Abstract
During development, angiogenesis occurs as a controlled series of events leading to neovascularization that supports changing tissue requirements. Several pro- and antiangiogenic factors orchestrate a complex, dynamic process to allow initial sprouting and invasion, subsequent pruning and remodeling, and finally maturation and survival of blood vessels. In the last decade, a new class of small RNA molecules termed micro-RNAs (miRs) have emerged as key regulators of several cellular processes including angiogenesis. Micro-RNAs such as miR-132, miR-126, miR-296, miR-145, and miR-92a have been shown to play pro- and antiangiogenic roles in the vasculature of both endothelial cells and perivascular cells. However, in pathological situations such as cancer or inflammation, the same angiogenic signaling pathways and miRs are dysregulated and exploited, typically resulting in poorly organized vessels with leaky and tortuous properties. This article is a brief overview of specific miRs that have been reported to play a role in the vasculature. The authors explore emerging principles that suggest miRs insulate cellular processes from external perturbations and provide robustness to biological systems in the context of angiogenesis.
Introduction
An adult human has about 1 to 10 × 1012 endothelial cells lining the blood vessels with only about 1 in 10,000 undergoing cell cycle at any given time.1,2 This fraction can dramatically increase during specific physiological requirements such as wound healing and tissue repair or pathological conditions such as tumorigenesis. The quiescent endothelium resumes an active proliferation program that results in growth of new blood vessels governed by a complex milieu of growth factors and signaling networks to generate new blood vessels in a process broadly termed
A Brief Primer on miRs
miRs are small 18- to 25-nucleotide sequences most often found in the intronic or intergenic regions. They are transcribed by RNA polymerase II, processed into pri-miRs by an RNAse Type III enzyme Drosha, and exported out of the nucleus by exportin-5 and then cleaved by another RNAse Dicer. 4 In a process that is not entirely clear, one of the strands of the miR duplex selectively assembles an miR-induced silencing complex (miRISC) involving members of the Argonaute protein family (AGO). The miR strand then binds to target mRNA’s 3′ untranslated regions (UTRs), often forming contiguous base-pairing in the “seed sequence” between nucleotides 2 and 10 of the miR and partial base- pairing with other nucleotides of the miR. This triggers the recruitment of a host of other proteins that can modify RNA molecules, and the target mRNA is deadenylated, decapped, and degraded by exonucleases. In general, it is thought that miR-mediated repression of translation contributes to a decrease in protein output that often correlates with the decrease in mRNA levels of the targets. 5
Global Loss of miRs Affects Development and Function of the Vasculature
The role of miRs in cardiovascular development was immediately apparent with the generation of Dicer hypomorphic mice. These mice had severe vascular deformation in the embryo and yolk sac and died at E12.5-E14.5.
6
The knockdown of both maternal and zygotic Dicer in zebrafish also resulted in pericardial edema and lack of proper circulation.
7
Suarez
Specific miRs Implicated in Angiogenesis
Several excellent reviews have comprehensively addressed the different miRs that play a role in angiogenesis and cardiovascular development.12,13 Therefore, we will provide a few illustrative examples of specific miRs and present a broad evolutionary perspective on the emergence of miRs as an adaptation to insulate cardiovascular systems from external stress.
miR-1/miR-133 in Cardiovascular Development
miR-1 and miR-133 were among the first miRs to be characterized as regulators of muscle proliferation and differentiation both in cardiac and skeletal muscles.
14
Zhao
miR-143/miR-145 in Vascular Smooth Muscle Biology
Several groups have shown miR-143 and miR-145 to play fundamental roles in vascular smooth muscle cell function.20-23 These miRs are co-transcribed in response to serum response factor and target a network of transcription factors including Kruppel-like factor (KLF) genes, Elk1, and cytoskeletal proteins to regulate the proliferation, differentiation, contraction, and migration of smooth muscle cells. 24 Although mice without either miR-145 or both miR-143 and miR-145 show normal development, they have significantly less neointima formation after vascular injury. Also, these mice have a decrease in blood pressure and enhanced development of atherosclerotic lesions. Taken together, these observations point to the role of miR-143/145 in smooth muscle cell responses to insults and injuries in the vasculature.
miR-126 as a Regulator of VEGF Signaling in the Endothelium
Targeted deletion of miR-126 causes a loss of vascular integrity in mice and zebrafish during development and leads to defective angiogenesis. 25 Mice that survive the loss of miR-126 during development show defective neovascularization following myocardial infarction. It has been shown that miR-126 inhibits sprout-related protein SPRED1, a negative inhibitor of VEGF signaling. 26 Recent work has shown that blood flow–induced upregulation of miR-126 by a mechanosensitive transcription factor Klf2 in endothelial cells activated VEGF signaling pathways and led to sprouting and remodeling of the aortic arch in developing zebrafish. 27 These studies highlight a unique role for an miR in translating physical or mechanical stress into a biological response.
miR-132 as a Genomic “First Responder” to Activation
miR-132 is encoded on human chromosome 17 and is transcribed by the transcription factor cAMP-response element binding protein (CREB) in multiple cell types.
28
We recently characterized miR-132 as a key regulator of pathological neovascularization that affects endothelial activation (Fig. 1) by downregulation of p120RasGAP.
29
Angiogenic growth factors such as VEGF, bFGF, and conditioned media from a variety of tumors lead to phosphorylation of CREB and rapid transcription of miR-132 that peaks about 3 to 6 hours after activation. Using gain and loss-of-function studies, we showed that miR-132 affected endothelial proliferation, tube formation
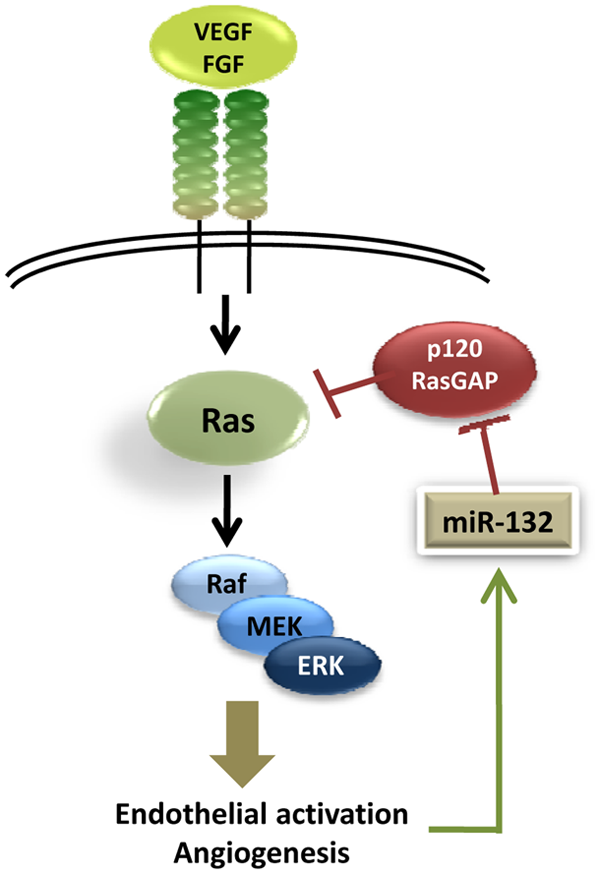
Micro-RNA (miR)-132/p120RasGAP as an angiogenic switch. miR-132 is upreg- ulated by growth factor signaling in endothelial cells and downregulates p120RasGAP levels. This in turn stabilizes active Ras and amplifies endothelial activation leading to angiogenesis. VEGF = vascular endothelial growth factor; FGF = fibroblast growth factor; MEK = MAP kinase or ERK kinase; ERK = extracellular signal-regulated kinase.
miRs That Affect Angiogenic Signaling Cascades Indirectly
miR-296 has been shown to be induced in brain endothelial cells during glioma progression. 31 miR-296 directly decreased the levels of hepatocyte-growth factor regulated tyrosine kinase substrate (HGS) and indirectly affected the levels of VEGFR2 and PDGFRβ by altering their trafficking to the cell surface. Hypoxia has been shown to trigger miR-424 expression, which leads to degradation of an ubiquitin ligase scaffold protein cullin-2. Degradation of cullin-2 prevents HIF-1α downregulation and facilitates a cascade of proangiogenic signaling. 32
miR-17~92 Cluster as Intrinsic Antiangiogenic miRs in the Endothelium
miR-17~92 is a well-characterized group of miRs that are transcribed as a single polycistronic RNA. Several studies have reported that this cluster of miRs functions as an oncogene and drives key physiological responses during development and disease.
33
Bonauer
This representative list of miRs that regulate different aspects of endothelial/smooth muscle growth and differentiation highlights the biological complexity and perhaps redundancy in the myriad network of factors that shape the structure and function of blood vessels. Although the presence of pro- and antiangiogenic factors has been appreciated for decades, it is becoming clear that miRs provide a unique layer of checks and balances because of their ability to alter biological responses both on a broader and subtler scale.
miRs as Canalization Factors during Development
Decades ago, C.H. Waddington
36
introduced the term
Some of the early evidence for the role of miRs in canalization of development came from studies in
miRs as Modulators of Morphological Complexity during Evolution
The circulatory system is one of the first organs to develop in an embryo so that organogenesis can be sustained with the delivery of oxygen and nutrients. During early development, blood vessels arise
Pathological Angiogenesis Recapitulates Developmental Angiogenesis
Pathological neovascularization shares fundamental mechanisms with developmental angiogenesis at multiple levels, including receptor signaling cascades, migration, invasion, proliferation, and tube formation.
53
A developing tumor begins to secrete angiogenic factors partly in response to hypoxia. This leads to the activation of quiescent endothelial cells that proliferate, migrate, and establish a robust capillary network. However, in contrast to normal vasculature, pathological neovascularization in tumors results in leaky immature vessels with poor pericyte coverage. This phenomenon of a small dormant tumor acquiring a vascular network has been historically referred to as the
Conclusions
Since the discovery of miRs, we have accumulated a large body of knowledge about their transcription, processing, and expression in tissues and organisms during development and disease. Target prediction programs and high-throughput genomics/proteomics methods to identify miR targets are being refined. Ongoing efforts are focused on understanding the functions of different miRs in a context-dependent manner. Although miRs represent a unique, potentially exciting therapeutic agent for targeting angiogenesis, uncovering the biological role of miRs holds the clues to understanding the equilibrium between quiescence and activation of the endothelium. Such understanding will ultimately benefit and inform the development of miR therapeutic agents for treatment of pathological neovascularization across several human diseases, including cancer.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Work in the Cheresh laboratory is funded by NCI and NHLBI. S.A. is supported by a postdoctoral fellowship award (09POST2040038) by American Heart Association.
