Abstract
Changes in the management of cancers such as colorectal cancer (CRC) are urgently needed, as such cancers continue to be one of the most commonly diagnosed cancers; CRC accounts for 21% of all cancers and is responsible for mortalities second only to lung cancer in the United States. A comprehensive science-driven approach towards markedly improved early detection/screening to efficacious targeted therapeutics with clear diagnostic and prognostic markers is essential. In addition, further changes addressing rising costs, stemming from recent health care reform measures, will be brought about in part by changes in how care is reimbursed. For oncology, the advances in genomics and biomarkers have the potential to define subsets of patients who have a prognosis or response to a particular type of therapy that differs from the mean. Better definition of a cancer’s behavior will facilitate developing care plans tailored to the patient. One method under study is episode-based payment or bundling, where one payment is made to a provider organization to cover all expenses associated with a discrete illness episode. Payments will be based on the average cost of care, with providers taking on a risk for overutilization and outliers. For providers to thrive in this environment, they will need to know what care a patient will require and the costs of that care. A science-driven “personalized approach” to cancer care has the potential to produce better outcomes with reductions in the use of ineffectual therapies and costs. This promising scenario is still in the future, but progress is being made, and the shape of things to come for cancer care in the age of genomics is becoming clearer.
Introduction
Advanced colorectal cancer (CRC) remains the third most common cancer in both men and women, with 141,210 new CRC cases diagnosed in the United States every year. Late-stage disease has a dismal survival rate: 45% of patients die of recurrence despite adjuvant therapy CRC. 1 Despite a long-standing slow decline in CRC incidence, costs associated with CRC care for the Medicare population are expected to increase 89% in constant dollars from 2000 to 2020. 2 This increase is driven in part by the rising median age of the population and higher cost of care, including chemotherapy. 3 In 2000, CRC care costs alone represented 3.4% of the total Medicare budget. 2 Most of this expense is concentrated in the first year following diagnosis (Fig. 1). 4 The health care debate that resulted in health care reform in the United States prompted The Lancet to point out that debates comparing British and American health systems became national pastimes on both sides of the Atlantic. 5 In CRC, the challenge of simultaneously saving lives and money while enacting reform could depend more on advances in a molecular genetics laboratory, whether in Helsinki or Houston or Tel Aviv, than in a clinical setting in New York, California, or any other state. In an ideal world, a simple blood test that identifies high-risk patients who would then undergo rigorous screening procedures to detect CRC before it reaches stage II or metastases would result in a cure. A first approach, from early detection to improved screening methods, could lead to the identification of high-risk populations and promotion of behavioral changes (e.g., diet modifications). Currently, the national guidelines indicate a 20% detection rate for adenomas in the general population through screening colonoscopic examinations. 6 Multiple studies indicate that this rate could be optimized by improved endoscopic techniques such as withdrawal times, imaging, and skilled endoscopists. 7 Optimizing these criteria can lead to markedly improved detection rates as well as the identification of high-risk patients, as in the Endoscopy Center at MD Anderson Cancer Center (Fig. 2). A comprehensive program analyzing tissues and blood for genomic markers to targeting later stage disease will be crucial. Such an approach will need to include defining predictive and prognostic biomarkers through genomics and proteomics models and developing mechanistic insight into new pathways that emerge from the data accrued.
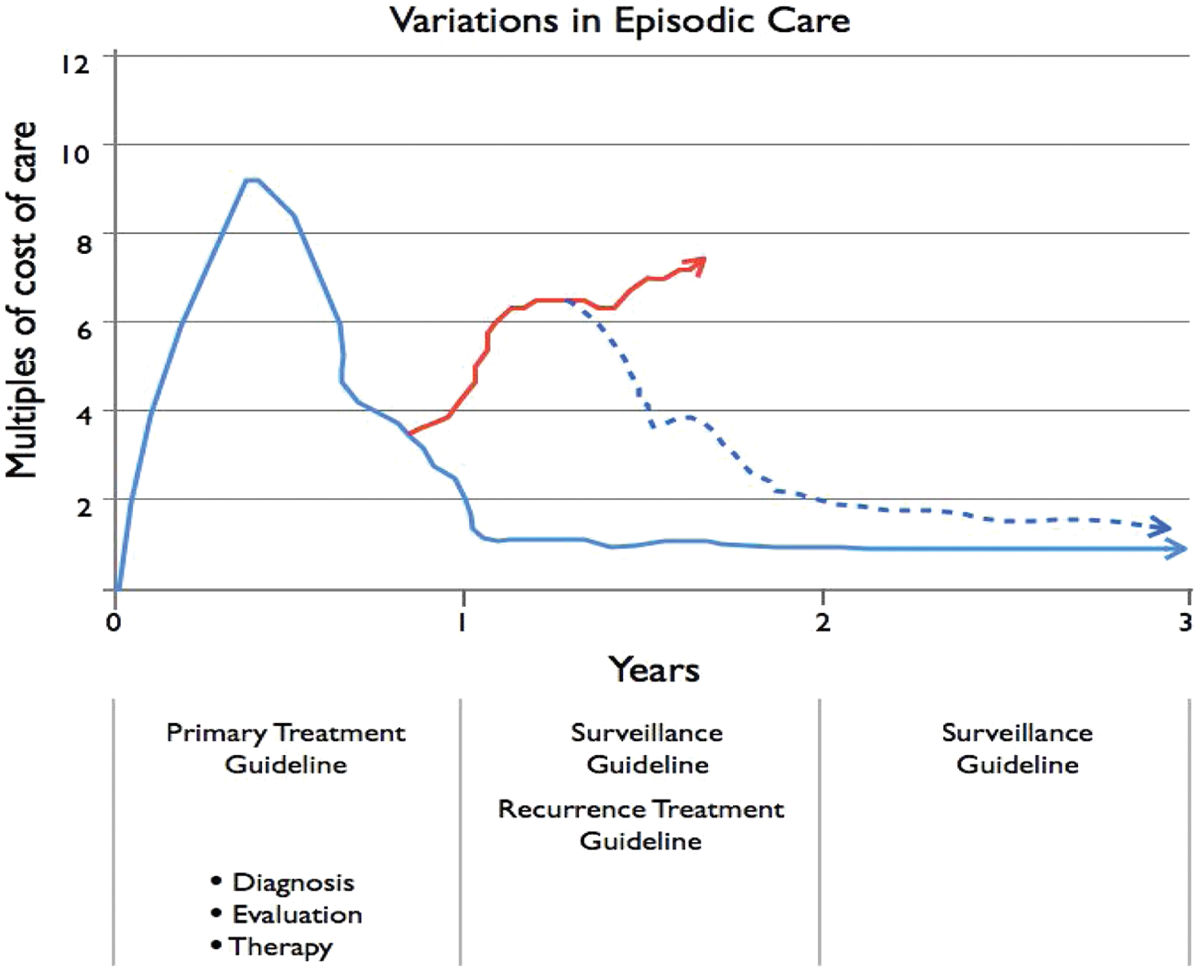
Challenge for bundling payments. In colorectal cancer, the period of greatest care provided and costs is the first year. In that year, costs may be approximately 10 times (year 1 with evaluation, diagnosis [Dx], and identification of primary) what they are in any other subsequent year. Accounting for the broad scope of patient comorbidities poses a challenge for bundling, as does predicting average outcomes. What percentage of cases will experience recurrence? What percentage of cases with recurrence will resolve and enter surveillance? Outliers among the cases may have an effect beyond the proportion of their numbers because of the cost of their care and the structure of bundling, which will require cooperating care providers to divide single payments. Understanding true costs that reflect variability in patient outcomes will be critical.
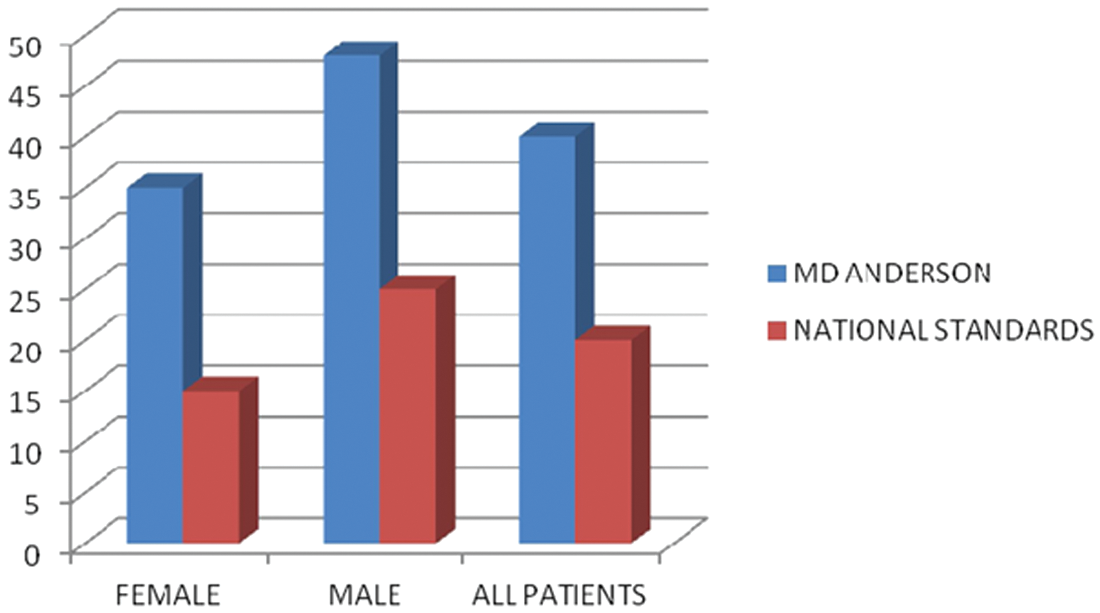
Adenoma detection rates by gender and overall in screening colonoscopies performed at MD Anderson Cancer Center in 2011 compared with current quality assurance standards.
Biomarkers’ Emerging Role in Enhancing Evidence-Based CRC Care
At its simplest level, the notion of bundling assumes that, whenever possible, there is evidence for performing a particular test or offering a specific therapy. In the case of biomarkers and responsiveness to chemotherapy, for example, the question becomes whether the difference in response rate is clinically relevant. Biomarkers in colon cancer have been shown to predict a response to chemotherapeutic regimens and allow more effective use of expensive agents. 8 However, the full potential of biomarkers to impact clinical care has yet to be realized.9,10 Recent work has demonstrated the complexity of genetic alterations associated with colon cancer and the need to test tumors for an array of biomarkers. 11 The development of biological chemotherapeutic agents that are highly targeted has offered major potential to improve responses and overall survival. One such agent, cetuximab, is an epidermal growth factor receptor (EGFR), which is a targeting monoclonal antibody. Because of this relative specificity, it has been appropriate to investigate mediators of EGFR activity. The RAS family of genes encodes GTP-binding proteins, and KRAS is a key effector of bound EGFR. KRAS primarily signals through BRAF and MAPK. Several studies of the efficacy of EGFR-targeting monoclonal antibodies have thus had occasion to evaluate the role of these mediators. Stratified analysis of the CRYSTAL (Cetuximab Combined with Irinotecan in First-Line Therapy for Metastatic Colorectal Cancer) trial showed a survival advantage with the addition of cetuximab to the existing multidrug regimen, but the clinical activity of cetuximab was limited to patients with wild-type KRAS. 12 Since KRAS is mutated in 30% to 50% of CRC, this large group of patients is not responsive to cetuximab. These findings were confirmed in the OPUS trial (Oxaliplatin and Cetuximab in First-Line Treatment of Metastatic Colorectal Cancer). 13 A position statement from the American Society of Clinical Oncology has essentially elevated this issue to one of standard of care by recommending routine testing of tumors from patients with advanced CRC for evidence of mutations in KRAS, going on to recommend against cetuximab administration in those with such mutations. 14 The European Society for Medical Oncology reached a similar conclusion. 15
Studies examining the effect of KRAS mutation status in treatment response have also evaluated the effect of the presence or absence of mutations in the BRAF gene. A lower frequency of mutations in BRAF has precluded definitive judgment, but there is a tendency toward poorer response in those with BRAF mutations, and this includes patients who would otherwise be considered responsive because of the presence of wild-type or nonmutated KRAS. In the phase III CAIRO2 study, mutated BRAF in tumors was a marker of poor prognosis in patients with metastatic CRC who received capecitabine, oxaliplatin, and bevacizumab, with or without cetuximab.16-18
Colorectal tumors arise from the modulation of multiple genes by mutations, epigenetic regulation, glycosylation, and microRNAs and transform from benign to advanced cancer over a 17-year period, yet these tumors take less than 2 years to metastasize.19,20 Hereditary cancer syndromes have provided powerful insights into our understanding of somatic mutations present in sporadic cancers as well as implicated cell signaling pathways.21-25 One clear example is the identification of germline, inactivating mutations in the APC gene, which encodes a 300-kD wnt pathway adaptor protein. 24 Although germline mutations in APC are responsible for familial adenomatous polyposis, a rare condition affecting about 1 in 7,000 individuals in the United States, somatic mutations in the APC gene are present in more than 70% of colonic adenomatous polyps and carcinomas. 25 Hereditary nonpolyposis colon cancer (HNPCC), or Lynch syndrome, involves a germline mutation in one of the DNA mismatch repair (MMR) genes, leading to defective DNA repair. Recent studies identifying inactivation of TGF-β signaling through loss of the Smad3 adaptor β-2 spectrin could potentially provide new insights into CRC development from stem-like tumor-initiating cells (STICs) for targeted chemoprevention. While more than 90% of tumors are considered to be clonal and arise from a STIC, the precise identification and mechanism for STIC formation remain poorly understood. Moreover, a functional role for TGF-β signaling in the intestinal stem cell niche has been demonstrated by the association of SMAD4 and BMPR1A/ALK-3 mutations with human juvenile polyposis syndrome. Studies delineating key functional pathways in colorectal STIC formation could help us effectively prevent the disease. The rapid advent of relatively inexpensive whole genome sequencing could, in the future, enable us to detect CRC early and treat it effectively, potentially reducing adverse events as well as the financial burden of late-phase treatment.
MMR Proficiency, Prognosis, and Treatment Response
Long before the MMR genes responsible for HNPCC were identified in 1993, survival data had suggested that HNPCC patients experienced a better outcome, stage for stage, than did sporadic CRC patients.26,27 Following discovery of the role of MMR in HNPCC, it became apparent that HNPCC tumors and about 15% of tumors from nonfamilial or sporadic cases of CRC showed microsatellite instability.26,28 Whether because of greater immunogenicity or other factors, tumors having high microsatellite instability (MSI-H) were associated with a better prognosis.
More recently, and as noted above in the discussion of KRAS-related treatment outcome differences, studies of treatment response came to include post hoc analyses in which responses were stratified according to various markers, including MSI. A smaller proportion of patients with advanced disease have evidence of MSI, so most data are limited to the adjuvant setting of patients with stage II and III disease. Such studies showed that, as expected, patients with MSI-H tumors had a better prognosis. However, the same studies found MSI-H tumors to be less responsive to traditional therapy with 5-fluorouracil (5-FU) than those with microsatellite-stable tumors.29,30
Here, it is important to distinguish between patterns in stage II and III disease. A recent pooled analysis from several adjuvant trials that included MSI data showed no benefit to stage II patients with MSI-H tumors; however, the more frequently occurring stage III setting differed in that a 5-FU benefit was seen in patients with MSI-H tumors when these patients were compared to controls not treated with 5-FU. Notably, in this analysis, the benefit appeared limited to those patients with MSI-H tumors and probable HNPCC, with sporadic cases (as evidenced by the presence of BRAF mutations) failing to benefit from 5-FU–based adjuvant therapy. 31 The findings appear to support the prevailing recommendation that stage II MSI-H colon cancer cases should not receive 5-FU–based adjuvant therapy but that stage III MSI-H cases should.
These data on prognosis and (un)responsiveness to certain treatments in patients with MSI-H tumors compose one part of a growing sentiment for the routine, even universal, use of MSI testing in newly diagnosed CRC. 32 The other part of the argument for routine MSI testing is the potential for diagnosing HNPCC in patients in whom such clues as family history positivity or early age at diagnosis are lacking. Several cost analysis studies have been done and appear to support universal MSI testing, driven primarily by the importance of detecting HNPCC. 33 It would be of interest to see more complex modeling performed in which both HNPCC detection and treatment planning were considered.
Biomarkers therefore represent an active area of research that could enhance our ability to stage, treat, and prognosticate. 34 Biomarkers associated with prognoses that vary from the norm have been described.35,36 Patients with colon cancer can now be classified by the molecular profile of their tumor. 37 As clinical data accumulate, these various profiles can be associated with well-defined responses to therapy and prognosis. 38 This subclassification of the CRC population would allow finer determination of the care required and the cost to be incurred, making episode-based payment more acceptable to all parties. What is more important is that such well-defined treatment regimens (“personalized medicine”) would reduce the variability and waste seen in current practice while producing better patient outcomes. 39
Health Care Reform and Bundled Payments
Will these advances in our understanding of the molecular pathogenesis of CRC facilitate adapting to a different payment model for CRC care? The Patient Protection and Affordable Care Act of 2010 included provisions for Medicare to study different payment models to reimburse providers. The current fee-for-service system fosters high-volume care with no positive incentive to provide quality care. 40 New payment models being considered have 2 general aims: to reduce cost and to enhance quality. One such model is episode-based payment, or bundling, in which one payment is made to a provider organization to reimburse for all the care involved in a discrete episode of illness, for example, newly diagnosed CRC. The payment is based on the average cost of an evidence-based treatment plan rendered over a defined period of time. The one payment is divided among the providers of care: hospital, surgeon, pathologist, radiologist, oncologist, and other providers. This straightforward concept runs into a myriad of difficulties in implementation from the onset. 41 How is an episode defined? How is the payment determined? Does that determination take into account comorbidities or other risks that have the potential to increase costs? How is the reimbursement to be divided among the providers and the various settings where care is provided? 42
From the provider side, it becomes apparent that there has to be a level of coordination of care far beyond what is generally practiced and a level of financial integration that is rare. Also, providers will have to understand their true costs to provide a service, vital information that their current cost accounting systems frequently are not able to provide. 43 Finally, providers need to have a complete understanding of the prognosis and most effective therapy for a given patient. The legislation encourages the development of “accountable care organizations” (ACOs) that will be able to provide services to the patient through the entire continuum of care for an episode and be responsible for costs incurred. The ACO will take on the responsibility of total care for the episode as well as the financial risk if excessively high costs are incurred. For an episode-based payment system to work, the data on patient care costs have to be highly developed so as to accurately reflect changes in expenses that occur when patients have comorbidities and suffer a severity of disease different from the average. If payments fail to reflect this variability in costs among patients with the same condition, then providers run the risk of being inadequately compensated for costs incurred, and the system will ultimately fail.
The aforementioned health care reform bill allocated $10 billion over 9 years for establishing a Center for Medicare and Medicaid Innovation, whose task will be to study various payment models. Episode-based payments are just one of the models under study, but one with a track record, albeit a limited one. Such a system transfers the risk of incurring excess costs from outliers or complications to the provider. Risk of increasing incidence of the condition being bundled remains with the payer. 44 Episode-based payments are believed to be potentially applicable from half to up to two thirds of total health care costs. 45 A prior pilot study of episode-based payment was carried out in the 1990s at a handful of centers for coronary artery bypass grafting. The results demonstrated cost savings, but the program was not extended. 46 Current legislation insulates the new pilot programs from political interference, as funding is long term and the secretary of Health and Human Services (HHS) sets the ground rules. The Obama administration has filled leadership positions in HHS with advocates of highly integrated health care organizations like ACOs that would facilitate adoption of an episode-based payment scheme. 47 This push to foster greater integration echoes similar attempts in the past and shares with these prior movements the hope that numerous benefits will flow from better coordination between various providers.
Benefits from integration are readily documented outside of health care; however, previous attempts at vertical integration in health have not been shown to have any consistent benefit in terms of quality or costs.48-50 So while bundled payments appear to be a future reality, the organizational infrastructure to make it work effectively is largely not in place, and the benefits in terms of producing low-cost quality care are speculative. Therefore, with these caveats in mind, how do these impending changes impact care for the patient with CRC?
Bundling in CRC Care
Regardless of the role of tumor markers in prognosis and tumor treatment responsiveness, it will be increasingly necessary to bundle episodic cancer treatment in a fashion similar to that considered for other major diseases. One element in the provision of bundled services is the use of clinical care pathways (CCPs). Use of such pathways is helpful in characterizing usual and expected processes and procedures for treatment plans that are both high volume and complex. Regular adherence to such CCPs for the management of other cancers has been demonstrated to lead to performance improvement, resulting in shorter length of stay and lower cost of disease management. 51
What remains the same in the midst of change is that while our understanding grows of the molecular processes involved in CRC, the translation into routine clinical practice will take time. 52 Bundling of payments may arrive before the data emerge that will allow individualized therapy and fine prognostic precision. As episode-based payments are likely to be based on average costs of large subsets of undifferentiated patients with condition X, the provider organization assuming the risk must have a group of patients with the condition of sufficient size so that 1 or 2 outliers do not skew their costs to such an extent as to produce a deficit. The optimal patient population for an ACO will vary depending on the condition. The need to have sufficiently large risk pools to mitigate the risk of outliers will be countered by the loss of economies of scale in administrative costs as the organization grows.44,53 Finances will drive the movement to ever larger integrated provider organizations to act as ACOs in the episode-based payment scheme. We can anticipate greater scrutiny of costs and more emphasis on quality measures. While in the business world, low costs and high quality are often compatible, in health care, the 2 primary aims of the new payment model may not be complementary and may be in competition. 54 Challenging times are ahead, but the advances in biomarkers may reduce the uncertainty about diagnosis, treatment, and prognosis of CRC to the benefit of all concerned, whether they look east across the Atlantic or west.
As this article goes to press, recent events emphasize the questions confronting the implementation of alternative payment plans and the ultimate utility of genetic profiling of tumors. The Supreme Court has just heard arguments regarding the constitutionality of the universal mandate intrinsic to the Obama health care reform. 55 In addition, the genetic heterogeneity within a tumor has been highlighted in a recent The New England Journal of Medicine article. 56 The implications of this intratumoral heterogeneity for personalized medicine are profound and not favorable. 57 Nevertheless, the parsing of the genetic alterations of cancer will improve our understanding of the process of malignant transformation and will ultimately yield useful clinical tools.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Institutes of Health grants RO1 CA106614-06 (L. Mishra), RO1 CA042857-06 (L. Mishra), PO1 CA130821-04 (L. Mishra), RC2 AA019392 (H. Tsukamoto), P30 CA016672-36 (R. DePinho), and P30 DK56338-10 (M. Estes).
