Abstract
Background
Allergic fungal rhinosinusitis (AFRS) is an endotype of chronic rhinosinusitis (CRS) with nasal polyps characterized by eosinophilic mucin laden with fungal hyphae entrapped in expanded sinus cavities with an exaggerated hypersensitivity to fungal elements. The last decade has elucidated fungi-driven inflammatory pathways contributing to the pathophysiology of chronic inflammatory respiratory diseases. In addition, novel therapeutic biologic options have become available for CRS over the last several years.
Objective
To review the current literature examining AFRS, focusing on recent developments in our understanding of its pathophysiology and implications for treatment options.
Methods
Review article.
Results
Fungi-driven respiratory inflammation has been linked to fungal proteinases and toxin activity. In addition, AFRS patients demonstrate a local sinonasal immunodeficiency in antimicrobial peptides and hence limited antifungal activity, along with an exaggerated type 2 inflammatory response, highlighting a possible imbalanced type 1, type 2, and type 3 profile. The elucidation of these dysregulated molecular pathways has highlighted novel potential therapeutic targets. As such, the clinical management of AFRS, which once included surgery and extended courses of oral corticosteroids, is transitioning away from long courses of oral corticosteroids to incorporate novel delivery mechanisms of topical therapeutic targets and biologics for recalcitrant disease.
Conclusion
AFRS is an endotype of CRS with nasal polyps (CRSwNP) for which the molecular pathways leading to its inflammatory dysfunction are beginning to be illuminated. In addition to affecting treatment options, these understandings may shape necessary changes to diagnostic criteria and the extrapolated effects of environmental changes on AFRS. More critically, a better appreciation of fungi-driven inflammatory pathways may have implications for the understanding of broader CRS inflammation.
Keywords
Introduction to Allergic Fungal Rhinosinusitis
Chronic rhinosinusitis (CRS) is an inflammatory condition that affects the paranasal sinuses and has many representative subtypes. Allergic fungal rhinosinusitis (AFRS) is a subtype of CRS with nasal polyps (CRSwNP) that is characterized by eosinophilic mucin within expanded sinus cavities and immunoglobulin E (IgE)-mediated type 1 hypersensitivity to fungal elements. This noninvasive disease process typically affects immunocompetent, younger patients in geographic regions with warm temperatures and high humidity, which facilitate fungal proliferation.1–4 These patients typically report minimal sinus symptoms, which commonly include nasal congestion, thick nasal drainage, and anosmia/hyposmia. Severe cases can report visual changes, or even facial deformities as the disease progresses. These minimal symptoms contrast with the often-striking radiographic findings of paranasal sinus opacification with significant polyp burden and expanded paranasal sinuses with heterogenous intrasinus densities, and possible demineralization and erosion of the sinus walls seen on computed tomography (CT). 5 AFRS can present with unilateral disease. Additionally, over 30% of patients with AFRS can have a noninvasive expansion or erosion of the skull base or orbits, leading to telecanthus or other facial deformities.5,6 As symptoms can often be mild in these patients, they typically present later in their disease course with extensive disease characterized by thick, eosinophil-laden mucus. This makes these patients a challenge to treat with studies citing rates of nasal polyp recurrence after medical/surgical treatment as high as 60%. 7
Diagnostic Dilemma
The Bent and Kuhn criteria have been used to diagnose AFRS since its conception in 1994. The major criteria consist of: (1) type 1 hypersensitivity (IgE-mediated) to fungal elements, (2) nasal polyposis, (3) characteristic CT findings, (4) eosinophilic mucin without invasion of fungi, and (5) positive fungal stain. 8 Using Bent and Kuhn to diagnose AFRS, a patient must demonstrate all 5 of the major criteria. Despite their widespread use, these criteria have been criticized in recent years. Specifically, the nonspecific nature of each of its components. Type 1 hypersensitivity has been cited as something specific to AFRS; however, elevated fungal IgE can be found in patients with allergic rhinitis with fungal sensitization or those with CRS with fungal allergy.9,10 Limitations in technology at the time the Bent and Kuhn criteria were initially published further question their validity in the current treatment of AFRS. Montone et al examined the immunohistology of surgical samples for noninvasive fungal elements. In 25% of patients who would have met the Bent and Kuhn diagnostic criteria for AFRS, no fungus was identified. 11 However, if collected mucus is treated with a reducing agent prior to analysis, up to 97.5% of CRS subjects without AFRS have been shown to demonstrate growth on fungal cultures and the presence of fungal elements by staining nasal secretions.12,13 Hence, the criteria to demonstrate positive fungal stain within nasal secretions do not seem to be critical in AFRS.
Currently, there is a paucity of clinical trials investigating AFRS and future studies with AFRS patients may be limited due to the lack of agreed-upon diagnostic criteria. Previously published studies focused on AFRS have been criticized for their heterogeneous inclusion of patients based on the use of varying modified Bent and Kuhn criteria. One particular criterion that is difficult to satisfy is the demonstration of the presence of eosinophilic mucin without fungal invasion. Clinical trials that do not incorporate surgery may not provide a means of collecting the sinus secretions necessary to meet this criterion. In addition, it remains unclear how to demonstrate the lack of tissue invasion in AFRS, where CT findings may have bony erosion. These challenges demonstrate some of the limitations of the Bent and Kuhn criteria and highlight the need to re-address the diagnostic criteria for AFRS.
Pathophysiology Update
In recent years, our understanding of the pathophysiology of CRS as well as AFRS has continued to progress. The initial advancement was the appreciation that CRS represented an inflammatory disease state. Inflammatory responses associated with CRS fall under 3 possible types (types 1, 2, or 3). In Western countries, CRSwNP is typically associated with type 2 inflammation characterized by tissue eosinophilia and elevated type 2 (T2) cytokines, such as IL-4, IL-13, and IL-5.14,15 However, a study by Stevens et al 16 demonstrated heterogeneity of inflammatory profiles making up CRSwNP patients. Among CRSwNP patients, approximately 87% had T2 inflammation, either solely or mixed with type 1 (T1) or type 3 (T3) inflammation (with 60% showing single T2 inflammation). The other 40% consisted of single or mixed type 1, type 2, and/or type 3 inflammation. 16 Type 1 is characterized largely by type 1 innate lymphoid cells (ILC1) and adaptive T helper 1 (Th1) cells that secrete inflammatory mediators such as IFN-γ and stimulate phagocytic activity. Type 3 inflammation is differentiated by type 3 ILCs (ILC3s) and T helper 17 cells (Th17). Th17 cells are physiologically associated with protection against extracellular bacteria and fungi. The various subtypes of CRS, including AFRS, have shown varying expression levels of these markers. 17
In a study designed to molecularly differentiate AFRS from other CRSwNP patients, Tyler et al 18 conducted a comparative microarray analysis of diseased sinus tissue from patients undergoing sinus surgery for recalcitrant CRS. 19 While there was significant overlap between the 2 groups in terms of gene expression, AFRS demonstrated a differential upregulation of many mediators and pathways associated with Th2 cell differentiation and activation of IgE production. A novel finding in this study was the near absence of histatin gene expression in AFRS patients. Histatins consist of a family of 5 peptides differentially spliced from 2 histatin genes, initially described in the oral cavity 20 associated with wound healing as well as antibacterial and antifungal properties. There appears to be a negative correlation between histatin gene expression and many type 2 inflammatory markers in patients with AFRS. 19 In addition to histatins, other antimicrobial peptides (AMPs) such as LL-37 have been found to be decreased in AFRS as compared to other CRSwNP patients (Luong, unpublished data). This decrease in AMPs in AFRS patients creates an environment conducive to fungal overgrowth, as histatins have been shown to limit colonization of fungal species, including C. albicans on epithelial surfaces. 21 As such, methods for increasing endogenous AMPs or supplementation with exogenous AMPs may be a potential novel therapeutic target in the treatment of fungal diseases such as AFRS.
The understanding of Th17 and its response to fungal elements is another area of advancement in recent years. As the primary regulators of AMP expression, Th17 cells, like Th1 and Th2, are important mediators in immune responses, particularly against fungi. They are largely found in the intestinal and epidermal tissues, and their dysregulation is implicated in many local inflammatory disorders like cancer, chronic obstructive pulmonary disease, and inflammatory bowel diseases. 22 Particularly, the Th17 response is important in responses to C. albicans, and C. albicans-specific Th17 cells are found in the blood of virtually all healthy patients. 22 Thus, it is likely that a combination of reduced Th17 and T3 responses in conjunction with increased T2 and normal T1 responses characterize the inflammatory milieu of patients with AFRS, though the exact examination of this relationship is a topic that requires further investigation.
Type 2 fungi-linked respiratory diseases like AFRS and allergic asthma seemingly are due to a combination of both infectious processes and noninfectious hypersensitivity to fungal elements. In recent years, the understanding of the enzymatic mechanisms that influence these processes has been elucidated. Several key molecular pathways activated by secreted fungal proteinases and toxins triggering a type 2 airway hyperreactivity characteristic of asthma have recently been described.23–25 Fungal proteinases induce both the allergic response to fungi as well as the activation of macrophages and parts of the innate immune system intended to limit fungal growth and tissue invasion. 26 Both pathways are driven by fibrinogen cleavage products, formed from proteinase-cleaved fibrinogen. These cleavage products interact with Toll-like receptor 4 (TLR4) as well as Mac-1 to initiate a signaling cascade that results in both innate fungal immunity and airway hyper-responsiveness, neutrophilia, and eosinophilia seen in allergic airway disease. 25 Although proteinases are essential virulence factors for mold, yeast C. albicans induces allergic asthma via secretion of the peptide toxin candidalysin. Activating platelets to release the Wnt antagonist Dickkopf-1 (Dkk-1), candidalysin ultimately drives Th2 and Th17 responses aimed similarly to contain fungal tissue invasion while driving airway hyperreactivity. 23 This understanding helps develop the link between fungal disease and chronic respiratory inflammation and fungal disease and presents possible novel targets for intervention.
As previously mentioned, IgE plays a large role in AFRS and is also an area of recent investigation. A recent study by Haruna et al 25 generated monoclonal antibodies produced by IgE-secreting cells harvested from nasal polyps of patients with AFRS. These antibodies were compared to those from patients with both eosinophilic CRS with nasal polyps (eCRSwNP) and healthy controls. 27 AFRS patients had higher serum antifungal IgE and IgG levels; however, those with AFRS did not show higher serum IgE titers for fungal cell wall polysaccharides compared to healthy subjects or those with eCRSwNP. In addition, 55% AFRS patients demonstrated serum reactivity to more than 1 fungal genus, as well as a higher reactivity to major fungal antigens. In addition, the nasal mucosa of AFRS patients produced IgE, which specifically targeted surface cell wall polysaccharides in a similar manner to IgGs in patients with fungal infections. Interestingly, these IgEs did not contribute to the serum IgE reactivity in individuals with AFRS, suggesting a nasal production of IgE antibodies as a response against local bacteria or fungus. 27 The last several years have seen novel investigations into the pathophysiology of fungi-driven inflammatory respiratory diseases.
Climate Change and AFRS
Aside from the physiologic factors that can influence fungal infection, there are important environmental changes to consider. It is known that fungal spores are more prevalent in warm, humid conditions.3,4 Though data are limited, regions like the southern United States and the Mississippi Basin have reported the highest prevalence of AFRS. 3 A recent systematic review by AlQahtani et al looked at 18 studies comparing AFRS prevalence in 35 cities with different climates. 4 Their results again demonstrated significantly higher average temperatures in areas of high AFRS prevalence. They also found that mid-latitude moist climates with cold winters (eg, Cape Cod, MA; Pittsburgh, PA) had a significantly lower prevalence of AFRS and no significant relationship between humid climate types and AFRS incidence. In fact, areas with low AFRS prevalence were found to have significantly higher humidity. It should be noted, however, that the studies compiled in the analysis were limited to 35 cities, and the previously mentioned mid-latitude moist climates were also subcategorized as humid continental climates. Additionally, 9/11 (82%) of cities with high incidence of AFRS (defined as prevalence >10.1%) were in tropical, subtropical, or wet equatorial climates. Unfortunately, due to dearth of data, it is difficult to make many conclusions about climate and AFRS outside of the United States. Regardless, it is known that weather changes can have an impact on environmental levels of fungal spores. Not surprisingly, climate change has and will likely affect the distribution of fungal infections as regions experience changes in temperature and humidity. We have already seen an increased incidence of infections from the fungal species Cryptococcus gattii in western Canada and the Pacific Northwest United States, a species typically associated with tropical and subtropical climates. 26 It is possible that other fungal species may see similar changes in their geographic spread through gradual changes in climate. Furthermore, acute weather events linked to climate change can further disrupt the geographic tendencies of AFRS. Dust storms, tornadoes, hurricanes, and other large weather events have been linked with dispersing fungal elements from endemic areas in the past. 26 It is thought that climate change will not only alter weather patterns but also potentially give rise to such extreme weather events. 27 Together, these effects of climate change will likely result in increases in rates of AFRS in regions with historically low incidence, further heightening the need for improved treatment modalities.
Treatment Update
In the treatment and management of patients with AFRS, surgery combined with topical and oral corticosteroids is the standard of care (Table 1). Endoscopic sinus surgery to open the paranasal sinuses allows for the removal of eosinophilic mucin and debris as well as the penetration of irrigations and topical medications in the postoperative period. 28 In the past, patients were also managed with long-term, high-dose oral corticosteroids. Recent studies have highlighted multiple doses and duration-dependent complications associated with oral corticosteroids, including hyperglycemia, infection, poor wound healing, avascular necrosis, and increased intraocular pressures, among others.29,30 As we appreciate these adverse effects of long-term use, the paradigm has shifted away from oral corticosteroids and now focuses on improved delivery of topical corticosteroids. A recent randomized controlled trial (RCT) demonstrating no additional benefit of postoperative prednisone over placebo for CRS without nasal polyp patients highlighted the worse psychological scores in those receiving prednisone. 31 Given these heightened concerns about adverse effects, oral corticosteroids now have a diminished role in the current treatment paradigm for patients with CRS and AFRS.
Comparison of the Benefits and Utilization of Each Treatment Methodology for AFRS.
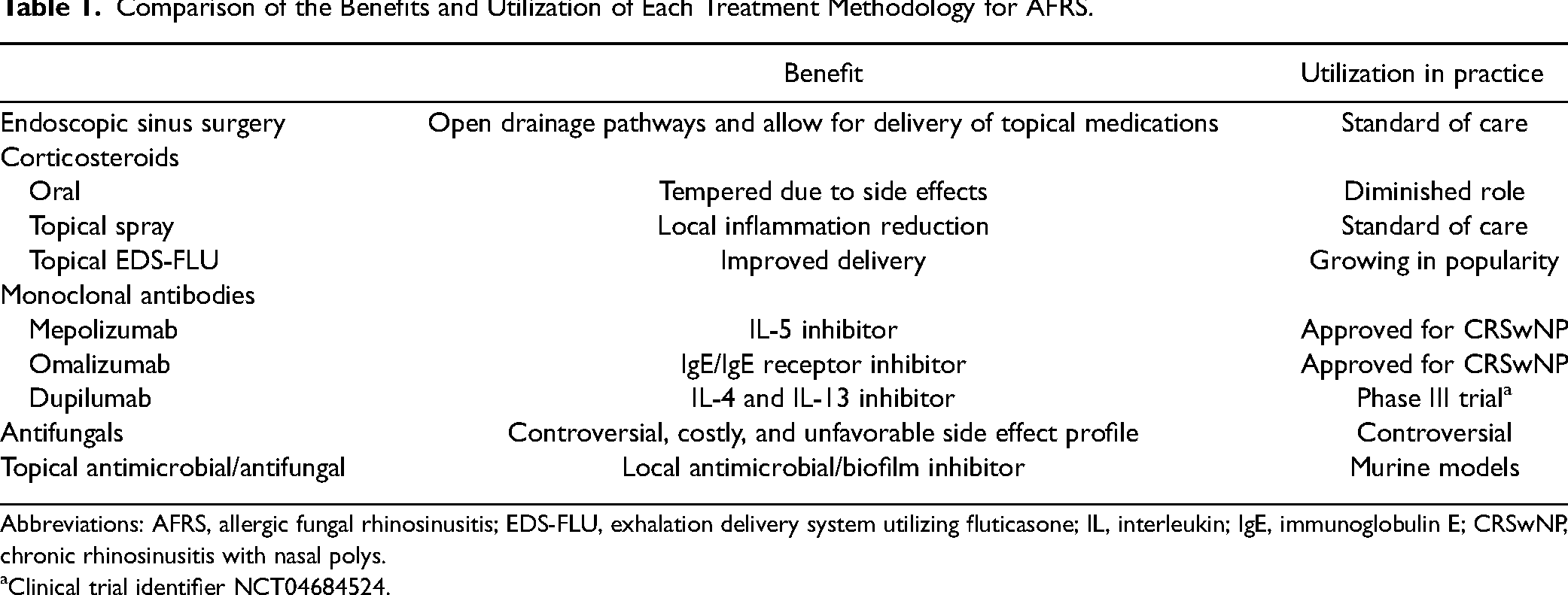
Abbreviations: AFRS, allergic fungal rhinosinusitis; EDS-FLU, exhalation delivery system utilizing fluticasone; IL, interleukin; IgE, immunoglobulin E; CRSwNP, chronic rhinosinusitis with nasal polys.
Clinical trial identifier NCT04684524.
Topical corticosteroids remain a mainstay as they have few systemic effects. However, topical corticosteroids in unoperated patients show very limited distribution and thus little effectiveness in treating CRS patients with nasal polyps due to anatomic obstructions preventing medication delivery.32,33 Topical steroids after endoscopic sinus surgery, however, show significant improvement in delivery and symptoms in CRS patients.33,34 While nasal topical steroids are beneficial, in recent years, it has been found that traditional methods of delivery (sinus rinses and sprays) have limited deposition in the upper and posterior aspects of the paranasal sinuses.33,35 An exhalation delivery system utilizing fluticasone (EDS-FLU) has shown significant promise in recent years. EDS-FLU utilizes a nosepiece that forms a seal with the vestibule and a mouthpiece, which the patient uses to blow medications into the nose. By utilizing a closed palate during exhalation, the EDS-FLU has demonstrated expanded delivery in postoperative patients with open sinuses. 36 This extended delivery of corticosteroids into the sinuses has resulted in improved symptoms and decreased polyp burden in RCTs involving CRSwNP patients.37,38 Importantly, exhalation delivery systems have shown improvement in these metrics even in patients with persistent symptoms despite prior use of traditional nasal sprays and/or prior surgery. 39
Although we have made improvements in the management of CRS and AFRS through improved delivery of nasal topical corticosteroids, the treatment of the underlying fungal pathology requires further refinement. To meet this need, the role of antifungal medications has been investigated. To date, however, the use of such medications has not been optimized. This is likely due to a combination of their exorbitant cost and harmful side effect profile, both of which make oral antifungals untenable for many patients. 40 There have been, however, recent RCT investigating perioperative antifungals in patients with AFRS with mixed results. One such study compared postoperative systemic and topical steroids with postoperative oral itraconazole. 41 These 2 therapies demonstrated no significant difference in change in absolute eosinophil count, serum IgE levels, or sinonasal outcome test (SNOT) scores between the 2 groups. 41 In another RCT, preoperative versus postoperative itraconazole use was examined in AFRS patients. This study compared Lund Mackay and nasal endoscopy grades at 24 weeks after surgery to patients using steroids alone. 42 At 24 weeks, preoperative itraconazole resulted in significantly better nasal endoscopy and Lund Mackay scores compared to postoperative itraconazole and both groups had significantly higher scores than steroids alone. 42 To address the systemic issues of utilizing oral antifungals, topical AMPs have recently been investigated as a potential therapy for chronic sinus disease. These peptides show promise as general antimicrobial therapies and have anti-biofilm properties that are particularly beneficial to those with chronic sinus disease.43,44 A recent study examined the effects of synthetically derived AMPs on both inflammatory markers and bacterial load in a murine model for rhinosinusitis. They found that topical application of these peptides not only showed a significant reduction in nasal bacterial load, but also a significant reduction in inflammatory marker IL-6, which is associated with nasal polyp formation. 44 As biofilm formation likely plays a role in CRS and AFRS, these therapies offer a promising, potentially low-risk treatment for patients with chronic sinus disease and suggest there may be a place for antifungals in the management of AFRS, though further investigation is needed.
Alternative therapies are emerging in the treatment of CRS and AFRS. These biologics target type 2 inflammatory mediators: IgE, IL-4, IL-5, and IL-13. Omalizumab, dupilumab, and mepolizumab are currently approved for the treatment of CRSwNP based on phase III studies.45–47 However, AFRS patients were excluded from these trials. Dupilumab, a biologic agent that inhibits IL-4 and IL-13 signaling, is currently undergoing phase III clinical trials in AFRS patients (NCT04684524). As of now, the published literature as to its effectiveness is limited to case reports and case series. Multiple case reports demonstrate a benefit in both objective CT scans and subjective SNOT-22 scores in previously recalcitrant AFRS patients after several weeks of dupilumab.48,49 A recent retrospective case series by Bulkhi et al 50 examined the use of dupilumab in 4 patients with partially or poorly controlled asthma and previously operated AFRS resistant to maximal medical management. At 4 to 6 months after biweekly dupilumab therapy, all 4 patients reported large improvements in SNOT-22 scores (average decrease of 56.25), asthma control tests, pulmonary function tests, and serum eosinophils. This study was limited by the small sample size, and statistics were not available. 50
Mepolizumab is an IL-5-inhibiting monoclonal antibody that is also currently under investigation. The only published study on its effectiveness retrospectively examined 27 recalcitrant AFRS patients with concurrent asthma who received monthly mepolizumab. 51 These patients were followed every 4 to 6 weeks for 3 post-treatment visits. Over the course of their treatment, patients experienced significant improvements in endoscopic modified Lund–Kennedy (MLK) scores (P = .04), SNOT-22 scores (56 vs 43, P = .02), and eosinophil counts (P < .01). These improvements were found to be greater for patients with more significant disease, as patients who had a baseline polyposis score of 1 or more showed a significant reduction in MLK scores (P = .02) compared to those without polyposis (P = .90). Mepolizumab was also well tolerated, and patients receiving mepolizumab required less rescue doses of prednisone compared to control (P = 1). 51
While IL-4 and IL-5 can be elevated in several subtypes of CRS, AFRS is differentiated from other subtypes by its elevated, often exaggerated, serum IgE levels. Omalizumab is a monoclonal antibody that binds free IgE and reduces its receptors on granulocytes, making it a good candidate for patients with AFRS. A single-blind, case-controlled RCT of recalcitrant AFRS patients who had previous surgeries and failed medical management compared 10 patients who received a single dose of postoperative omalizumab with 10 patients who received intranasal corticosteroids for 6 months. 52 After 6 months, the omalizumab group found significantly improved SNOT-20, total nasal symptom scores (P = .02 and P = .001, respectively), and IgE levels (P = .02 at 12 weeks), though there was no difference between the 2 in endoscopic staging scores (Philpott–Javer, P = .1). Additionally, there was no significant difference in the complications between the 2 groups. 52
Conclusion
AFRS is a subset of CRSwNP that is characterized by IgE hypersensitivity to fungal elements, type 2 inflammatory reaction, and eosinophil-laden mucin. Recent developments have elucidated additional details in the pathophysiology of this disease process. The interplay of Th1, Th2, and Th17 cells is now better understood, along with the molecular pathways activating these immune responses by key fungal proteinases and toxins. These advancements identify novel potential therapeutic targets, adding to current therapies that focus primarily on surgery and nasal topical corticosteroids. The growing role of monoclonal antibodies in the treatment of AFRS is in its infancy but is showing promise in recalcitrant patients. Further investigation into these therapies is warranted.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Amber U. Luong—Consultant: Acclarent, Lyra Therapeutics, Maxwell Biosciences, Stryker ENT, Medtronic, ENTvantage and Sanofi; Advisory boards: AstraZeneca and GlaxoSmithKline; Grant support: Sanofi; Speaker honorarium: GSK and Aerin Medical.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
