to 1 nM Dex or Dex-free culture. Extracellular matrix accumulation was evaluated after 15 days of culture, while PGE2 secretion was quantified for up to 21 days of culture. The contribution of cyclooxygenase-2 (COX-2) to PGE2 secretion and chondrogenesis was evaluated using celecoxib (Sigma-Aldrich, Saint Louis, MO), a nonsteroidal anti-inflammatory drug that selectively inhibits COX-2. 19 Experiments evaluating the effects of timing of administration of Dex were performed using 1 or 100 nM Dex. The effects of Dex withdrawal were evaluated by removing Dex from the culture medium after 1, 3, or 6 days of a 15-day culture period. For the Dex withholding study, 1 nM Dex was added to the culture medium after 1, 2, or 3 days, or 100 nM Dex was added to the culture medium after 3, 4, 5, or 6 days of a 15-day culture period.
Statistical Analysis
Data were analyzed for analysis of variance with mixed model using animal as a random effect. Pairwise comparisons were analyzed using least squares means with Tukey-Kramer adjustment.
Results
Preliminary Studies
Two experiments were conducted with a small sample size and limited analysis to serve as a basis for designing the primary experiments in this study. First, we evaluated the effect of decreasing 100 nM Dex over several orders of magnitude. When normalized to wet weight, GAG accumulation in 100 nM Dex was not significantly different from 10, 1, or, 0.1 nM Dex (
Dexamethasone Titration
Comparisons among 0, 1, and 100 nM Dex were conducted using MSC from 8 horses, with ECM accumulation and histology analyzed after 15 days of culture.
ECM accumulation
When normalized to wet weight, GAG (
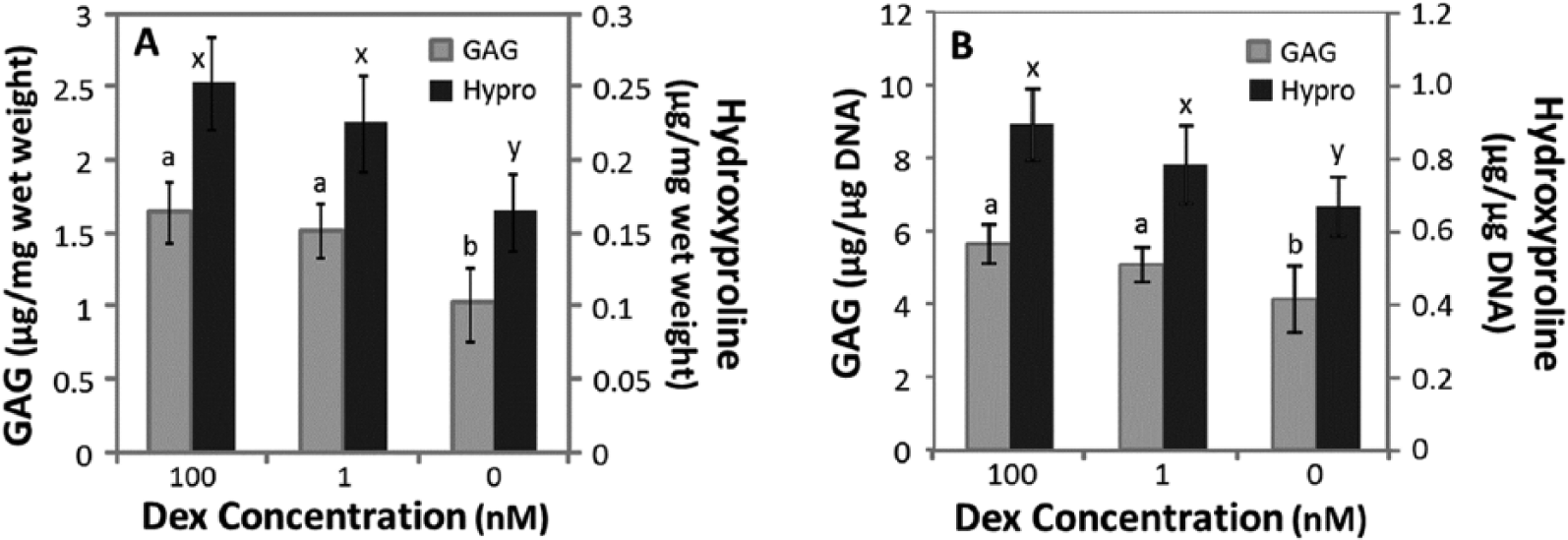
Glycosaminoglycan (GAG) and hydroxyproline (Hypro) accumulation after 15 days of culture in chondrogenic medium containing 100, 1, or 0 nM dexamethasone (Dex). (
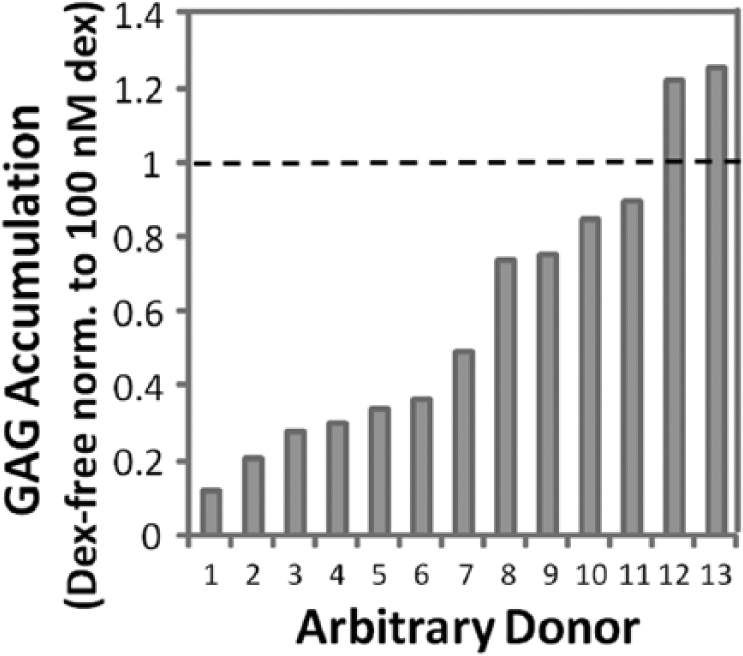
Glycosaminoglycan (GAG) accumulation normalized to wet weight from 13 horses after 15 days of culture without dexamethasone (Dex) or with 100 nM Dex. Data from cultures without Dex were normalized to 100 nM Dex for each individual horse.
Histology
Type II collagen (Fig. 3A) and toluidine blue staining (Fig. 3B) was present in all conditions from 15-day culture samples. Grossly, the distribution of both stains appeared similar between 1 and 100 nM Dex, with the most prominent staining in pericellular spaces. Staining in Dex-free cultures was largely localized to pericellular spaces. In Figure 3B, toluidine blue staining for Dex-free samples for which GAG accumulation was relatively high (1.29 μg/mg wet weight, Fig. 3B-i) or low (0.27 μg/mg wet weight, Fig. 3B-ii) are presented. Decreasing GAG accumulation in Dex-free cultures was associated with fewer cells that were surrounded by robust pericellular toluidine blue staining.
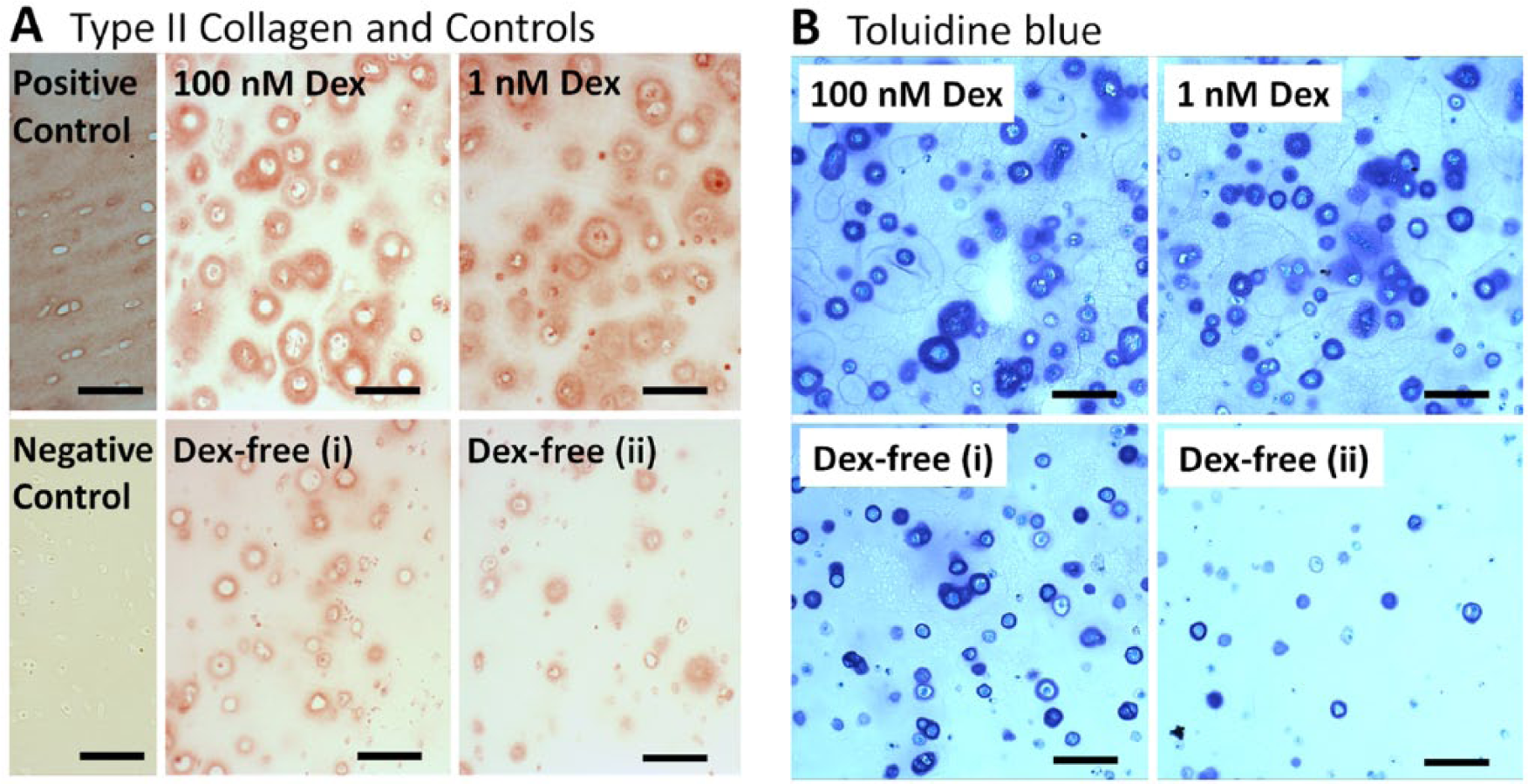
Representative staining of mesenchymal stem cells (MSCs) encapsulated in agarose gel cultured in 100, 1, and 0 nM dexamethasone (Dex) after 15 days of culture. (
Gene Expression
Gene expression was evaluated for MSCs from 5 horses after 3, 6, 9, and 15 days of culture (Fig. 4A-G). In Figure 4A, expressions of each gene in 100 nM Dex culture were normalized to mean expression of day 3 and in Figure 4B-G, expressions in Dex-free and 1 nM Dex cultures were normalized to 100 nM Dex at each time point.
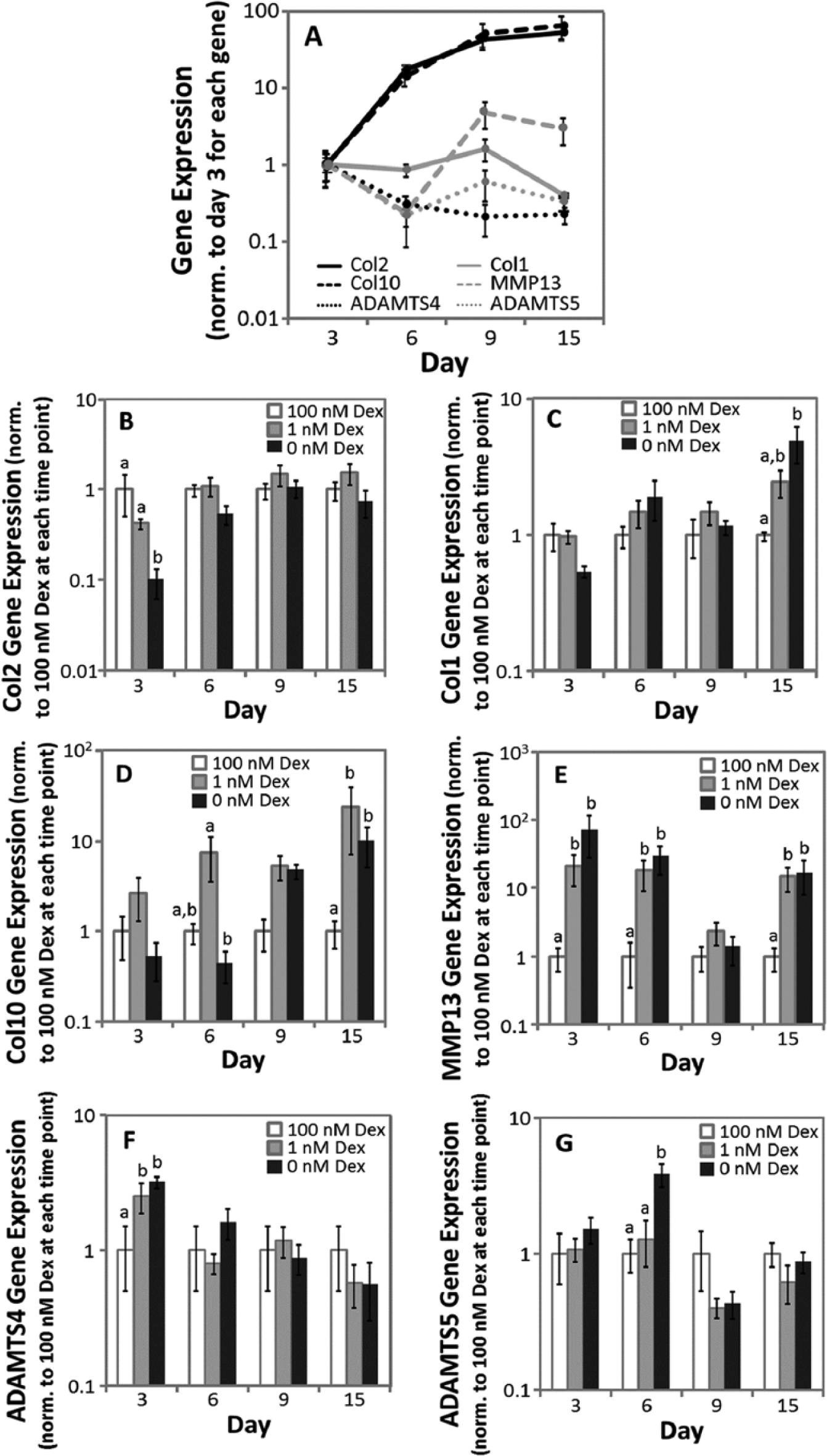
Gene expression from days 3, 6, 9, and 15 of cultures. (
Collagen
Type II and I Collagen
In 100 nM Dex (Fig. 4A), type II collagen expression increased with time in culture, with an overall 52-fold upregulation between days 3 and 15. Type I collagen expression in 100 nM Dex did not change with time in culture between days 3 and 9 (
Type X Collagen
In 100 nM Dex (Fig. 4A), type X expression increased 13.9-fold between days 3 and 6 but was not significantly different for the rest of the time course. The temporal pattern of increasing type X collagen expression with time in culture was consistent with previous reports for human MSCs in pellet culture. 20 When considering relative expression among conditions at each time point (Fig. 4D), Dex-free and 1 nM Dex cultures were not significantly different from 100 nM through 9 days of culture, although on day 6 type X collagen expression in 1 nM Dex was 16.6-fold higher than Dex-free cultures. On day 15, type X collagen expression in Dex-free and 1 nM Dex cultures was 9.9- and 24-fold, respectively, higher than 100 nM Dex.
Matrix Metalloproteinase
In 100 nM Dex (Fig. 4A), MMP13 expression did not change with time in culture (
A Disintegrin and Metalloproteinase with Thrombospondin Motifs
In 100 nM Dex (Fig. 4A), ADAMTS4 expression decrease with time in culture with an overall 4.5-fold down-regulation between days 3 and 15, while ADAMTS5 expression did not change with time in culture (
Alkaline Phosphatase
Alkaline phosphatase activity was evaluated for MSCs from 5 horses. Culture media was analyzed on days 3, 6, 9, and 15, and in Figure 5 the results were normalized to the wet weight of the samples. In 100 nM Dex, alkaline phosphatase activity increase for 1.7-fold between day 3 and day 6, and did not change with time for the remainder of the culture period (
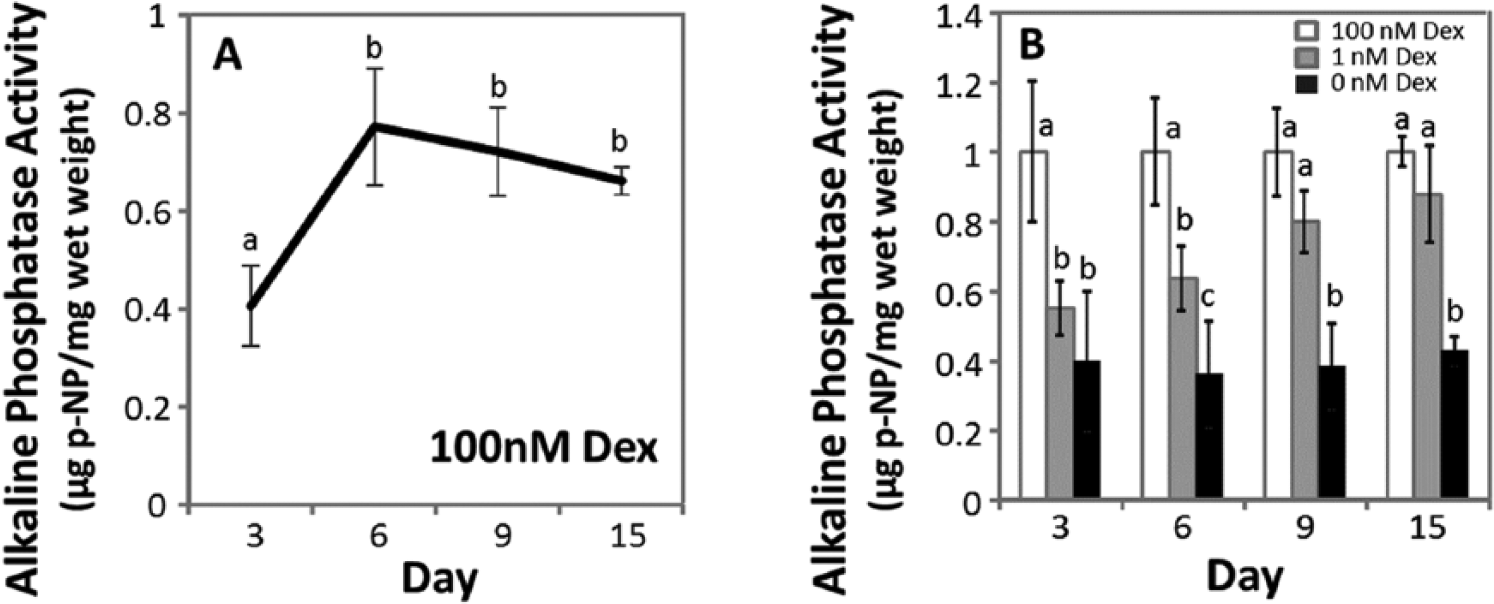
Alkaline phosphatase activity. (
Prostaglandin E2
Prostaglandin E2 secreted into the culture medium was quantified between 1 and 21 days of culture for MSCs from 6 horses. In Figure 6, the rate of PGE2 secretion was calculated by dividing the total amount of PGE2 in the medium by the time elapsed since the previous medium change, which was 3 days for all time points except for day 1. The rate of PGE2 secretion was normalized to the wet weight of the sample. In 100 nM Dex (Fig. 6A) the rate of PGE2 accumulation in the medium was initially low, then increased more than 200-fold by days 3 and 6 of culture. Between days 6 and 9, the rate of PGE2 secretion decreased approximately 30-fold, and did not significantly change for the remainder of the timecourse (
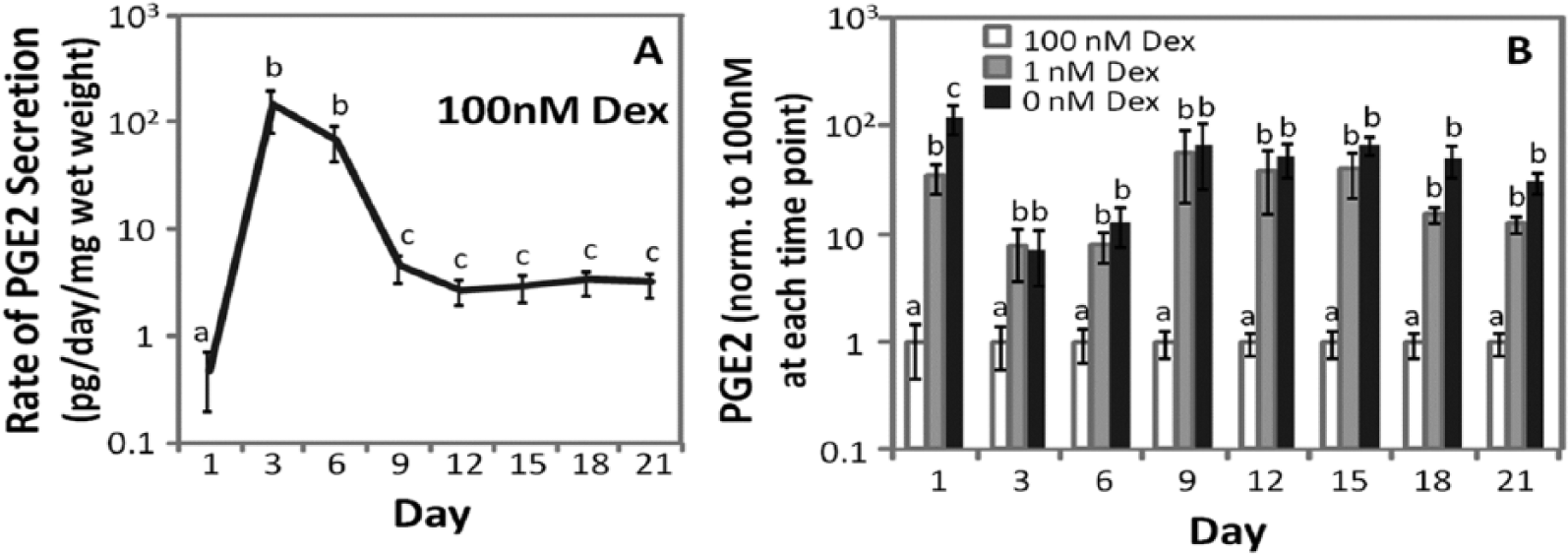
Prostaglandin E2 (PGE2) secretion. (
Extracellular matrix accumulation normalized to sample wet weight on day 21 resembled that of Figure 1 as GAG (
Celecoxib
Celecoxib was evaluated at a concentration of 10 μM, with a final dimethyl sulfoxide (DMSO) concentration of 0.025%, for MSCs from 3 horses. Control cultures were supplemented with 0.025% DMSO. All cultures were maintained in 1 nM Dex. When normalized to wet weight, GAG accumulation in celecoxib cultures (2.24 μg/mg wet weight) was not significantly different from controls (2.04 μg/mg wet weight,
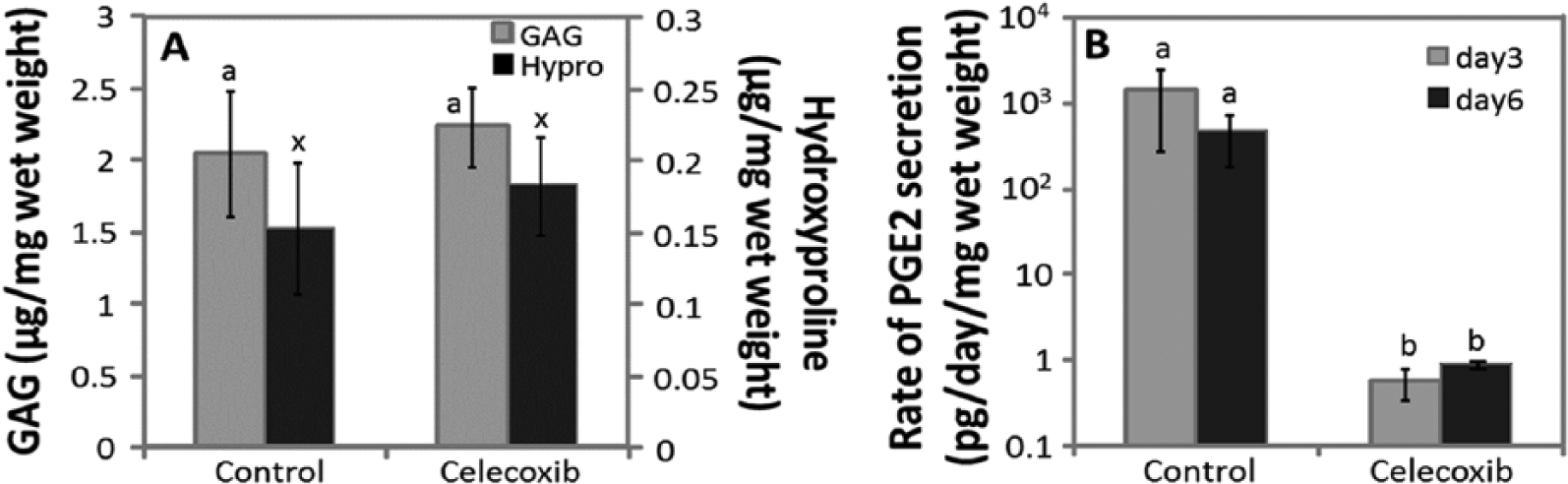
Extracellular matrix (ECM) accumulation and prostaglandin E2 (PGE2) secretion in the presence of 10 µM celecoxib. (
Timing of Dex Exposure
The effects of withdrawing Dex from chondrogenic culture over time, or temporarily withholding Dex from the start of chondrogenic culture, were tested using a Dex concentration of 1 or 100 nM over 15-day culture period. In both experiments, control cultures were maintained in the presence or absence of Dex for 15 days. ECM accumulation in control cultures were consistent with Figure 1 as GAG and hydroxyproline in 1 and 100 nM Dex was higher than in Dex-free cultures (Fig. 8A-C). In Figure 8, data were normalized to sample wet weight, which resulted in comparable statistical outcomes when normalized to DNA.
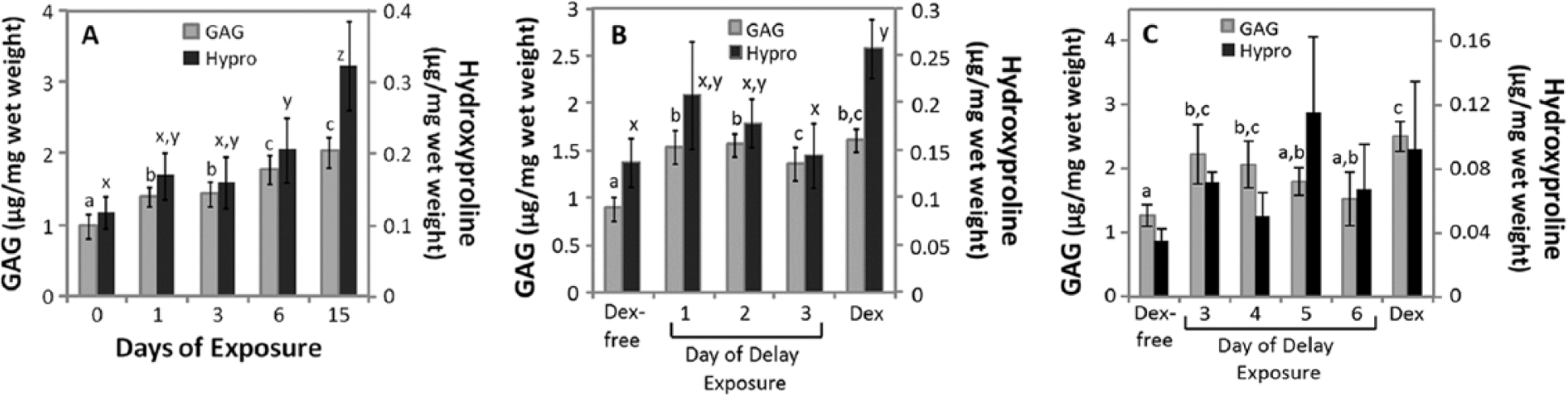
Glycosaminoglycan (GAG) and hydroxyproline (Hypro) accumulation after 15 days of culture with withdrawing or withholding dexamethasone (Dex) during chondrogenic culture. (
Dex Withdrawal
Dex was withdrawn after 1, 3, or 6 days of 15-day culture period. In 1 nM Dex culture, withdrawing Dex after 1 and 3 days resulted in approximately 70% and 51% in GAG and hydroxyproline accumulation relative to controls that received Dex for 15 days (Fig. 8A, n = 7). Withdrawing Dex after 6 days of exposure resulted in a significant decrease in hydroxyproline, but not GAG (
Dex Withholding
In medium containing 1 nM Dex, withholding Dex for up to 3 days did not significantly suppress GAG accumulation relative to the Dex control culture (
Histology
For both experiments, staining for type II collagen and toluidine blue was present in all conditions, with ECM accumulating largely in pericellular spaces as in Figure 3A and
Discussion
Withholding Dex from chondrogenic cultures of adult equine MSCs resulted in a decrease in ECM accumulation of ~40% compared with 100 nM Dex, which was consistent with previous studies involving chondrogenesis of bovine MSCs,6-8 human bone marrow MSCs in pellet culture,
5
and human adipose-derived stromal cells cultured in alginate hydrogel.
21
Furthermore, the modest increase in type I collagen expression in Dex-free samples on day 15 may suggest greater fibrocartilage-like differentiation in the absence of Dex with time in chondrogenic culture. The effects of withhold Dex was variable across donors in that MSCs from certain donors were minimally affected, while others accumulated only modest amounts of GAG in the absence of Dex. Donor-to-donor variability may be a concern for autologous human therapies as chondrogenesis has been shown to vary greatly among donors
Suppression of ECM accumulation in Dex-free culture was reversed with 1 nM Dex. Furthermore, measures of cartilage-like phenotypic expression were not significantly different between 1 and 100 nM Dex as both cultures showed robust staining for type II collagen and toluidine blue, while gene expression of type I and II collagen were not significantly different. These data indicate that Dex concentrations may be reduced 100-fold from the conventional laboratory dose of 100 nM while still supporting robust MSC chondrogenesis, which is consistent with reports that Dex concentrations less than 100 nM stimulated robust chondrogenesis in multipotent rat calvaria cells, 23 and sox-9 expression in chondrocytes. 24
While Dex-free culture resulted in moderate suppression of ECM accumulation, collagen gene expression did not convincingly differentiate between Dex and Dex-free cultures as type II collagen expression in Dex-free culture was significantly different than 1 or 100 nM Dex on day 3 only. While these data may indicate that Dex enhances the rate of differentiation during early chondrogenesis, it is not known whether a lag in type II collagen expression during a period of low ECM synthesis 20 is sufficient to account for the large discrepancies in ECM accumulation between Dex and Dex-free conditions after 15 days of culture. Furthermore, given that gene expression does not necessarily translate to protein synthesis, as documented for aggrecan during MSC chondrogenesis,15,25 it is possible that posttranslational regulation of ECM synthesis may account for the relative low accumulation of GAG and hydroxyproline in the absence of Dex.
A second possibility is that Dex acts to support ECM accumulation through potent anti-inflammatory properties that suppress catabolism in chondrogenic culture. This concept is supported by studies reporting increased aggrecanase activity when Dex was withheld in chondrogenic bovine MSC cultures,
8
and MMP cleavage of aggrecan in human MSC cultures maintained in chondrogenic medium containing 100 nM Dex.
7
When considering gene expression of catabolic enzymes, decreasing or withholding Dex resulted in modest (ADAMTS4) or moderate (MMP13, MMP1) upregulation of gene expression at early timepoints, and moderate upregulation of MMP13 on day 15, although minimal differences between 1 nM Dex and Dex-free cultures on day 3 does not strongly support the differences in ECM accumulation between these 2 groups. As a second measure of inflammation, we measured PGE2 secretion, which when induced from activation of COX-2 has been associated with degradation in osteoarthritic cartilage,
26
and cartilage when cultured with pro-inflammatory cytokines
An ongoing concern about the use of MSCs to resurface articular cartilage defects is the propensity of MSCs to acquire markers of hypertrophy during chondrogenesis. Hypertrophic markers have been identify over time in MSC chondrogenic culture in 100 nM Dex,
30
while reducing the concentration of Dex in culture has been used to promote hypertrophy.
31
In Dex-free culture in the current study, upregulation of MMP13 and type X collagen relative to 100 nM Dex may suggest advanced hypertrophic differentiation in the absence of Dex. One nanomolar Dex did not suppress these markers of hypertrophy, and in fact increased type X collagen expression over Dex-free cultures on day 6. Alkaline phosphatase activity did not suggest increased hypertrophy with reduction or elimination of Dex, although these data are consistent with human MSC pellet cultures in which alkaline phosphatase activity was modestly lower in Dex-free culture relative to 100 nM Dex.
5
By day 15 of chondrogenic culture, moderate levels of alkaline phosphatase secretion may be expected,30,32 although secretion may increase significantly with additional time in culture.30,32,33 In addition, beyond 15 days of chondrogenic culture it has been shown that medium conditions that promote hypertrophy can further increase alkaline phosphatase secretion up to 3-fold.
33
Therefore it is possible that additional time in culture, over which alkaline phosphatase secretion may increase with concomitant increases in type I and X gene expression,
30
would better delineate the effect of Dex concentration on hypertrophic markers. While additional assays are necessary to more thoroughly characterize commitment to hypertrophy, the gene expression data suggest that high levels of Dex may be beneficial to reduce differentiation to this undesirable phenotype. It is possible that Dex influences hypertrophic differentiation through the regulation of COX-2 as suppression of hypertrophic markers has been associated with COX-2 inhibition during chondrogenesis
Given that the temporal effects of Dex exposure on MSC chondrogenesis have not been reported, we performed experiments in which Dex was withheld or withdrawn to determine how the timing of exposure influence chondrogenesis. At a concentration of 1 nM, Dex could be withheld for up to two days without significantly affecting the accumulation of ECM, while withdrawal studies suggested that Dex plays an important role in ECM accumulation beyond day 6. A similar temporal pattern was noted for 100 nM Dex, although the high variability in hydroxyproline accumulation suggest that a larger samples size is necessary to better delineate the effect of withholding or withdrawing 100 nM Dex. Taken together, these data suggest that strategies for delivering Dex
When considering the design of delivery strategies for Dex to support MSC chondrogenesis
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by College Research Council (CRC) of Colorado State University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study used cells from bone marrow that had been previously obtained using a university-approved animal care and use protocol.
