Abstract
The paxillin family of intracellular scaffold proteins includes paxillin, Hic-5, and leupaxin, and all have been identified as key regulators of the cellular migration machinery in both 2- and 3-dimensional microenvironments. Herein, we provide insight into the roles of these proteins during tumorigenesis and metastasis, highlighting their functions in cancer initiation as well as tumor cell dissemination and survival. Furthermore, we speculate on the potential of paxillin family proteins as both future prognostic and therapeutic targets.
Keywords
Introduction
Cell adhesion either to neighboring cells or to the surrounding extracellular matrix (ECM) or stroma plays a critical role in tumor progression by regulating proliferation and survival as well as enabling tumor cell dissemination and metastasis. 1 The local tumor microenvironment, including the molecular composition and organization, density, and rigidity of the stromal ECM, has been shown to influence tumor malignancy. This information is relayed to the tumor cells through ligation of transmembrane integrin molecules on the tumor cell surface. Integrins lack enzymatic activity and thus communicate with the intracellular signal transduction pathways to regulate cell migration and/or changes in gene expression via the recruitment of multiple structural and signaling proteins to their cytoplasmic tails. 2 Scaffold proteins, including members of the paxillin family,3-5 perform critical functions in coordinating these signaling events downstream of integrin-ECM ligation.
The paxillin family comprises 3 proteins: paxillin, Hic-5, and leupaxin (Fig. 1). Paxillin was first identified in a screen for substrates for the oncogene tyrosine kinase v-src in Rous sarcoma virus (RSV)–transformed fibroblasts.6,7 Consistent with its function as a molecular adaptor/scaffold protein, paxillin comprises multiple discrete protein binding modules, including numerous tyrosine as well as Ser/Thr phosphorylation sites that, when modified, contribute further to the complexity of the protein interactome. 5 Many of these phosphorylated residues have been detected in tumor samples and/or cancer cell lines (Fig. 1). The amino terminal LD motifs and the C-terminal LIM domains, are highly conserved in the 2 other family members, Hic-5 and leupaxin (Fig. 1). As a result, the 3 proteins share many of the same binding partners,8-17 although their interactions can be spatiotemporally regulated by the balance of Rho family GTPase activity. 8 However, it is important to note that each family member has its own unique set of interactions, for instance, the association of Hic-5 with members of the Smad family of transcription factors18,19 and paxillin with bcl-2. 20 In-depth functional analysis of domain mutants has revealed a critical role for the paxillin family in coordinating cell migration by acting primarily as a hub for the regulation of Rho GTPase signaling.3,4 In addition, roles for paxillin in cell survival, mRNA processing,21,22 and Hic-5 in regulating gene expression through its function as a steroid receptor coactivator have also been reported.
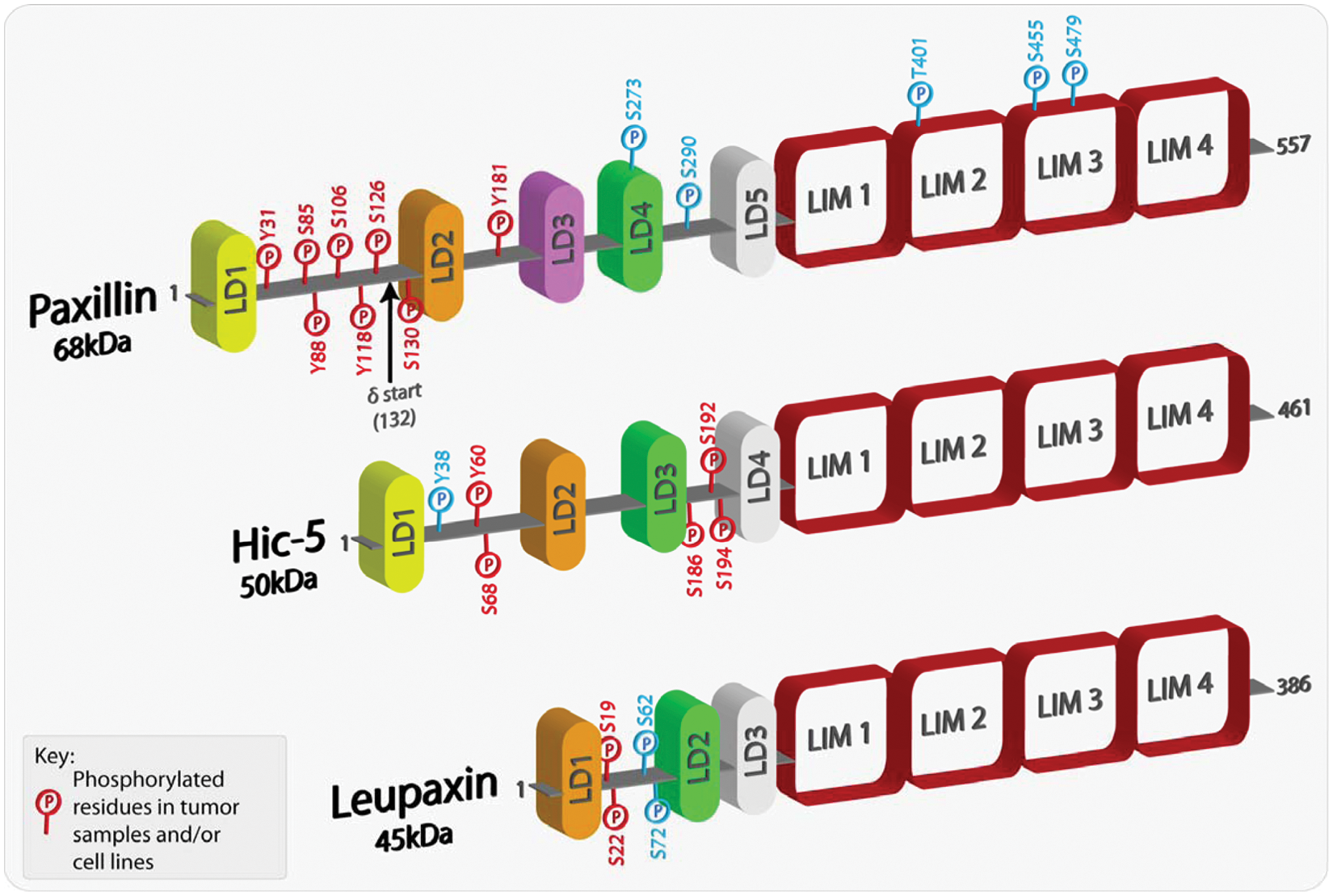
Schematic representation of paxillin family members highlighting phosphorylated residues. Paxillin and its family members comprise a variable number of amino-terminal LD domains, which are short leucine-rich motifs that are responsible for mediating protein-protein interactions. At the carboxyl-terminus, each family member contains 4 highly homologous LIM domains, which also serve as protein-protein binding moieties as well as being responsible for appropriate protein subcellular localization, including sites of integrin-mediated adhesion and the nucleus. Paxillin family members can also be phosphorylated at multiple sites, with the majority located in the less conserved regions between the LD domains. Furthermore, a number of the phosphorylated residues have been revealed as mediating and regulating paxillin family member protein interactions. The phospho-residues highlighted in red have been shown in multiple studies to be phosphorylated in both tumor samples and/or cancer cell lines. The amino acids highlighted in blue have been shown to be phosphorylated in a variety of studies but thus far have not been identified in cancer cells or tissues.
Paxillin is ubiquitously expressed, although protein levels in the nervous system are low.23-25 In contrast, Hic-5 is enriched in smooth muscle tissues, in particular the vasculature,26,27 and is upregulated in response to transforming growth factor (TGF)–β during epithelial-mesenchymal transition (EMT).27,28 Leupaxin, the least well-characterized member of the family, was originally thought to be restricted to cells of the leukocyte lineage, 29 but more recent studies suggest a broader expression profile.30-32 Gene ablation studies in mice have generated surprisingly diverse phenotypes, with paxillin ablation causing early embryonic lethality, 10 while the Hic-5 knockout mouse is viable and exhibits only very mild vascular defects. 33 Importantly, paxillin, Hic-5 and leupaxin expression and/or phosphorylation changes have been linked to a number of human malignancies. In this review, we will describe some of the more recent studies that have begun to explore how disruption of paxillin family function contributes to increased cancer malignancy and speculate on their potential as therapeutic targets.
Paxillin Family Member Expression and Mutation in Human Cancers
Paxillin, Hic-5, and leupaxin have all been linked to malignant progression and have been shown, through both tissue microarray and histologic analyses, to be differentially expressed in a variety of invasive/metastatic cancers, including but not limited to breast, lung, and prostate tumors (Fig. 2).34-51 As there is a range of expression profiles of paxillin family members observed in different cancers (Fig. 2) and cancer subtypes, it precludes the classification of any individual protein as a traditional oncogene or tumor suppressor. However, given the extensive crosstalk and synergy between paxillin family members,8,13,28,52,53 it is likely that variations in the expression of any one protein will influence the downstream signaling of the other closely related family members to dictate tumor progression, invasion, and malignancy. For example, enhanced ectopic expression of either Hic-5 or leupaxin prevents paxillin phosphorylation and direct protein interactions to alter downstream signaling and cell function.13,53
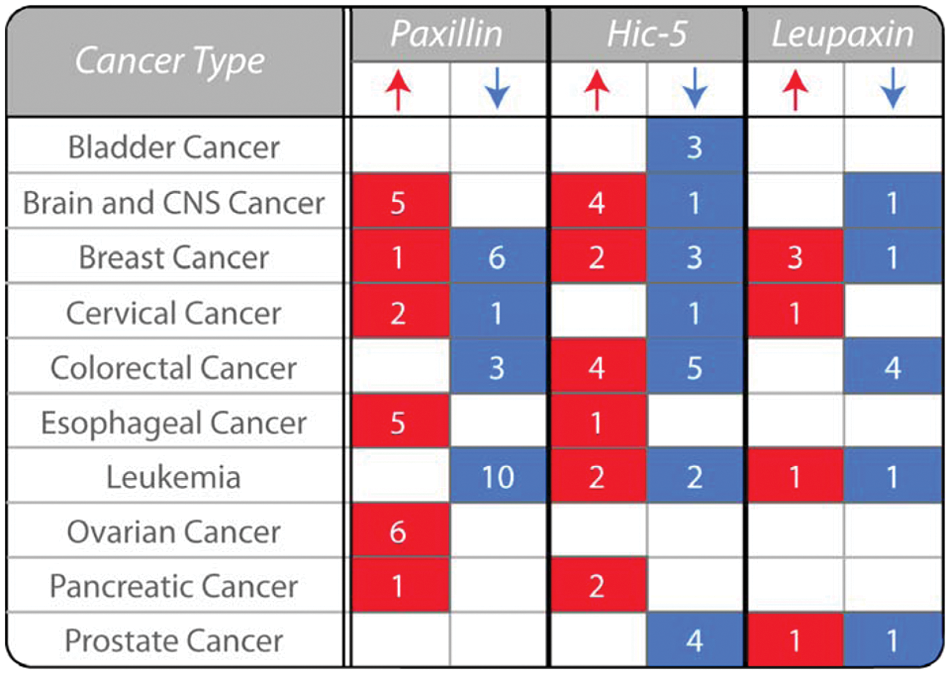
Paxillin family expression is variable in human cancers. The table highlights the variability in the expression of paxillin, Hic-5, and leupaxin in human cancers as determined by analysis of their mRNA levels in tumor samples relative to normal tissue (P < 0.00001). The number of studies that reveal a significant change in expression in tumor samples relative to normal tissues is indicated. Expression data are collated from the Oncomine cancer gene microarray database (Compendia Biosciences; Ann Arbor, MI, USA; www.oncomine.org). CNS, central nervous system.
To date, no somatic mutations in Hic-5 or leupaxin have been reported in human cancers. In contrast, analysis of lung adenocarcinoma samples identified a number of paxillin gene sequence aberrations, which were located between the LD1 and 2 motifs, the most common being an A127T mutation, which was linked to increased tumor growth and invasion.40,54 However, it is important to note that the presence of somatic mutations of paxillin in human cancers remains controversial55,56 and thus requires further study in a variety of different cancers.
Paxillin, Hic-5, and leupaxin are all downstream targets of activated hormone and growth factor receptors associated with aggressive cancers and decreased patient survival.27,28,30,57-61 For example, Hic-5 is phosphorylated in response to epidermal growth factor (EGF) receptor activation. 62 Furthermore, Hic-5 is expressed in prostate stroma, where it functions as a TGF-β–induced coactivator of the androgen receptor and is linked to prostate tumorigenesis. 63 Likewise, paxillin is phosphorylated downstream of EGF, TGF-β, platelet-derived growth factor (PDGF), and androgen receptor activation in a variety of cancers and affects both tumor formation and invasion.61,64-67 Furthermore, paxillin expression levels have also been shown to correlate with HER2 levels in breast cancer cells and patient samples and thus may be a predictor of therapeutic efficacy.58,60
The Roles of Paxillin and Hic-5 in Epithelial-Mesenchymal Transition (EMT)
Epithelial-mesenchymal transition is an evolutionarily conserved process that is essential for normal organism development and involves the dissolution of cell-cell adhesions, induction of cell-matrix adhesions, loss of apical-basolateral polarity, and a reorganization of the cytoskeleton to promote cell motility. Although currently controversial,68,69 the process of EMT is widely believed to be highly analogous to the changes observed during cancer initiation, in which there is a loss of cell cohesion, enhanced migration, tissue stroma invasion and subsequent metastasis. 70 The conversion of epithelial cells to the more malignant mesenchymal phenotype can be stimulated by numerous factors, including activation of receptor tyrosine kinases and TGF-β–mediated signaling pathways. Cells that have undergone an EMT share various properties with a population of multipotent, self-renewing tumor cells termed cancer stem cells (CSCs), which have tumor-initiating potential.71-73 For example, cells stimulated to undergo a TGF-β–mediated EMT exhibit CD44+/CD24+ cell surface markers, upregulation of transcription factors such as Twist and Snail, 74 and resistance to chemotherapy treatments, 75 all of which are properties of CSCs. 76
Importantly, paxillin family members appear to be differentially regulated during EMT. The paxillin gene contains an internal translation initiation site that can produce a truncated form, termed paxillin δ, which is preferentially expressed in epithelial cells but is downregulated upon TGF-β–stimulated EMT. 28 Although no change in paxillin expression is observed, paxillin tyrosine phosphorylation is increased in response to TGF-β. 28 In contrast to paxillin, Hic-5 is not expressed in epithelial cells but is upregulated in response to EMT, whereby TGF-β promotes both Hic-5 expression and its Src-dependent phosphorylation.66,77 Furthermore, ectopic expression of Hic-5 is sufficient to promote normal mammary cells to undergo an EMT in the absence of TGF-β.66,77 In contrast, the overexpression of paxillin is unable to induce a transition to the mesenchymal phenotype. 78 It is also noteworthy that Hic-5 can stimulate the production of TGF-β through an autocrine feed-forward loop in fibroblasts, 79 which may enable more epithelial cells to undergo a malignant transition and thus promote tumor growth and dissemination. It will be important to determine if, similar to EMT, paxillin family members are disregulated in CSCs at the level of protein expression and/or posttranslational modifications or indeed if they play a role in the development of stem cell–like properties in tumor cells.
Paxillin and Hic-5 Regulate Invadopodia Function
In order for tumor cells to disseminate from the primary solid tumor, they must degrade the surrounding ECM-rich basement membrane barrier to gain access to the surrounding tissues and subsequently metastasize. Analysis of invasive tumor cells in vitro and in vivo has revealed the presence of ECM-degrading protrusions, termed invadopodia.80-82 These structures aid in the process of invasion by focally disrupting the dense meshwork of ECM barriers through localized activity of membrane-bound and soluble matrix metalloproteinases (MMPs) to promote tumor cell dissemination.83,84
Invadopodia comprise an actin core that contains actin-binding and nucleating proteins as well as a surrounding ring of scaffold and adhesion proteins, including paxillin and Hic-5 (Fig. 3). 85 Paxillin was identified as a component of invadopodia in both cancer cells and RSV-transformed fibroblasts,86,87 where it forms a complex with cortactin, protein kinase C (PKC) µ, 86 and the ArfGAP AMAP1. 88 In addition, paxillin tyrosine phosphorylation is necessary for promoting invadopodia dynamics. 89
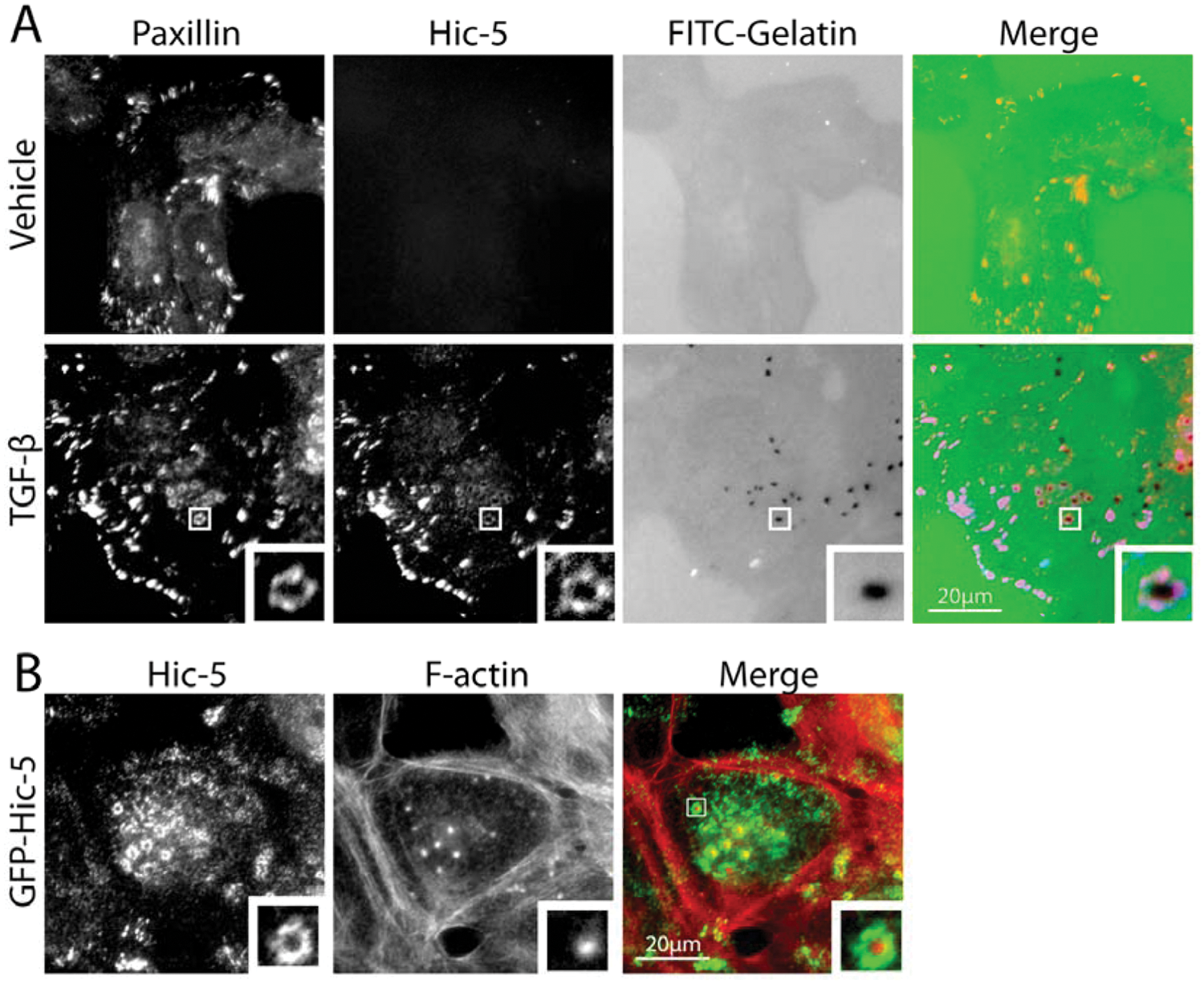
Paxillin and Hic-5 localize to invadopodia. (
We have recently identified Hic-5 as an essential component of invadopodia formed in cells that have undergone a TGF-β–mediated EMT (Fig. 3A). 77 Interestingly, overexpression of Hic-5 in epithelial cells in the absence of TGF-β can also induce the development of invadopodia (Fig. 3B), suggesting that it is both necessary and sufficient for their formation and for subsequent cell invasion. 77 Interestingly, in this system, paxillin expression is dispensable for invadopodia formation. 77 Although a role for leupaxin in cancer cell invadopodia has not been assessed, leupaxin has been shown to be associated with invadopodia-like podosomes at the sealing zone of osteoclasts, 90 where it associates with several proteins, including the non–receptor tyrosine kinases Pyk2 91 and Src, 31 and thus it is plausible that leupaxin plays a similar role in tumor cells.
The regulation of the formation of invadopodia and their maturation into matrix-degrading structures has been shown to require a fine balance of Rho GTPase signaling.92,93 Furthermore, Rho GTPase family proteins have been shown to be upregulated in a variety of cancers where they regulate various aspects of tumorigenesis, including invasion and cell proliferation. 93 RhoC, in particular, has been shown to be upregulated during EMT94,95 and overexpressed in multiple invasive cancer types96-99 to enhance malignancy.100-103 Multiple Rho GTPase family members, along with their regulatory proteins, guanine nucleotide exchange factors (GEFs) and GTPase activating proteins (GAPs), have been shown to localize to invadopodia. Indeed, RhoC has recently been shown to be a component of invadopodia, where the discrete localization of p190RhoGEF and p190RhoGAP regulates its activity. 104 Interestingly, invadopodia formation in TGF-β–treated cells also requires RhoC activity, which in turn is regulated by the upregulation of Hic-5. 77 Thus, the localization of paxillin family members to the ring compartment of invadopodia positions them to function in their capacity as hubs for GTPase regulation 4 through interactions with multiple GAPs and GEFs, including cdGAP 105 and βPIX, 17 to spatiotemporally integrate RhoGTPase activity and so coordinate invadopodia structure and function.
Paxillin and Hic-5 Regulate Tumor Cell Invasion, Plasticity, and Metastasis
The ability of paxillin and, to a lesser extent, Hic-5 and leupaxin to regulate 2-dimensional (2D) cell motility through coordinating the migration machinery is well established.3,4 However, in vivo, migrating cells rarely meet a 2D ECM substrate but instead encounter a wide array of structurally and compositionally diverse 3-dimensional (3D) microenvironments. In vitro models of 3D cancer cell invasion have revealed important roles for paxillin, 52 Hic-5,63,77 and leupaxin 106 in regulating motility in this more in vivo–like environment.
Intravital and in vitro imaging studies have revealed that cancer cells are able to invade through the tissue ECM microenvironment either as collective multicellular sheets or as individual cells, contributing to lymphatic and hematogenous infiltration, respectively.107-117 In addition, cancer cells can employ 2 distinct and interchangeable modes of single-cell motility, referred to as either mesenchymal or amoeboid migration,115,118 that are determined by numerous factors ranging from ECM architecture and rigidity to the balance of intracellular Rho GTPase family signaling. 109 Mesenchymal migration is characterized by an elongated cell morphology requiring extracellular proteolysis and dynamic integrin-mediated interactions with the ECM. In contrast, amoeboid motility is independent of protease activity and is associated with a rounded or ellipsoid morphology.113,115 Cells exhibiting amoeboid migration squeeze through preexisting gaps in the heterogeneous ECM milieu using actin-rich membrane blebs or filopodial-like projections and display highly transient, weak interactions with their surroundings.113-115,119 The spontaneous interconversion between the 2 forms of migration, known as plasticity, is considered one of the primary reasons for cancer cell evasion of current invasion-targeted therapeutics.120-123
Paxillin and Hic-5 have been identified as critical determinants of breast cancer cell morphology and plasticity during invasion. 52 The balance of paxillin and Hic-5 expression and/or signaling is required for efficient invasion and plasticity, with paxillin depletion promoting the mesenchymal mode of invasion and Hic-5 RNAi inducing an amoeboid phenotype (Fig. 4), while their respective overexpression has the reciprocal effect. 52 Importantly, depletion or overexpression of either protein was found to inhibit plasticity, invasive migration, and transendothelial migration, culminating in a lack of metastasis. 52 Furthermore, analysis of 3D cell-ECM adhesion contacts revealed an absolute requirement for Hic-5 for their formation and identified paxillin as a regulator of adhesion dynamics during 3D invasive migration. 52 These data suggest that for optimal, efficient invasion and metastasis, cancer cells must balance the signaling and/or expression of these paxillin family members.
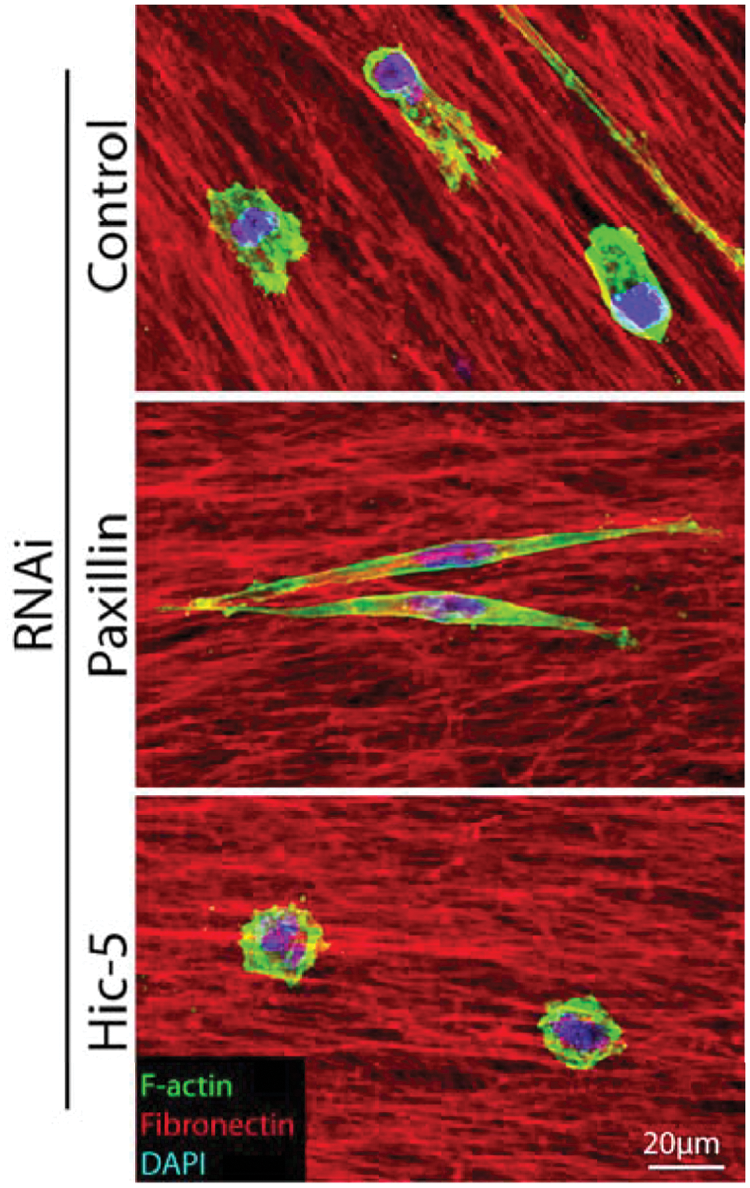
Paxillin and Hic-5 regulate breast cancer cell morphology in 3D environments to coordinate plasticity during invasion. Representative immunofluorescence images of MDA-MB-231 breast cancer cells invading through a 3D cell-derived matrix (CDM). Cells were fixed and stained for F-actin (green), fibronectin (red), and DAPI (blue). RNAi of paxillin promotes a highly elongated mesenchymal phenotype, whereas Hic-5 depletion induces the amoeboid phenotype. Importantly, depletion of either paxillin or Hic-5 prevents invasion, plasticity, and metastasis.
Paxillin is widely expressed in normal tissue, whereas Hic-5 and leupaxin expression appears more spatially restricted.24,25,27,29,32 A balance of Hic-5 and paxillin expression by tumor cells may be achieved through TGF-β–mediated upregulation of Hic-5.28,77 Intravital imaging has revealed that cells exhibiting constitutive TGF-β activation, and thus presumably elevated Hic-5 expression, invade readily as single cells and intravasate to promote distant metastasis. 111 Therefore, it is plausible that the efficiency of invasion and plasticity in vivo may be determined by spatiotemporally localized TGF-β–driven fluctuations in Hic-5 expression.
Control of Cell Survival and Apoptosis
In order for solid tumor cells to metastasize to distant sites, they must survive and proliferate outside their normal tissue microenvironment. These normally adhesive cells must also endure the lack of cell adhesion–related survival signals during the process of lymphovascular infiltration.124,125 Indeed, the ability of tumor cells to exhibit anchorage-independent growth correlates with their metastatic potential and thus malignancy. 126 Paxillin has been found to promote transformation and facilitate anchorage-independent cell growth through its interaction with the bovine form of the human papillomavirus (HPV) E6 protein. 127 The HPV virus is known to cause multiple malignancies, including cervical, oropharyngeal, and lung cancers,128-130 through promoting the degradation of the tumor suppressor p53. 131 Indeed, it has been suggested that nearly all cervical cancer patients are positive for HPV infection. 132
Interestingly, paxillin has also been implicated in regulating anchorage-independent growth and cell survival through its regulated tyrosine phosphorylation as well as by its direct interactions with p210BCR/ABL, FAK, and vinculin.67,133-136 Furthermore, paxillin is able to promote cell survival signaling through its direct interaction with the pro-survival, proto-oncogene bcl-2, 20 which is known to function to prevent apoptosis in the absence of cell adhesion in both normal and cancerous cells, in part through maintaining adhesion-related signaling through FAK.137,138 Importantly, neither Hic-5 nor leupaxin interacts with bcl-2 family members, although the former may regulate cell survival by other mechanisms, for example, through regulating the nuclear function of the pro-survival protein cyclin D1. 139 Furthermore, Hic-5 interacts with the heat shock protein 27 (hsp27) to increase cell death in response to heat shock. 140 Interestingly, the role of paxillin and Hic-5 in regulating cell survival represents another example of their reciprocal relationship, with paxillin predominantly functioning to promote cell survival, whereas increased Hic-5 expression appears to correlate with cell death in cancer and normal cell populations.63,140-143
Prognostic Marker and Therapeutic Potential
The elevated phosphorylation and/or expression of paxillin has been implicated as a prognostic marker for long-term patient survival in ovarian cancer, 144 breast carcinoma,58,145 hepatocellular carcinoma, 146 and lung adenocarcinoma. 39 However, it is noteworthy that elevated paxillin expression has also been shown to exhibit no, or a negative correlation with patient outcome and recurrence-free survival.147,148 In contrast to paxillin, the potential for Hic-5 and leupaxin as prognostic indicators is enigmatic, although it is important to note that increased leupaxin and reduced Hic-5 expression have been observed in prostate cancers and that Hic-5 may be linked to treatment responsiveness and recurrence-free survival.30,41,42,63,149Importantly, none of the above analyses concurrently assessed the relative levels of the other paxillin family members. Indeed, cell-based studies suggest that a balance of paxillin family members is critical for tumor cell survival, proliferation, and invasion. Taken together, these studies indicate the possibility that the expression of paxillin and Hic-5 is variable in different cancers and that their prognostic value may depend on their relative, rather than individual, levels of expression.
The single worst prognostic indicator in cancer is the presence of distant metastases, which is associated with decreased therapeutic effectiveness and the vast majority of cancer-related deaths. Therefore, paxillin and Hic-5 represent attractive targets for anti-metastatic drug development as their inhibition not only prevents breast cancer cell plasticity but also impairs invasion, transendothelial migration, and metastasis. 52 Furthermore, malignant cancer cells can often survive for extended time periods in a dormant state at distant secondary sites, only to begin proliferating long after treatment and removal of the primary tumor. 150 Given the ability of paxillin family members to regulate cell survival, as well as invasion mechanisms, they may also represent viable candidates for therapeutics targeting against these elusive dormant tumor cell populations and thereby improve patient long-term survival.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Studies from the authors’ laboratory are supported by grants from the National Institute of Health, (NIH) GM47607 and CA163296 (C.E.T), as well as a Susan G. Komen for the Cure Postdoctoral Fellowship (N.O.D).
