Abstract
Objectives:
To provide a contemporary update on the pathophysiology, diagnosis, and treatment of allergic fungal rhinosinusitis (AFRS).
Methods:
A review article on the pathophysiology, diagnosis, and treatment of AFRS was performed.
Results:
AFRS is a subtype of chronic rhinosinusitis with nasal polyps characterized by noninvasive fungal hyphae, eosinophilic mucin, and a type 2 inflammatory response, resulting in significant clinical and radiographic presentations. Although a diagnostic criterion has been used since the 1990s, advances in the pathophysiology and treatment of AFRS question the specificity and applicability of such criteria. Furthermore, the development and progression of AFRS are closely associated with the social determinants of health (SDOH) with a possible genetic component that limits sinonasal antifungal activity. Surgery followed by topical corticosteroids and nasal saline irrigations remains the mainstay of treatment with immunotherapy, antifungals, and biologics having a potential role.
Conclusions:
AFRS is a complex sinonasal inflammatory disease influenced by socioeconomic, genetic, and environmental factors. Future research is needed to investigate the influence of the SDOH on AFRS, advance diagnostic methods to optimize utility in everyday clinical practice and research, and elucidate the role of potential novel medical therapies in the current treatment pathway.
Introduction
Since the recognition of a new form of chronic rhinosinusitis (CRS) by Katzenstein et al,1,2 the understanding of allergic fungal rhinosinusitis (AFRS) has continued to evolve. While much has been published about this sinonasal inflammatory disease over the past 4 decades, the past 10 years have been fruitful in producing advances in the epidemiology, pathophysiology, and management of AFRS with questions even surrounding traditional diagnostic measures. Prior to the 2010s, understanding of AFRS focused on the diagnostic criteria created by Bent and Kuhn (type 1 hypersensitivity to fungi, presence of nasal polyps, characteristic Computed Tomography (CT) scan findings, eosinophilic mucus without fungal invasion into the sinonasal tissue, and positive fungal stain of sinonasal specimens), 3 the epidemiological factors recognizing increased prevalence in immunocompetent individuals living in warmer climates, the pathophysiologic understanding of an exaggerated type 2 inflammatory response, and treatment modalities, including sinus surgery, oral/topical steroids, antifungals, and immunotherapy.4-7
With advances in immunology, epidemiologic understanding, and pharmacology over the past decade, AFRS is now thought to be a noninvasive form of CRS with nasal polyps (CRSwNP) via an exaggerated inflammatory response to fungi possibly due to altered gene expression and interactions with the social determinants of health (SDOH). The SDOH are factors that determines a person’s health and well-being, including economic stability, education, health care access, race, and ethnicity. 8 Additionally, the recent development of novel biologics targeting specific inflammatory mechanisms could significantly impact how AFRS is managed medically in addition to surgical intervention. 9 Finally, although the Bent and Kuhn criteria has been the most-traditionally-accepted diagnostic standard of AFRS, the specificity and practicality of such diagnostic measure has been questioned in recent years. 10
This review has several unique components aimed at enhancing value in the realm of AFRS literature today. The primary objective was to evaluate the most up-to-date literature on AFRS, which includes analysis on the efficacy of biologic medications, the radiographic nuances of AFRS, and comparison of adult and pediatric disease. In doing so, the present review provides a contemporary and holistic understanding of AFRS via several avenues in addition to providing direction for future research.
Methods
A review of the recent literature from 2014 to 2024 was performed investigating the pathophysiology, diagnosis, and treatment of AFRS. Relevant articles were identified through the following databases: PubMed, Scopus, and Cochrane Library. Emphasis was placed on articles that provided novel findings not included in previously-published review articles, systematic reviews, and meta-analysis.
Results
Epidemiology
The prevalence of AFRS has been reported to be about 8% with a range of 0.2% to 27% among CRSwNP patients.11-13 Compared with the overall population of CRS patients, AFRS patients have unique demographic characteristics that are in close association with the SDOH. 8 While CRS patients have been reported to be predominately Caucasian, female, and more likely to have adequate health insurance, AFRS patients are more likely to be African American, male, and either uninsured or under-insured.8,14 Additionally, AFRS patients are reported to be younger (usually <30 years old), atopic, and present with more objective signs of advanced disease than CRSwNP patients.10,15 However, AFRS patients may minimize sinonasal symptoms compared with their CRSwNP counterparts.10,16 Despite the difference in objective disease severity, AFRS patients are more frequently lost to follow up, which further alludes to the link between this disease process and the SDOH. 16 Moreover, geographic location has classically been associated with AFRS, 17 with a higher prevalence of this pathology in locations of higher temperatures, less humidity, and less windy climates. 13 Although AFRS has been reported worldwide, most cases of AFRS have been reported in the southern United States, especially along the Mississippi River basin. 17
Pathophysiology
AFRS is considered a distinct, more severe, subclass of CRSwNP,15,18,19 characterized by noninvasive fungal hyphae, eosinophilic mucin, and an exaggerated type 2 inflammatory response.9,15,20 The pathophysiology is thought to be a complex interplay of genetic, environmental, and immunologic factors. 21 Recent studies have suggested that the pathophysiology of AFRS is in part due to the decreased expression of certain histatin genes (HTN1 and HTN3) associated with a deficiency in IL-22 and IL-17 producing T cells within diseased sinonasal mucosa, leading to the decreased production of antimicrobial peptides with antifungal properties.9,22,23 Specifically, histatins are one class of antimicrobial peptides that were originally found in saliva and are notable for antifungal and wound closure activity. 23
Additionally, Connell et al found that AFRS patients harbor a low diversity, dysbiotic sinonasal microbiome dominated by Staphylococcus aureus, Streptococcus pneumoniae, and Haemophiles influenzae, in association with abundant fungi, including Malassezia, Aspergillus, and Curvularia. 21 S. aureus has been previously reported to possibly contribute to the pathophysiology of AFRS through superantigen potential to upregulate the type 2 immune response. 21 However, S. pneumoniae and H. influenzae, both known to have virulent bacterial properties may also contribute to the pathological response. 21
Although the exact pathologic mechanisms of AFRS remain unclear, the theory is that limited antifungal activity in the sinonasal mucosa via decreased production of antimicrobial peptides due to impaired gene expression allows for the persistence and expansion of sinonasal mycosis.9,15,23 In response, the fungi, either alone or synergistically with virulent bacteria, upregulate the type 2 inflammatory response, which is characterized by tissue eosinophilia and elevated type 2 cytokines (IL-4, IL-13, and IL-5), 9 leading to airway remodeling with nasal polyposis, thick eosinophilic mucin secondary to goblet cell hyperplasia, and increased IgE production. 23 This type 2 inflammatory response is thought to have antifungal properties mediated by adaptive immune cells resulting in the recruitment of neutrophils, eosinophils, and macrophages, all of which have unique strategies to provide antifungal immunity, thus limiting the mucosal invasion of fungi. 23 However, without the aid of antimicrobial peptides, the fungi are unable to be cleared. Thus, the inflammatory response continues with the resultant symptoms of AFRS. 23 Environmental and socioeconomic factors also play a role in the development and progression of AFRS that have yet to be fully understood. 15
Diagnosis
Classically, AFRS is diagnosed by the 5 major criteria set forth by Bent and Kuhn in 1994, which includes the following: (1) type 1 hypersensitivity to fungi, (2) nasal polyps, (3) characteristic CT findings (serpiginous areas of high attenuation in affected paranasal sinuses), (4) eosinophilic mucus without fungal invasion into the sinonasal tissue, and (5) positive fungal stain of sinonasal specimens removed from surgery. 3 Although AFRS patients may report minimal sinonasal symptoms, 15 workup often reveals severe nasal polyps (refer to Supplemental Video 1) and CT findings, with the latter including expanded paranasal sinuses filled with heterogenous densities and erosion of the bony walls of the paranasal sinuses 24 that may present as unilateral or bilateral disease 25 (Figure 1). Moreover, about 30% of patients present with skull base or orbital expansion and erosion, which can lead to visual disturbance and facial deformity in severe cases.15,26,27 African American male patients were found to have more bony erosion with intracranial and intra-orbital extension than Caucasian patients. 28 Similarly, AFRS in the pediatric population has a comparable clinical presentation to those seen in adults but with a more aggressive course. 29 More than 50% of pediatric AFRS patients present with signs of facial skeletal changes, including proptosis, telecanthus, or malar flattening.11,29 Additional radiographic features common to AFRS compared with CRS include concha bullosa and longer distance between the low hanging anterior ethmoid artery and skull base, both of which may have implications during surgery.27,30,31 Within the paranasal sinuses, the eosinophilic mucus is thick with a consistency of peanut butter (refer to Supplemental Video 1) and contains necrotic and degranulating eosinophils with mucin, Charcot-Leyden crystals, and fungal hyphae.10,15,32
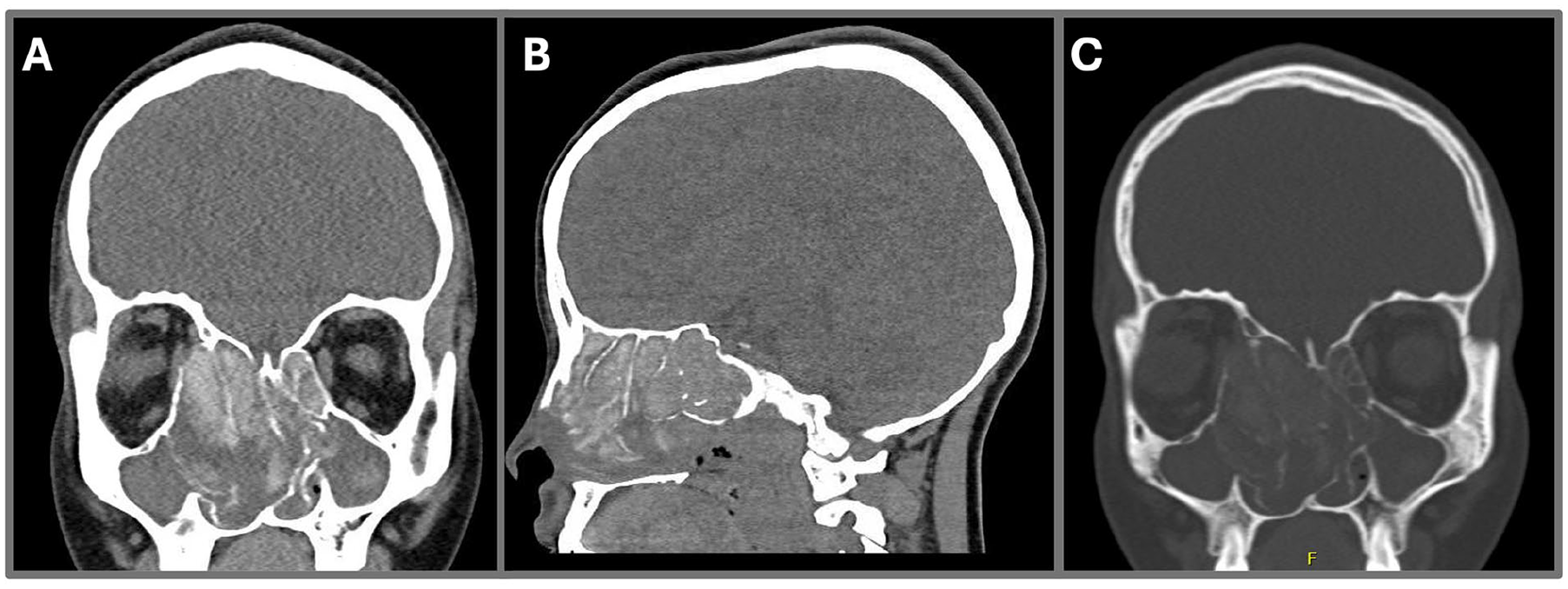
(A, B) Coronal and sagittal computed tomography in the soft tissue window demonstrating the right-sided AFRS causing expanded paranasal sinuses filled with hyper-attenuated densities, severe leftward septal deviation, thinning of the right skull base, and distortion of the right anterior ethmoid artery. (C) Coronal computed tomography in the bone window showing right orbital expansion from AFRS. AFRS, allergic fungal rhinosinusitis.
Although the Bent and Kuhn criteria has performed well in accurately diagnosing AFRS, limitations exist when applying the diagnostic criteria in both the clinical and research settings. 10 Currently, all 5 Bent and Kuhn criteria must be met to officially diagnose AFRS. However, when taken individually, some of the Bent and Kuhn features are not specific to AFRS and may overlap with other inflammatory sinonasal diseases. Additionally, in order to meet the Bent and Kuhn criteria, surgical intervention is often needed prior to an official diagnosis. 10
In regard to fungal stains and cultures, Ponikau et al found that 81% of CRS patients (both with and without polyps) had fungal elements on histologic specimens. 33 Additionally, Porter et al found that 68% of CRSwNP patients had positive fungal cultures.33,34 Even healthy controls without sinonasal disease have been found to harbor fungi in sinonasal secretions greater than 90% of the time.33,35 Additionally, no single fungus is pathognomonic for AFRS. While Aspergillus may have been historically synonymous with AFRS, Ponikau et al identified 40 different genera of fungi in CRS and AFRS patients. 33 However, a recent study by Connell et al observed that the AFRS mycobiome predominately contains Malassezia, Curvularia, and Aspergillus, while non-fungal CRSwNP patients harbor mainly Malassezia. 21 This difference observed in the mycobiome between AFRS and CRSwNP presents an opportunity to increase the specificity of AFRS diagnosis.
The difficulty in diagnosing AFRS within a population of CRSwNP individuals is further compounded by the fact that in addition to nonspecific fungal stains and cultures, eosinophilic mucin is found in both groups. 10 Given that type 2 inflammation, characterized by the recruitment of eosinophils and mast cells, is a common pathway for both CRSwNP and AFRS, eosinophilic mucin is frequently observed for both inflammatory sinonasal diseases. 10 Furthermore, type 1 hypersensitivity to fungi can be seen in allergic rhinitis with fungal sensitization. If these patients also had CRSwNP, the constellation of signs and symptoms could be confused for AFRS. 10
Finally, surgical intervention is often required to obtain sinus tissue samples, which is an additional limitation of the Bent and Kuhn criteria. With the need for surgery prior to an official diagnosis, recruitment of AFRS patients for research studies may be hindered as investigators have to balance the fine line between enrolling preoperative subjects to test possible medical interventions and being criticized for not strictly adhering to the Bent and Kuhn standard.
Treatment/Management
Surgical Management
Despite the improved understanding of the pathophysiology of AFRS as a unique disease entity, no disease-specific interventions exist for AFRS. Instead, the current mainstay of AFRS therapy is similar to that of CRSwNP. Sinus surgery is used to clear fungal eosinophilic mucin causing sinonasal inflammation, debride nasal polyps, and open the paranasal sinuses for improved penetration of subsequent sinonasal saline irrigations and topical steroid therapy.9,11,15,24 Clearing all gross fungal eosinophilic mucin is imperative. Therefore, preoperative planning with imaging is critical, as AFRS patients often have distorted anatomy due to bony erosion and progression of disease and are more likely than CRS patients to have concha bullosa on the affected side that is possibly harboring recurrent or residual disease.27,3 Additionally, AFRS patients are more likely to have a low-lying anterior ethmoid artery, which is associated longer distances from the skull base compared to other CRSwNP patients. 30 This characteristic makes AFRS patients at potentially higher risk for injury to this critical vascular structure during sinus surgery. 30
No studies have specifically compared surgical intervention versus medical management in the treatment of AFRS. 15 Regardless, endoscopic sinus surgery remains the most accepted initial intervention for AFRS with about 30% of patients needing revision surgery.15,36 In pediatric AFRS, surgical intervention with postoperative topical corticosteroids is also the standard of care. However, the recurrence of AFRS in children has been reported to be higher than that in adults. 29 A period of postoperative waiting may be beneficial between primary and revision surgery as a mean time of around 36 months was associated with postoperative osteogenesis, specifically at the orbit. 37 Additionally, although AFRS was traditionally thought to be a unilateral disease process, evidence has suggested that bilateral disease is just as common. Evolution of the disease process often causes disease recurrence in the contralateral paranasal sinuses, possibly due to subclinical involvement of the contralateral sinus at the time of initial presentation or progression of unilateral disease to the contralateral side. 25 Thus, upfront bilateral surgical and medical interventions has been proposed, although data are limited. 25
Medical Management
Systemic/Topical Steroids
Postoperatively, AFRS was once commonly managed with high-dose systemic corticosteroids on a long-term basis, 9 even though no studies exist to elucidate the optimal duration of such treatment. 15 However, given the complications associated with long-term oral corticosteroids, including hyperglycemia, infection, suboptimal wound healing, psychiatric manifestations, and glaucoma,9,38 topical corticosteroids are now the mainstay. This includes traditional nasal sprays, steroid nasal irrigations, and novel delivery methods, such as exhalation delivery system utilizing fluticasone (EDS-FLU). 9 Although, traditional nasal sprays are commonly used as the first-line delivery method, nasal sprays have limited exposure to the upper and posterior aspects of the paranasal sinuses.9,39 Improved delivery methods, like budesonide nasal irrigations, have proven to provide better exposure to the sinonasal mucosa than traditional nasal sprays, thus improving postoperative outcomes.40,41 Additionally, the EDS-FLU system improves delivery to the paranasal sinuses by using a closed palate during exhalation for expanded delivery of topical corticosteroids in postoperative patients with open sinuses, resulting in improved sinonasal symptoms and decrease in polyp burden.9,39 Although budesonide nasal irrigations and the EDS-FLU system may provide enhanced medication delivery compared with nasal sprays, these medications are often more costly and difficult to prescribe, which may be a barrier to use for some patients.
Nasal Saline Irrigations
In addition to topical corticosteroids, daily-nasal saline irrigations are routinely recommended as a postoperative care strategy. Previous reviews, including international consensus statements, have found that daily-postoperative nasal saline irrigations may improve early-postoperative symptoms and endoscopic appearance of the sinuses.15,42 Harm was minimal and only included local irrigation, nasal burning, headaches, and ear pain.15,42
Antifungal Therapy
Although fungus is thought to be the main offending pathogen in AFRS, antifungals have played an unclear role in the primary treatment of AFRS. This is historically due to insufficient evidence from only a few low-level studies investigating the role of antifungal therapy in AFRS patients. 15 Nevertheless, these previous studies have suggested that itraconazole may mildly improve postoperative sinonasal symptoms and decrease time to recurrence, although with a potential risk of liver enzyme elevation.43-45 The most recent study on antifungal therapy was a pre-post interventional study performed by Shah et al involving 34 patients. This study found that oral itraconazole 200 mg twice daily for 3 months without oral steroids was associated with symptomatic and radiological improvements in AFRS patients and could be used in place of steroids. 46 However, Shah et al noted that surgery should remain the primary intervention for AFRS patients and not antifungals. 46 The major limitation to this study was the lack of a control group to assess for possible confounders and the inability to assess the target serum concentration of itraconazole. 46
Immunotherapy
Given the pathophysiology of AFRS includes type 1 hypersensitivity to fungi, immunotherapy has been investigated as a potential medical regimen. To date, the literature is limited on the use of immunotherapy for AFRS, with only 1 systematic review revealing that immunotherapy may be of value to reduce mucosal inflammation and the amount of topical/systemic corticosteroids required. 47 However, due to the lack of high-quality studies (small sample sizes, absence of control groups, mix of immunotherapy and other medical treatments), no clear recommendation for or against immunotherapy can be made at this time. 15 Nevertheless, immunotherapy remains a potential treatment option, especially if the patient has other allergic symptoms.15,47,48
Biologics
Biologics, including omalizumab (binds free IgE), dupilumab (inhibits IL-4 and IL-13 signaling), and mepolizumab (inhibits IL-5), have been approved by the Food and Drug Administration (FDA) for the treatment of CRSwNP aged 12 years and older.9,48-51 However, these clinical trials excluded AFRS patients, thus leaving the literature on treating AFRS with biologics historically to case reports and case series. Nevertheless, more robust studies have been published in recent years with a systematic review and meta-analysis by Aljubran et al summarizing the results of biologic use specifically in AFRS patients. 52 Six studies were included with a total of 64 patients. 52 Of the 6 studies, 3 used dupilumab, 2 used omalizumab, and 1 used mepolizumab. Pooled analysis revealed that the use of biologics significantly improved SNOT-22 scores by 63%. 52 Additionally, total IgE levels, serum eosinophil count, and Lund-Mackay scores significantly improved after the use of biologics in AFRS patients. 52 However, exact conclusions from this systematic review and meta-analysis are limited due to the high heterogeneity among the studies included. Additionally, Bentan et al using data from the TriNetX Research Network database found that only dupilumab reduced the rate of sinus surgery, inpatient admission, emergency department visits, and acute sinusitis in AFRS patients compared with omalizumab and mepolizumab. 53 Currently, biologics are not approved for use by the FDA in AFRS patients. However, dupilumab is currently undergoing phase III clinical trial for AFRS patients who are at least 6 years old (NCT04684524). At this time, due to paucity of data, biologics can be considered for use in adult recalcitrant AFRS patients, 15 although further investigation is warranted.
Follow Up and Monitoring
Due to the high recurrence rate of AFRS, patients often require multiple interventions over a number of years. 11 However, given the link between AFRS and socioeconomic disparities, Gutierrez et al observed that AFRS patients are more likely to be lost to follow-up than CRSwNP patients. Among AFRS patients, young males are more likely to be lost to follow-up. 16 Further research is needed in elucidating the specific barriers to care for AFRS patients.
Discussion
As a subclass of CRSwNP with ample prevalence contributing to significant deterioration in quality of life, AFRS is a common topic in the literature with systematic reviews, review articles, and meta-analyses written yearly. Review articles and systematic reviews most recently published in 2023, including those by Chua et al, 10 Cameron et al, 9 and Gutierrez et al, 8 do a fine job in providing a comprehensive overview of AFRS, including the epidemiology, pathophysiology, diagnosis, and treatment. As such, this review article not only gives foundational knowledge of AFRS but builds on the previous literature to provide the reader with the most up-to-date advances and discoveries pertaining to AFRS not included in other reviews. Highlights of novel AFRS topics incorporated in this review article include the following: (1) radiographic nuances of AFRS compared with those of CRSwNP, such as greater prevalence of concha bullosa and longer distance between the low-hanging anterior ethmoid artery and skull base, which has surgical implications 30 ; (2) differentiating between pediatric and adult AFRS patients, noting the more aggressive course observed for pediatric patients 29 ; (3) highlighting nasal saline irrigations as an important part of the postoperative medical regimen, which some previous review articles and guidelines fail to do; and (4) overviewing the most updated published literature of biologic efficacy for AFRS patients, including those by Aljubran et al 52 and Bentan et al. 53
Although AFRS has been a known entity for the past 40 years with advances in the diagnosis, pathophysiology, and treatment, knowledge gaps still exist. First, the SDOH have been firmly established to be associated with AFRS as evidence of greater prevalence and severity of this disease in patients who are male, African American, and of lesser financial means. 8 As such, further research is needed to investigate how the SDOH influence the pathophysiology of AFRS, effects on treatment, and specific barriers to care to improve diagnosis and management for this patient population.
Second, even though the Bent and Kuhn criteria has withstood the test of time in being an accurate diagnostic tool for AFRS, the non-specificity of certain criteria and the need for surgical intervention 9 may prevent timely diagnosis and development of novel therapeutic interventions, especially with the creation of more powerful and promising medical options. Thus, diagnostic criteria that are unique to AFRS without the need for surgery are needed to improve research and management of this disease process.
Third, while biologics have shown promise in effectively improving sinonasal symptoms and objective markers of disease, 52 questions remain in the specific role biologics should play in AFRS in addition to the potential side effects with long-term use for both adult and pediatric patients.
Conclusion
The understanding of AFRS has continued to progress since the discovery of this unique sinonasal pathology. However, despite advances in the diagnosis, pathophysiology, and management of AFRS, much has yet to be understood about this subtype of CRSwNP. Thought to be in part caused by decreased gene expression of antifungal peptides in conjunction with a dysbiotic sinonasal microbiome, AFRS is closely associated with the SDOH, suggesting an environmental component to disease formation and progression. The current diagnostic criteria, although comprehensive, is limited in ability to make a timely and definitive diagnosis without surgical sampling, thus potentially restricting proper day-to-day clinical management and research. While surgery with postoperative topical steroids remains the standard of treatment, novel medical therapies, including biologics, may play an increasing role in the future. However, a deeper understanding of the effectiveness, duration of therapy, and long-term adverse effects of biologics in AFRS patients is required before routine use is recommended.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Philip Chen is a consultant for Medtronic, advisory board for Sanofi and GlaxoSmithKline, speaker’s bureau for GlaxoSmithKline and Optinose, and has research funding from Aerin Medical. None affect this research.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
