Abstract
Prostate cancer is a frequently occurring disease and is the second leading cause of cancer-related deaths of men in the United States. Current treatments have proved inadequate in curing or controlling prostate cancer, and a search for agents for the management of this disease is urgently needed. Survivin plays an important role in both progression of castration-resistant prostate cancer and resistance to chemotherapy. Altered expression of survivin in prostate cancer cells is associated with cancer progression, drug/radiation resistance, poor prognosis, and short patient survival. In the present study, the authors performed a cell-based rapid screen of the Prestwick Chemical Library consisting of 1120 Food and Drug Administration–approved compounds with known safety and bioavailability in humans to identify potential inhibitors of survivin and anticancer agents for prostate cancer. Sanguinarine, a benzophenanthridine alkaloid derived primarily from the bloodroot plant, was identified as a novel inhibitor of survivin that selectively kills prostate cancer cells over “normal” prostate epithelial cells. The authors found that sanguinarine inhibits survivin protein expression through protein degradation via the ubiquitin-proteasome system. Sanguinarine induces apoptosis and inhibits growth of human prostate cancer cells and
Introduction
Prostate cancer is a frequently occurring disease and is the second leading cause of cancer-related deaths of men in the United States. About 1 out of every 6 men will be diagnosed with prostate cancer in their lifetime. Current treatments have proved inadequate in curing or controlling prostate cancer, and searching for agents for the management of this disease has become a priority for researchers.
Survivin is a unique member of the inhibitor of apoptosis (IAP) protein family.
1,2
In addition to the observation that its expression appears to be both cell cycle dependent
3
and independent,
4,5
survivin has been shown to be involved in both control of cell division
6
and inhibition of apoptosis. It is highly expressed in all human cancers but is undetectable in most normal adult tissues.
7
Survivin is the top fourth “transcriptome” universally enhanced in cancer tissues compared to normal tissues in genome-wide searches.
8
Accumulating data indicate that altered expression and/or function of survivin in cancer cells are associated with cancer progression, drug/radiation resistance, poor prognosis, and short patient survival.
2
Importantly, although some human normal tissues or cells with proliferative potential express survivin, its expression level is usually very low, and less than 5% of cells in the tissues show positive.
9
More important, growing evidence indicates that the regulation and/or functional mechanism of survivin in normal cells versus cancer cells appears to be different.
10
All these characteristics about survivin make it an important cancer therapeutic target without induction of drug toxicity to normal tissues. Antisense oligonucleotides,
6,11-13
dominant-negative mutants,
6,14-18
ribozymes,
19,20
triplex DNA formation,
21
RNA interference,
22,23
and pharmacological inhibitors have been used
There is increasing evidence suggesting that survivin plays an important role in both progression of castration-resistant prostate cancer and resistance to chemotherapy. 26-30 Several small-molecule inhibitors and natural compounds that suppress survivin expression have been developed and shown to be effective in suppressing prostate cancer tumor growth and enhancing taxotere-induced apoptosis. 31,32 Therefore, targeting survivin signaling may be an effective therapeutic approach for castration-resistant prostate cancer.
In the present study, we performed a cell-based rapid screen of the Prestwick Chemical Library consisting of 1120 Food and Drug Administration (FDA)–approved compounds with known safety and bioavailability in humans to identify potential survivin inhibitors and anticancer agents for prostate cancer. Sanguinarine was identified as a novel inhibitor of survivin. Sanguinarine, which is derived primarily from the bloodroot plant, induces apoptosis and inhibits tumor formation and growth of human prostate cancer cells.
Results
Rapid Compound Screening
In an attempt to identify novel potential therapeutic agents for prostate cancer, DU145 human prostate cancer cells that express high levels of survivin were treated with compounds from the Prestwick Chemical Library (Illkirch, France) for 24 h. The Prestwick Chemical Library consisted of 1120 FDA-approved compounds with known safety and bioavailability in humans. Over 85% of the compounds in the library are off-patent drugs that are marketed in a wide range of therapeutic areas. DU145 cell viability was detected using the MTS assay. PZ-HPV7 immortalized prostate epithelial cells were used as “normal” cell control because they are nontumorigenic when injected into nude mice. Compounds that decreased DU145 cell viability by >50% but decreased PZ-HPV7 cell viability by <10% were selected for further characterization. One of the selected compounds is sanguinarine. Sanguinarine (13-methyl[1,3]benzodioxolo[5,6-c]-1,3-dioxolo[4,5-i]phenanthridinium) (Fig. 1A), which is derived primarily from the bloodroot plant, is a benzophenanthrene alkaloid. It has been shown to possess antimicrobial, antioxidant, anti-inflammatory, and antitumor properties and is widely used in toothpaste and mouthwash to prevent/treat gingivitis and other inflammatory conditions of the mouth. 33-35
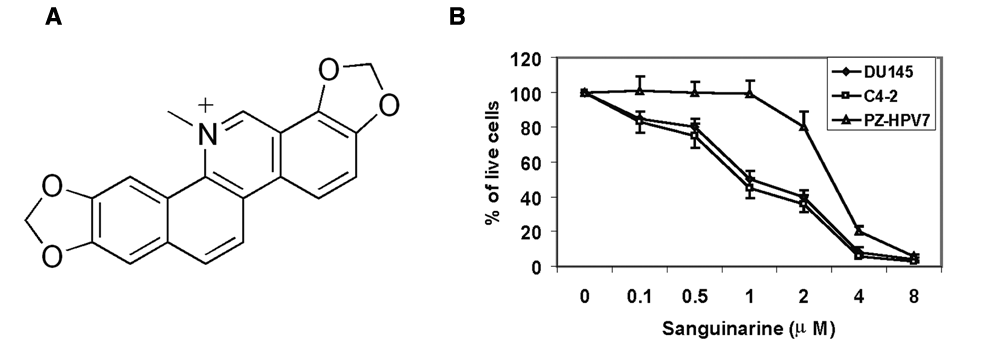
Sanguinarine inhibits the growth of prostate cancer cells. (
Sanguinarine Selectively Kills Prostate Cancer Cells over “Normal” Cells
To further characterize the efficacy of sanguinarine in inhibiting the growth of prostate cancer cells, we treated prostate cancer cell lines C4-2 and DU145 and immortalized PZ-HPV7 prostate epithelial cells with different concentrations of sanguinarine. The number of live cells was counted after 24 h of treatment. As shown in Figure 1B, although 1 µM sanguinarine killed ~50% of the C4-2 and DU145 cells, it did not affect the growth of PZ-HPV7 cells. The IC50 for the PZ-HPV7 cells was about 3 times higher than that for C4-2 and DU145 cells, suggesting that sanguinarine selectively kills cancer cells versus “normal” cells. However, most of the cells were killed at 4 µM concentration.
Sanguinarine Sensitizes DU145 Cells to Paclitaxel Toxicity
Paclitaxel is a chemotherapy drug classified with the taxane group and used in the treatment of advanced prostate cancer and recurrent prostate cancer. We then tried to examine whether sanguinarine would facilitate cytotoxicity of paclitaxel in prostate cancer cells. DU145 cells were treated with 5 nM paclitaxel with or without 0.5 µM sanguinarine for 48 h. Although sanguinarine at 0.5 µM had little inhibitory effect on cell growth, the combination of 5 nM taxol and 0.5 µM sanguinarine significantly decreased cell proliferation (Fig. 2A). To further validate the effect of sanguinarine on paclitaxel sensitization, we performed a cell death enzyme-linked immunosorbent assay (ELISA), detecting internucleosome degradation of genomic DNA. As shown in Figure 2B, compared with paclitaxel alone or sanguinarine alone, the amount of apoptosis was significantly increased by the paclitaxel and sanguinarine combination treatment. Sanguinarine induces apoptotic cell death in a dose-dependent manner in both C4-2 and DU145 cells (Fig. 2C).
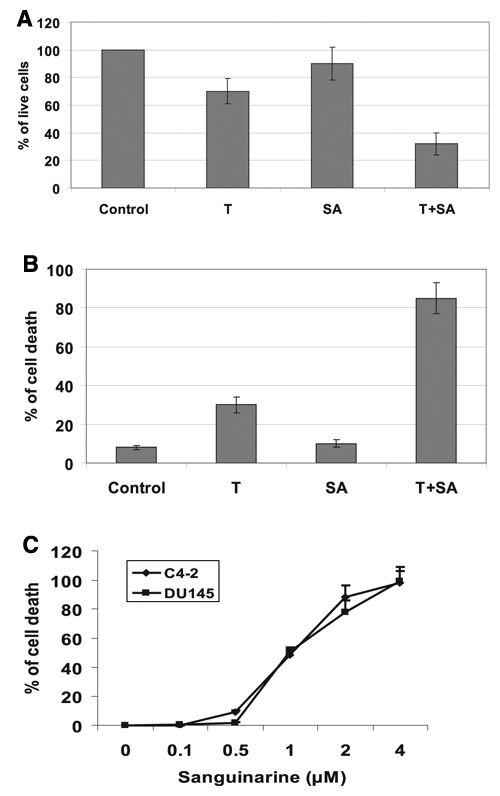
Sanguinarine sensitizes DU145 cells to paclitaxel-mediated growth inhibition. DU145 cells were treated with 5 nM paclitaxel (T) with or without 0.5 µM sanguinarine (SA) for 48 h. (
Sanguinarine Decreases Survivin Protein Expression and Increases Cleavage of Poly(ADP-Ribose) Polymerase
Survivin is a unique member of the IAP family. To examine whether sanguinarine affects survivin expression, DU145 cells expressing high levels of survivin protein were treated with sanguinarine, and the levels of survivin protein were analyzed by Western blot using antibodies against survivin. As shown in Figure 3A, sanguinarine decreases the protein expression of survivin in a dose-dependent manner but has no effect on the levels of Bcl-2 and XIAP proteins. The levels of survivin protein started to decrease at 30 min after sanguinarine treatment (Fig. 3B). Sanguinarine induced strong cleavage of poly(ADP-ribose) polymerase (PARP) at 5 µM concentration (Fig. 3A), suggesting that sanguinarine induces apoptotic cell death. Because normal immortalized PZ-HPV7 prostate epithelial cells are insensitive to sanguinarine treatment compared to DU145 and C4-2 prostate cancer cells, we determined whether sanguinarine treatment regulates survivin expression in PZ-HPV7 cells. As shown in Figure 3C, sanguinarine treatment did not change the levels of survivin protein expression in PZ-HPV7 cells, suggesting that the degree of survivin degradation may be responsible for the tumor-specific cytotoxic effects of sanguinarine.

Sanguinarine decreases survivin protein expression and induces cleavage of poly(ADP-ribose) polymerase (PARP). (
Sanguinarine Decreases Survivin Protein Stability by Enhancing Protein Degradation
We then studied how sanguinarine decreases survivin expression. First we examined the effects of sanguinarine on survivin expression at the transcriptional level. LNCaP cells were transfected with a survivin promoter luciferase reporter construct, pLuc-1430, following increasing doses of sanguinarine treatment for 24 h. As shown in Figure 4A, sanguinarine did not affect promoter reporter luciferase activity, suggesting that sanguinarine did not affect survivin expression at the transcriptional level. This experiment was repeated in C4-2 and DU145 cells, and the data remained consistent (not shown). We then used real-time PCR to determine the effect of sanguinarine on the level of survivin mRNA. Consistent with luciferase data, sanguinarine did not affect survivin mRNA expression (data not shown). These data indicate that downregulation of survivin expression by sanguinarine is not at the transcriptional level.
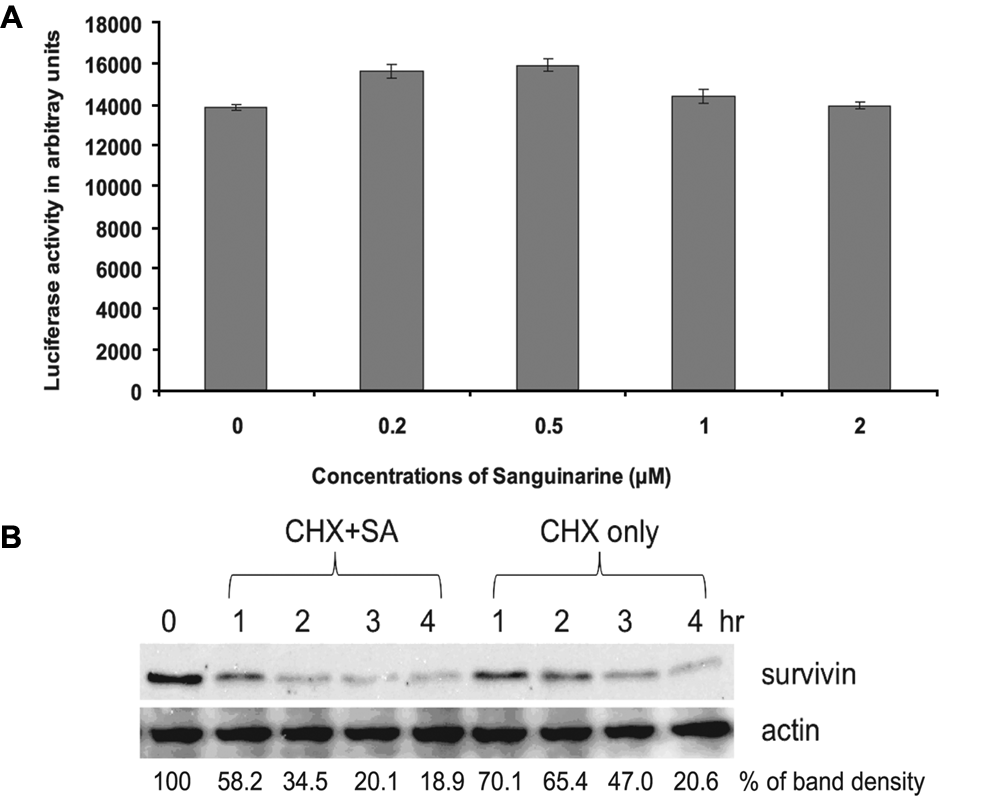
(
We next examined the effect of sanguinarine on survivin protein degradation after new protein synthesis was blocked by cycloheximide as a potential mechanism for downregulation of survivin protein level. The protein synthesis inhibitor cycloheximide (50 µg/mL) was added with or without 2 µM sanguinarine at time 0. At specified time points, cells were harvested, and the levels of survivin protein were measured by Western blot using antibodies specific against survivin. In sanguinarine-treated cells, the half-life of survivin protein was reduced to 1 h from 4 h in the control cells (Fig. 4B), suggesting that survivin protein degradation was greatly enhanced in the presence of sanguinarine.
Sanguinarine Enhances Survivin Degradation through the Ubiquitin-Proteasome System in DU145 Cells
Systematic protein degradation by the ubiquitin-proteasome system plays an important role in the maintenance of protein stability. Protein ubiquitination provides the recognition signal for the 26S proteasome, leading to protein degradation. 36,37 The ubiquitin-proteasome pathway plays a central role in regulating important cellular processes such as cell cycle progression and apoptosis. Survivin protein degradation is mediated through the ubiquitin-proteasome system, as shown in the human embryonic kidney cells. 38 We therefore treated DU145 cells with the proteasome inhibitor MG132 in the presence and absence of sanguinarine. As expected, MG132 was able to partially reverse the degree of inhibition of survivin expression by sanguinarine, as shown in Figure 5A, suggesting that sanguinarine induced survivin degradation via a proteasome-dependent pathway. We then examined the ubiquitination status of survivin with or without sanguinarine. Figure 5B shows that the ubiquitination of survivin protein is significantly increased in cells treated with 2 µM sanguinarine (in the presence of MG132). These data suggest that sanguinarine enhances survivin degradation via the ubiquitin-proteasome system.
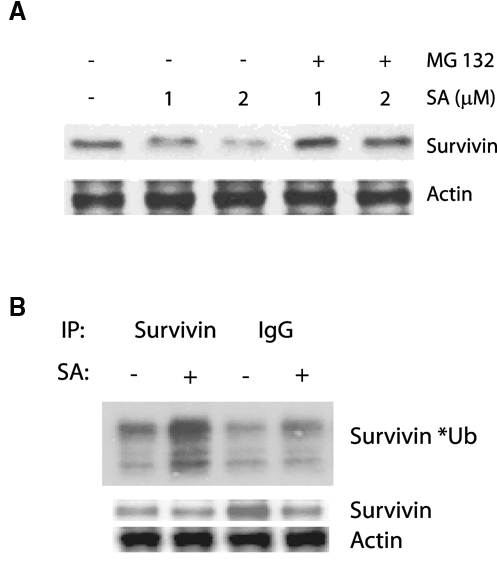
Sanguinarine enhances survivin degradation through the ubiquitin-proteasome pathway. (
Sanguinarine Inhibits DU145 Xenograft Growth in Nude Mice
To determine whether sanguinarine is capable of suppressing prostate tumor growth
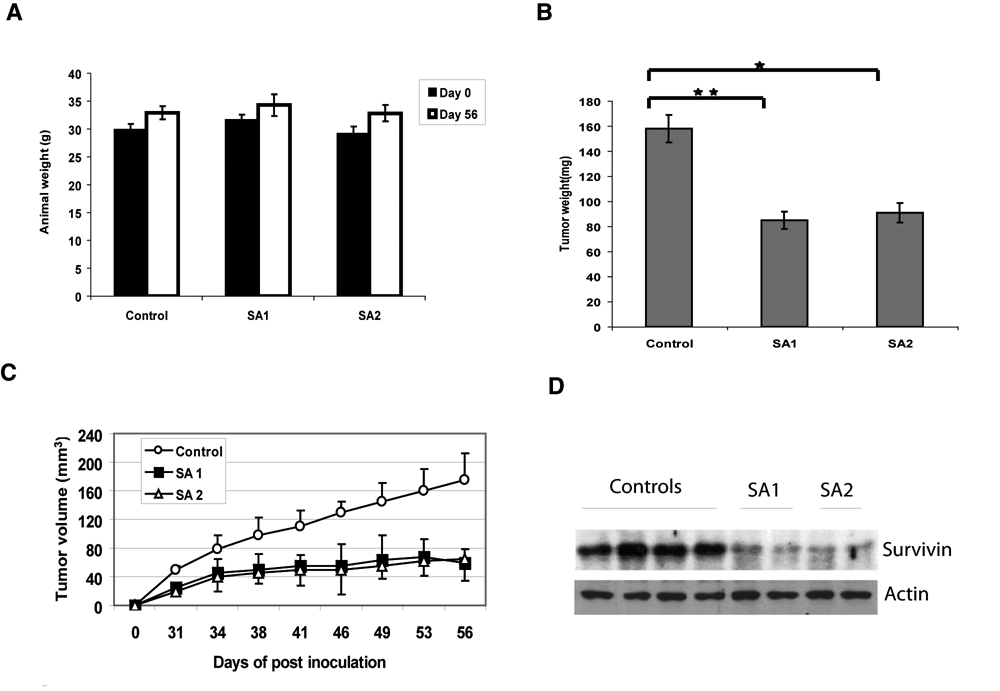
Sanguinarine (SA) suppresses DU145 xenograft growth in nude mice. Male nude mice were injected subcutaneously with 3 × 106 cells/flank of DU145 cells. The mice were randomly divided into 3 groups with 10 mice each. One group received vehicle only as a control, one group received dose of sanguinarine (0.25 mg/kg of body weight, SA1), and the third group received dose of sanguinarine (0.5 mg/kg of body weight, SA2). The treatment (intraperitoneal injection) was performed every other day starting 3 days post cell inoculation for 7 weeks. (
Discussion
In the present study, we performed a cell-based rapid screen of the Prestwick Chemical Library consisting of 1120 FDA-approved compounds with known safety and bioavailability in humans to identify potential anticancer agents for prostate cancer. Sanguinarine was identified as a potential inhibitor of survivin that selectively kills prostate cancer cells over “normal” prostate epithelial cells. Sanguinarine, which is derived primarily from the bloodroot plant, induces apoptosis and inhibits tumor formation and growth of human prostate cancer cells. Administration of sanguinarine, beginning 3 days after ectopic implantation of DU145 human prostate cancer cells, delays tumor formation by 2 weeks and reduces both tumor weight and volume. In addition, sanguinarine inhibits survivin protein expression through protein degradation via the ubiquitin-proteasome system.
The benzophenanthrene sanguinarine is a naturally occurring alkaloid present in plants such as
Hormone refractory prostate cancers are treated with paclitaxel. Previous study has revealed that paclitaxel (Taxol) transcriptionally upregulates survivin expression independent of G2/M arrest, and knocking down survivin expression by specific RNAi could sensitize MCF-7 cells to paclitaxel-induced apoptosis.
48
Because sanguinarine inhibits survivin expression, it is possible that sanguinarine could enhance paclitaxel anticancer activity by suppressing paclitaxel-induced survivin expression in prostate cancer. Our data support this hypothesis by showing that sanguinarine sensitizes paclitaxel-mediated growth inhibition and apoptosis. Although the efficacy of combination treatment was evaluated
In conclusion, we have identified sanguinarine as a potential inhibitor of survivin and an anticancer agent for prostate cancer by a cell-based rapid screen of the Prestwick Chemical Library that consists of 1120 FDA-approved compounds. Sanguinarine exhibits anticancer efficacy both
Materials and Methods
Reagents and Cell Culture
LNCaP, DU145, and C4-2 cells were maintained in RPMI 1640 supplemented with 10% fetal bovine serum (FBS) and 100 U/mL penicillin and 0.1 mg/mL streptomycin (PS). PZ-HPV7 cells were cultured in Keratinocyte-SFM medium supplemented with L-glutamine, epidermal growth factor (EGF), and bovine pituitary extract (BPE). The cells were maintained at 37°C in a humidified incubator with 5% carbon dioxide. The Prestwick Chemical Library, consisting of 1120 off-patent FDA-approved compounds, was purchased from Prestwick Chemical, France. Sanguinarine chloride was purchased from Sigma-Aldrich (St. Louis, MO) and dissolved in methanol for
Compound Screening
DU145 and PZ-HPV7 cells were seeded onto 96-well plates at 5000 cells per well (200 µl) and allowed to grow for 24 h. The cells were then treated with the compounds at a final concentration of ~5 µM and incubated at 37°C for another 24 h. The CellTiter 96 Aqueous One Solution Cell Proliferation Assay, MTS (Promega, Madison, WI) was used to detect cell viability according to the manufacturer’s instructions.
Luciferase Reporter Assay
The survivin promoter luciferase reporter construct pLuc-1430 was generated as described previously. 49 Cells were plated in 12-well plates (1 × 105 cells per well) and grown to about 70% confluence. The cells in each well were transfected with 0.5 µg of pLuc-1430 in the presence of Tfx-20 (Promega) in serum-free RPMI 1640 medium. After 1 h, the medium was removed, and the cells were cultured in regular RPMI 1640 with 10% FBS. On the next day, the cells were treated with various doses of sanguinarine for 24 h. Cell lysates were used for measurement of luciferase activity in a luminometer.
Real-Time PCR Analysis
Total RNA was extracted using Trizol (Invitrogen, Carlsbad, CA) reagent. Then, 1 µg of RNA was digested with DNase and reverse transcribed with random primers and Im-Prom II Reverse transcriptase (Promega). The cDNA was subjected to RT-PCR using specific primers for survivin (forward, 5′-AGAACTGGCCCTTCTTGGAGG-3′; reverse, 5′-CTTTTTATGTTCCTCTATGGGGTC-3′) and β-actin (forward, 5′-CCCAGCCATGTACGTTGCTA-3′; reverse, 5′-AGGGCATACCCCTCGTAGATG-3′). Quantitative real-time RT-PCR was performed in 25 µL reaction mixture with 12.5 µL SYBR Green (Bio-Rad, Hercules, CA), 1 µL of 5′ primer, and 1 µL of 3′ primer. Expression levels of survivin were normalized to β-actin. The experiment was repeated with duplicates 3 times.
Western Blot Analysis
Whole-cell protein extracts were resolved on 12% or 15% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). Proteins were then transferred to nitrocellulose membrane. After blocking for 1 h at room temperature in 5% milk in phosphate-buffered saline (PBS)/0.1% Tween-20, membranes were incubated overnight at 4°C with the indicated primary antibodies. Following secondary antibody incubation, immunoreactive proteins were visualized with an enhanced chemiluminescence detection system (Amersham Pharmacia Biotech, Buckinghamshire, England).
Ubiquitination Assay
MG132 was used to block proteasome-mediated degradation of ubiquitin-conjugated proteins. DU145 cells were treated with 5 µM MG132 in the presence or absence of 2 µM sanguinarine for 4 h. Cells were lysed in a buffer containing 10 mM HEPES (pH 7.9), 250 mM NaCl, 1% NP-40, 1 and mM EDTA with the addition of protease inhibitors (Roche, Basel, Switzerland). The whole-cell lysates were precipitated with antibodies against survivin or IgG overnight. The precipitates were collected by incubation with protein A/G-agarose, washed, eluted with SDS sample buffer, resolved on SDS-PAGE, and transferred to nitrocellulose membranes. Ubiquitin-conjugated survivin was visualized by blotting the membranes with antiubiquitin antibodies. The membranes were then blotted with antisurvivin antibodies to confirm the presence of survivin.
Cell Growth Assay
C4-2, DU145, or PZ-HPV7 cells were seeded on 12-well plates at a density of 1 × 105 cells/well in RPMI 1640 media containing 10% FBS. The cells were treated with increasing doses of sanguinarine (0.1-10 µM), and live cells were counted using Trypan blue exclusion after 24 h.
Cell Death ELISA
C4-2 or DU145 cells were seeded on 12-well plates (1 × 105 cells/well) and treated with vehicle alone or different doses of sanguinarine or different combinations of sanguinarine and paclitaxel in RPMI 1640 with 10% FBS. The treatment lasted for 48 h. Mono- and oligonucleosomes in the cytoplasmic fraction were measured at 405 nm using the Cell Death Detection ELISA kit (Roche).
In Vivo Tumor Growth Assay
DU145 prostate cancer cells were used for this experiment. Thirty male nude mice were randomly arranged into 3 groups. The mice were inoculated subcutaneously in the right and left flanks with 3 × 106 viable cells. Three days after injection, one group of mice was treated with 0.5 mg/kg body weight sanguinarine, another group of mice was treated with 0.25 mg/kg sanguinarine, and mice in the control group were injected with 10% DMSO. Treatment was performed intraperitoneally every other day for 7 weeks. Body weights were measured before and after the treatment. Tumor volume measurements were taken twice per week after visible tumors appeared. Animals were sacrificed 7 weeks after initial injections, and a final measurement of tumor volume was taken. The volume of the growing tumors was estimated by measuring their dimensions (length × width × depth) with calipers. Tumors were harvested and frozen in liquid nitrogen. Tumor proteins were extracted as described previously. The level of survivin was determined by Western blot.
Statistics
Data are shown as mean ± SD. The tumor weights were compared by one-tailed Student
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
This work was supported in part by grants from National High Technology Joint Research Program of China 2006DFB32330, NIH CA118887, and CA109441.
