Abstract
Background
Respiratory epithelial adenomatoid hamartoma (REAH) is a benign tumor-like lesion that appears in the sinonasal tract. Due to the lack of well-defined distinctive features of REAH, specifically clinical and molecular characteristics, this lesion is often misdiagnosed as an inflammatory nasal polyp or sinonasal malignancy.
Objective
This study aimed to systematically review the existing literature to determine the distinguishing attributes of REAH.
Methods
In accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines, a Medline, EMBASE and Web-of-Science literature review was undertaken to evaluate studies published till July 1, 2024. Forest and funnel plots were used to present prevalence and potential small-study effects, respectively. The estimates were calculated using random-effects methodology. The I2 value was determined to assess the interstudy heterogeneity. The quality of the included studies was evaluated using National Heart, Lung, and Blood Institute Study Quality Assessment Tools.
Results
A total of 39 studies were included, of which 10 investigated molecular characteristics. Several studies have shown the involvement of inflammatory processes in REAH. In addition, 37 studies with 1127 patients met the inclusion criteria for the meta-analysis. The findings of these studies showed that REAH was most commonly identified in the olfactory cleft (66.7% [49.8%-83.7%]), often bilaterally (66.4% [54.0%-78.7%]). Furthermore, REAH was associated with enlarged olfactory cleft widths on computed tomography scans, with an estimated width of 10.58 mm [9.41-11.75 mm].
Conclusion
Our study showed that inflammatory responses likely play a role in REAH. Notably, the symptoms of REAH are often indistinguishable from those of chronic rhinosinusitis with nasal polyposis. Thus, a high index of suspicion is required to diagnose REAH, typically when patients present with bilateral olfactory cleft masses with an enlarged olfactory cleft width on a computed tomography head scan.
Keywords
Introduction
Respiratory epithelial adenomatoid hamartoma (REAH) is a histopathologic diagnosis first described by Wening and Heffner in 1995 as an indurated polypoid mass lesion with prominent glandular proliferation surrounded by stromal hyalinization. 1 REAHs are masses containing disorganized but mature differentiated cells or tissues that commonly originate from the olfactory cleft (OC). 1
REAHs are found in the nasal cavity, paranasal sinuses, and nasopharynx and frequently appear as an incidental finding after endoscopic sinus surgery (ESS) for suspected chronic rhinosinusitis (CRS).2–5 To date, the etiology of REAH has not been fully elucidated. Some authors have suggested that REAHs are caused by hyperplasia of the respiratory epithelium from chronic sinonasal inflammation on the basis of histological features showing multi-layered and ciliated respiratory epithelium, often with mixed mucocytes alongside pseudogland formation. 6 Others postulate that REAHs may be neoplastic entities, considering the occasional concomitant presentation with sinonasal adenocarcinoma and the abnormally high allelic loss observed at the molecular level.7,8
In addition to its etiology, identification of REAH remains a challenge. In particular, no well-established clinical predictors of REAH are available to help clinicians distinguish it from other diagnoses. Therefore, this study aimed to systematically review the existing literature to determine clinical predictors of REAH and summarize the molecular findings related to these lesions.
Materials and Methods
This systematic review and meta-analysis was performed in accordance with the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). 9
Data Sources and Searches
Electronic bibliographic searches were systematically performed in Medline, EMBASE, and Web of Sciences up to July 1, 2024. Our search string was “respiratory epithelial adenomatoid hamartoma” OR “REAH.” Two independent authors (F.L. and Y.F.L.) screened the titles and abstracts in accordance with the following inclusion and exclusion criteria: Studies that analyzed symptomatic presentations and/or molecular characteristics of histologically confirmed REAH were included. All included studies were required to have reported the prevalence of at least one symptom, clinical presentation, or molecular feature. Abstracts, books, conference articles, preprints, reviews, case reports, and case series with fewer than five patients were not eligible. All potentially relevant studies were identified for a full-text review (Figure 1).
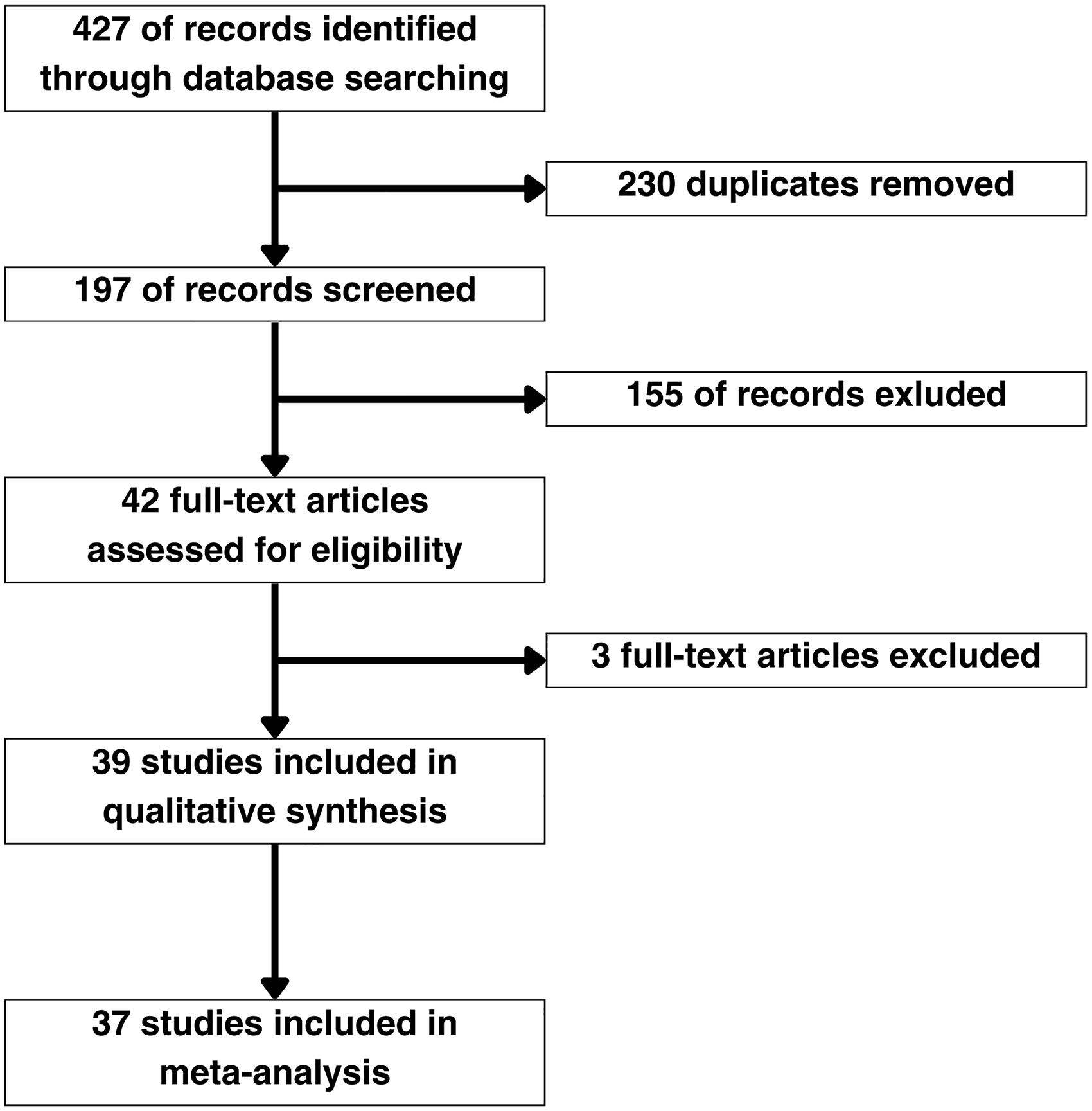
PRISMA Flow Diagram for the Systematic Literature Search and Study Selection.
The primary endpoints were molecular markers, clinical presentation, and REAH predictors. The secondary endpoints were evaluation of the study design and reporting quality.
Data Extraction and Quality Assessment
Two authors (F.L. and Y.F.L.) independently extracted demographic, methodological, molecular, and clinical presentation data from the included studies. A data-extraction spreadsheet specific to the interests of this study was designed. This Excel spreadsheet included information regarding the study type, country, sample size, patient characteristics, molecular markers, incidence, prevalence of symptoms, duration of symptoms, associations, sites, imaging features of REAH, surgical interventions, olfactory outcomes, and postoperative recurrence rates. The graphical presentation of values was pooled using WebPlotDigitizer. 10 The quality of the studies was assessed using the National Heart, Lung, and Blood Institute (NHLBI) Study Quality Assessment Tools for observational cohort, cross-sectional, and case-series studies. 11 An overall rating of good (all key domains met), fair (≥1 key domain unclear or missing, but not enough to invalidate results), or poor (≥1 critical flaw) was determined for each study.
Data Synthesis and Analysis
A random-effects model was used for the meta-analysis, and heterogeneity was assessed using I2 values. Forest plots were used to determine the pooled prevalence of each clinical manifestation. Characteristics vulnerable to selective reporting (eg, symptoms, locations, and associated presentations) were only analyzed if they were presented in 10 or more studies. These restrictions were not applied to other parameters, such as olfactory outcomes or CT findings. For the meta-analysis, the median age or OC width with range was converted to mean and standard deviation using the approach proposed by Wan et al. 12 In addition, only studies with a specified surgical intervention and postoperative follow-up period were included for outcome data meta-analysis. Characteristics reported in 10 or more studies were also analyzed using funnel plots and Egger's tests to assess potential small-study effects. The R programming language was used for all statistical analyses.
Results
A total of 190 articles were included after duplicates were removed. After abstract screening, 42 articles were identified as potentially relevant to the present study. Three studies were excluded after full text review. The reason for exclusion was the lack of data regarding the REAH features evaluated in this review. A flow diagram of the screening and exclusion of articles in accordance with the PRISMA statement is shown in Figure 1. Ten studies involving 183 samples investigated the molecular characteristics of REAH (Table 1). Three studies13,16,18 proposed molecular findings to support the involvement of inflammatory pathways in REAH. Three other studies7,8,15 presented molecular evidence potentially suggestive of the neoplastic characteristics of REAH. Finally, four studies5,14,17,19 confirmed the benign nature of REAH.
Insights into the Molecular Landscape of REAH.
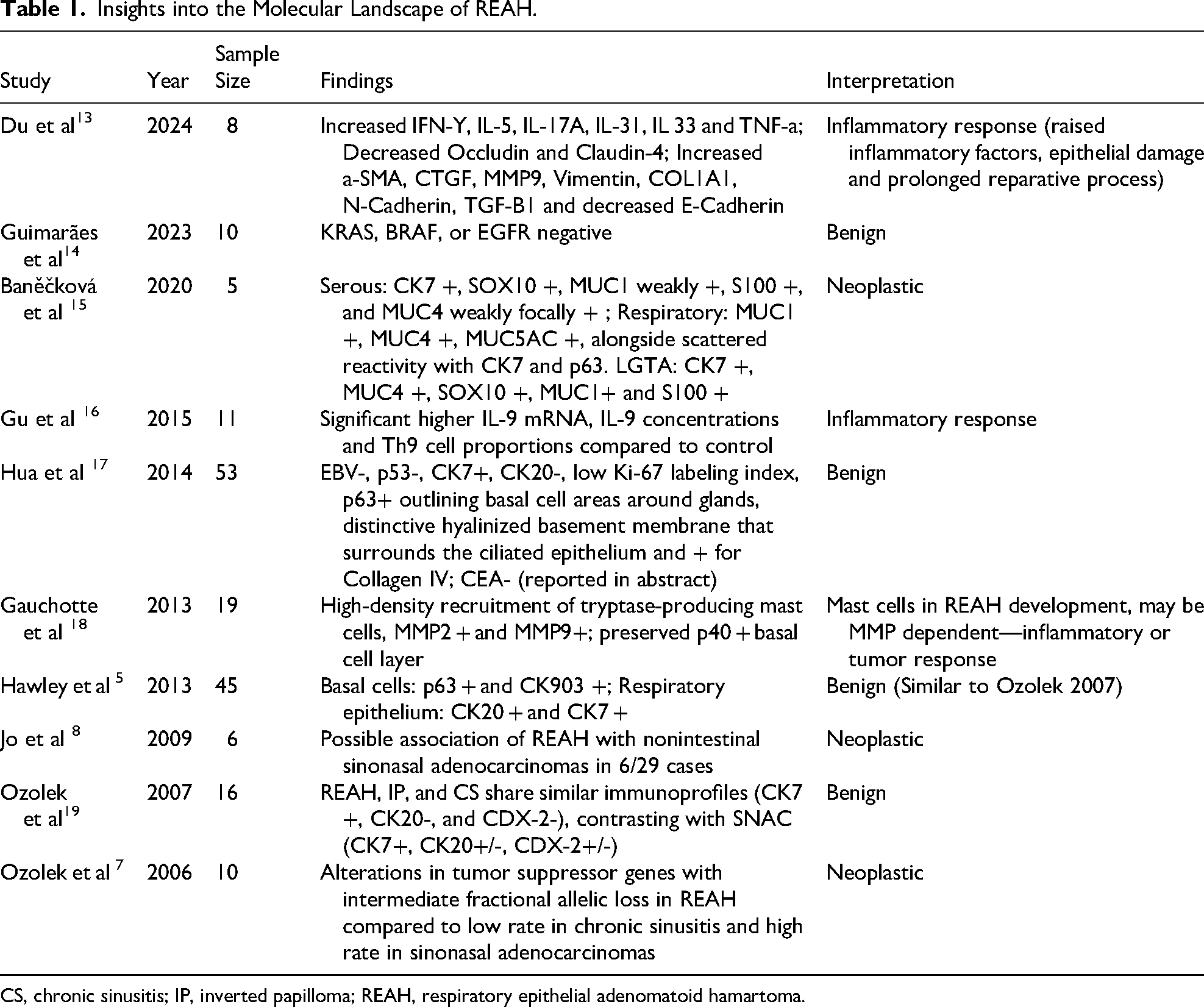
CS, chronic sinusitis; IP, inverted papilloma; REAH, respiratory epithelial adenomatoid hamartoma.
The 37 included studies for meta-analysis included a total of 1127 patients (Table 2). The majority of the studies had good quality ratings with no major concerns (Supplementary file Table S1-S4). The results of heterogeneity analysis (Table S5) and small-study effect assessment (Table S6, Figure S1) of the studies can also be found in the supplementary file. The funnel plots and Egger's tests showed small-study effects of some categories, namely for sex, nasal obstruction, paranasal cavity, and nasal polyp data distribution (Table S6, Figure S1).
Characteristics of the Included Studies.
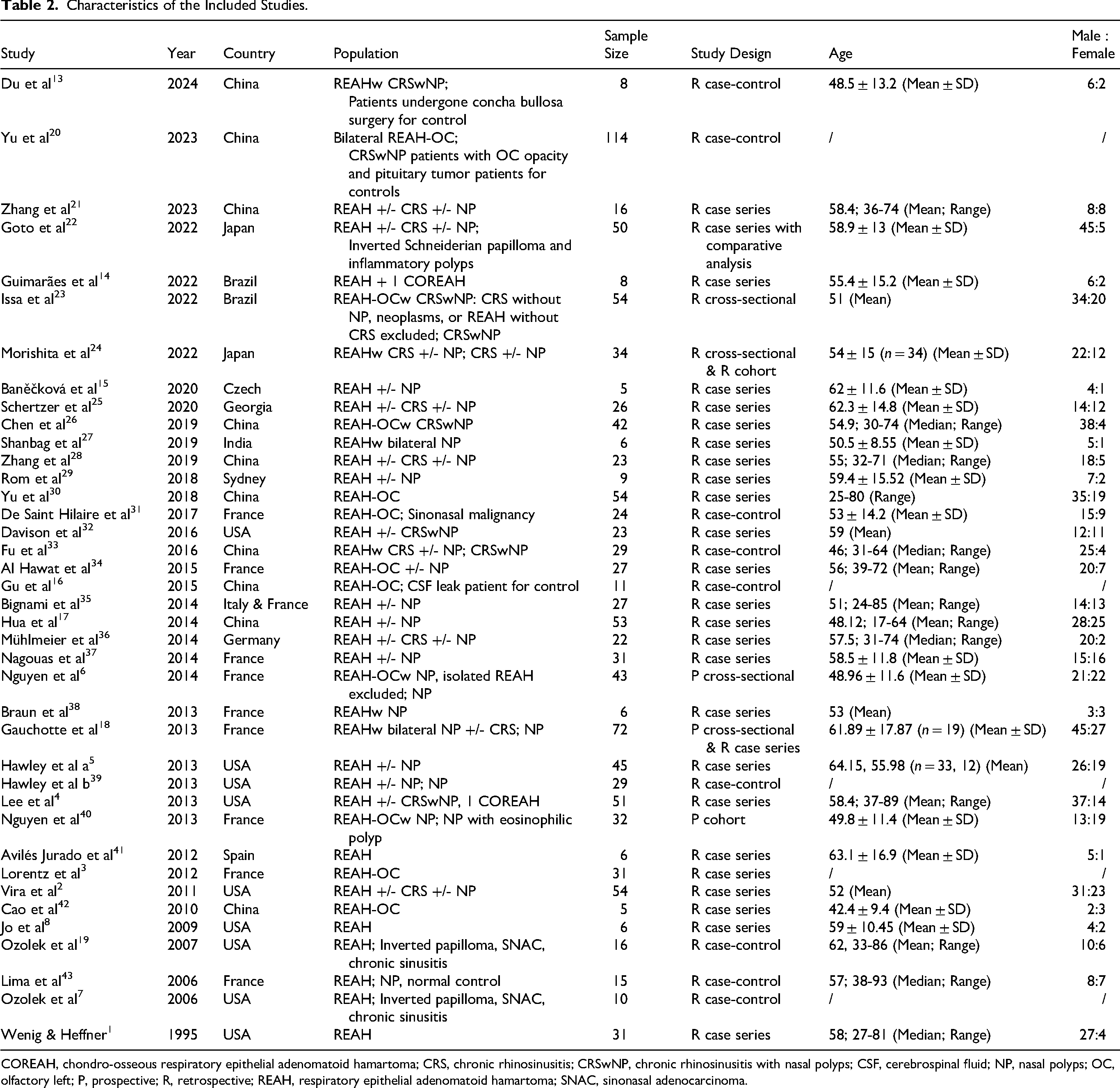
COREAH, chondro-osseous respiratory epithelial adenomatoid hamartoma; CRS, chronic rhinosinusitis; CRSwNP, chronic rhinosinusitis with nasal polyps; CSF, cerebrospinal fluid; NP, nasal polyps; OC, olfactory left; P, prospective; R, retrospective; REAH, respiratory epithelial adenomatoid hamartoma; SNAC, sinonasal adenocarcinoma.
Regarding demographics, REAH showed an estimated incidence of 42.0% [36.9%-47.2%] among patients with nasal polyposis (NP) undergoing ESS (Figure 2A).3,6,18,23,40 Specifically, patients in these five studies underwent systematic examination during surgery and biopsies from the OC. Regarding sex distribution, an estimated 67.0% [61.6%-72.5%] of patients with REAH were male, indicating a significantly higher occurrence in males than in females (Figure 2B).1,2,4–6,8,13–15,17–19,21–38,40–43 Moreover, the estimated mean age of patients with REAH was 54.86 years [52.93-56.79 years] (Figure 2C).1,4,6,8,13–15,17–19,21,22,24–29,31,33–37,40–43
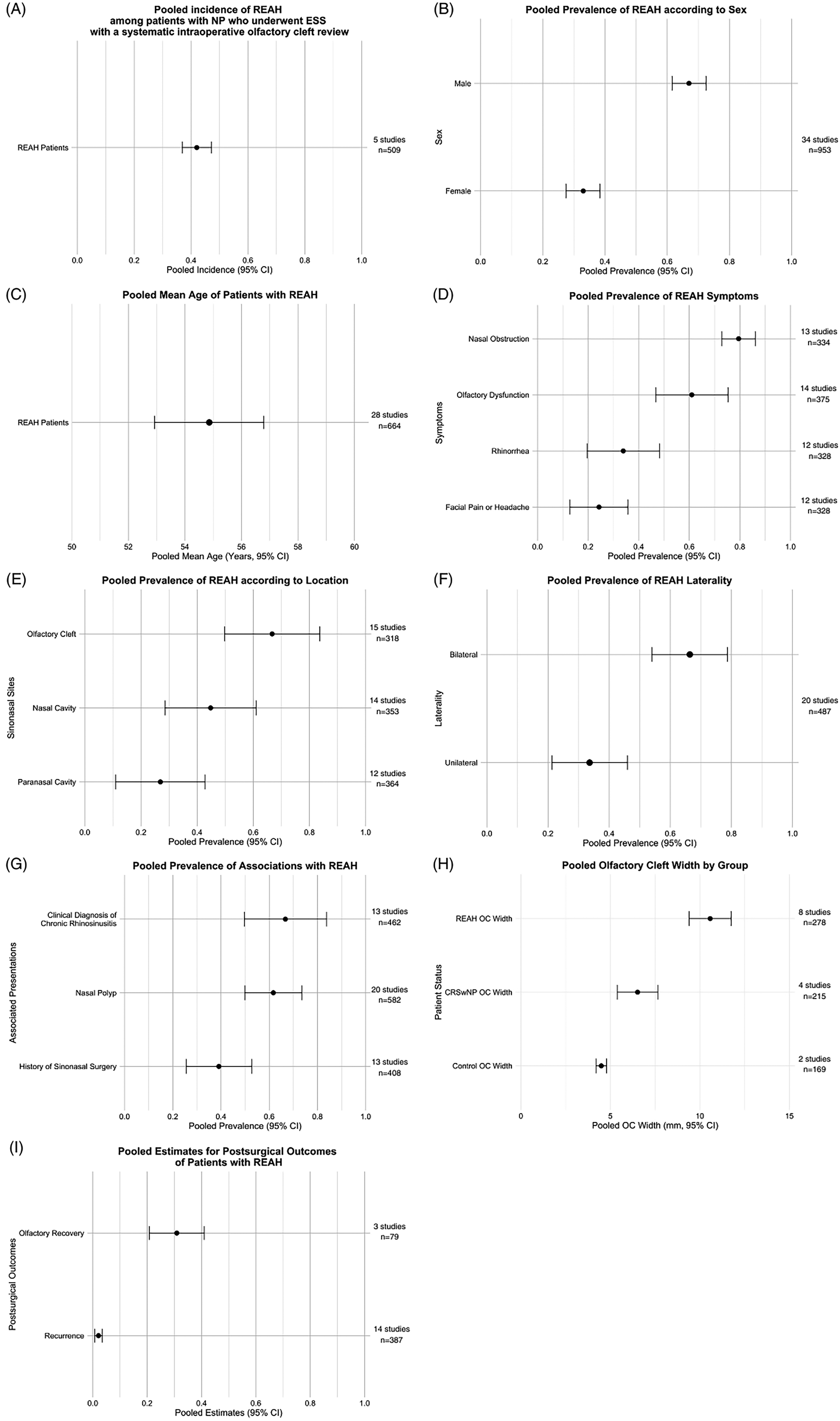
A, pooled incidence of REAH among patients with NP who underwent ESS with a systematic intraoperative olfactory cleft review. B, Pooled prevalence of REAH according to sex. C, Pooled mean age of patients with REAH. D, Pooled prevalence of REAH symptoms. E, Pooled prevalence of REAH according to the location. F, Pooled prevalence of REAH laterality. G, Pooled prevalence of associations with REAH. H, Pooled olfactory cleft width by group. I, Pooled estimates for postsurgical outcomes of patients with REAH. CRSwNP, chronic rhinosinusitis with nasal polyps; ESS, endoscopic sinus surgery; NP, nasal polyps; OC, olfactory cleft; REAH, respiratory epithelial adenomatoid hamartoma.
The prevalence of these symptoms showed notable variability (Figure 2D). Nasal obstruction demonstrated the highest pooled prevalence, with an estimate of 79.5% [72.9%-86.1%].4,5,17,18,21,24,27,28,34–36,41,42 Olfactory dysfunction was the second-most common symptom, with an estimated prevalence of 61.0% [46.7%-75.3%].4,5,17,18,21,24,27,28,34,35,37,40–42 In contrast, rhinorrhea had a moderate prevalence of approximately 33.9% [19.6%-48.2%],4,5,17,18,21,24,28,34–36,41,42 while facial pain or headache exhibited a lower pooled prevalence of approximately 24.3% [12.8%-35.7%].4,5,17,18,21,24,27,28,34–36,42 These findings underscore the prominence of nasal obstruction and olfactory dysfunction as key symptoms, whereas facial pain and headache appear to be less consistently reported.
Regarding locations, REAH was most commonly identified in the OC in approximately 66.7% [49.8%-83.7%] of the cases.4,5,14,18,21,22,24,25,28,29,35–38,42 Other known locations of REAH include the general nasal cavities and paranasal cavities, with estimates of 44.8% [28.6.%-61.0%]1,2,4,5,14,15,17,21,22,24,28,29,36,41 and 26.9% [11.0%-42.8%],1,2,4,14,17,22,24,25,28,35,36,41 respectively (Figure 2E). Additionally, REAH commonly showed bilateral presentation in the sinonasal tracts (66.4% [54.0%-78.7%]), rather than appearing unilaterally (Figure 2F).3–5,14,18,21,24–28,31–33,35,37,38,41–43
In relation to associated presentations, REAH was more likely to present with nasal polyps (61.7% [49.9%-73.5%]) than appearing independently.2,4,5,15,17,21,24–26,28–30,32–38,42 Moreover, patients with REAH often had a history of previous sinonasal surgeries, which were performed in approximately 39.1% [25.6%-52.7%] of the cases.6,13,21,22,25,26,28–30,33–35,37,40 Furthermore, up to 66.7% [49.7%-83.8%] of the REAH patients were clinically diagnosed with CRS (Figure 2G).2,4,17,18,21,22,25,26,28,30,32,33,36
Eight studies have also investigated the image features indicative of REAH, and they found that patients with REAH had an enlarged OC width with a pooled average of 10.58 mm [9.41-11.75 mm].20,21,31–33,37,39,43 OC width was measured as the widest linear distance between the medial aspects of the bilateral middle turbinates on a coronal CT head scan. The measured width was substantially greater than that in patients showing CRS with NP (CRSwNP) (6.52 mm [5.39-7.65 mm])20,33,39,43 or healthy individuals (4.49 mm [4.19-4.79 mm]) (Figure 2H).20,43
Fourteen studies have shown that REAH has a very low recurrence rate of 2.1% [0.7%-3.5%] after surgical resection, with follow-up periods ranging from 1 month to 128 months in 13 studies,1,2,4,5,18,21,24,28,35–38,41 while one study 22 reviewed recurrences across patient records over a 13-year-period (Figure 2I). In addition, three studies with a postoperative follow-up period of 1.5 to 80 months evaluated the olfactory outcomes of patients with REAH. Of these patients, 30.9% [20.8%-41.0%] reported olfactory recovery (postoperative normosmia from previous hyposmia or anosmia), of which two studies were subjective patient reporting34,37 and one study utilized the n-butanol odor threshold test alongside the Sniffin’ Sticks test (Figure 2I). 40 No explicit concerns have been raised regarding olfactory deterioration following surgery in the available literature.
Discussion
To the best of our knowledge, this is the first systematic review and meta-analysis focusing on the clinical and molecular presentations of REAH, highlighting its distinct features and diagnostic challenges. The reported incidence of REAH varied broadly across studies,2,3,6,16,18,22–24,34,36,40 ranging from 0.3% to 47.7% among different groups of patients who underwent ESS. These variations can be attributed to the introduction of systematic examinations, a better understanding of the disease, and differences in the study population.
Lorentz et al 3 recorded the incidence of REAH among patients who underwent ESS for NP in tertiary centers across different time periods. During the first period (2003-2006), the detection rate of REAH was 0% in 2003-2004, rising to 1.6% in 2005 (1/64) and 12.5% in 2006 (10/80). In the second period (2007), the implementation of systematic endoscopic examination of OCs during ethmoid labyrinth surgery increased the detection rate to 27% (27/100). By the third period (2008), the addition of systematic biopsies of the abnormal mucosa in the OCs during ethmoid surgery further increased the detection rate to 47.7% (31/65). Similar findings were observed by Gauchotte et al, 18 who reported an incidence rate of 35.3% (53/150 patients with NP-indicated ESS) when polyps removed from the ethmoid labyrinth and the OC were sent separately for review. However, they found that the incidence of REAH in the previous 18-month cohort of 160 patients, who underwent operations for bilateral NP with routine tissue processing, was 3.9%. Prior routine tissue processing techniques include limited sampling, single-level HE block, unseparated polyps originating from the OC, and no specifically checked OC. More structured approaches have been introduced recently, including systematic examination and specimen collection from targeted areas (the OC).3,24 In addition, REAH has been gradually recognized and included in the differential diagnosis by pathologists. These changes may have plausibly contributed to the increased incidence of REAH.
Other factors contributing to the vast incidence variation across studies include different selections and spectra of the sample population. Since nasal polyps are correlated with REAH, and the diagnostic criteria for CRS are closely comparable with the presenting symptoms of REAH, the incidence of REAH in patients with NP or CRS undergoing ESS would be significantly higher than that in the general population or patients undergoing ESS for any indication.
On the other hand, since REAH is a relatively new histopathological diagnosis, the high incidence among patients with NP-indicated ESS does not necessarily represent the true disease prevalence, but may be a correction of prior under-recognition and accumulation of undiagnosed cases of REAH. Therefore, more studies and more time are required to accurately determine the incidence of the REAH. Nevertheless, the pooled incidence showed that REAH is common in patients with NP and an important option in differential diagnosis.
In addition, our findings showed clinical challenges in distinguishing REAH from CRSwNP based solely on symptoms and clinical examinations. Both conditions commonly present with nasal obstruction, olfactory dysfunction, rhinorrhea, and non-distinctive endoscopic findings, which may lead to misdiagnosis. In fact, 66.7% of patients with REAH had a clinical diagnosis of CRS. This overlap highlights the need for histological confirmation, since REAH is rarely diagnosed preoperatively on the basis of clinical findings alone.
Importantly, our review emphasizes that REAH is most frequently localized to the OC, a finding that might have important implications on surgical management. A lesion identified at the OC prompt intraoperative frozen section analysis for diagnosis. Unlike the mucosa-preserving techniques 44 and long-term steroid douching required for CRSwNP, 45 an intraoperative diagnosis of REAH justifies complete surgical excision to healthy margins, which is often curative given REAH's low recurrence rate. This is in stark contrast to CRSwNP, particularly the eosinophilic subtype, which is associated with a persistently high recurrence rate despite medical and surgical interventions.46,47 Therefore, despite REAH is generally regarded as benign, these distinctions underscore the clinical importance of obtaining an accurate diagnosis, since it guides surgical management and influences patient outcomes, postoperative follow-up, and patient counseling. Furthermore, REAH tends to be present bilaterally, which could be a distinguishing factor from malignant tumors. This is again important, since REAH does not require extensive resection owing to its noninvasive nature. Together, our findings showing the tendency of REAH to present bilaterally in the OC could explain the OC widening observed on coronal CT head scans, which is an imaging feature that has been proposed to facilitate REAH identification.
Three studies examined the olfactory outcomes of patients with REAH after surgical intervention. Approximately 30.9% of the patients showed olfactory recovery, regaining self-reported or olfactory-tested normosmia from preoperative hyposmia or anosmia. Moreover, no records of olfactory deterioration following the surgery were noted. Specifically, Ngyuen et al 40 clarified that none of the patients experienced postoperative worsening of the sense of smell. However, these results must be interpreted with caution because the findings for olfactory outcomes were based on only 79 patients among three studies with mixed methodologies. Nevertheless, with the low recurrence rate, currently available evidence indicates that complete surgical excision is the optimal first-line treatment for REAH. However, more research is required to consolidate olfactory outcomes and investigate why a certain proportion of patients do not achieve olfactory improvement despite REAH excision. Additionally, current available data does not permit robust subgroup analysis regarding whether isolated REAH and those with CRSwNP warrant distinct postoperative olfactory outcomes. To refine management recommendations, future studies should report outcomes stratified by isolated REAH versus concurrent CRSwNP.
Another valuable finding of this review was the propensity of REAH to coexist with nasal polyps. This observation suggests that REAH may be overlooked in patients undergoing ESS for presumed CRSwNPs. Up to 39.1% of patients with REAH had undergone previous sinonasal surgery, and retrospectively confirming the presence of REAH when the first surgery was performed was impossible. Regardless, since REAH has a low recurrence rate after ESS, we can rule out earlier surgeries as a risk factor and interpret that a history of sinonasal surgery indicates a missed diagnosis. However, as REAH studies were derived from surgical cohorts, the reported frequency of REAH-NP association might be inflated by selection bias. Symptomatic REAH patients, potentially attributing to the concomitant NP, are more likely to seek medical attention, undergo surgery and thus histological sampling; while asymptomatic isolated REAH may remain undetected. Together with the presence of small-study effects, publication bias cannot be excluded, and the association rate requires careful interpretation (Table S6, Figure S1). Nevertheless, recognizing this association with NP is vital for unmasking the underlying diagnosis and improving diagnostic rates.
Furthermore, this review identified a growing body of molecular research on REAH. Multiple studies13–18 have demonstrated that inflammatory processes characterize the molecular landscape of REAH. Cytokine profiling demonstrates upregulation of pro-inflammatory mediators (IFN-γ, IL-5, IL-17A, IL-31, IL-33, TNF-α) 13 and enrichment of the IL-9/Th9 axis. 16 Simultaneously, reduced tight-junction proteins (occludin, claudin-4), increased expression of epithelial-mesenchymal transition related factors (α-SMA, CTGF, MMP9, vimentin, COL1A1, N-cadherin, TGF-β1) and high-density recruitment of tryptase-producing mast cells support epithelial barrier disruption with a prolonged reparative response.13,18 This aligns with the clinical overlap observed between the REAH and CRSwNP groups. However, the exact etiology of REAH remains unknown, limiting the scope to clarify whether inflammation is a primary driver or secondary effect of frequent concurrent CRSwNPs.
Three molecular studies have provided evidence potentially hinting at the neoplastic nature of REAH. This hypothesis was first proposed by Ozolek et al, 7 who found alterations in tumor suppressor genes and intermediate fractional allelic loss in REAH, comparable to the high rate of such alterations in sinonasal adenocarcinomas. Furthermore, Baněčková et al 15 found a similar immunohistochemistry profile between REAH and low-grade tubulopapillary adenocarcinomas, while Jo et al 8 proposed associations of REAH with nonintestinal sinonasal adenocarcinomas in 6/29 cases.
The remaining four molecular studies confirmed the benign nature of REAH.5,14,17,19 REAH typically shows CK7 positivity, basal cell markers (p63, CK903) and a characteristic hyalinized basement membrane positive for collagen IV; while EBV and p53 are generally negative and Ki-67 is low, reinforcing a benign profile.5,17,19 Moreover, common oncogenic drivers (KRAS, BRAF, EGFR) have been reported as absent. 14 In addition, among the patients with REAH included in this review, only 10 presented with malignancy.5,8,18 This finding indicates that adequate excision of REAH can cure this condition.
However, further research is required to elucidate the etiology and pathogenesis of REAH. Investigating the molecular differences between REAH and other inflammatory nasal conditions such as CRSwNP could provide valuable insights into potential biomarkers and targeted therapies for patients who are unsuitable for surgical intervention.
The limitations of this study include the potential bias in observational studies. Of the 37 studies selected for the meta-analysis, 24 were case series without comparative analysis and were more prone to reporting bias or confounding factors (Table 2). In addition, three studies did not use continuous sampling, making them vulnerable to biased patient selection with certain distinctive features (Supplementary file Table S4).14,15,38
Moreover, the sample sizes in these studies were relatively small. Nine of the studies included fewer than 10 patients each, and small sample sizes can lead to variability and less reliable estimates (Table 2). With random-effects statistical analysis, these small-sample studies may have overestimated the results. This and the above-mentioned restraints of observational studies are likely the reasons for the small-study effects detected from the Egger's test and funnel plots in some of the results of the meta-analysis.
Although the included studies were limited and the results may be subject to small-study effects, this review and meta-analysis provides an important preliminary synthesis of the available evidence regarding REAH. It presents a framework to support future research and highlights the key areas in which more robust data are required.
In conclusion, this systematic review and meta-analysis highlights the unique clinical and molecular characteristics of REAH, emphasizing its overlap with CRSwNP features and the need for a high level of suspicion alongside histological confirmation for accurate diagnosis. By reviewing the current knowledge, this study provides a foundation for improving diagnostic accuracy and optimizing the identification of REAH.
Supplemental Material
sj-docx-1-ajr-10.1177_19458924261440421 - Supplemental material for Clinical and Molecular Presentation of Respiratory Epithelial Adenomatoid Hamartoma: A Systematic Review
Supplemental material, sj-docx-1-ajr-10.1177_19458924261440421 for Clinical and Molecular Presentation of Respiratory Epithelial Adenomatoid Hamartoma: A Systematic Review by Frederick Lam, Alvin Po-Ngai Chu, Fergus Kai-Chuen Wong, Jason Ying-Kuen Chan, Eddy Wai-Yeung Wong and Yuk-Fai Lau in American Journal of Rhinology & Allergy
Supplemental Material
sj-xlsx-2-ajr-10.1177_19458924261440421 - Supplemental material for Clinical and Molecular Presentation of Respiratory Epithelial Adenomatoid Hamartoma: A Systematic Review
Supplemental material, sj-xlsx-2-ajr-10.1177_19458924261440421 for Clinical and Molecular Presentation of Respiratory Epithelial Adenomatoid Hamartoma: A Systematic Review by Frederick Lam, Alvin Po-Ngai Chu, Fergus Kai-Chuen Wong, Jason Ying-Kuen Chan, Eddy Wai-Yeung Wong and Yuk-Fai Lau in American Journal of Rhinology & Allergy
Footnotes
Acknowledgments
The manuscript was professionally edited for English language by Cambridge University Press Editing Services.
Ethical Considerations
This study synthesized publicly available published data and did not involve human participants or identifiable private information; therefore, Institutional Review Board approval was not required.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
