Abstract
Background
Eosinophilic chronic rhinosinusitis (eCRS) is a type 2 inflammatory disease that frequently recurs after surgery. In recent years, dupilumab has been available for the treatment of refractory chronic rhinosinusitis since 2020 in Japan. Although there are some reports of its usefulness, there are not enough reports of its clinical efficacy for longer than 1 year, especially for olfactory recovery.
Methods
Twenty patients with eCRS who had recurrence after surgery and had been receiving dupilumab were enrolled retrospectively. The nasal polyp score (NPS), computed tomography (CT) score, T&T olfactometer, and olfactory cleft opacification on CT were evaluated at baseline, at an average of 5.1 months later (short term), and at an average of 18.3 months later (long term).
Results
At the short-term evaluation, there were significant improvements in the NPS and CT scores (P < .001, P = .008, respectively). The CT score was further improved at the long-term evaluation compared to the short-term evaluation (P = .018) and baseline (P = .008). T&T detection/recognition thresholds and olfactory cleft opacification showed significant improvements only at the long-term evaluation compared to baseline (P = .002, P = .006, and, P = .006, respectively).
Conclusion
The NPS remained improved, and the CT score showed further improvement with long-term treatment, whereas olfactory function and olfactory cleft opacification showed significant improvement only after long-term treatment. There was a dissociation between the time to improve in the NPS and CT scores and the time to improve in olfactory function and olfactory cleft opacification. Based on these results, dupilumab should be administered for longer than 1 year, especially for olfactory function.
Keywords
Introduction
Chronic rhinosinusitis (CRS) is defined as the presence of symptoms such as nasal blockage, obstruction, congestion, nasal discharge, facial pain/pressure, and reduction or loss of smell for longer than 12 weeks. CRS is divided into primary and secondary CRS, and each is subdivided into localized or diffuse diseases based on the anatomic distribution. When the disease is classified by endotype dominance, primary CRS is divided into either type 2 or non-type 2 inflammatory endotype. Eosinophilic CRS (eCRS) is classified as primary diffuse CRS with type 2 inflammation. 1
In Japan, the concept of eCRS was first proposed by Haruna et al 2 in 2001 based on the presence of a phenotype characterized by eosinophilic infiltration of nasal polyps and sinus mucosa, bilateral nasal polyps, and olfactory disturbance in cases of refractory CRS that recurred after endoscopic sinus surgery (ESS). Although the concept of eCRS was widely known, the absence of diagnostic criteria had been a clinical problem. In 2015, a Japanese multicenter study established diagnostic criteria for eCRS requiring both a Japanese Epidemiological Survey of Refractory Eosinophilic Chronic Rhinosinusitis (JESREC) score of 11 or higher and a mucosal eosinophil count of 70/HPF (high-power field) or higher. 3 Some patients with eCRS have polyp recurrence despite local corticosteroids and nasal lavage, and systemic corticosteroids are needed. Although systemic corticosteroids are effective for recurrence, which does not continue over the long term, and are an important treatment option, there are several side effects associated with their long-term administration. 4
There was no effective treatment for these refractory cases of eCRS other than systemic corticosteroids until dupilumab, an anti-IL-4/IL-13 receptor monoclonal antibody, was approved in 2020 as a new treatment for refractory CRS with nasal polyps (CRSwNP) in Japan, following its approval for atopic dermatitis and bronchial asthma. In SINUS-52, a phase III study of dupilumab, the nasal polyp score (NPS), computed tomography (CT) score, and nasal congestion or obstruction score showed trends toward improvement early in treatment and a significant difference at 24 weeks. The results showed gradual improvements thereafter, with significant improvement compared to the placebo group at 52 weeks. 5 In addition, subjective symptoms of olfactory impairment improved on day 3 of treatment, and significant improvement in the University of Pennsylvania Smell Identification Test (UPSIT) score was already observed at 2 weeks, and both subjective symptoms and the UPSIT score showed significant improvements compared to placebo control at 24 weeks. 6
Dupilumab was introduced into clinical practice for bronchial asthma, a similar airway disease, earlier than CRS, and its efficacy has been shown to persist beyond the first year of administration. 7 However, the observation period for SINUS-52 was up to 52 weeks, 5 and its long-term efficacy beyond 1 year is unknown. The European position paper on rhinosinusitis and nasal polyps 2020 (EPOS2020) 1 recommends efficacy assessment at 16 weeks and 1 year after initiation of biological treatment, but there is no information on treatment guidelines after 1 year. In addition, there are still few reports on the efficacy of dupilumab outside of the clinical trial setting. Therefore, it is necessary to establish evidence regarding the efficacy of dupilumab for longer than 1 year. Because the effect of long-term treatment on eCRS is unknown, we hypothesized that long-term treatment with dupilumab is effective in improving CT, endoscopic, and olfactory-related outcomes in patients with refractory eCRS beyond the first year of administration. In this study, the effects of long-term treatment with dupilumab for refractory CRS, including olfactory-related outcomes, for more than 1 year were investigated.
Materials and Methods
Study Population
Appropriate IRB approval from the Ethics Committee of Dokkyo Medical University Hospital was obtained for this study (R-58-C109). The electronic medical records of patients who started treatment with dupilumab after April 2020 and received it for more than 1 year were reviewed. Of these patients, patients who had undergone CT, endoscopy, and olfactometer (T&T olfactometer) testing prior to administration (baseline), at an average of 5.1 months later (short-term), and at an average of 18.3 months later (long-term) were included. All patients underwent ESS for CRSwNP and subsequently had polyp recurrence, which was poorly controlled with existing conservative treatments. In addition, dupilumab was started in patients with the NPS of at least 5 of 8 bilaterally (at least 2 in each nasal cavity), the score for nasal obstruction of at least 2 of 3, and the presence of olfactory disturbance and nasal discharge at the time of recurrence. Seventeen of 20 patients had been receiving continuous oral corticosteroid therapy, and oral corticosteroids were stopped in all patients when dupilumab administration was started. The use of nasal corticosteroid spray and nasal lavage was continued.
Outcomes
The NPS, CT score (Lund–Mackay score),
8
and detection/recognition thresholds by T&T olfactometer testing were investigated. The NPS was assessed by nasal endoscopy with a 4.0-mm 0-degree rigid endoscope. In addition, to evaluate the anti-inflammatory effect of dupilumab on the olfactory cleft, the opacification area of the olfactory cleft on CT was measured at each evaluation. For olfactory cleft opacification, the imaging conditions were identical to ensure measurement reproducibility, and the images used were defined as 3 slices based on anatomical features. Considering the anatomical location of the olfactory mucosa,
9
the position of the olfactory mucosa in coronal sections was defined as (1) the position where the crista galli appears the largest, (2) 6 mm behind the slice in (1), and (3) 6 mm further behind the slice in (2) (Figure 1A). In measuring the occlusion rate, the FIJI package of ImageJ (National Institutes of Health (NIH), Bethesda, MD, USA) was used to trace the medial part of the superior and middle turbinate and the cribriform plate, and the space created by linearly connecting the lower edge of the middle turbinate mucosa was set on both sides as the olfactory cleft. After the images were binarized, the percentage of area other than air was measured as olfactory cleft opacification (Figure 1B). The images were anonymized and randomly measured to eliminate examiner bias. Since there is no established method for evaluating olfactory cleft opacification on CT, it was evaluated with reference to previous reports.
10
The differences from previous reports were (1) the interval between the 3 slices was kept constant; (2) the anatomical index (crista galli) was used as the merkmal; and (3) the area below the olfactory cleft was enlarged. In addition, by using the crista galli as the anatomical merkmal, the reproducibility of the slices was ensured. In a previous report, the olfactory cleft was defined as an area 1 cm from the base of the skull in the vertical direction.
10
However, since the thickening of the olfactory cleft mucosa may cause conductive olfactory dysfunction, a larger area of the olfactory cleft was defined, and changes in the olfactory cleft opacification after dupilumab administration were investigated. Olfactory function was measured by the T&T olfactometer using 5 odorants: (A) b-phenyl ethyl alcohol, which smells like a rose; (B) methyl cyclopentenolone, which smells like burning; (C) iso-valeric acid, which smells like sweat; (D) g-undecalactone, which smells like fruit; and (E) skatole, which smells like garbage (Takasago Industry, Tokyo, Japan). Both detection and recognition thresholds for each odorant were obtained and averaged. The minimum clinically important difference (MCID) refers to the smallest change in a patient that can be considered beneficial before and after treatment. In T&T olfactometry, the distribution-based technique was employed, specifically entailing the computation of the minimum detectable change (MDC) via the utilization of the standard error of measurement (SEM) at a 95% confidence interval, in order to determine the MCID: MDC = SEM *
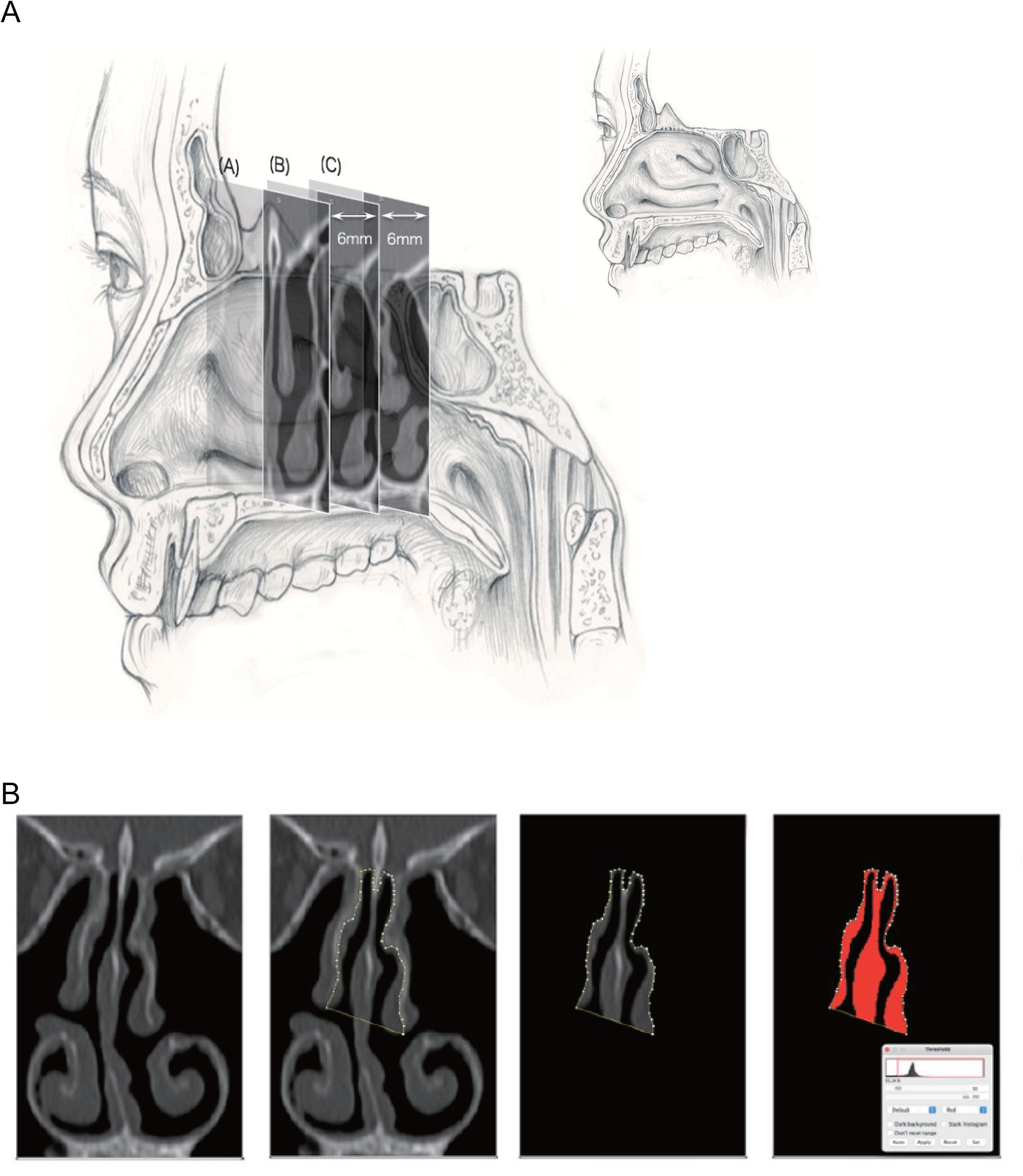
Olfactory cleft no pacification on computed tomography (CT): (A) slices were made at regular intervals using the crista galli as the merkmal and (B) olfactory cleft opacification is measured using the FIJI package of ImageJ.
Statistical Analysis
All data are expressed as means ± standard deviation. All analyses were performed using Prism version 9 (GraphPad Software, La Jolla, CA, USA). Differences between more than 2 groups were assessed by the Friedman test with Dunn's post hoc test. A P value of <.05 was considered significant.
Results
Twenty cases (13 males and 7 females) diagnosed with eCRS were enrolled in the study. Their mean age was 50.6 ± 14.0 years, 18 (90.0%) had bronchial asthma, and 7 (35.0%) had nonsteroidal anti-inflammatory drug-exacerbated respiratory disease (NSAID-ERD) (Table 1). The mean period from surgery to administration was 56.6 ± 48.2 months, and the number of previous surgeries was 1 for 17 patients and 2 for 3 patients. The baseline NPS before dupilumab administration was 6.0 ± 0.2, and the CT score was 12.1 ± 4.9. The T&T olfactometer detection threshold was 2.9 ± 2.0, the recognition threshold was 3.6 ± 1.8, and olfactory cleft opacification was 72.7 ± 19.2% at baseline. The patients were evaluated at 5.1 ± 2.9 months (short-term evaluation) and 18.3 ± 2.9 months (long-term evaluation).
Patients’ Characteristics.
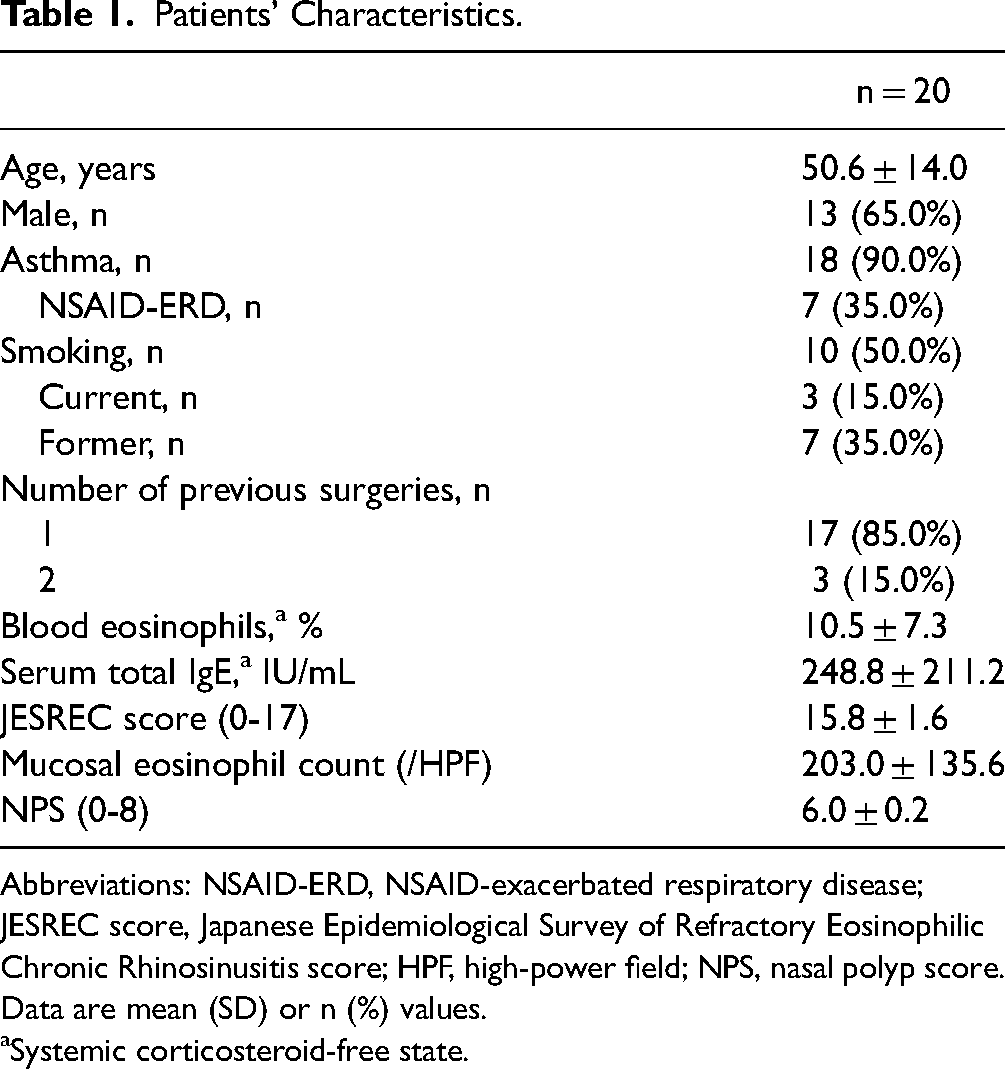
Abbreviations: NSAID-ERD, NSAID-exacerbated respiratory disease; JESREC score, Japanese Epidemiological Survey of Refractory Eosinophilic Chronic Rhinosinusitis score; HPF, high-power field; NPS, nasal polyp score.
Data are mean (SD) or n (%) values.
Systemic corticosteroid-free state.
The NPS improved significantly at both the short-term and long-term evaluations compared to baseline (6.0 ± 0.2 vs 0.8 ± 1.9; P < .001 and 6.0 ± 0.2 vs 0.2 ± 0.7; P < .001, respectively) (Figure 2A). Nasal polyps disappeared in the majority of cases at the short-term evaluation, and there was no significant difference between the short-term and long-term evaluations (P = .999). The CT score showed improvement not only at the short-term and long-term evaluations compared to baseline (12.1 ± 5.0 vs 4.8 ± 2.4; P = .008 and 12.1 ± 5.0 vs 2.2 ± 2.1; P < .001, respectively), but also between the long-term evaluation and the short-term evaluation (P = .018) (Figure 2B).
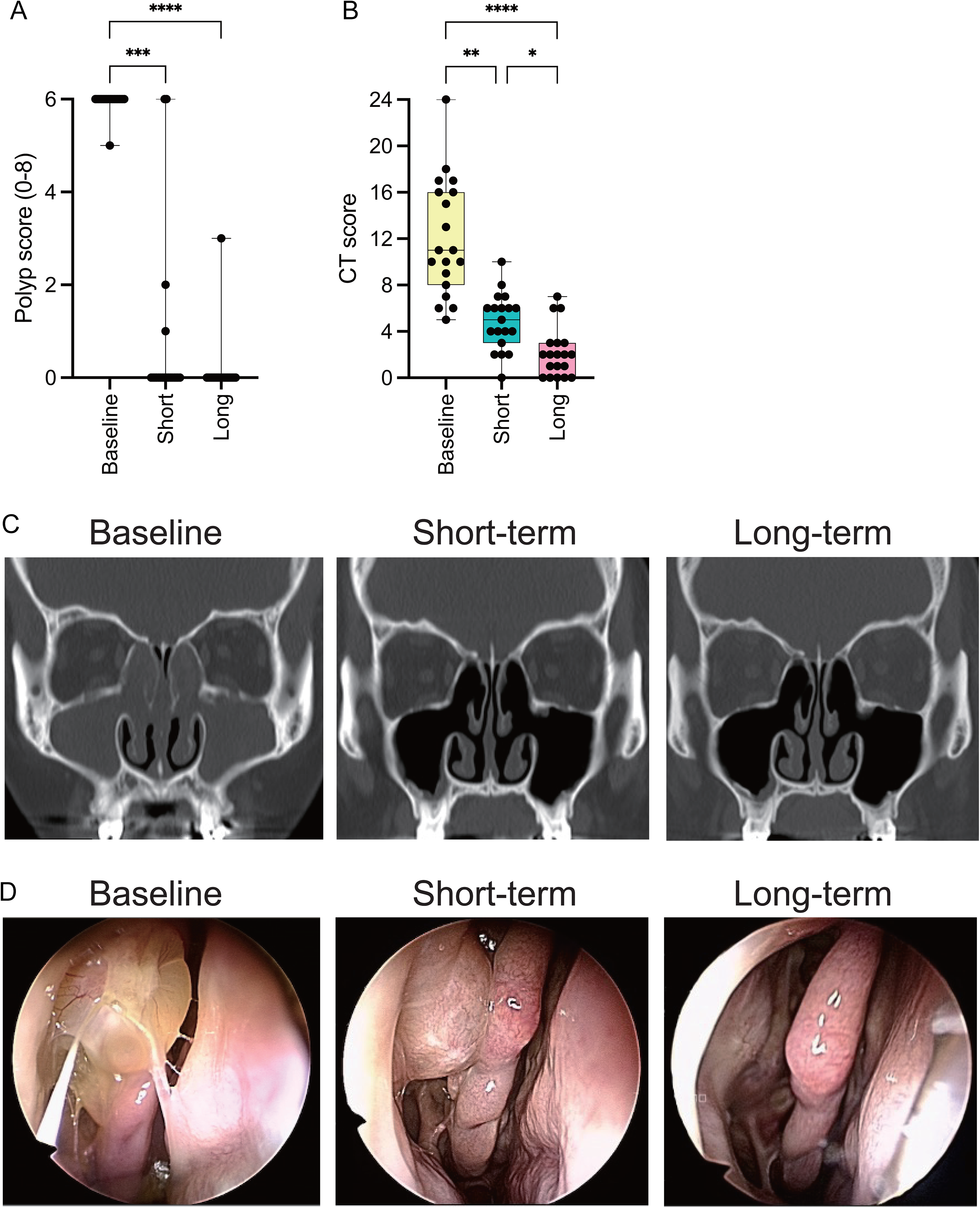
Changes of the NPS and CT score at each evaluation: (A) NPS improves significantly at both the short-term and long-term evaluations compared to baseline (6.0 ± 0.2 vs 0.8 ± 1.9; P < .001 and 6.0 ± 0.2 vs 0.2 ± 0.7; P < .001, respectively), (B) the CT score shows improvement, not only at the short-term and long-term evaluations compared to baseline (12.1 ± 5.0 vs 4.8 ± 2.4; P = .008 and 12.1 ± 5.0 vs 2.2 ± 2.1; P < .001, respectively), but also between the long-term and short-term evaluations (P = .018), (C) representative CT images at each evaluation and (D) representative endoscopic images at each evaluation. * P < .05, ** P < .01, *** P < .001, **** P < .0001.
The T&T olfactometer detection threshold showed no significant difference between the baseline and the short-term evaluation (3.1 ± 2.1 vs 1.5 ± 1.4; P = .595), but a significant difference between baseline and the long-term evaluation (3.1 ± 2.1 vs 1.1 ± 1.4; P = .006) (Figure 3A). Similarly, the T&T olfactometer recognition threshold showed a significant difference only between the baseline and the long-term evaluation (3.8 ± 1.9 vs 2.3 ± 1.3; P = .006). At the long-term evaluation, the MCID for the detection and recognition thresholds of the T&T olfactometer were 1.4 and 1.1, respectively, indicating the improvement surpassing the MCID. The olfactory cleft opacification was significantly different only between baseline and the long-term evaluation, like olfactory function (72.7 ± 19.8% vs 55.6 ± 8.5%; P = .002).
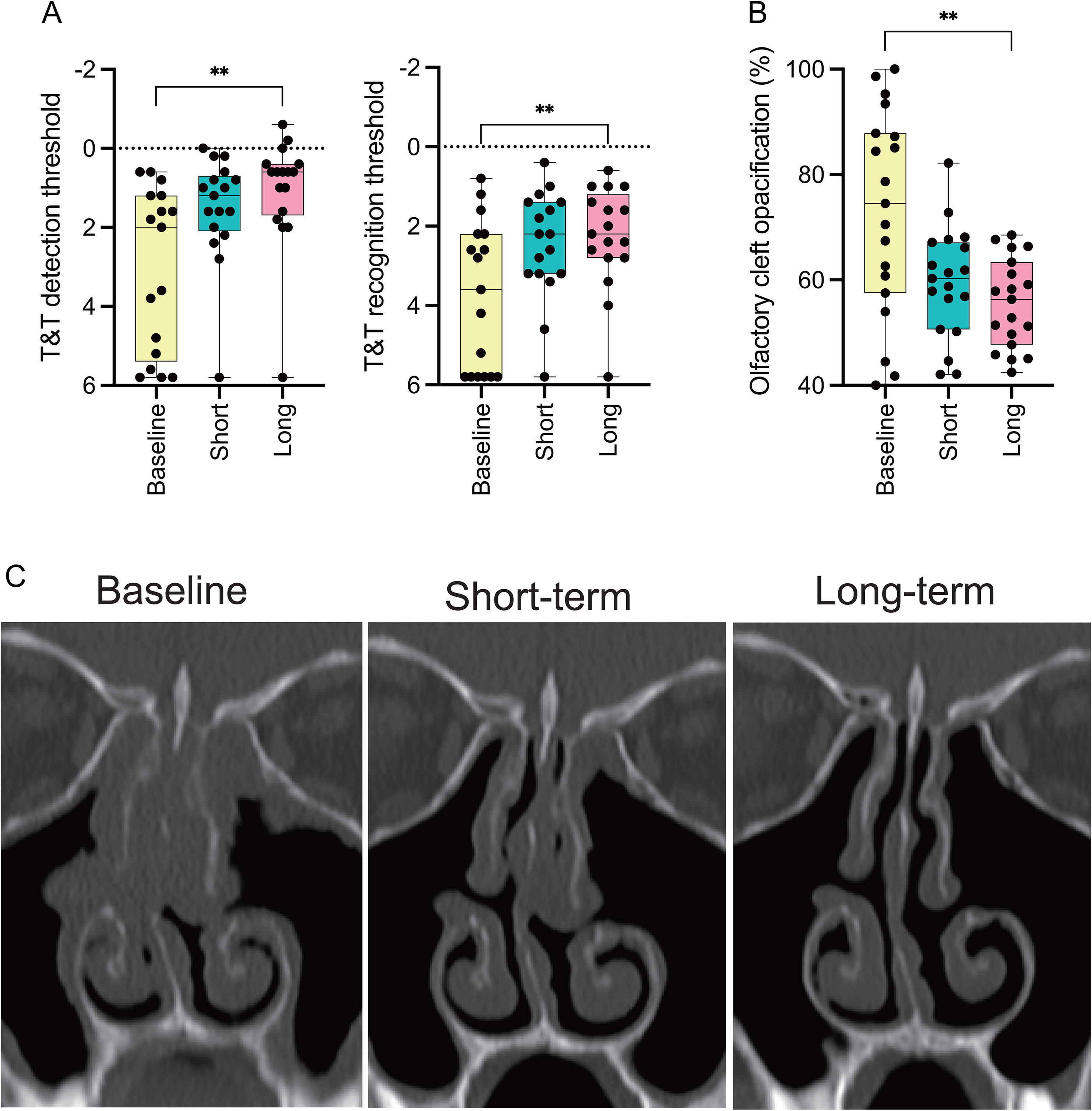
Changes in the T&T score and olfactory cleft opacification at each evaluation: (A) the T&T olfactometer detection/recognition threshold shows significant differences only between baseline and the long-term evaluation (3.1 ± 2.1 vs 1.1 ± 1.4; P = .006, 3.8 ± 1.9 vs 2.3 ± 1.3; P = .006, respectively), (B) olfactory cleft opacification is significantly different only between baseline and the long-term evaluation, like olfactory function (72.7 ± 19.8% vs 55.6 ± 8.5%; P = .002) and (C) representative images of olfactory cleft opacification on computed tomography (CT) at each evaluation. ** P < .01.
Discussion
Various studies have shown that eCRS is a type 2 inflammatory disease that involves type 2 cytokines such as IL-4, IL-5, and IL-13 induced by innate and acquired immunity.11,12 For eCRS patients with polyp recurrence after surgery, systemic corticosteroids have been used as an effective treatment, but repeated short courses of systemic corticosteroids, as well as continuous use, induce several side effects, and there is a major concern. Dupilumab is a monoclonal antibody against IL-4Ra, which is a common receptor for IL-4 and IL-13, and thus inhibits important IL-4 and IL-13 pathways in type 2 inflammation. Dupilumab has been approved in Japan since 2020 for the treatment of refractory CRSwNP. Nasal polyps recurring after appropriate and radical surgery are the most characteristic features of eCRS, and all patients in this study had eCRS that met the JESREC score and tissue eosinophil count.
Similar to the results of SINUS-52, dupilumab showed efficacy for the NPS and CT score at the short-term evaluation in the present study. Furthermore, the efficacy for the NPS and CT scores was still observed during the long-term evaluation, confirming longer efficacy than in the previous reports. In particular, CT, which had improved in the short term, improved even more at the long-term evaluation than at the short-term evaluation. CT allows a more precise assessment of sinus condition than endoscopic findings, and further improvement suggests the necessity for long-term dupilumab treatment (Table 2). In SINUS-52, the reductions in the NPS and CT score were −1.7 and −5.2, respectively, at 24 weeks, whereas in the present study, they were −5.2 and −7.3, respectively, at 5.1 months. Although the time periods evaluated were slightly different, the reason the present results were better than in the clinical trial would be due to the differences in the patients’ background characteristics. In SINUS-52, the concomitant rates of asthma and NSAID-ERD were 59.1% and 28.1%, respectively, compared to 90% and 35%, in the present study. In SINUS-52, the percentage of patients with a history of surgery was 78.7%, whereas that in the present study was 100% because the indication for dupilumab is only for CRSwNP patients with recurrence after surgery in Japan. On the other hand, CT scores were milder in the present study. Although the severity of the present patients appeared to have been milder at first glance, this result is due to the fact that 85% of the patients received continuous systemic corticosteroid therapy immediately prior to dupilumab administration. In addition, all patients who received dupilumab had no residual cells on CT, which is the risk factor for recurrence.13,14 These results suggest that almost all the present patients had type 2 inflammatory endotypes, which may have resulted in the higher efficacy of dupilumab. Even more noteworthy is that all these patients improved without the use of systemic corticosteroids.
The Outcome of Dupilumab Treatment for Chronic Rhinosinusitis With Nasal Polyps.
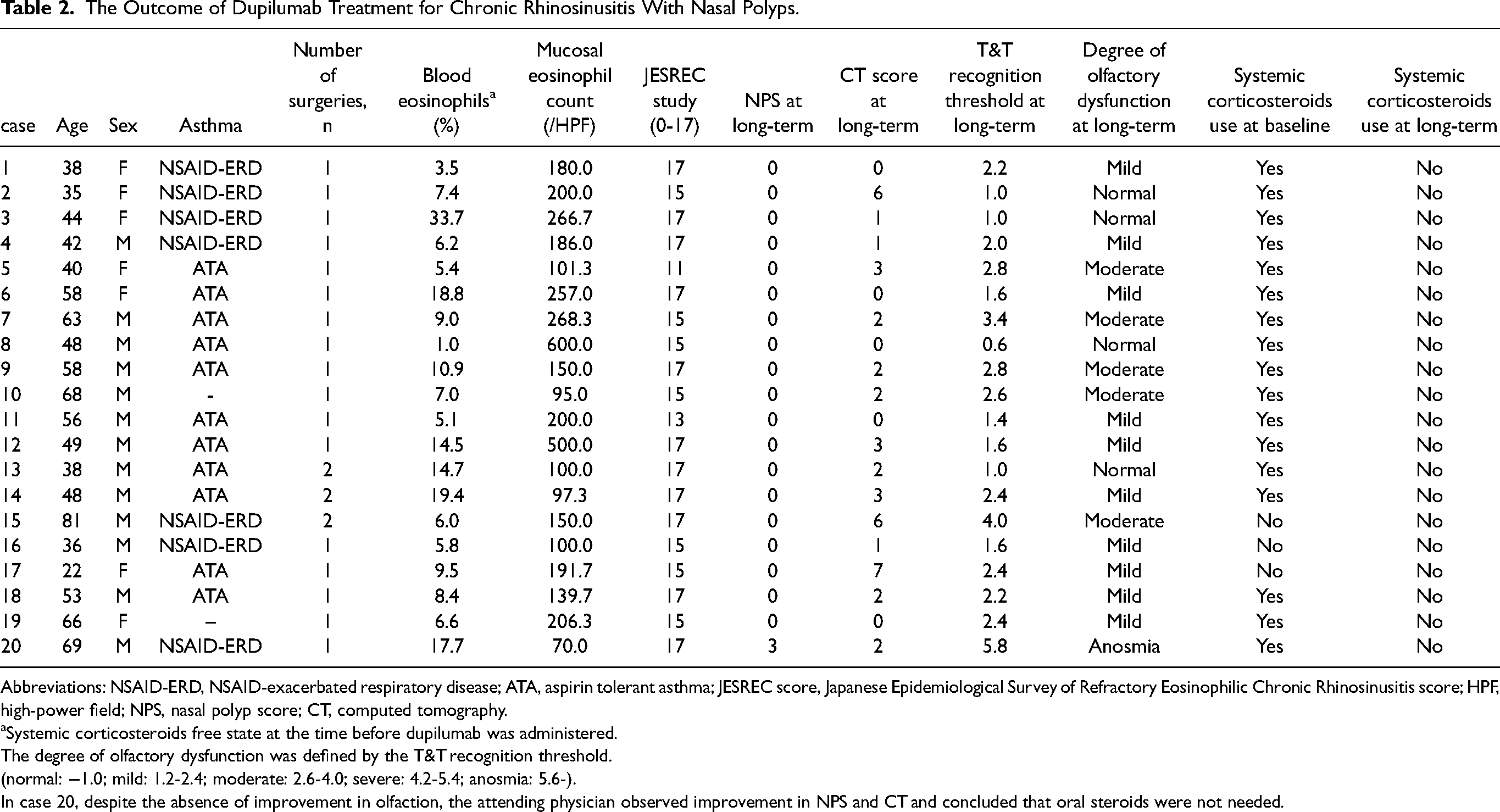
Abbreviations: NSAID-ERD, NSAID-exacerbated respiratory disease; ATA, aspirin tolerant asthma; JESREC score, Japanese Epidemiological Survey of Refractory Eosinophilic Chronic Rhinosinusitis score; HPF, high-power field; NPS, nasal polyp score; CT, computed tomography.
Systemic corticosteroids free state at the time before dupilumab was administered.
The degree of olfactory dysfunction was defined by the T&T recognition threshold.
(normal: −1.0; mild: 1.2-2.4; moderate: 2.6-4.0; severe: 4.2-5.4; anosmia: 5.6-).
In case 20, despite the absence of improvement in olfaction, the attending physician observed improvement in NPS and CT and concluded that oral steroids were not needed.
Olfactory dysfunction in eCRS is one of the major symptoms that improves at 3 months postoperatively, but it is difficult to maintain the improvement at 1 year postoperatively.15,16 However, the present study showed that long-term administration resulted in a significant improvement from baseline, indicating the usefulness of long-term administration. Although the mechanism by which type 2 inflammation causes olfactory dysfunction in the olfactory mucosa is still unclear, 2 mechanisms are considered to contribute to olfactory dysfunction in eCRS: conductive olfactory dysfunction and sensorineural olfactory dysfunction. 17 In the present study, the opacification of the olfactory cleft on CT was improved in the long term, as was the olfactory function. This means that inflammatory mucosal edema in the olfactory cleft is ameliorated by long-term administration, which may ameliorate conductive olfactory dysfunction. The olfactory mucosa of CRS, especially eCRS, is infiltrated with eosinophils, which are the characteristic immune cells of type 2 inflammation,18,19 as well as respiratory mucosa, and there is a decrease in mature olfactory sensory neurons. 15 The effect on sensorineural olfactory dysfunction is still unknown, but it has been suggested that dupilumab improves olfactory function at least through ameliorating conductive olfactory dysfunction by blocking IL-4 and IL-13 signaling, thereby suppressing type 2 inflammation in the olfactory mucosa.
The present study showed that there is a difference in the time period to improvement in NPS and CT scores and in the time period to improvement in olfactory function and olfactory cleft opacification on CT. One of the characteristics of eCRS symptoms is that, of the various symptoms, olfactory disturbances appear early. The reason for this is that the posterior ethmoid sinus inflammation, which is a characteristic feature of eCRS, affects the olfactory cleft via the superior turbinate and vice versa, 20 and the olfactory cleft is narrow due to its anatomical structure. Therefore, the anti-inflammatory effect of dupilumab may be slower to develop than its effect on the sinus as assessed by the polyp score or sinus CT. In fact, the degree of recovery of olfactory function after dupilumab treatment is independent of NPS reduction. 21 Furthermore, 6 months after dupilumab administration, despite marked improvement of the NPS, the olfactory function was reported to be improved in only two-thirds of patients. 22 These reports support the discrepancy between the time to NPS and CT score improvements and the time to olfactory improvement observed in the present study. However, the reason for this is unclear at this time, and further study is needed.
There are several limitations in this study. First, the time period in which the effect of dupilumab was evaluated was not constant, because it was a retrospective study. A prospective study would be warranted to evaluate the long-term administration of dupilumab. Second, the T&T olfactometer showed that olfactory function did not improve in some patients with olfactory anesthesia, despite improvement in the NPS and CT, including olfactory cleft opacification. Such patients are suspected to have irreversible olfactory neurogenic dysfunction, and this might have had an impact on the effect of dupilumab in the present. Third, despite the early improvement in olfactory function in SINUS-52, the improvement in olfactory function with the T&T olfactometer was not significant during the short-term evaluation. The reasons for this are the small number of patients and the difference in the tests used; SINUS-52 used the UPSIT, which is an olfactory identification test in suprathreshold olfactory tests, whereas the present study used the T&T olfactometer, which is an olfactory threshold test. If the number of patients were to be increased, there is a possibility that a significant difference would be observed. However, the NPS and CT scores improved even with a small number of patients at the short-term evaluation, and it is highly possible that there is a difference in timing between the improvement in olfactory function and the improvement in the NPS and CT scores.
Conclusion
Dupilumab was demonstrated to have longer-term efficacy than in SINUS-52 for the NPS, CT score, olfactory function, and olfactory cleft opacification. In particular, the time to improvement in olfactory function and olfactory cleft opacification differed from the time to improvement in the NPS and CT scores, indicating that long-term treatment is necessary to improve olfactory function. It is becoming clear that dupilumab will now become an alternative treatment option to systemic corticosteroids for refractory eCRS.
Footnotes
Acknowledgments
The authors would like to thank all the individuals who offered help and advice on this study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TN reports lecture fees and research grants, and TK and SH report lecture fees from Sanofi.
Ethical Approval
This study was conducted with the approval of the Ethics Committee of Dokkyo Medical University Hospital (R-58-C109).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
