Abstract
Objective:
The purpose of this study was to analyze the morphological imaging characteristics and incorporation of TruFit bone graft substitute (BGS) plugs using cartilage-sensitive magnetic resonance imaging (MRI) and quantitative T2 mapping.
Design:
Twenty-six patients (mean age, 28.72 years; range, 11-56 years) underwent osteochondral autologous transplantation (OATS) for chondral defects with filling of the knee joint donor sites using Trufit BGS plugs. The mean follow-up interval between implantation and MRI analysis was 21.3 months (range, 6-39 months). During this period, 43 cartilage-sensitive and 25 quantitative T2-mapping MRI studies were performed. The donor sites were assessed for plug and interface morphology, displacement, hypertrophy, subchondral edema, presence of bony overgrowth, percentage fill, and degree of incorporation. T2 relaxation times were measured for the superficial and deep layers of the repair tissue. A linear regression and correlational analysis was performed with Bonferroni correction, and P < 0.05 was defined as significant.
Results:
Longitudinal analysis revealed favorable plug appearance at early follow-up (≤6 months), with 75% of plugs demonstrating flush morphology and 78% demonstrating near complete to complete fill. Plug appearance deteriorated at intermediate follow-up (~12 months), with only 26% of plugs demonstrating flush morphology and 52% with near complete or complete fill. Plug appearance substantially improved with longer follow-up (≥16 months), with 70% of plugs demonstrating flush morphology and 90% demonstrating near complete or complete fill. Interface resorption was common at ~12 months (P < 0.0001) and was associated with older age (P = 0.01) or a single-plug configuration (P = 0.04). T2 values for the repair cartilage approached that of normal cartilage with increasing duration after surgery (P < 0.004), more so for single- compared with multiple-plug configurations (P = 0.03).
Conclusions:
The Trufit BGS plug demonstrates a predictable pattern of postoperative maturation on MRI images that parallels its biological incorporation. An intermediate postoperative interval can be associated with unfavorable MRI findings. However, the plug appearance significantly improves with greater postoperative duration and has mean T2 relaxation times that approach those of normal articular cartilage.
Introduction
The treatment of articular cartilage defects continues to be one of the most challenging clinical problems for orthopedic surgeons. Articular cartilage is a highly organized, complex tissue with distinct biomechanical properties and substantial durability. It has poor intrinsic capacity for healing, however, and damage from trauma or degeneration can result in significant morbidity. 1-6 Untreated chondral lesions can lead to debilitating joint pain and dysfunction. 1-6
Biodegradable scaffolds offer a novel therapeutic strategy for articular cartilage repair. Synthetic scaffolds can have a number of favorable characteristics, including mechanical properties and a biphasic design that matches the modulus of adjacent native cartilage and subchondral bone. 7-19 Furthermore, scaffolds can be used alone or as delivery vehicles for cells, mitogens, and chondrogenic growth factors. 7-19
The TruFit bone graft substitute (BGS) plug (Smith & Nephew Endoscopy, San Antonio, TX) is a synthetic, polylactide-co-glycolide (PLG)–based implant that is commercially available for filling bone void defects. PLGs are often chosen for cartilage tissue engineering applications as their ability to support chondrocytes and the production of a cartilage extracellular matrix is well documented. 20-25 Currently, the TruFit BGS plug is approved only for the fill of bone void lesions and can be used to fill donor bone voids during an autologous osteochondral transfer procedure. Although there is a paucity of clinical outcome studies, animal studies have demonstrated excellent tissue fill, host incorporation, and the formation of a hyaline-like cartilage repair tissue with more favorable histological and biomechanical characteristics compared with fibrocartilage. 26,27 The purpose of this study was to analyze the morphological characteristics and incorporation of TruFit BGS plugs used for a backfill indication (surrounded by “normal” articular cartilage) using cartilage-sensitive magnetic resonance imaging (MRI) and quantitative T2 mapping, the latter of which has been shown to correlate to collagen orientation in the extracellular matrix. 19,28
Methods
This study was approved by the Institutional Review Board at the Hospital for Special Surgery. Twenty-six patients (mean age 28.72 years; range, 11-56 years) underwent osteochondral autologous transplantation (OATS) for chondral defects of the knee (n = 21) or talus (n = 5). All donor sites were filled with Trufit BGS plugs. Sixteen patients had OATS for knee chondral defects, 5 for osteochondritis dissecans, and 5 for talar osteochondral defects. Forty-three cartilage-sensitive MRI studies and 25 T2-mapping MRI studies were performed at various postoperative intervals (range, 6-39 months; mean, 21.3 months). All donor sites were localized to the medial or lateral trochlear margin, with a mean of 2 plugs (range, 1-5) used per knee. Typically, donor sites were at least 3 mm in distance from adjacent sites along the trochlear ridge. The average plug size was 9 mm (range, 6-11 mm).
All MRI images were obtained on a clinical 1.5T or 3T MRI unit (Signa HDx, General Electric Healthcare, Milwaukee, WI) using a standard receive-only 8-channel phased array or quadrature knee coil (InVivo extremity coil; InVivo Corporation, Orlando, FL). Cartilage-sensitive morphologic MR images were obtained using a previously validated fast-spin echo pulse sequence. 29,30 Coronal, axial, and sagittal fast-spin echo images were performed using a repetition time (TR) of 3500 to 6000 ms, echo time (TE) of 28 (3T) to 34 (1.5T) ms (effective), field of view (FOV) of 14 (axial) to 16 (sagittal) cm, matrix of 512 × 320-416, slice thickness of 3 to 3.5 mm with no gap, receiver bandwidth (over the entire frequency range) of 31.25 kHz at 1.5T and 62.5 kHz at 3T, at 2 excitations. An additional fat-suppressed fast-spin echo sequence (Chemsat; General Electric Healthcare) was obtained in the sagittal plane using an effective TE of 45 ms, matrix of 256 × 224, and slice thickness of 4 mm with no gap. Echo train length varied between 6 and 14. The cartilage morphological characteristics were evaluated using a previously reported series of imaging parameters, including signal intensity of the repair cartilage relative to the surrounding cartilage (hypointense, isointense, or hyperintense, measured using a standardized region of interest [ROI] in the center of the repair with a standard of deviation on an MRI workstation), gross appearance (depressed, flush, or proud), the presence or absence of hypertrophy or displacement, subchondral edema (mild [<1 cm2], moderate [1-2 cm2], or severe [>2 cm2]), bony overgrowth (absence or presence), interface with adjacent cartilage (absence, presence, size of fissure [<2 mm or >2 mm]), percentage of fill based on both coronal and sagittal images (0%-33%, 34%-66%, or 67%-100%), integrity of adjacent cartilage (modified International Cartilage Repair Society [ICRS] classification), integrity of opposing cartilage (modified ICRS classification), fat pad scarring (mild, moderate, or severe), and synovial reaction (none, mild, moderate, severe). 31,32 Plug incorporation into the native trabecular architecture was graded as poor, partial, or complete. The cartilage pulse sequences used to evaluate these repairs has been previously validated based on a suitable standard of arthroscopy 29 and were applied to several preclinical models of cartilage repair with histologic validation. 11
Quantitative T2 mapping was performed using a multislice, multiecho, modified Carr Purcell Meiboom Gill pulse sequence, which uses interleaved slices and tailored refocusing pulses to minimize contribution from stimulated echoes. 33,34 Standard pulse sequence parameters used were a TR of 800 to 1000 ms, 8 echoes sampled using sequential multiples of the first TE (9-10 ms), FOV of 16 cm2, matrix of 256 to 384 × 256, providing a minimum in-plane resolution of 254 µm in the frequency direction by 312 µm in the phase direction, by slice resolution of 2.0 to 3.0 mm with no gap, and a receiver bandwidth of 31.25 (1.5T) to 62.5 (3T) kHz. After image acquisition, data sets were analyzed on a pixel-by-pixel basis with a 2-parameter weighted least-squares fit algorithm, assuming a monoexponential fit (Functool 3.1; General Electric Healthcare). Quantitative T2 relaxation times or values were calculated by taking the natural logarithm of the signal decay curve in a selected ROI. The ROIs were obtained in a standardized fashion from the articular cartilage over the center and periphery of the plug, both within the deep and superficial 50% of the plug, at the plug-native cartilage interface, as well as of the adjacent and opposite articular cartilage surfaces. Care was taken not to sample close to the tidemark/subchondral plate to avoid partial volume effects of sampling any misregistration due to residual chemical shift. Image assessment was performed by 2 senior musculoskeletal radiologists (L.F.F. and H.G.P.) who had no knowledge of the patient or the treating surgeon. T2-mapping software has also been validated based on histology using polarized light microscopy. 28
A linear regression and correlational analysis was completed with Bonferroni correction and P < 0.05 defined as significant.
Results
Analysis of the TruFit BGS plugs on MR images in multiple planes on all sequences revealed no evidence of plug displacement or hypertrophy at the donor sites for all knees across all time points. Fat pad scarring was minimal or mild in all cases. Similarly, no evidence of a inflammatory synovitis or effusion was appreciated on any images throughout the period of study ( Fig. 1 ).
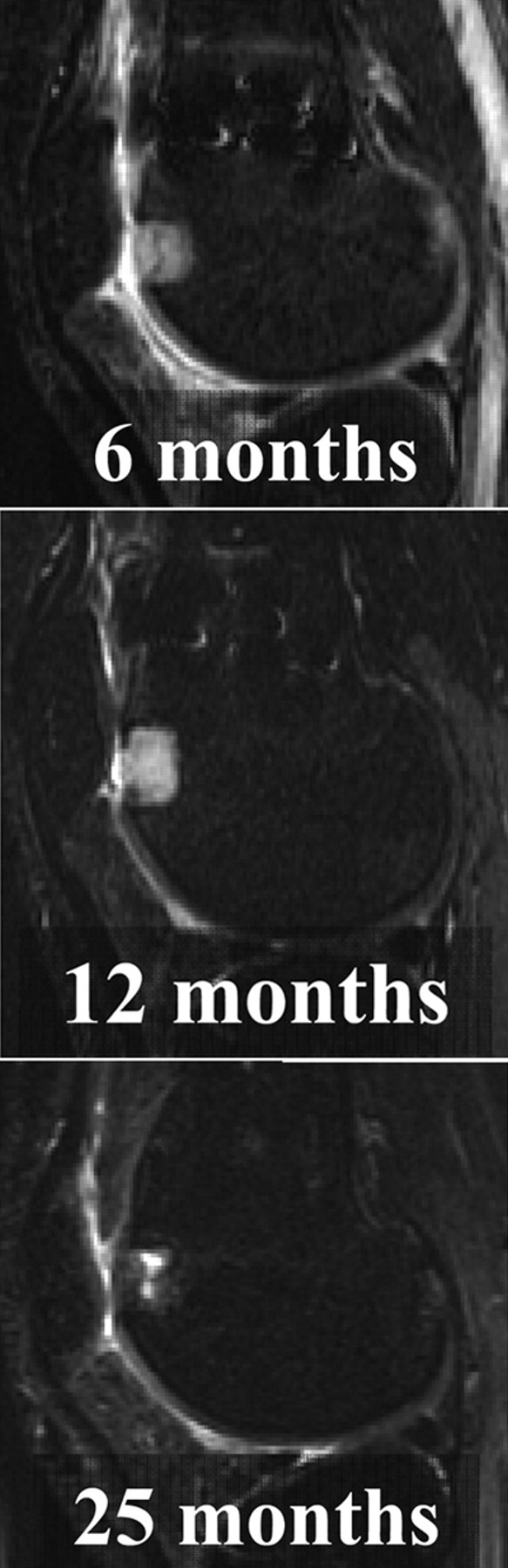
Sagittal fat-suppressed cartilage-sensitive, fast-spin echo magnetic resonance images of a TruFit bone graft substitute plug at 6 months, 12 months, and 25 months postoperatively demonstrates absence of adjacent bone marrow edema pattern, at even 6 months postoperatively. Images also demonstrate no displacement, effusion, or an adverse synovial reaction.
TruFit BGS plugs demonstrated a favorable appearance on MR images in the early postoperative period (≤6 months). Seventy-five percent (33 of 46) of plugs demonstrated a flush morphology, and 78% (36 of 46) of plugs exhibited near complete or complete fill of the defect. Subchondral edema was rarely observed and occurred with minimal or mild severity in only 30% (14 of 46) of the plugs during this early postoperative period. No bony overgrowth was seen. Eighty percent of plugs, however, demonstrated a hyperintense signal compared with the adjacent articular cartilage. Furthermore, no plug demonstrated complete incorporation, with 64% (29 of 46) and 37% (17 of 46) demonstrating partial or poor incorporation into the native trabecular architecture, respectively (
Figs. 1
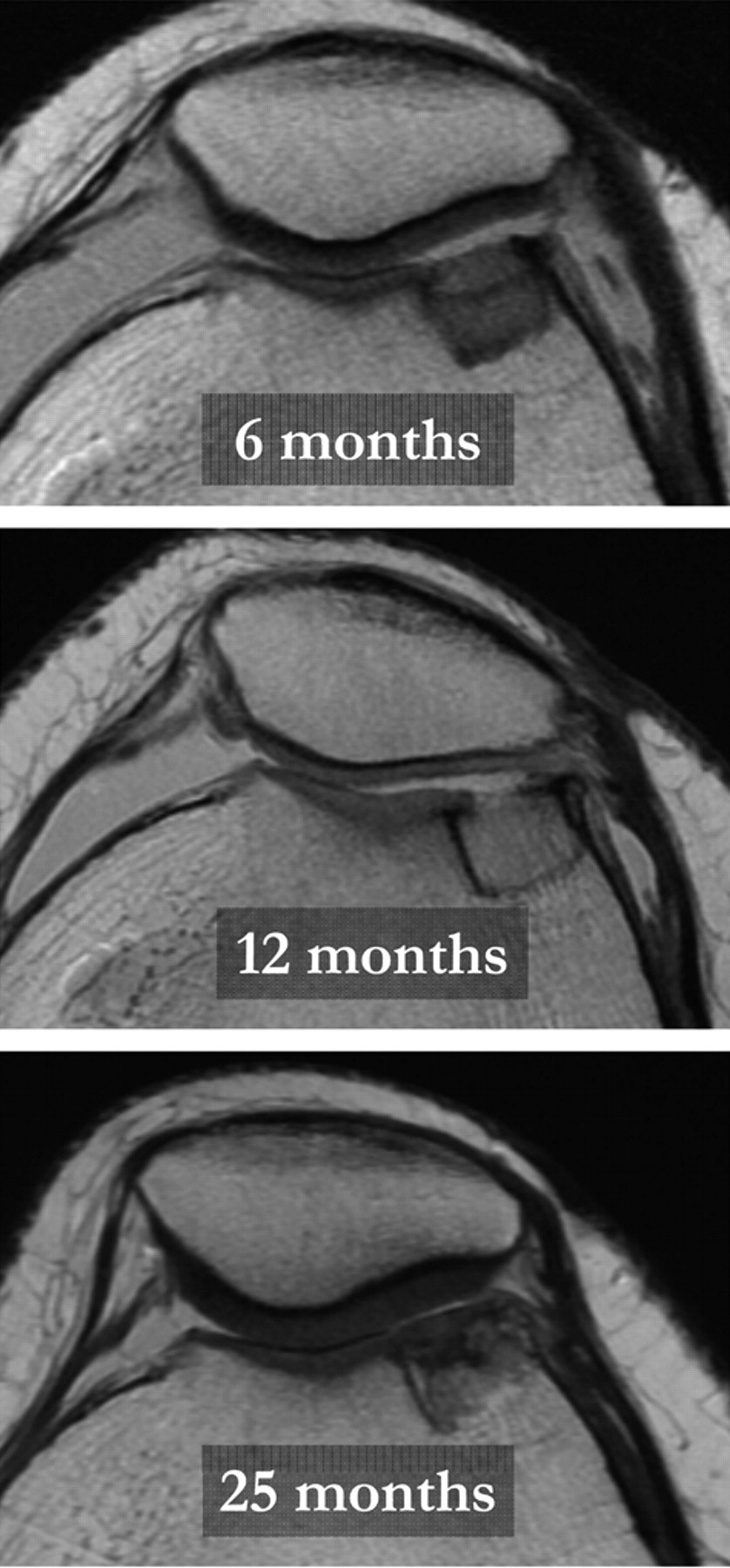
Axial cartilage-sensitive, fast-spin echo magnetic resonance images of a TruFit bone graft substitute plug at 6 months, 12 months, and 25 months postoperatively. Plugs demonstrate a favorable appearance in the early postoperative period, with greater than 75% of plugs demonstrating a flush morphology and near complete or complete fill of the defect. Plug appearance deteriorates by approximately 12 months postoperatively. Only 26% of plugs demonstrated a flush morphology, and only 52% of plugs exhibited near complete or complete fill of the defect. Plug appearance substantially improves, however, with longer postoperative duration (≥16 months). Seventy percent of plugs (32 of 46) demonstrated flush morphology, and 90% of plugs (40 and 46) exhibited near complete or complete fill.
TruFit BGS plug appearance deteriorated on MR images by approximately 12 months postoperatively. Only 26% of plugs (12 of 46) demonstrated a flush morphology, and only 52% (24 of 46) of plugs exhibited near complete or complete fill of the donor site defect. Resorption at the interface between the plug and native bone, defined by a larger measured diameter of the nonossified plug matrix compared with the known plug size, was common and affected 61% of plugs (28 of 46) at intermediate follow-up ( Fig. 3 ). Ninety percent of plugs (40 of 46) had hyperintense signal compared with the surrounding articular cartilage. Mild subchondral edema was observed below 15% of the plugs (7 of 46). No bony overgrowth was seen. Only 3% of the plugs demonstrated complete incorporation, while 83% (38 of 46) demonstrated partial incorporation into the native trabecular architecture ( Fig. 4 ).
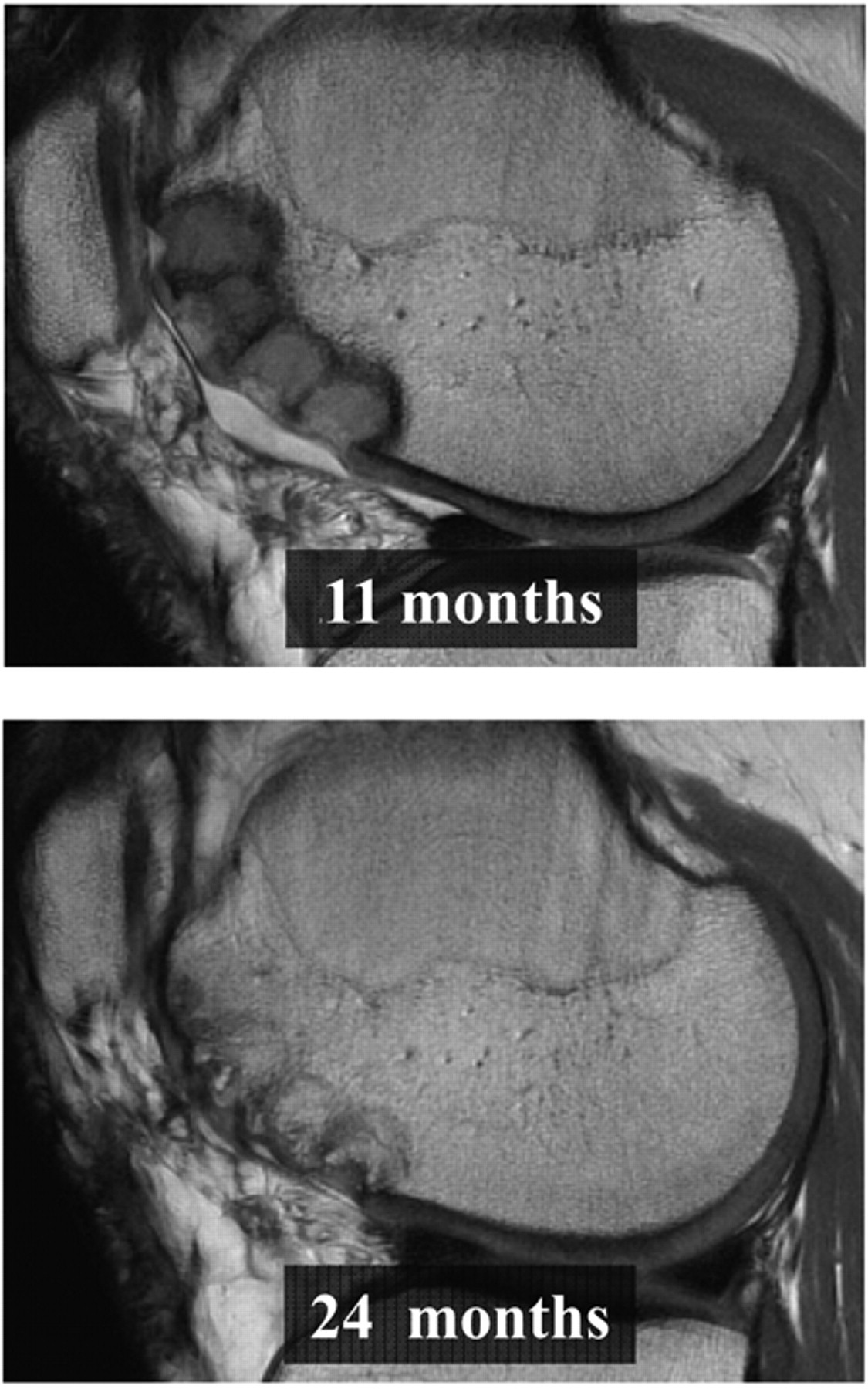
Sagittal cartilage-sensitive, fast-spin magnetic resonance images of 4 TruFit bone graft substitute plugs used to fill donor defects at 11 months and 24 months postoperatively. Mild bony overgrowth was observed and strongly correlated with a middle position in a multiple-plug configuration (P < 0.0001) and with postoperative duration greater than 16 months (P < 0.0001).
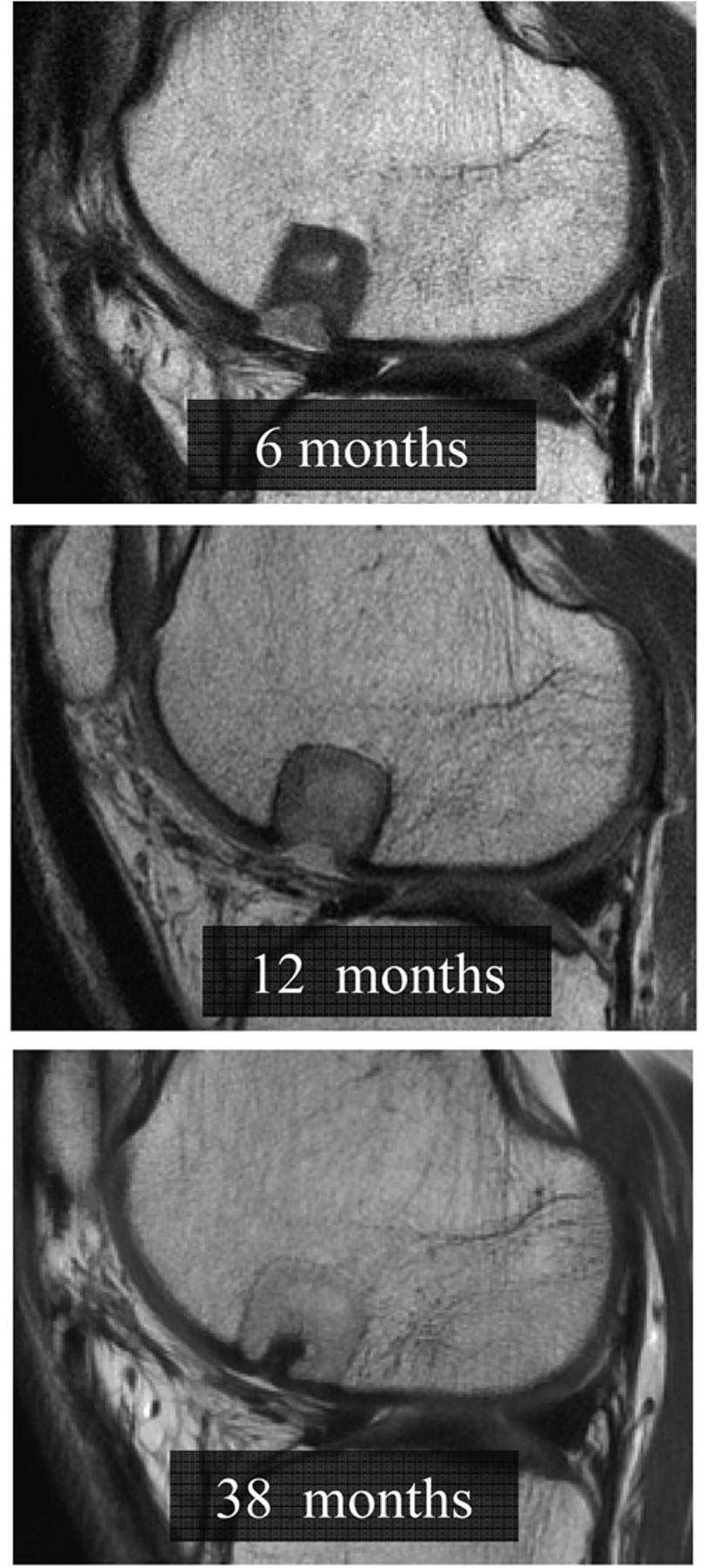
Sagittal cartilage-sensitive, fast-spin echo magnetic resonance images of a TruFit bone graft substitute plug (BGS) at 6 months, 12 months, and 38 months postoperatively. The appearance of the plug parallels its biological incorporation, demonstrating unfavorable characteristics during the phase of resorption and early incorporation at intermediate postoperative follow-up. Resorption at the plug-native bone interface was seen in greater than 60% of TruFit BGS plugs at approximately 12 months.
The MR appearance of TruFit BGS plugs substantially improved, however, with longer postoperative duration (≥16 months). Seventy percent of plugs (32 of 46) demonstrated flush morphology, and 90% of plugs (40 and 46) exhibited near complete or complete fill of the donor lesion. Isointense signal was appreciated in 44% of the plugs (20 of 46), and no subchondral edema was seen ( Figs. 1 - 4 ). Bony overgrowth was observed in 26% of the plugs (12 of 46) but was evident only in the setting of multiple plugs used to fill 3 or more donor lesions. Complete incorporation was observed in 64% of the plugs (29 of 46), while 26% of plugs (12 of 46) demonstrated progressing partial incorporation into host bone ( Fig. 4 ).
Linear regression and correlational analysis revealed important relationships between the measured MR parameters of the repair cartilage and preoperative, intraoperative, and postoperative variables. The presence of subchondral edema was uncommon but significantly associated with a short postoperative duration (P = 0.02). Abnormal, hyperintense signal of the repair tissue was associated with a short postoperative interval (P = 0.02) and multiple-plug configuration (P = 0.01; Figs. 1 - 4 ). Mild bony overgrowth was observed and strongly correlated with a middle position in a multiple plug configuration (P < 0.0001) and with postoperative duration greater than 16 months (P < 0.0001; Fig. 5 ). Depressed or proud plug morphology was associated with a short postoperative interval (P = 0.02) and large plug size (P = 0.003). Significant resorption at the plug-native bone interface was associated with older age (P = 0.01), intermediate postoperative duration (~12 months; P < 0.0001), and a single-plug configuration (P = 0.04; Fig. 3 ). Both incomplete fill of the defect and poor plug incorporation were associated with short to intermediate postoperative duration (P = 0.02 and P = 0.006, respectively). Poor ICRS scores of adjacent cartilage were significantly associated with the use of multiple plugs compared with a single plug to fill donor lesions (P = 0.01).
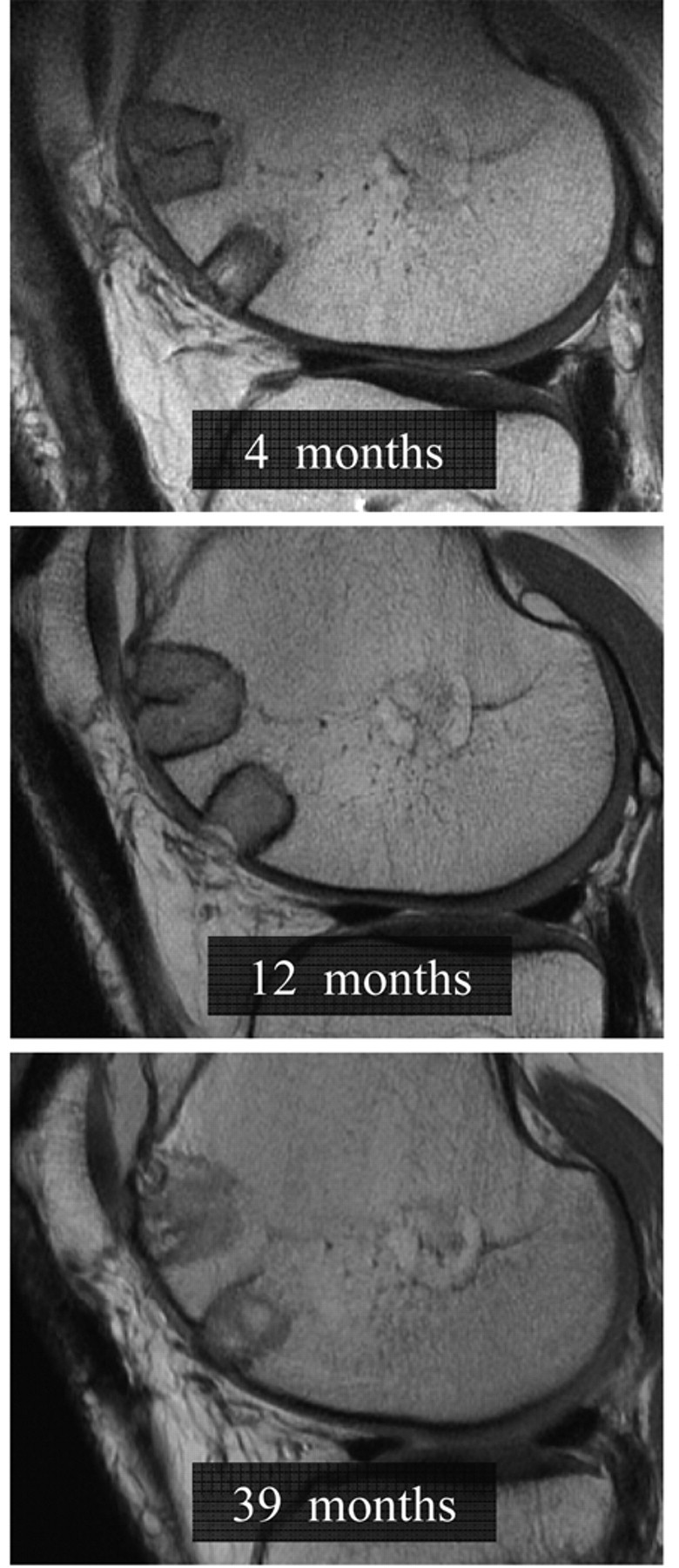
Sagittal cartilage-sensitive, fast-spin echo magnetic resonance images of a TruFit bone graft substitute plug at 4 months, 12 months, and 39 months postoperatively. Complete incorporation of the plug into host bone was observed in greater than 60% of cases at final follow-up (≥16 months).
T2 relaxation times (milliseconds) for the superficial and deep cartilage layers of the central and peripheral plug demonstrated significant shortening with increasing duration after surgery (P < 0.004;
Figs. 6
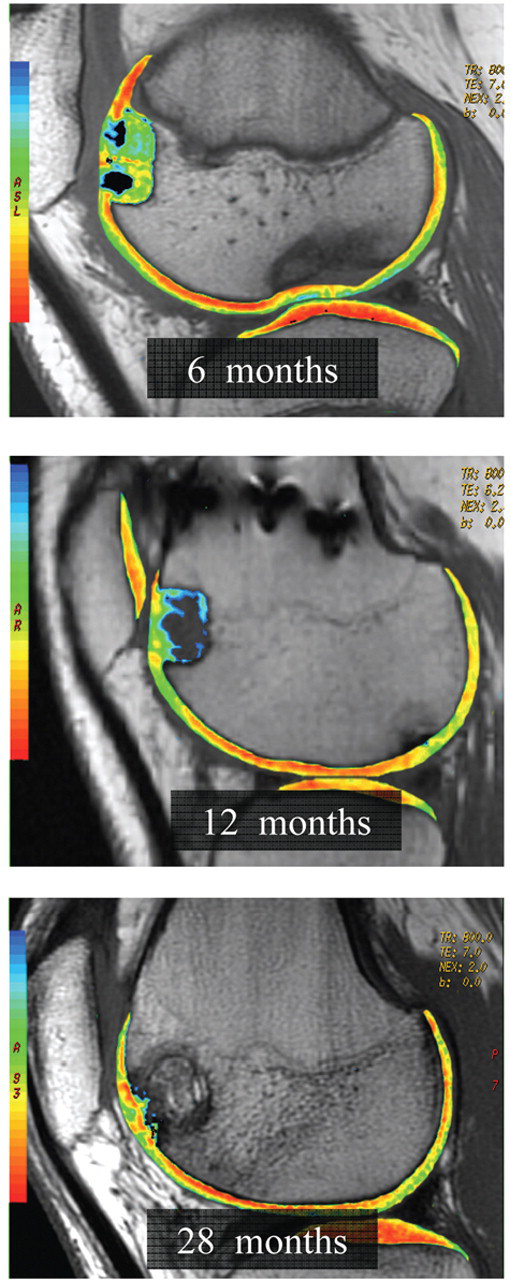
Sagittal quantitative T2-mapping of a TruFit bone graft substitute plug used to fill a donor void at 6 months, 12 months, and 28 months postoperatively. T2 relaxation times (milliseconds) for the superficial and deep cartilage layers of the central and peripheral plug demonstrated significant shortening with increasing duration after surgery (P < 0.004). More stratified T2 repair tissue with shorter values appears at the area of peripheral integration rather than the subchondral repair tissue.
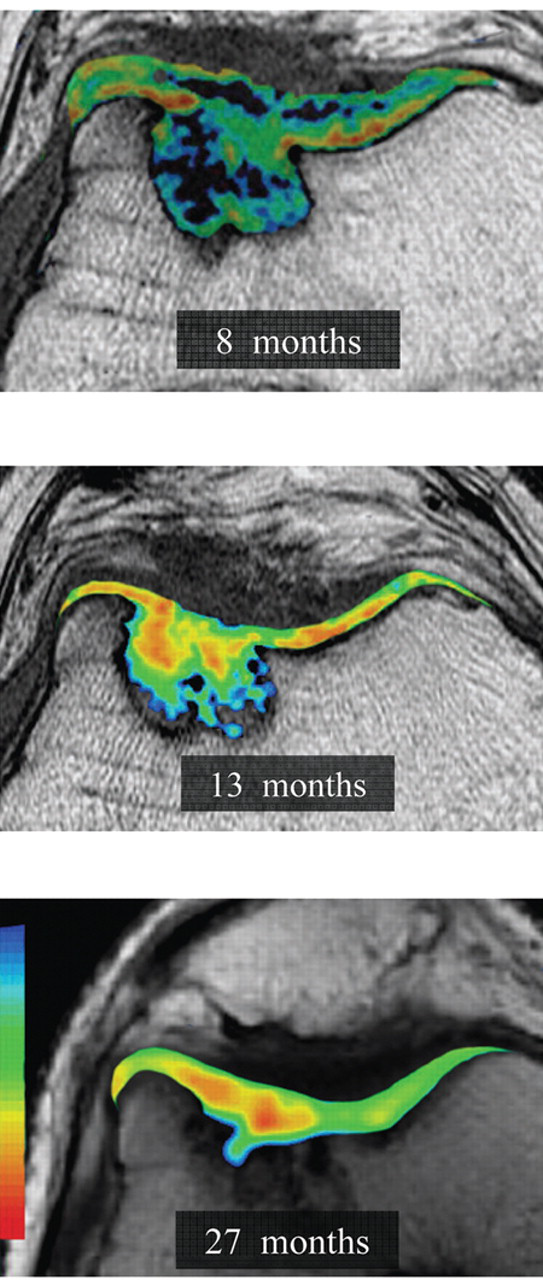
Axial quantitative T2 mapping of a trochlear TruFit bone graft substitute plug at 8 months, 13 months, and 27 months postoperatively. T2 relaxation times (milliseconds) for the superficial and deep cartilage layers of the central and peripheral plug demonstrated significant shortening with increasing duration after surgery (P < 0.004). These values approached but always remained prolonged compared with those of normal articular cartilage by final follow-up evaluation.
Discussion
The purpose of this study was to define the longitudinal morphological characteristics and incorporation of TruFit BGS plugs with cartilage-sensitive MRI and T2-mapping techniques. In this study, we found that the Trufit BGS plug demonstrates a predictable pattern of postoperative maturation on MRI images that parallels its biological incorporation. 26,27 The intermediate postoperative interval (6-12 months following implantation) is frequently associated with unfavorable MRI findings, including hyperintense signal, depressed morphology, interface resorption, incomplete fill, poor incorporation, and prolonged T2 values of the repair cartilage. These findings do not reflect a surgical failure, however, and are rather a part of the natural history of their host incorporation. Morphologic plug appearance significantly improves with increasing postoperative duration and is also associated with progressive shortening of T2 relaxation times of repair tissue, approaching that of native articular cartilage. Patient age, plug size, and plug configuration are other important variables that affect imaging outcomes.
The treatment of articular cartilage defects continues to be one of the most challenging clinical problems for orthopedic surgeons. Articular cartilage is a highly organized, complex tissue with well-defined biomechanical properties and substantial durability. It has poor intrinsic capacity for healing, however, and damage from trauma or degeneration can result in significant morbidity. 1-6 Unaddressed chondral lesions can lead to debilitating joint pain and dysfunction. Despite tremendous improvement in the available treatment strategies for articular cartilage lesions, no validated algorithm to manage these lesions exists, resulting in substantial variability in decision making between practicing clinicians. Currently applied cartilage repair strategies include marrow stimulation, cell-based, scaffold-based, cells plus scaffold-based, and whole-tissue transplantation techniques. 1-6,27
The use of bioabsorbable scaffolds for repair of chondral and osteochondral defects has recently been explored in numerous laboratory investigations. 7-9,15-18 Such matrix scaffolds, implanted alone or in combination with cells, immediately fill a defect and support local migration of chondrogenic and osteogenic cells that synthesize new ground substance. 7-9,15-18 Bioresorbable implants offer substantial advantages, including the avoidance of donor site morbidity associated with autologous osteochondral transplantation, as well as avoidance of residual foreign materials. In addition, these implants are matched in modulus to the adjacent articular cartilage and subchondral bone to create a mechanical environment that supports neocartilage, repair tissue. The optimal scaffold for articular cartilage repair, however, has not been identified but should ideally (1) provide sufficient initial mechanical strength, (2) support the migration and attachment of chondrogenic cells, (3) facilitate ingrowth of reparative tissue, and (4) ultimately incorporate into host tissue without residual foreign material or a secondary inflammatory reaction. 7-9,15-18
The TruFit BGS plug is a bioresorbable implant composed of semiporous 75:25 PLG copolymer, calcium sulfate, polyglycolide fibers, and surfactant. This biphasic implant is designed to approximate the mechanical properties of the adjacent subchondral bone (deep phase) and articular cartilage (superficial phase). Furthermore, the ability of PLG to support chondrocytes and their production of extracellular matrix has been well established. 20-27 Industry-supported animal studies in the femoral condyles and trochlea of goats have been completed to evaluate the efficacy of these implants (Osteobiologics Inc.). 20-27 TruFit BGS plugs were press fit into surgically created 5 × 5-mm osteochondral defects via bilateral arthrotomies in 12 Spanish goats. Histological and biomechanical evaluation was completed at 6 weeks, 6 months, and 12 months postoperatively. Gross examination revealed good incorporation of the plugs with no evidence of an inflammatory synovitis or foreign body reaction. The plug-native cartilage interfaces were smooth, and the defects were almost entirely filled in all cases. The stiffness of the repair tissue ranged from 93% to 101% of native cartilage, demonstrating favorable biomechanical properties that resembled hyaline more than fibrocartilage. Repair tissue thickness was close to that of adjacent native cartilage, with a high percentage of hyaline-like cartilage and good bony restoration on histological analysis. 26,27 Interestingly, staged resorption of the plug occurred in a controlled manner; the calcium sulfate dissolved in the first 3 months, followed by resorption of the polymer over the next 9 months. 26,27
The results of our study demonstrate characteristic MR changes of the TruFit BGS plug that parallel its biologic incorporation. 35 During the first 6 months postoperatively, the plug largely retains its mechanical structure and therefore demonstrates flush morphology and good fill of the donor lesion on MRI. As the scaffold dissolves and resorbs in the intermediate postoperative phase (~9 to 12 months), the MR images of the plug demonstrate an amorphous appearance with resorption at the host-plug interface, depressed morphology, and incomplete fill of the chondral lesion. As the incorporation process progresses, however, plug appearance on MRI substantially improves to demonstrate a high percentage of lesion fill, good incorporation, and restoration of a flush morphology at repair surface. Our finding of more stratified T2 repair tissue with shorter values appearing at the area of peripheral integration rather than the subchondral repair tissue suggests that the scaffold recruits chondrocytes from the periphery to produce matrix, rather than from multipotential stem cell in the subchondral bone. This also correlates with the histological findings noted in the preclinical goat model. 26,27 In addition, the quality of repair tissue as assessed by T2 relaxation times approaches native hyaline cartilage, suggesting that the collagen orientation is more typical of hyaline cartilage. This information is of importance to clinicians who are evaluating the efficacy of the plug with postoperative MRI. The relatively poor filling characteristics of these synthetic plugs at an intermediate postoperative interval (~12 months) should not be misinterpreted as a failure of the cartilage repair procedure but rather a normal phase in the natural history of incorporation of the scaffold into host bone. MRI is not uncommonly performed at 1 year following repair, and caution should be used so as not to misinterpret delayed biologic incorporation as implant failure. The MR appearance of bone incorporation likely reflects the delayed biologic incorporation of this particular implant. There is an expected maturation of relaxation times that both the bone and the cartilage phases undergo, which is most likely a function of a host-dependent response to the scaffold.
The results of our study also provide important information regarding the theoretical concerns with the use of synthetic scaffolds for cartilage repair. In the early postoperative period, potential concerns of an inflammatory synovitis, reactive effusion, or acute plug displacement have been raised. 26,27 None of these complications were observed throughout our study duration. While the appearance and maturation of the TruFit plug were favorable by final follow-up evaluation, our study did identify some potentially important, long-term concerns. These were primarily observed when multiple plugs were used to manage a large donor lesion. Bony overgrowth was relatively uncommon but usually affected the middle plug in a multiple-plug configuration used to fill 3 or more donor lesions. In addition, the use of multiple plugs for a donor lesion was associated with poor ICRS scores of the adjacent, native cartilage. These results suggest the potential for increased morbidity with the use of multiple TruFit plugs and should provide caution to surgeons when managing the donor defect for a large mosaicplasty procedure. The authors hypothesize that incorporation and maturation of the synthetic scaffolds is dependent on creeping substitution from viable host bone and that the limited interface of a plug with surrounding host bone that occurs in a multiple-plug configuration may delay or impair its successful incorporation. 36 Furthermore, the lack or paucity of adjacent normal hyaline cartilage may affect the ability to produce matrix across the articular phase.
Our study is not without limitations. While the MR data at various postoperative intervals help to define the natural history of TruFit plug incorporation, clinical outcomes scores related to the backfill of the donor lesion cannot be separated from the primary OATS procedure to correlate the significance of the imaging findings on functional outcomes. In addition, this study reports on a best-case scenario in which the synthetic scaffold was used to fill a surgically created defect that was surrounded by viable, native articular cartilage along the trochlear margins. The biological milieu surrounding a focal chondral defect may be considerably different and certainly alter the maturation and incorporation of a TruFit BGS plug used to treat the primary chondral lesion. Studies are ongoing to define both the clinical and imaging outcomes of TruFit plugs used to address primary chondral lesions in the knee.
Footnotes
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
