Abstract
Stem cells are a unique population that lies at the summit of any, or at least most, biological systems. They can differentiate in a variety of mature cell types, but they also have the ability to self-renew, that is, the capacity to divide and retain all the features of the mother cell. The regulation of self-renewal has been studied for many years, but several aspects of this regulation are still vague. The combined decision to divide and self-renew or differentiate suggests that the mechanisms that regulate self-renewal and cell cycle activity are intermingled. While inactivation of many cell cycle regulators impacts the physiological and pathological biology of stem cells, the exact mechanisms that link the decision to enter the cell cycle and the choice of the cellular fate are poorly understood. The multiplicity of signals and pathways regulating self-renewal add to the complexity of the phenomenon. Here, I will review the described links between the cell cycle and self-renewal and discuss the role of the niche in the regulation of both mechanisms.
Introduction
Stem cells lie at the apex of a biological system, and they have the unique capacity to either self-renew or differentiate to ultimately produce
Human and mouse embryonic stem cells have been extensively studied during the last 2 decades.1,2 A major difference between embryonic and adult stem cells lies in the regulation of cell cycle activity: while embryonic stem cells display extensive proliferation, 3 adult stem cells are often highly quiescent, 4 although there are variations among different organs. Indeed, stem cells in intestinal 5 and skin 6 epithelia, for example, exhibit a robust cell cycle activity.
In addition to differentiation into mature lineages, stem cells also have the choice to symmetrically or asymmetrically divide into new daughter stem cells, a phenomenon commonly described as self-renewal. 7 Self-renewal can be considered not only as an active property of stem cells to retain their identity by activating a program of genes that maintain their stem cell status, but it can also be seen as the active repression of genetic programs that promote cellular differentiation. 7 In both cases, the result is the generation of new daughter cells that exhibit similar self-renewal and differentiation capacities compared to the mother cell.
An important and inherent aspect of differentiation is the capacity for cells to enter and appropriately regulate cell division. A complex network of genes and pathways regulates the progression of cells through cellular division, and cell cycle activity is often divided into 4 separate phases. 8 Quiescent cells are in the G0 phase, and a variety of proliferative stimuli can trigger the entry of cells into the G1 phase, which is followed by the S and G2/M phases. While the different phases are independently controlled by distinct cell cycle regulators, the G1 phase is under the most developed regulation by strongly conserved genes. 9 This regulation is enforced by the Rb pathway, which includes the Rb family members as well as various members of cyclins and cyclin-dependent kinases. 10 Regulation of cell cycle status is an important feature of stem cells, and the recent literature suggests that both cell cycle and self-renewal activities are regulated by common sets of genes and pathways. 11
Here, we will review the role of established cell cycle regulators in the control of self-renewal activity. We will focus on embryonic and adult hematopoietic stem cells, as their cell cycle activity is very different, suggesting different ways to regulate self-renewal. As we will see, both extrinsic and intrinsic factors regulate self-renewal status, and we will investigate the connections between cell cycle regulation and these extrinsic/intrinsic factors.
Role of Cell Cycle Regulators in Stem Cell Maintenance
Embryonic Stem Cells
Mouse embryonic stem (ES) cells originate from the inner cell mass of the blastocyst at the very early step of embryonic development, and they are totipotent, which means that they can differentiate into all cell types found in the body. 12 While mouse ES cells have been studied for over 30 years, the last decade has seen tremendous progress in our understanding of human ES cells. 2 Both human and mouse ES cells display somewhat similar mechanisms of self-renewal and cell cycle regulation, and we will mostly describe the regulation of these 2 biological activities in mouse ES cells.
External Factors
Mouse ES cells can easily be cultured
Internal Factors
Transcription factors: Oct4, Nanog, and Sox2 take center stage
ES cells rely on a limited number of transcription factors to ensure the maintenance of their undifferentiated state. Within that short list, Oct4, Sox2, and Nanog are critically important to promote self-renewal in ES cells. 18 These 3 transcription factors bind to overlapping as well as distinct promoter regions to repress the expression of genes that promote cellular differentiation. 19 In addition, recent work has identified Ronin as another factor whose expression is critical for ES cell maintenance. 20 In a noncanonical mode, it has been shown that caspase-3 can cleave both Ronin and Nanog, thereby inducing differentiation. 21
miRNAs specific to ES cell maintenance
The overall role of miRNAs in ES cells is to promote proliferation and differentiation.22,23 This was suggested by results generated in Dicer- and Dgcr8-deficient ES cells: these ES cells have a lower proliferation index and maintain their pluripotent potential, even in culture conditions that promote differentiation.23,24 In fact, a reduced number of miRNAs are expressed in ES cells. 25 One of the main goals of mir-290 and mir-302 is to promote high cell cycle activity by targeting numerous components of the cell cycle regulatory machinery, including members of the cyclin-dependent kinase inhibitor and Rb family.22,26-28 As a result, the G1/S transition, the main point of control for the cell cycle, is accelerated, and ES cells have high proliferative activity. Not surprisingly, these miRNAs are under positive transcriptional regulation by the pluripotency factors Oct4, Sox2, and Nanog. 29 The same 3 transcription factors also repress the expression of miRNAs that promote differentiation of ES cells, including mir-145, mir-134, mir-296, mir-470, mir-200c, and mir-183, and members of the Polycomb group of proteins (see below) are involved in the silencing of these target miRNAs. 29 Interestingly, target genes of these miRNAs include Oct4, Sox2, and Nanog, thereby generating a negative feedback loop. 30 Finally, the expression of let-7 promotes differentiation, and let-7 expression is repressed by pluripotency factors, such as Lin28B.31,32
Regulation of chromatin state
Expression of factors that promote differentiation in ES cells is also tightly regulated at the chromatin level. Two groups of chromatin regulators compete for an appropriate methylation status: Polycomb and Trithorax proteins modulate the state of chromatin by methylating different lysine residues, H3K7 (repression) and H3K4 (activation), respectively. Indeed, it has been shown that loss of the expression of key Polycomb genes promotes ES cell differentiation.33,34
Cell Cycle Regulators
Cell cycle activity is very high in ES cells, with a shortened G1/S transition. Rb family proteins are heavily phosphorylated and therefore functionally inactivated.3,35 As a consequence, genes that are involved in cell division are constantly “on,” leading to a reduced population in the G0 quiescence state. Interestingly, while the p53 expression level is high in ES cells, the capacity for p53 to activate its canonical program in a normal condition is still unclear, and ES cells are uniquely insensitive to DNA damage–induced, p53-mediated cell cycle arrest. 36 However, several lines of evidence point to an important role for p53 in the control of self-renewal and differentiation in ES cells. The first evidence comes from its ability to repress Nanog expression (as well as Oct4 in human ES cells).37,38 Decreased expression of these factors is sufficient to impair self-renewal in ES cells. However, p53 also promotes differentiation by activating the expression of several miRNAs. An important effector of p53 is the mir-34 family, which directly targets Nanog for silencing,39,40 as well as Lin28, 41 which maintains self-renewal by repressing let-7 expression. Other miRNAs transcriptionally activated by p53 include mir-145, mir-200, mir-107, and mir-194. 42 These miRNAs target multiple components of the machinery that maintains ES cells in an undifferentiated state. While all these elements point to the prodifferentiation activity of p53, it is also of interest to note that p53 can promote ES cell maintenance by activating the expression of several Wnt ligands. 43 These data suggest an important role for p53 in the control of ES cell self-renewal. In contrast, surprisingly little is known about the role of Rb proteins and the different components of the Rb pathway in the same activity. 44
Adult Stem Cells
Hematopoietic Stem Cells
Hematopoietic stem cells (HSCs) have served as a paradigm for the study of adult stem cell biology. Since their identification more than 15 years ago, the scientific community has identified a hierarchy of stem and progenitor cells that contribute to the constant replacement of cells from the lymphoid and myeloid lineage. 45 In contrast to ES cells, HSCs are mostly quiescent and only exit cell cycle arrest to either self-renew or differentiate into the various mature populations of the hematopoietic system. 4 It is generally thought that the disruption of HSC quiescence is associated with a decrease in self-renewal (see below), but exceptions have been found to this rule, and the mechanisms that connect cell cycle status and self-renewal activity are only poorly understood. 11 Under physiological conditions, HSCs are mostly found in the bone marrow. It is now established that long-term HSCs are settled in a specific environment, frequently described as a niche, which regulates HSC functions through different signals. 46 In addition to these external signals, previous studies have shown that several factors could influence stem cell biology in a cell-autonomous manner. 47 As the external signals influence the internal circuitry of stem cells, an interesting perspective is the regulation of niche signaling by these internal factors (see Figure 1).
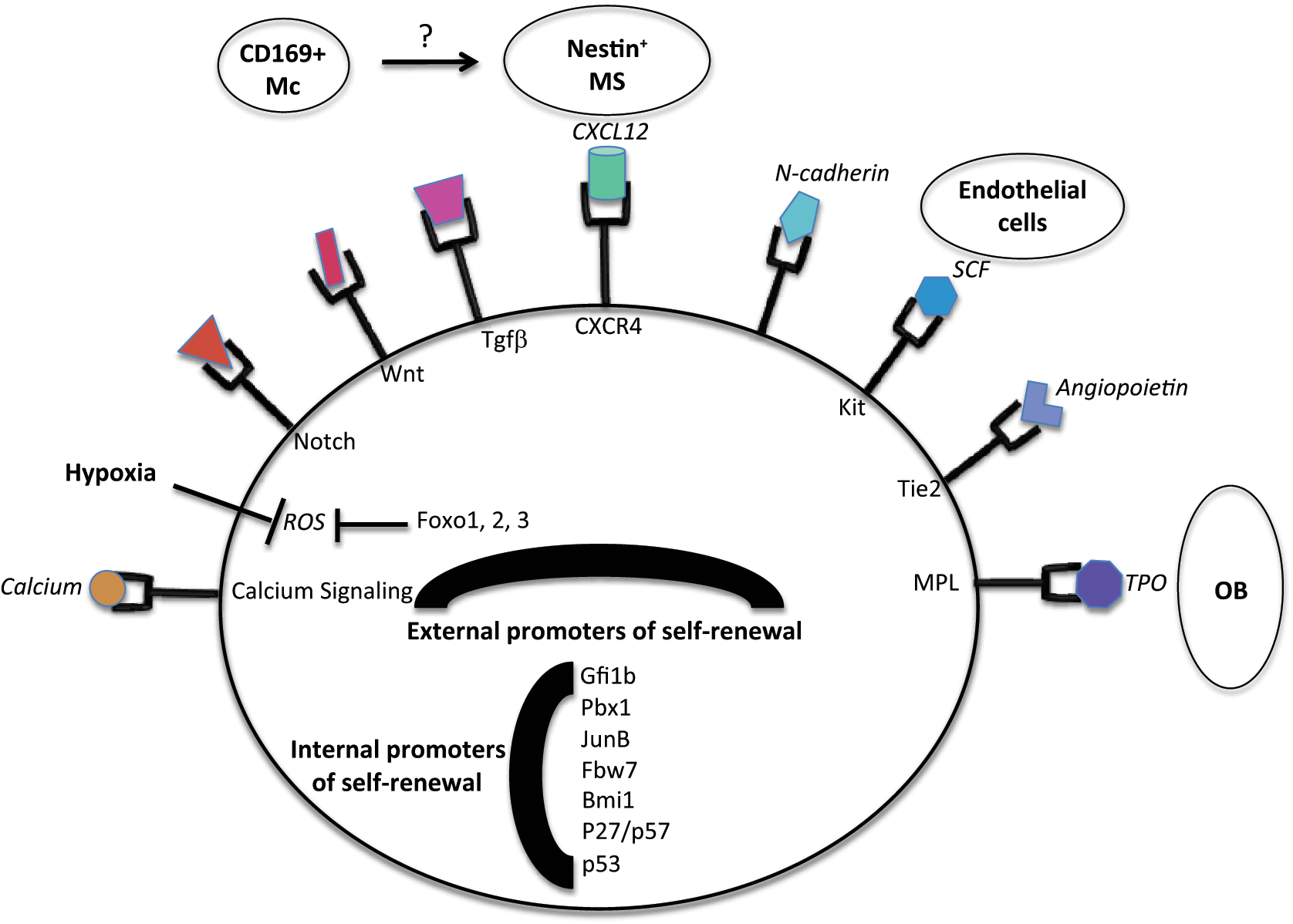
External and internal promoters of self-renewal in HSCs. As illustrated, multiple cell types are involved in the regulation of self-renewal of HSCs, including osteoblasts (OB), endothelial cells, mesenchymal cells (MS), and a specific subpopulation of macrophages (Mc). See text for further details on their respective contributions.
External factors
The HSC niche is a highly complex environment that is only partially described. It appears to be constituted by different cell types (recently reviewed in Ehninger
In addition to these signals that are produced by different populations of the niche, it has also been observed that the general environment of the niche matters. Indeed, the niche is located in an extremely hypoxic environment. 59 A low concentration of oxygen and anaerobic metabolism reduce the production of reactive oxygen species (ROS) by stem cells. High intracellular levels of ROS promote the differentiation of HSCs by activating JNK, Pi3K signaling, and inactivating FOXO factors. Indeed, inactivation of FOXO factors leads to the increased production of ROS in stem cells, which is detrimental to the homeostasis of the stem cell pool. 60 As a consequence, FOXO1/2/3-deficient stem cells are decreased in number, and their cell cycle activity is promoted. This phenotype can be corrected by N-acetylcysteine (NAC) treatment, which reduces the level of ROS. Interestingly, FOXO1/2/3-deficient mice also develop mild myeloproliferation, which suggests that cell cycle entry is conjugated to the activation of a genetic program that promotes the abnormal development of the myeloid lineage. Quite intriguing is actually the fact that often loss of quiescence is associated with the development of a myeloid disorder (see below). ROS also promote DNA damage, which activates the p53 pathway (see below). The p53 pathway is important for HSC homeostasis, as the ELF4 effect on HSCs is mostly mediated by the MDM2/p53 axis. 61 In addition, DNA damage also recruits the ATM kinase, and ATM-deficient mice exhibit higher levels of ROS and a decrease in their HSC population. 62 Last, HSCs are also sensitive to a Ca2+ gradient, and the endosteal niche exhibits a high concentration of Ca2+ ions. Accordingly, HSCs that are deficient in the calcium receptor have a defect in their migration and homing properties. 63
Recent advances in the field have shed light on the phenomenon of asymmetric cell division, that is, the ability of a stem cell to divide into 2 differentiated cells, 2 stem cells, or 1 differentiated cell and 1 stem cell. 7 Within the niche, the mother cell is physically connected to the signaling that promotes its maintenance (Wnt, TPO, etc.). Upon its division, based on the spatial availability of the niche, 1 or 2 daughter cells may retain a direct access to signals that will promote its stem cell status. Yet, it is also possible that daughter cells may be physically separated from the source of the signal (osteoblast, mesenchyme cells, etc.), which will induce its differentiation. As a consequence, one mother cell may give birth to 2 stem cells (both connected to the niche), 1 stem cell (connected) and 1 differentiating cell (not connected), or 2 differentiating cells (that are pushed away from the niche). There again, it is not clear whether this model is dynamic, with cells shuttling constantly between different regions of the niche and a different status of differentiation (and self-renewal), or whether it is similar to a 1-way road, where cells can only evolve into a state that is associated with a lower self-renewal status.
Another guest has recently made its entrance in the circle of cells that form the HSC niche. It has been known for a while that granulocyte colony-stimulating factor (G-CSF) treatment can induce the mobilization of HSCs in the periphery, which obviously means to exit the niche. 4 As HSCs do not express the receptor for G-CSF, it was recently shown that a population of macrophages serves as an intermediate. Macrophages in the niche promote the activity of mesenchyme and osteoblastic cells, and G-CSF treatment induces the depletion of macrophages from the niche. Therefore, a differentiated population within the hematopoietic compartment can impact the status of HSCs in their niche. 64
Internal factors
Many factors have been shown to play a role in the regulation of self-renewal in HSCs. Gfi1 was one of the first genes identified for its role in the maintenance of HSCs. Mice that are deficient in Gfi1 display increased cell cycle activity of HSCs and are outcompeted in transplantation protocols. 65 Recent work suggests that Gfi1 promotes HSC maintenance by repressing Bax expression. 66 Pbx1 deficiency also induces the cell cycle activity and exhaustion of HSCs, and it is currently suggested that Pbx1 supports HSC self-renewal by promoting TGFβ signaling in HSCs. 67 Another factor that promotes TGFβ signaling in HSCs is JunB, and JunB-deficient HSCs have impaired TGFβ and Notch signaling, leading to enhanced cell cycle activity and the onset of a myeloid disorder in these mice. 68 Myc is another factor that controls stem cell quiescence, and inactivation of Myc in the hematopoietic compartment leads to cytopenia and an accumulation of HSCs. 69 Recent work has shown that deletion of Fbw7 leads to the proliferation and exhaustion of HSCs, and further analysis has shown that the main relevant target for Fbw7 in HSCs is cMyc. Indeed, inactivation of Fbw7 in HSCs leads to an increase in the amount of Myc. 70 Finally, deletion of Evi1 is detrimental to the maintenance of HSCs, and the effect appears to be mainly mediated by the regulation of GATA-2 expression by Evi1. 71
Cell cycle regulators
The Rb pathway is a critical regulator of the G1/S sequence of the cell cycle. The majority of the components of the pathway have been specifically inactivated in the hematopoietic compartment. Components of the Polycomb group have been shown to promote self-renewal in ES cells (see above). In HSCs, it has also been demonstrated that Bmi1 expression promotes self-renewal in HSCs by repressing the expression of p16 and p19.72,73 This activity is unique among members of the Polycomb groups, suggesting a specific role for Bmi1. Bmi1 also represses the expression of genes that promotes differentiation, such as Ebf1 and Pax5, as well as Ikaros, for the B cell lineage. 74 Within the CIP/KIP family, p21 deficiency leads to the extensive proliferation and exhaustion of HSCs. 75 However, recent data suggest that p21 activity is only relevant in stressed HSCs. 76 In contrast, p27 inactivation did not lead to a defect in the HSC compartment but impacts progenitor proliferation. 77 There probably exists a compensation phenomenon within the CIP/KIP family, as double deficiency for p57 and p27 leads to a loss of HSC quiescence. TPO and TGFβ signaling in HSCs induce the expression of p57, which sequesters a complex formed by Hsc70 and cyclin D1 in the cytoplasm. Upon the deficiency of p57 and p27, this complex shuttles to the nucleus to promote the phosphorylation of the Rb family and entry into the cell cycle.78,79
p16’s role in HSC maintenance is somehow specific, as only aged HSCs are impacted by the loss of p16. p16 seems to promote an age-related decline in the functions of HSCs, probably in response to an accumulated amount of ROS. 80 While the general trend associates cell cycle entry to loss of self-renewal, the loss of p18 translated into increased cell cycle activity and concomitant unaltered self-renewal activity. In this case, p18 deficiency would considerably shorten the length of the G1 phase, thereby bypassing the decision to differentiate. 81 Finally, inactivation of the Rb gene is not detrimental to HSC status. 82 The Rb family genes (Rb, p107, and p130) display strong overlapping activities, and it is therefore not surprising that the concomitant inactivation of the 3 Rb family members (TKO) is sufficient to lead HSCs to cell cycle re-entry. However, the self-renewal status of TKO HSCs is not yet clear, and additional experiments are required to assess the long-term self-renewal status upon inactivation of the Rb family of genes. As often observed when HSCs are forced to enter the cell cycle, TKO mice display a myeloid disorder. While more work is also required to understand the molecular basis of the phenotype, several key myeloid genes had up-regulated expression in TKO KLS (Kit+, Lineage–, and Sca1+ cellular fraction, enriched in stem and early progenitor cell activity) cells, and a possible explanation is that the Rb pathway directly controls their expression. This model would mechanically link proliferation with promotion of the myeloid lineage, at least in adult HSCs. 83 Indeed, GATA-2, whose expression in human progenitor cells promotes the enforced differentiation into the eosinophilic lineage, 84 seems to be directly regulated by the Rb pathway, through direct binding of E2F transcription factors to its promoter sequence.
Last, p53 promotes the quiescence of HSCs, not so much by inducing p21 expression, whose role in physiological hematopoiesis is limited, but by inducing the expression of Gfi1 and Necdin. 85 In addition, p53 limits the self-renewal ability of HSCs, a phenomenon that is also observed in other biological compartments. Interestingly, irradiated HSCs, with a high expression of active p53, have a reduced ability to engraft and contribute to hematopoiesis upon transplantation. While this defect has been associated with a defect in the cell cycle (as p53 promotes cell cycle quiescence), 85 another possibility is that p53 impairs the capacity of HSCs to connect and respond to the signal provided by the niche for engraftment.
Other Stem Cell Compartments
In
Concluding Remarks
Stem cells are particularly sensitive to the dysregulated activity of canonical cell cycle regulators. While these cells are highly quiescent, the inactivation of several cell cycle regulators is sufficient to lead them to cell cycle re-entry. Interestingly, and although beyond strictly the scope of this review, while alteration of the Rb pathway is sufficient to trigger stem cell proliferation in the hematopoietic system and in the liver, the same genetic alteration does not modify the cell cycle status of mature populations in the same systems.83,88 While this is not very surprising in the case of myeloid cells, which are highly quiescent and have terminally exited the cell cycle, the finding that mature hepatocytes cannot re-enter the cell cycle upon a loss of the Rb family of genes is more surprising, especially given the potential of hepatocytes to re-enter the cell cycle when challenged by specific stimuli (such as partial hepatectomy). Alternatively, it was shown recently that loss of Rb and p19 ARF was sufficient to introduce cell cycle activity in postmitotic myocytes, and this was associated with dedifferentiation of these cells. 89 Similarly, inactivation of p53 increases the efficiency of induced pluripotent stem cell derivation from various differentiated cell types. 90 These data would suggest that components of the cell cycle machinery may link the regulation of cell proliferation to differentiation, although the exact mechanism is certainly context dependent. This hypothesis may be connected to what we have observed in the stem cell compartment. Clearly, stem cells are uniquely sensitive to the deficient expression of components of the cell cycle machinery, and stem cells often exit their quiescence state upon inactivation of several regulators of this machinery. The trade-off seems to be a loss of self-renewal potential of these proliferating stem cells, at least upon the inactivation of several cell cycle regulators, although not all. Indeed, p18 deficiency does not appear to be associated with a loss of self-renewal, and inactivation of the Rb family of genes does not seem to induce a decrease of self-renewal either (see above). As suggested before, this discrepancy in the activity of several cell cycle regulators in the control of self-renewal may be associated with the particular phase of the cell cycle that they are controlling. 11 This is illustrated by ES cells, where proliferative ES cells are kept in an undifferentiated state, and this high self-renewal activity is linked to the very short G1/S phase that is unique to these ES cells.
Regulators of self-renewal status are often classified as external regulators (signals produced by the niche to promote self-renewal) and internal factors. The role of these internal factors is often described as mostly involved in the regulation of intracellular processes. However, work on the Lnk gene, for example, has shown that the main activity of this factor is to precisely modulate TPO signaling, therefore integrating the external with internal maintenance of self-renewal. 52 In the case of components of the cell cycle machinery, it is probable that they directly control the decision to self-renew or differentiate (as illustrated by the capacity for Bmi1 to repress p16 and p19 expression, but also factors that promote the B cell lineage). However, given the ever-expanding role of these genes in various cellular activities, it is also plausible that cell cycle genes directly interact and regulate the external signaling that promotes self-renewal. In the complexity of self-renewal regulation, the fact that a mature hematopoietic population can regulate the self-renewal activity of HSCs suggests that cell cycle regulators (or other genes that control self-renewal) may also play a distinct role in different populations of the same system. 64 As traditional methods lead to the deletion of a particular gene of interest in an entire compartment, this phenomenon may have been overlooked in the past. Clearly, future studies will require the deletion of genes of interest in very specific populations of the niche and the different cell types of a biological system. These studies should shed light on the role of cell cycle regulators in the control of stem cell self-renewal, which probably includes a mix of direct regulation of genes involved in differentiation and fine-tuning of signals that modulate self-renewal.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: The Viatour laboratory is supported by institutional division funds from the Center for Childhood Cancer Research at The Children’s Hospital of Philadelphia and the WW Smith Charitable Trust Foundation.
