Abstract
Despite the involvement of genetic alterations in neoplastic cell transformation, it is increasingly evident that abnormal epigenetic patterns, such as those affecting DNA methylation and histone posttranslational modifications (PTMs), play an essential role in the early stages of tumor development. This finding, together with the evidence that epigenetic changes are reversible, enabled the development of new antineoplastic therapeutic approaches known as epigenetic therapies. Epigenetic modifications are involved in the control of gene expression, and their aberrant distribution is thought to participate in neoplastic transformation by causing the deregulation of crucial cellular pathways. Epigenetic drugs are able to revert the defective gene expression profile of cancer cells and, consequently, reestablish normal molecular pathways. Considering the emerging interest in epigenetic therapeutics, this review focuses on the approaches affecting DNA methylation, evaluates novel strategies and those already approved for clinical use, and compares their therapeutic potential.
Introduction
Epigenetics is the study of somatically heritable and reversible modifications of genome functions (gene expression and genomic stability) not involving changes in DNA sequence. DNA methylation, together with histone posttranslational modifications (PTMs), represents the most characterized epigenetic modification and is known to participate in the regulation of gene expression by modifying the chromatin structure and, consequently, the accessibility of transcription factors to regulatory regions of genes. Aberrant epigenetic patterns are involved at early stages of cancer initiation and, altering both global gene expression and genomic stability, strongly contribute to cancer progression. 1
DNA methylation involves the covalent addition of a methyl group (-CH3) to the C-5 position of cytosine, mostly in the context of the CpG dinucleotide. This chemical reaction is catalyzed by enzymes belonging to the DNA methyltransferase family (DNMTs). DNMT1 is predominantly responsible for maintaining the preexistent methylation pattern during DNA replication, whereas DNMT3a and DNMT3b are required for de novo DNA methylation. 2,3 CpG dinucleotides are clustered in regions, named CpG-islands, present in almost 60% of all gene promoters. 4 DNMT activity regulates global gene expression by hypermethylation of promoter CpG-islands and, as recently shown, of regulatory sequences near the promoter (CpG island shores), thus causing gene silencing. Not surprisingly, the DNA methylation pattern is altered in cancer. Paradoxically, tumor cells exhibit global genome hypomethylation and increased levels of DNMT1 expression at the same time. Moreover, both DNA hypo- and hypermethylation occur at specific gene loci, leading to the overexpression of proto-oncogenes and the silencing of tumor suppressor genes, respectively. These phenomena are thought to be promoting factors of cellular transformation. 5,6
Evidence regarding the involvement of DNA methylation in tumorigenesis gave rise to the development of new therapeutic strategies that target this modification. In the past few years, many drugs have been proposed. Some have already been approved for clinical use, and many others are still under evaluation. In this review, we focus on antineoplastic strategies affecting DNA methylation, with particular attention to their mechanism of action and the molecular pathways involved. In addition, we compare novel approaches, such as the use of nonnucleoside inhibitors and DNMT silencing with azanucleoside administration, the only demethylating strategy approved for clinical therapy.
The Azanucleosides: Azacytidine and Decitabine
Mechanism of Action
Azanucleoside 5-azacytidine (azacytidine [AZA]) and its derivative 5-aza-2′-deoxycytidine (decitabine [DAC]) were synthesized almost 50 years ago with the original purpose of being used as classical cytostatic drugs. 7 Only some years later, Jones and Taylor 8 correlated the differentiating effect to the demethylating activity of the 2 compounds. Consequently, a series of clinical studies was aimed to ascertain the role of demethylating agents as “epigenetic” antineoplastic drugs, and AZA and DAC received approval by the US Food and Drug Administration for the treatment of myelodysplastic syndrome (MDS). 9,10
The 2 cytidine analogs, once internalized into the cell, are converted into the corresponding active triphosphorylated nucleotides and then incorporated into DNA or RNA as cytosine substitutes. In particular, DAC is exclusively incorporated into DNA, whereas AZA is mainly incorporated into RNA, causing defects in RNA and protein synthesis and tRNA functions. Only about 10% of AZA is incorporated into DNA after its conversion to deoxynucleotide. Under normal conditions, DNMTs establish a covalent bond with the carbon-6 atom of cytosine that is resolved by β-elimination through the carbon-5 atom. The presence of a nitrogen atom at the C-5 position of AZA and DAC prevents DNMT release, causing the covalent trapping of the enzymes to DNA and the subsequent depletion of the DNMT pool. 8 Interestingly, DNMT loss seems to be alternatively driven: Ghoshal and colleagues, 11 for example, demonstrated that DNMT1 levels also diminish in the absence of DAC genomic incorporation due to its induced degradation through the proteasome pathway.
The peculiarity of DNA methylation patterns in cancerous cells has been widely demonstrated. Generally, global genome hypomethylation, especially at highly and moderately repeated sequences, coexists with hypo- and hypermethylation near and at gene promoter regions. These modifications are responsible, respectively, for the high genomic instability that characterizes cancer cells and the deregulation of genes with crucial roles in differentiation, proliferation, and apoptosis processes. 12,13 Although the application of a DNA demethylating strategy in the already demethylated status of cancer cells could be considered paradoxical, the main function of AZA and DAC treatments is in the reexpression of aberrantly hypermethylation-silenced genes, thus restoring important tumor suppressor activities.
Azanucleoside activity has also been linked to the formation of covalent adducts between DNMTs and DNA, which, directly or indirectly, induce DNA double-strand breaks and lead to the activation of both ATM and ATR DNA damage pathways. 14,15 Moreover, the DNMT-DNA complex is chemically unstable and can be resolved by the hydrolysis of DAC ring structure. The resulting open ring is the candidate mediator of C:G→G:C transversions frequently observed after DAC administration. 16
The prevalence of one mechanism or the other seems to be strictly dependent on the tumor model considered and, most important, the drug dosage. At low doses, indeed, the main effect is the reexpression of methylated genes, possibly associated with reduced proliferation, cell differentiation, apoptosis, and senescence, whereas the DNA damage response, which triggers an apoptotic pathway, is predominant at high doses (Fig. 1). 17
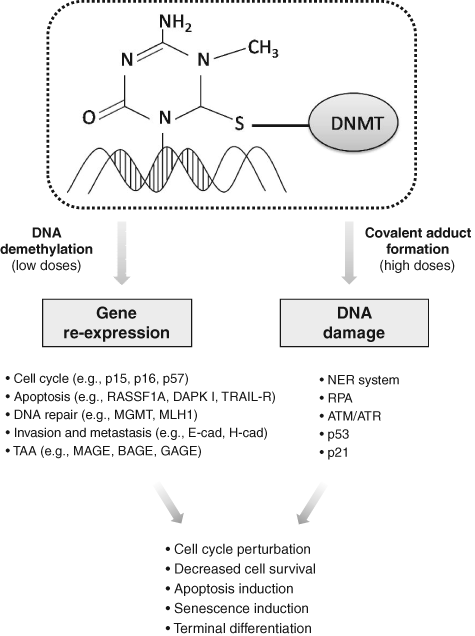
Major effects and cellular responses mediated by azanucleosides.
Another AZA- and DAC-mediated effect that warrants further in-depth examination pertains to organization of the centromeres. Centromeres, which comprise highly repeated sequences named alpha-satellites, are packaged in constitutive heterochromatin through mechanisms involving DNA methylation and posttranslational histone modifications. Such epigenetic properties prevent rearrangements of repetitive sequences and transposable elements, thus contributing to the maintenance of genome integrity and stability. 18 AZA and DAC, by direct incorporation into centromeric DNA sequences, could lead to heterochromatin decondensation and altered centromeric structure, resulting in genome destabilization and impaired kinetochore formation. Under these conditions, cells would suffer from altered chromosome alignment and incorrect sister chromatid segregation and, finally, would be irreversibly triggered to die, possibly through mitotic catastrophe.
Azanucleosides can be efficiently administered subcutaneously (SC) or intravenously (IV). Following SC administration, these molecules are rapidly adsorbed and, once in blood circulation, widely distributed. Unfortunately, drug elimination is also rapid because of its low stability. The half-life of AZA, indeed, is 1.5 ± 2.3 h, whereas the half-life of DAC is 20 ± 5 h in aqueous solutions. 19,20 The high instability, together with the cell cycle dependency of azanucleoside activity, implies the requirement of prolonged administration schedules. Moreover, due to its prevalent incorporation into DNA and its higher stability, DAC is more effective than AZA in inducing DNA demethylation, and thus it can be used at lower concentrations (100-135 mg/m2/course vs 525 mg/m2/course). 9,21-25
Reexpression of Silenced Genes
As mentioned above, low doses of AZA and DAC lead to the reexpression of genes silenced by aberrant DNA methylation. Depending on the function of the reactivated genes, different molecular pathways disrupted in cancer cells, varying from those involved in terminal differentiation and decreased proliferation to those implicated in apoptosis and premature senescence, have the potential to be reestablished. The ability of AZA and DAC to reactivate the expression of genes involved in cell cycle control that have been aberrantly silenced in cancer upon their hypermethylation has been widely demonstrated. Several CDK inhibitors, such as p16, p15, and p57, which regulate G1/S progression, as well as GADD45, involved in G2/M transition, have been found to be reexpressed after DAC exposure in different tumor models. 26-30 Loss of CDK inhibitors activity, which contributes to the accumulation of unrepaired DNA damage and the deregulation of cellular proliferation, is strongly involved in tumor development and progression. Restoring their expression and, consequently, their regulatory activity in cell cycle checkpoints impairs the neoplastic potential of cells and limits tumor growth. On the contrary, upregulation of the cell cycle inhibitor p21, although frequently observed following azanucleoside exposure, is independent of the methylation status of the gene and is more likely associated with activation of the DNA damage pathway. 31
Azanucleoside treatments are also able to sensitize tumor cells to programmed cell death by restoring the defective expression of genes involved in the apoptotic pathways. Exposure to DAC treatments has been shown to restore the expression of several silenced apoptotic effectors and mediators such as DAPK I, RASSF1A, XAF1, and TRAIL receptor-1, all of which are involved in interferon-induced apoptosis, and APAF1, a downstream element of the p53 apoptotic pathway. 32-38 Similarly, the reexpression of the hypermethylated genes HSPA9B, PAWR, PDCD5, NFKBIA, and TNFAIP3, with roles in regulating p53-, Bcl-2-, and NF-κB-related apoptosis, was observed in both osteosarcoma cells and xenografts. 30 Recently, it has been shown that DAC is able to induce the reexpression of p300/CBP factor (PCAF), known to be involved in the activation of p53, suggesting a contribution of this phenomenon in increasing the apoptotic responsiveness of cancer cells. 39
It has also been shown that DAC can regulate the reexpression and activity of genes involved in DNA repair pathways, such as the MLH1 mismatch repair gene that is reexpressed in cellular and xenograft tumor models. 40,41 However, this aspect requires careful attention because, considering their contribution in increasing resistance to DNA-targeting antineoplastic drugs, the reactivation of the DNA repair pathways may not necessarily be considered a therapeutic advantage, as in the case of the MGMT gene in relation to alkylating agents. 42,43 The ability of azanucleosides to restore molecular pathways involved in tumor invasiveness and metastatic capacity reduction, through the reexpression of genes such as E-cadherin and H-cadherin, involved in cell-cell adhesion; the antiangiogenic VEGF 189b variant; and thrombospondin-1 also has been shown. 44-49
Interestingly, AZA and DAC can augment the expression of molecules specifically recognized by targeted therapies. For example, loss of estrogen receptor (ER) expression in breast cancer induces a more aggressive tumor and results in inefficacy of therapy with selective ER modulators (SERM). Pretreating breast cancer cells with DAC can restore ER expression and function, thereby sensitizing ER-negative cells to endocrine therapy. 50 DAC exposure can also induce the reexpression of retinoid acid receptor-β2 (RAR-β2) in human head and neck squamous cell carcinoma cells, sensitizing these tumors to all-trans-retinoic acid (ATRA). 51 Comparable effects have been recently observed in prostate cancer cells, in which DAC exposure increased the efficacy of treatment with the growth inhibitor somatostatin by inducing the upregulation of its receptor. 52
Similarly, AZA and DAC have been successfully exploited in solid cancers to enhance tumor immunogenicity. Aberrant expression of recognition antigens, one of the main mechanisms used by cancer cells to evade the host immune system, is frequently imputable to promoter hypermethylation of the corresponding genes. Cancer testis antigens (CTAs) constitute a family of tumor-associated antigens (TAA) normally found in testes and ovaries but aberrantly expressed in diverse tumor types. These antigens represent a promising target for immunotherapy because of their specific distribution and capacity of triggering cellular and humoral immune responses. DAC treatment causes a persistent expression of numerous CTAs and normalizes their levels within various populations of neoplastic cells. For example, MAGE, BAGE, and GAGE family members, as well as NY-ESO-1, are induced by DAC both in vitro and in murine melanoma xenografts. 53 Moreover, DAC has been shown to upregulate several other molecules that play a central role in immune system regulation, such as human HLA class I antigens and the cell adhesion molecules ICAM-1, found reexpressed in different tumoral models, as well as LFA-3, induced by DAC treatments in melanoma cells. 54-58 Due to their “expression-restoring” properties, azanucleosides are able to induce a plethora of effects that contribute to the reversion of the malignant phenotype of cancer cells and, most important, may sensitize tumor cells to other therapeutics as well as to immune system recognition. This plurality of activities likely represents the most interesting feature of these agents.
DNA Damage Response
DNA damage that results from the formation of adducts between DNMTs and DNA is thought to be the other major mechanism of AZA and DAC antitumoral activity. Although the molecular details associated with this response are still under investigation, evidence indicates the involvement of the DNA double-strand break (DSB) repair system. More precisely, the adduct is likely to be first identified by the nucleotide excision repair (NER) system that generates a rupture in one or both DNA strands while removing the adduct. 15 The DNA strand breaks are then recognized and coated by the replication protein A (RPA), forming a DNA-RPA complex that recruits the ATR protein and is able to activate p53 through phosphorylation. Activated p53 upregulates p21, leading to inhibition of cell proliferation. 59,60 However, in accordance with other studies, our group found that p21 accumulation can also be triggered in a p53-independent manner in malignant pleural mesothelioma (MPM) cells. Moreover, we found that stable silencing of p21 limits the effect of DAC on MPM cells, indicating that p21 is a bona fide effector of DAC antitumoral activity. 61 Interestingly, another study demonstrated the involvement of DNMT1, in association with ATM and ATR, in the damage response induced by azanucleosides. However, how DNMT1 contributes to this process is not completely clear, although its role in the regulation of interactions between chromatin and checkpoint kinase 1 (CHK1), an ATR and ATM substrate, has been hypothesized. 15
Other Nucleoside Inhibitors
Despite the clinical success in MDS treatment, AZA and DAC administration is limited by serious side effects that are predominantly related to hematological perturbations, as well as by the high chemical instability of the compounds under physiological conditions. For these reasons, at present, special attention is aimed at improving the demethylation strategy through the synthesis of other nucleoside analogs, the identification and synthesis of nonnucleoside inhibitors, and the evaluation of the efficacy of low-dose treatment in combination therapies.
Among the nucleoside derivatives (Table 1), one of the most promising molecules is zebularine (dZTP), a cytidine analog originally developed as a cytidine deaminase inhibitor that was subsequently found to inhibit DNMTs. dZTP shares the same steps of activation and mechanism of action of azanucleosides. Its incorporation into DNA leads to the reactivation of epigenetically silenced genes, such as the CDK inhibitors p15 in acute myelogenous leukemia (AML) cells and thymic T lymphomas in mice; p16 in bladder, colon, and pancreatic cells as well as in a bladder carcinoma xenograft; and p57 in myeloid leukemic cells. In addition, exposure to dZTP leads to reexpression of the cell cycle and apoptosis modulator RASSF1A after demethylation of its promoter in ovarian cancer cells and in a lymphoma mouse model. 62-64 However, higher doses of dZTP are required to obtain demethylation and gene reexpression levels comparable to those induced by azanucleosides. This may be because of its lower binding affinity for uridine/cytidine kinase, the enzyme responsible for nucleoside analog activation, or its sequestration by cytidine deaminase. Unlike AZA and DAC, this compound is very stable at both neutral and acidic pH and shows slight cytotoxicity in vitro and in vivo. 65,66 Because of its minimal toxicity, dZTP can be used in prolonged treatment protocols, yielding a long-lasting demethylating effect and the prevention of remethylation. 67 Furthermore, based on its stability, it was the first DNMT inhibitor to show an in vivo antitumoral activity against T cell lymphoma after oral administration. 68
Nucleoside Analogs and Nonnucleoside Molecules
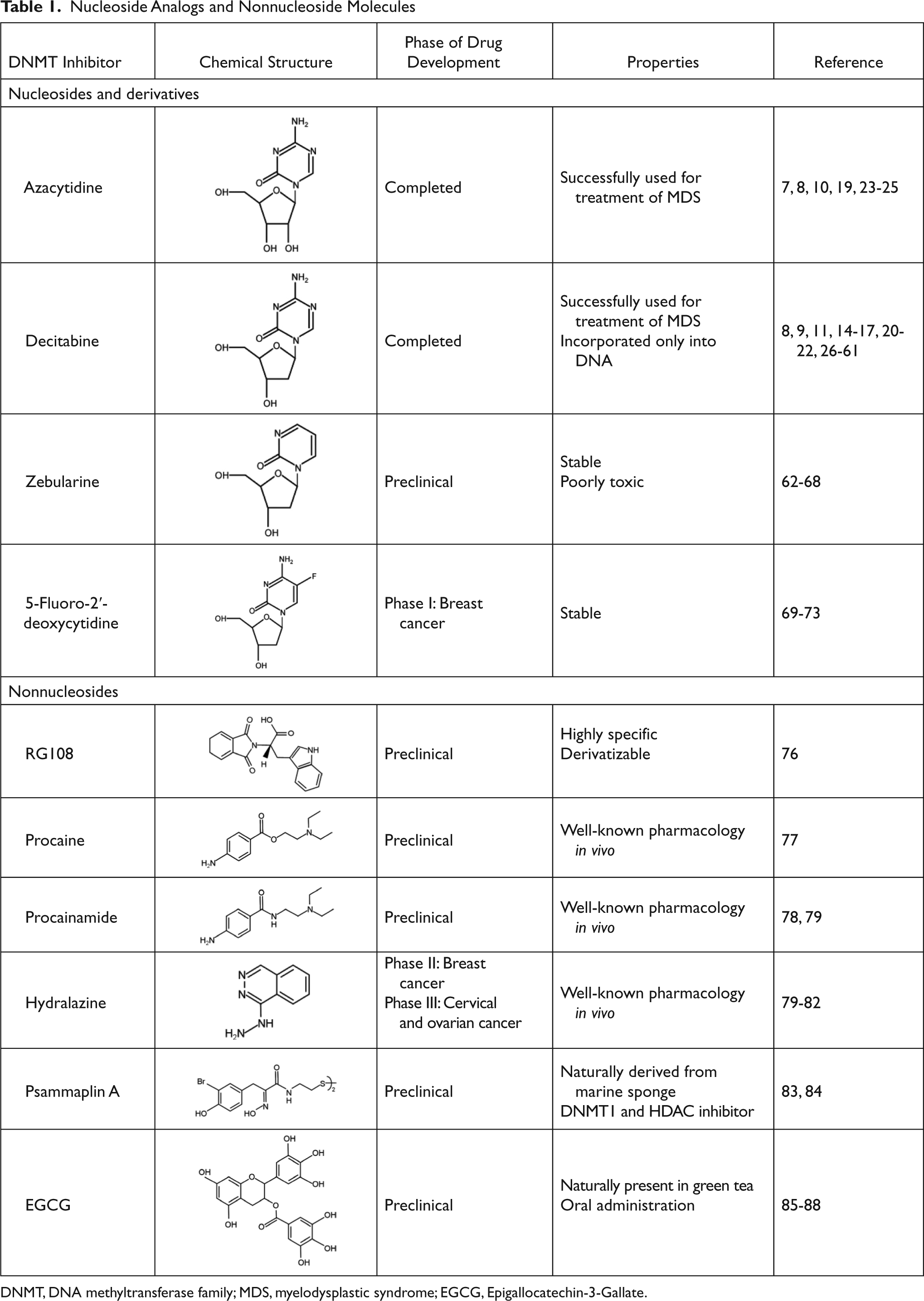
DNMT, DNA methyltransferase family; MDS, myelodysplastic syndrome; EGCG, Epigallocatechin-3-Gallate.
Another promising nucleoside analog is 5-fluoro-2′-deoxycytidine (FdCyd), a fluoropyrimidine that is stable in aqueous solutions and inhibits DNA methyltransferases once incorporated into DNA in its triphosphorylated form. Preliminary results indicate that FdCyd causes the demethylation and consequent reexpression of p16 and RASSF1A in bladder carcinoma cells, as well as of the CTA MAGE-1 genes in a melanoma cell line. 69-71 Even in these cases, the compound activation and the mechanism of action are the same as azanucleosides. However, the intracellular metabolism of FdCyd involves several different enzymatic pathways, some of which generate active metabolites that do not contribute to DNA demethylation. To diminish the accumulation of collateral metabolites and to promote the incorporation into DNA, FdCyd can be coadministered with tetrahydrouridine (THU), a cytidine deaminase inhibitor. Taking advantage of this combination, FdCyd is under investigation in breast cancer patients in a phase I trial, with encouraging preliminary results. 72,73
Because of the limited data from clinical studies, to date, it is impossible to determine whether these novel compounds are more or less efficacious than azanucleosides. Indeed, it has to be considered that improving in vitro efficacy of therapeutic molecules does not necessarily result in an improvement in their antitumoral activity in vivo. This is the case for 5,6-dihydro-5-azacytidine and fazarabine, which reached phase II of clinical trials for several types of solid tumors but were not examined further because of poor outcomes. 74,75
Nonnucleoside DNMT Inhibitors
Recently, attention has been focused on the identification of compounds that are able to inhibit DNMT enzymatic activity without trapping them on DNA (Table 1), with the primary aim being to avoid side effects caused by azanucleoside-mediated DNA adduct formation. This is the case for RG108, the first synthetic compound designed to directly inhibit DNMT1 enzymatic activity by targeting its catalytic domain. This molecule was found to induce the reexpression of different hypermethylation-silenced genes, such as p16 and the putative tumor suppressor genes SRFP1 and TIMP-3, in colon cancer cells. However, unlike azanucleosides, this compound does not affect the methylation status of repeated centromeric sequences, suggesting that the lack of effect on chromosomal stability could also contribute to reduced toxicity. Although RG108 does not seem to exert an appreciable antiproliferative effect on cancer cells per se, its possible exploitation as a safe demethylating agent in combination with other antineoplastic therapeutics renders this molecule worthy of particular attention. 76
In addition to RG108, several other nonnucleoside inhibitors of DNMTs have been discovered. These molecules are either drugs already used in the clinic for other therapeutic purposes, such as procaine, procainammide, and hydralazine, or natural compounds derived from marine sponges and plants, such as psammaplins and polyphenols.
Procaine and procainammide, both derivatives of 4-amminobenzoic acid, are already employed as local anesthetic and antiarrhythmic drugs, respectively. Recently, their ability to inhibit DNMT activity has been discovered and is associated with direct binding to CpG-rich sequences. More precisely, these molecules act as partial competitive inhibitors of DNMT1, decreasing the affinity of DNMT for its substrates (DNA and S-adenosyl-L-methionine), reducing the processivity of the enzyme and favoring the dissociation of DNMT1 from hemimethylated DNA. These activities result in global genome demethylation of both single-copy genes and centromeric repeats, as well as reactivation of tumor suppressor genes, such as p16, ER, and RAR-β, in bladder and breast cancer cells. In addition, ER expression was shown to be reinduced after hydralazine treatment in breast cancer xenografts. 77-79 Although these agents have not been tested in humans as antineoplastic drugs, their long-established and safe employment as pharmaceuticals suggests their possible use for cancer treatment with limited side effects.
The antihypertensive drug hydralazine was tested as a DNMT inhibitor as a consequence of its capability to induce (as a side effect) a lupus-like syndrome known to be related to disorders associated with DNA methylation. Although the details regarding its mechanism of action are still under investigation, some evidence indicates that hydralazine, similar to procaine and procainamide, binds to CpG-rich sequences and interferes with translocation of DNMTs along the DNA strand. 79,80 A phase I study has shown that hydralazine is able to induce reexpression of various tumor suppressor genes, including p16 and RAR-β, in cervical cancer patients, even at lower doses than those considered safe for the treatment of cardiovascular disorders. 81 Moreover, in a phase II study, hydralazine and magnesium valproate, a histone deacetylase inhibitor, were tested in combination with standard chemotherapeutics in patients with advanced and refractory solid tumors. 82 Because treatment was well tolerated and clinical benefits were observed in 80% of cases, a phase III clinical trial, which is still ongoing, was started to further evaluate the chemosensitizing potential of the combined therapy.
Different natural molecules have been tested for their ability to interfere with DNMT activity. Psammaplins, for example, are bisulfide bromotyrosines derived from a marine sponge and are able to inhibit both DNMT1 and histone deacetylase (HDAC) in vitro. Although psammaplin A administration at low doses was found to exert a strong cytotoxic effect in human tumor cell lines and to limit tumor cell growth in a A549 lung xenograft mouse model, DNMT inhibition was not followed by DNA demethylation and reexpression of tumor suppressor genes, suggesting that an intracellular target different from DNMT1 is responsible for the cytotoxic effect of the molecule. 83,84
Another natural molecule found to exhibit DNMT inhibitory activities is EGCG (Epigallocatechin-3-Gallate), a polyphenolic component of green tea. EGCG is methylated by the enzyme catechol-O-methyltransferase (COMT), which catalyzes O-methylation of various catecholic compounds. EGCG also inhibits COMT, which shares a common core structure at the active site with DNMT1. As a consequence of this observation, Fang and colleagues 85 demonstrated that EGCG is able to inhibit DNMT1 activity by obstructing entry of cytosine in the binding site of the enzyme. Furthermore, this compound was found to induce the demethylation and reactivation of the epigenetically silenced genes p16, RAR-β, MGMT, and MLH1 in esophageal cancer cells, together with inhibition of cellular proliferation. In addition, reversed silencing via promoter demethylation has also been reported for Wnt-inhibitory factor-1 (WIF-1), which is involved in the negative regulation of the Wingless-type (Wnt) oncogenic pathway in 2 different lung cancer cell lines, as well as for tissue factor pathway inhibitor-2 (TFPI-2), a member of the Kunitz-type serine proteinase inhibitor family, in renal cell carcinoma cells. 86,87 Interestingly, other dietary catechol-containing polyphenols, such as different tea catechins (catechin, epicatechin) and bioflavonoids (quercetin, fisetin), were also found to inhibit DNMT activity in vitro through mechanisms different from that of EGCG. 88 The results obtained from the usage of natural compounds are intriguing, especially considering their ease of use and low costs. However, further investigation is necessary to establish their real efficacy as DNMT inhibitors, as well as to evaluate the toxicity induced by their administration at pharmacological doses.
DNMT Silencing
Excluding some rare exceptions, 13 tumor cells generally show increased expression levels of DNMTs. 89,90 Although some experimental evidence indicates a role in neoplastic cell transformation, the mechanisms by which DNMTs participate in tumorigenesis are still uncertain and controversial. On one hand, their overexpression may explain promoter hypermethylation of tumor suppressor genes, whereas on the other hand, this is in contrast to the expression of proto-oncogenes and global genome hypomethylation that are frequently observed in cancer cells. Even with the hypotheses that attempt to explain this contradiction, this paradox is far from being resolved. Nevertheless, DNMT silencing has been demonstrated to reactivate the expression of genes that are aberrantly silenced in cancer cells and, most important, to have a strong antineoplastic effect.
Despite its role as maintaining DNMT, many authors reported that DNMT1 silencing simply causes a partial demethylation of aberrantly hypermethylated genes. On the contrary, the simultaneous silencing of DNMT1 and DNMT3b has been shown to induce genomic demethylation and the reactivation of aberrantly silenced genes, such as p16, TIMP-3, and insulin-like growth factor 2 (IGF2) in colorectal cancer cells, as well as RASSF1A and the tumor suppressor gene HIN-1 in ovarian cancer cells, although an antagonistic effect of the silencing of the 2 DNMTs was also reported. 91-95 Taken together, these findings cast serious doubt on the direct correlation between DNMT overexpression and promoter hypermethylation in tumor cells and, at the same time, underline the pressing need to further elucidate the mechanism of action of these enzymes to specifically target their activities. 96
To date, the majority of the studies conducted have focused attention on the antineoplastic effects exerted by the silencing of DNMT1 and provided interesting results in both cellular and animal neoplastic models. For example, DNMT1 silencing using antisense approaches has been shown to revert the malignant phenotype of tumor cells by inhibiting anchorage-independent growth (tumorigenicity index), inducing the specific demethylation of aberrantly hypermethylated genes such as p16, and inhibiting tumor growth in vivo. 97 The apparent contrast between the limited DNA demethylation ability and the robust antitumor effects only can be explained by assuming the presence of other important regulatory abilities exerted by this protein in addition to DNA methylation. Considering this point of view, it is reasonable to hypothesize a DNA methylation-independent property that contributes to the antineoplastic effect caused by the silencing of DNMT1, and this is probably associated with the regulatory functions in which the enzyme is known to be involved.
In the past few years, increasing evidence regarding the efficacy of DNMT silencing has prompted interest in its application in cancer therapy. The most interesting results have been obtained using MG98, an antisense oligonucleotide directed against the 3′ untranslated region of the DNMT1 mRNA. MG98 is able to specifically knock down DNMT1 expression and to induce, at least in part, the demethylation of DNA associated with reexpression of hypermethylated genes such as p16 in bladder and colon cancer cell lines. Treatment of nude mice bearing lung and colon xenografts with MG98 also resulted in tumor growth inhibition. 98 Moreover, MG98 acts synergistically in combination with DAC in vitro and in nude mouse models. 99 These promising preclinical data provided the rationale for 2 subsequent phase I studies. Despite differences in the therapeutic schedules in terms of mode of administration and dosage, both treatment protocols resulted in significant side effects, such as transaminitis, anemia, weakness, fever, nausea, and anorexia. 100,101 The best-tolerated protocol was chosen for phase II studies but, unfortunately, did not show any antitumoral activity in patients with metastatic renal carcinoma. 102 In a recent phase I trial, MG98 has been also tested for the treatment of patients with high-risk MDS and AML. Even in this case, this approach failed to result in appreciable effects, and the dose escalation was stopped due to severe toxicity. 103 However, the negative results obtained in these trials do not diminish the rationale of DNMT silencing therapy. On the contrary, it can be considered proof in support of the likely theory that targeting DNMT1 alone may be insufficient to cause a relevant antitumoral response. Moreover, the efficacy of this strategy in combination with other therapeutic approaches has never been tested.
Interesting results have come from miR-29b, a microRNA (miRNA) that directly targets DNMT3A and DNMT3B expression. miR-29b induces a decrease in methylation levels and induces the reexpression of hypermethylated tumor suppressor genes FHIT and WWOX in lung cancer cells, as well as of p15 and ER in AML cells. Interestingly, the studies conducted show that miR29b is also able to indirectly inhibit DNMT1 expression in AML cells. Although preliminary, these results support the possibility of miRNA-based approaches. 104,105
Nucleosides vs Nonnucleoside Strategies
Because only a few studies have directly analyzed nucleoside and nonnucleoside antitumoral activities using the same model, protocols of treatment, and methods of analysis, it is difficult to compare all of these approaches. Furthermore, diversity of the strategies employed makes the comparison even more difficult. Nevertheless, some observations based on demethylating activity, gene reexpression ability, cellular response, and in vivo applications can be discussed.
To date, only 2 in vitro studies directly compared different hypomethylating agents. In the first study, Chuang and colleagues 106 estimated the demethylating effect of DAC and 3 nonnucleoside inhibitors—EGCG, hydralazine, and procainamide—in different cancer cell lines. The results of these studies showed a significant reduction of the methylation levels of Alu and LINE repetitive elements and the reactivation of the silenced genes MAGE-A1, MAGE-B2, RARβ, and p16 only in cells treated with DAC. In another study, Stresemann and colleagues 107 performed a comparative analysis in different human cancer cell lines using 3 nucleoside inhibitors (AZA, DAC, and dZTP) and 3 nonnucleoside inhibitors (procaine, EGCG, and RG108). Even in this case, azanucleosides were the strongest demethylanting agents and the most efficient in restoring the expression of the hypermethylated gene TIMP-3. However, RG108 and dZTP exhibited low levels of demethylating activity, whereas ECGC and procaine induced no significant effects on DNA methylation. Nevertheless, with the exception of RG108, all compounds showed the capability to induce, to different degrees, a robust apoptotic response in cancer cells. The results obtained in these studies, although indicative of a limited ability to induce DNA demethylation and tumor-associated gene reexpression, do not exclude the potential of both nonnucleoside and nucleoside inhibitors to be safer alternatives to AZA and DAC.
Despite these results, the data concerning the toxicity of these molecules are limited, and most of these agents have never been tested in humans. Excluding 5,6-dihydro-5-azacytidine and fazarabine, FdCyd and hydralazine are the unique compounds for which a clinical trial has been started. 73,81,82 Preliminary preclinical studies have been performed only with dZTP, procainamide, and 2 psammaplins. 68,78,84 Although all studies have yielded interesting results, additional studies are necessary to investigate both in vivo efficacy and toxicity, especially in comparison with DAC and AZA.
As previously discussed, DNMT downregulation strategies result in lower demethylating activity when compared to azanucleosides. For example, when evaluating methylation-sensitive restriction pattern data, DNMT1 silencing appears to be less effective than DAC in producing extensive DNA demethylation. Interestingly, DAC-treated cells show demethylation of satellite 2, 3, and alphoid sequences whereas only methylation of satellite 2 and 3 is affected in DNMT1−/− cells. As expected, the different demethylating ability corresponds to a different capability to restore silenced-gene expression. In fact, when comparing results obtained from microarray-based gene expression analysis, DNMT knockdown seems to be less effective in inducing reexpression of hypermethylated genes than DAC. 93
In contrast to these results, Jung and colleagues 94 found that DNMT1 downregulation by siRNAs is sufficient to restore expression of aberrantly hypermethylated genes, despite the fact that its demethylating effect is weaker than that induced by DAC treatment. Moreover, these authors demonstrated that siRNAs seem to inhibit cancer cell proliferation and induce lower levels of DNA damage compared to DAC. In accordance with these results, our group has recently demonstrated that DAC and DNMT1 silencing exerts their antitumoral activity by regulating different cellular responses, although both strategies drastically decrease the survival of malignant pleural mesothelioma cells. 61 However, the data regarding the efficacy of DNMT1 silencing in vivo are still controversial. Most notably, this strategy does not seem able to improve the efficacy of DAC and AZA treatments, mainly because of the problems associated with the administration of siRNAs and antisense oligonucleotides to humans; this includes toxicity, instability, risk of nonspecific effects, and complexity in developing a suitable delivery system. 108
The finding that DNMT1 silencing induces a different response when compared to DAC is likely linked to the absence of DNA damaging activity and suggests that DNMT silencing strategies are worthy of further investigation. Moreover, to date, DNMT1 has been the main target of such studies, although evidence indicates that this strategy might be improved by targeting other enzymes in addition to DNMT1, such as de novo DNMT3a and DNMT3b.
Combination Therapy
Recent studies have demonstrated that treatment with demethylating agents such as DAC is able to sensitize cells and overcome the resistance of tumor cells to traditional antineoplastic therapies (Table 2). Yang and colleagues 109 have shown that pretreatment with DAC induces the reexpression of MLH1, which is involved in the mismatch repair system (MMR), sensitizing ovarian and colon tumor xenografts to cisplatin, carboplatin, temozolimide, and epirubicin. Subsequently, a phase I trial showed that it is possible to safely combine low doses of DAC and carboplatin in different solid tumors (colon carcinoma, breast cancer, melanoma, sarcoma, gallbladder, pleural mesothelioma), achieving epigenetic changes equivalent to or even greater than those observed in tumor xenografts. 21 In another study, treatment of colon adenocarcinoma with a combination of DAC and the topoisomerase-I inhibitor irinotecan (CPT-11) was found to induce a marked suppression of tumor growth compared to DAC or CPT-11 alone, both in vitro and in vivo, and restored the expression of tumor suppressor genes such as p14 and p16. 110
Recent Studies on the Efficacy of Combination Therapy with Decitabine and Other Therapeutic Agents

Interesting results were also obtained from the combination of DAC with nonchemotherapeutic agents. Cotreatment of leukemia cells with DAC and valproic acid (a histone deacetylase inhibitor) results in a significant synergistic effect on cell viability reduction and apoptosis induction associated with the upregulation of p21 and p57. 109 More interestingly, the ability of DAC low-dose administration to enhance IL-2 efficacy against metastatic melanoma was also demonstrated in a phase I trial. 111
DAC has also been shown to enhance the sensitivity of gastric cancer cells to radiotherapy. This result is not surprising considering that DAC exposure arrests tumor cells in G2-M, a phase of the cell cycle known to be the most sensitive to irradiation. Moreover, this effect was specific for tumor cells, probably as a consequence of the sensitivity of rapidly dividing cells to the drug. These findings suggest that DAC might be advantageous as it will avoid side effects on the surrounding organs caused by X-ray irradiation. 112
Not surprisingly, cancer cells can acquire resistance also to DAC. In fact, increased activity of cytidine (CR) deaminase, a key enzyme in the metabolism of nucleoside analogs, may reduce viability and sensitivity to DAC, leading to insensitivity of tumor cells to the drug. 113 Cotreatment with CR deaminase inhibitors, such as dZTP, was demonstrated to overcome this resistance, improving the efficacy of DAC against resistant tumor cells. 63,114
Conclusion and Future Perspectives
DNA demethylating agents have recently arisen as a promising therapeutic tool for the treatment of cancer. In the past few years, the severe toxicities related to the administration of azanucleosides have prompted research into new DNA demethylating compounds and strategies. In this field, a plethora of drugs, even if they have different mechanisms of action that are not fully understood, have demonstrated interesting anticancer properties. Unfortunately, although most of the new compounds exhibit favorable toxicity profiles, their capability to act as DNA demethylating agents with antitumor properties that are comparable to azanucleosides has not yet been achieved. The use of DNMT-silencing strategies has also been problematic with respect to the in vivo delivery of siRNAs and antisense oligonucleosides. Nevertheless, research in this field is at a relative early stage: many compounds have not been sufficiently researched in vivo and, in many cases, have never been tested in humans.
With our current level of knowledge regarding their functions, DNA demethylating agents, in our opinion, are unlikely to be used as single agents in cancer therapy. However, this does not diminish their importance in the fight against cancer for different reasons: first, as shown for MDS, these agents are potent and effective therapeutics; second, at low doses, these compounds can induce the reexpression of aberrantly silenced tumor suppressor genes, allowing tumor cells to revert to a normal phenotype (terminal differentiation) and/or reacquire cellular pathways crucial for cell cycle regulation and apoptosis induction. Based on their ability to induce the reexpression of tumor suppressor genes, DNA demethylating drugs can be used as “biosensitizers” by increasing the sensitivity of cancer cells to standard therapeutics (e.g., cisplatin, carboplatin, irinotecan, IL-2) or therapeutic procedures (e.g., radiotherapy). This aspect is even more important when considering the frequency with which drug resistance, whether intrinsic or acquired after the first cycles of treatment, negatively influences patients’ outcomes. Another aspect underlining the importance of the gene expression restoring activity of these agents pertains to their ability to potentiate the innate immune response against tumors by inducing cancer cells to reacquire the expression of silenced TAAs and improve the efficacy of targeted therapies by increasing the expression of their target molecules.
Finally, it has to be considered that long-term, chronic disease has recently arisen as a somewhat viable option for the treatment of seemingly incurable patients. DNA demethylating approaches could have a central role in this context, especially considering their known ability to induce a premature cellular senescent response that is similar to that caused by low-dose treatments with cytotoxic drugs. 115 Hematological malignancies have provided a good starting point for the application of DNA demethylating agents, but interesting results have recently been obtained with different solid tumors. However, further studies are necessary to evaluate the efficacy and toxicity of known compounds as well as new agents. Despite the number of problems to be solved, combination therapy using DNA demethylating drugs and conventional therapeutics has a reasonably good chance to be used in the clinic in the near future.
Footnotes
Acknowledgements
We have tried to cite as many works to the best of our knowledge but apologize to those omitted because of space limitations.
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The authors did not receive grants or outside funding of any kind in support of their research for the preparation of this manuscript.
