Abstract
The interaction of cocktail of phytochemicals from broccoli with gold salt results in dual reduction and surface capping to produce well-defined stable and biocompatible gold nanoparticles (B-AuNPs). Broccoli phytochemicals–coated gold nanoparticles (B-AuNPs) have been fully characterized. Detailed in vitro stability in various biological fluids and affinity and selectivity for tumor cells have been investigated. The B-AuNPs showed significant in vitro cytotoxic effects against various cancer cells (MDA-MB-231, PC-3, U266, SkBr3, and T47D) as confirmed by 3-(4,5-dimethyl thiazol-2-yl)-2,5-diphenyl tetrazolium (MTT) and flow cytometry apoptosis assays. Surface encapsulation of cocktail of broccoli phytochemicals on AuNPs facilitates the cellular internalization, thereby validating the in vitro therapeutic effects of these nanoparticles. Detailed analyses performed by combination of gas chromatography–mass spectrometry (GC–MS) and liquid chromatography–tandem mass spectrometry (LC–MS–MS) have confirmed the presence of biologically active phytochemicals including glucoraphanin, phenethyl glucosinolates, quercetin, folic acid, vitamin C, allyl isothiocyanates, 2-phenylethyl isothiocyanates, and sulforaphane. The unique synergistic cocktail effects of B-AuNPs will provide new opportunities for generating biocompatible AuNPs for molecular imaging and therapeutic applications.
Keywords
Introduction
Cancer continues to be a vexing health problem worldwide, as it is the second leading cause of death where a cure from a single modality is seldom possible. 1,2 Because of the high death rates and serious side effects associated with conventional therapies, many cancer patients seek alternative and complementary methods of treatment involving plant-based formulations that employ a plethora of cocktail of phytochemicals from fruits, herbal, and vegetable sources. 3 –5 A wealth of epidemiological studies indicate that consumption of large quantities of fruits and vegetables, particularly cruciferous vegetables (e.g., cabbage, kale, taramira, Brussels sprouts), is associated with a reduced incidence of cancer. 6 –8 Among the latter, broccoli (Brassica oleracea) certainly deserves attention. Broccoli belongs to the Brassicaceae family and is originated in the Mediterranean regions and distributed in Europe and in the United States. It is a popular vegetable in the United States and Europe. Broccoli is rich in micronutrients—carotene, vitamin C, and folic acid—fibers, phytochemicals such as glucosinolates (GLs), and polyphenols. 9,10 These phytochemicals induce phase-2 detoxication enzymes and antioxidant activities. 11,12 The phytochemicals in broccoli including GLs and flavonoids have recently garnered great interest for their potential role in the maintenance of human health. It has been speculated that the isothiocyanates (ITCs), obtained from myrosinase hydrolysis of GLs (by chewing, cutting, or processing the vegetable), are in great part responsible for the protective effects of Brassica vegetable. 13,14 The pharmacological efficacy of plant-based products has created a significant interest and awareness among the scientific community. Therefore, there is renewed interest in the development of new generation of pharmaceuticals derived from natural products.
A major impediment on the application of phytochemical-based pharmaceuticals is concerned with the lack of optimum bioavailability and thus low dosage of active ingredients reaching the target sites. Based on our pioneering efforts in green nanotechnology and nanomedicine, 15 –18 we are exploring to enhance the therapeutic payloads of biologically active phytochemicals in broccoli through their conjugation with AuNPs. The surface chemistry of AuNPs brings about a paradigm advantage allowing conjugation of cocktail of phytochemicals on the surface of AuNPs. This approach improves delivery and also enhances the bioavailability of phytochemicals cocktail from various plants and herbal sources. The larger surface area and the extraordinary kinetics of surface atoms of AuNPs allow highly effective loading of phytochemical cocktails in their nascent form onto the nanoparticles. The strong propensity of phytochemicals-coated nanoparticles (in the 50–100 nm hydrodynamic size range) allows efficient internalization of “diseases-preventive” and “disease-curing” phytochemicals into tumor cells through a combination of matrix, endocytosis, and phagocytic mediated pathways. Nanotechnology in herbal medicine would therefore allow storage, efficient transport, and delivery of reservoir of phytochemical cocktails into the cellular matrix. As part of our continued efforts to apply green nanotechnology for the creation of phytochemical-based nanoceuticals, we describe herein: (i) the synthesis and complete characterization of biocompatible AuNPs using cocktail of broccoli extract in aqueous media; (ii) in vitro anticancer activities of broccoli phytochemical-coated AuNPs (B-AuNPs) against prostate (PC-3), breast (triple negative MDA-MB-231, T47D, and SkBr3), and multiple myeloma (U266) cancer cell lines, and (iii) complete analysis of cellular internalization for potential applications in cancer diagnostics and therapy.
Results
Synthesis and Characterization of B-AuNPs
B-AuNPs were prepared by mixing aqueous solution of commercially available sodium tetrachloroaurate with broccoli extract in deionized (DI) water. The absorbance profile of these nanoparticles is shown in Figure 1. The surface plasmon resonance appeared at ∼535 nm. Transmission electron microscopy (TEM) and differential centrifugal sedimentation (DCS) techniques were used to determine the core size, while dynamic light scattering (DLS) was used to evaluate the hydrodynamic size and potential charge of phytochemical-coated AuNPs (Table 1). As seen in the TEM image (Figure 1), the size and shape of the nanoparticles are homogeneous with an average diameter of 20 ± 2 nm. The hydrodynamic size (90 ± 5 nm) of broccoli-coated gold nanoparticles is greater than the core size measured by TEM and DCS (20 ± 2 nm) techniques (Figure 1 and Figure S1). The negative zeta potential (ζ) value (−29.0 mV) for the B-AuNPs provides the necessary repulsive forces for the particles to remain stable in solution. In general, ζ provides important information on the dispersion of nanoparticles, as the magnitude of the charge is an indication of the mutual repulsion between particles that can be used to predict the long-term stability of the nanoparticulate dispersion. The content of gold metal in B-AuNPs was estimated to be 0.480 mg/mL by furnace atomic absorption spectroscopy (Table 1).

UV–vis absorption spectra of B-AuNPs. The inset shows the TEM and size distribution histogram of the gold nanoparticle solution. TEM, transmission electron microscopy.
Physiochemical Data Parameters of B-AuNPs.

Note. AAS, atomic absorption spectroscopy; B-AuNPs, broccoli phytochemicals–coated gold nanoparticles.
In Vitro Stability Study
The stability of B-AuNPs was evaluated by monitoring the plasmon (λmax) signal in various biological fluids—0.5% cysteine, 0.2 M histidine, 0.5% human serum albumin (HSA), 0.5% bovine serum albumin (BSA), and 1% NaCl solutions at different time points (1 h, 4 h, 24 h, 48h, and 1 week). The stability of B-AuNPs has also been observed using phosphate-buffer solutions at pH 7 and 9. The plasmon resonance wavelength in all the above formulations showed minimal shifts of ∼1 to 2 nm. Our results from these in vitro stability studies have confirmed that the AuNPs are intact and thus demonstrate excellent stability in biological fluids at physiological pH (Figure 2). We have also confirmed the stability of B-AuNPs at different dilution. It is vitally important that dilutions of nanoparticle solutions do not alter their characteristic chemical and photophysical properties. In order to establish the stability of B-AuNPs under dilution, the plasmon resonance wavelength was monitored after every successive addition of 0.1 mL of doubly ionized water to 1 mL of B-AuNPs solution. The absorption intensity at λmax is found to be linearly dependent on the concentration of B-AuNPs, in accordance with Beer–Lambert law as shown in Figure 3. The results confirmed that λmax of AuNPs does not change at very dilute conditions. It is thus conceivable that the cocktail of phytochemicals in broccoli is acting synergistically in stabilizing AuNPs from any agglomerations in solution and remains stable in aqueous media for over a month.

In vitro stability of B-AuNPs by UV–vis absorption spectra. B-AuNPs, broccoli phytochemicals–coated gold nanoparticles.

Change in plasmon absorption maximum of B-AuNPs as a function of dilution. B-AuNPs, broccoli phytochemicals–coated gold nanoparticles.
In Vitro Cell Viability Assay
In order to study the cytotoxic effects in cancer cells, we selected a weakly invasive breast cancer cell line: T47D; highly invasive breast cancer cell lines: MDA-MB-231 and SkBr3; prostate cancer cell line: PC-3; and the multiple myeloma: U266 cell line. 19 T47D is estrogen receptor positive (ER+) and MDA-MB-231 and SkBr3 are estrogen receptor negative (ER−) cell line, whereas prostate cancer cell line is androgen receptor positive. 20,21 The cytotoxicity of B-AuNPs and broccoli extract under in vitro conditions against various cancer cells was examined in terms of cell proliferation by the 3-(4,5-dimethyl thiazol-2-yl)-2, 5-diphenyl tetrazolium (MTT) assay. In this assay, only cells that are viable after 24 and 48 h exposure to the sample are capable of metabolizing a dye (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) efficiently and produce purple-colored crystals, which are dissolved in a solubilizing detergent and analyzed by enzyme-linked immunosorbent assay (ELISA) reader at wave length of 570 nm. After treatment, B-AuNPs showed maximum cell growth inhibition as compared to broccoli extract against cancer cell lines (Figure 4). The B-AuNPs showed significant cytotoxic effects at 48 h as compared to the data obtained at 24 h (Figure 4 and Figure S2). Inhibitory concentration 50% (IC50) values of B-AuNPs against various cancer cell lines were as follows: MDA-MB-231: 160 µg/mL; PC-3: 150 µg/mL; U266: 125 µg/mL; SkBr3: 80 µg/mL; and T47D: 22 µg/mL. The cytotoxic effect of test samples was significantly (p < 0.01) different from untreated control and broccoli extract treated cells (Figure 4). The data obtained from cytotoxicity assay indicated the concentration and time-dependent efficacy of B-AuNPs. These results, taken together, clearly demonstrate that the coating of broccoli phytochemicals on AuNPs results in their effective delivery into the cellular matrix with consequent inhibition in the growth of cancer cells, and also provide evidence of strong antiproliferative activities.

Dose-dependent cytotoxicity (MTT assay) of (a) B-AuNPs and (b) broccoli extract against cancer cells with 24 h treatment. MTT, 3-(4,5-dimethyl thiazol-2-yl)-2,5-diphenyl tetrazolium. Each value is mean ± SD (n = 3), p < .05 versus untreated control.
The Effect of B-AuNPs on Induction of Apoptosis in Cancer Cells
We further addressed whether reduced survival of cancer cells as triggered by B-AuNPs correlates with apoptosis. Cell apoptosis features some morphological alterations, such as chromatin condensation, membrane blebbing, internucleosomal degradation of DNA, and apoptotic body formation. The PC-3 cancer cells in the test sample were subjected to flow cytometry analysis, whereby apoptotic bodies can be detected by Annexin V-FITC and 7-AAD staining. As shown in Figure S3 and Figure 5, significant time and concentration-dependent increases of the total percentage of dead cells, that is, early apoptotic cells (annexin V-FITC+/7-AAD−) plus necrotic cells (annexin V-FITC+/PI+), were observed in PC-3 cancer cells treated with B-AuNPs. The apoptotic cells resulting from B-AuNPs were quantified at different time points with concentrations of 50, 100, and 200 µg/mL. At 24 h, the percentage of Annexin V-positive cells was found to significantly increase as compared to control untreated and broccoli extract treated cells.

Images represent the apoptosis induction by B-AuNPs in PC-3 cancer cells at different time points and concentrations. (a) Control untreated, (b) 100 µg/mL B-AuNPs; 12 h treatment, (c) 200 µg/mL B-AuNPs; 12 h treatment, (d) 100 µg/mL B-AuNPs; 24 h treatment, (e) 200 µg/mL B-AuNPs; 24 h treatment, (f) 200 µg/mL Broccoli extact; 24 h treatment. B-AuNPs, broccoli phytochemicals–coated gold nanoparticles.
Cellular Internalization Study
Cellular internalization studies provide important information on whether B-AuNPs can be used in nanoparticle-based drug delivery for potential therapeutic and intracellular molecular imaging applications. 22,23 The cellular internalization of B-AuNPs against breast (triple negative) and prostate cancer cell lines were evaluated using dark field optical microscopy and also through TEM image analysis with different concentrations at two different time points. A number of studies have demonstrated that phytochemical-coated AuNPs have the ability to penetrate cell membranes and internalize within the cellular matrix. 16,24 The B-AuNPs were incubated with cancer cells and were washed vigorously with phosphate-buffered saline (PBS; pH 7.4) to remove any unbound AuNPs. Figure 6 provides Cyto viva dark field microscopic images showing internalization of AuNPs within PC-3 and MDA-MB-231 cells. The internalization of AuNPs is also corroborated independently by TEM image analysis of cancer cells. Figure 7 shows the TEM images of B-AuNPs after their internalization within PC-3 and MDA-MB-231cancer cells. These results clearly demonstrate that B-AuNPs are internalized into both prostate and breast cancer cells and that the identity of individual nanoparticles remained intact inside the cells.

Cellular internalization of B-AuNPs via dark field microscope (a) against PC-3 cells untreated, (b) against PC-3 cells after 4 h treatment, (c) against PC-3 cells after 8 h treatment, (d) against PC-3 cells after 18 h treatment, (e) against MDA-MB-231 cells untreated, (f) against MDA-MB-231 cells after 18 h treatment. B-AuNPs, broccoli phytochemicals–coated gold nanoparticles.
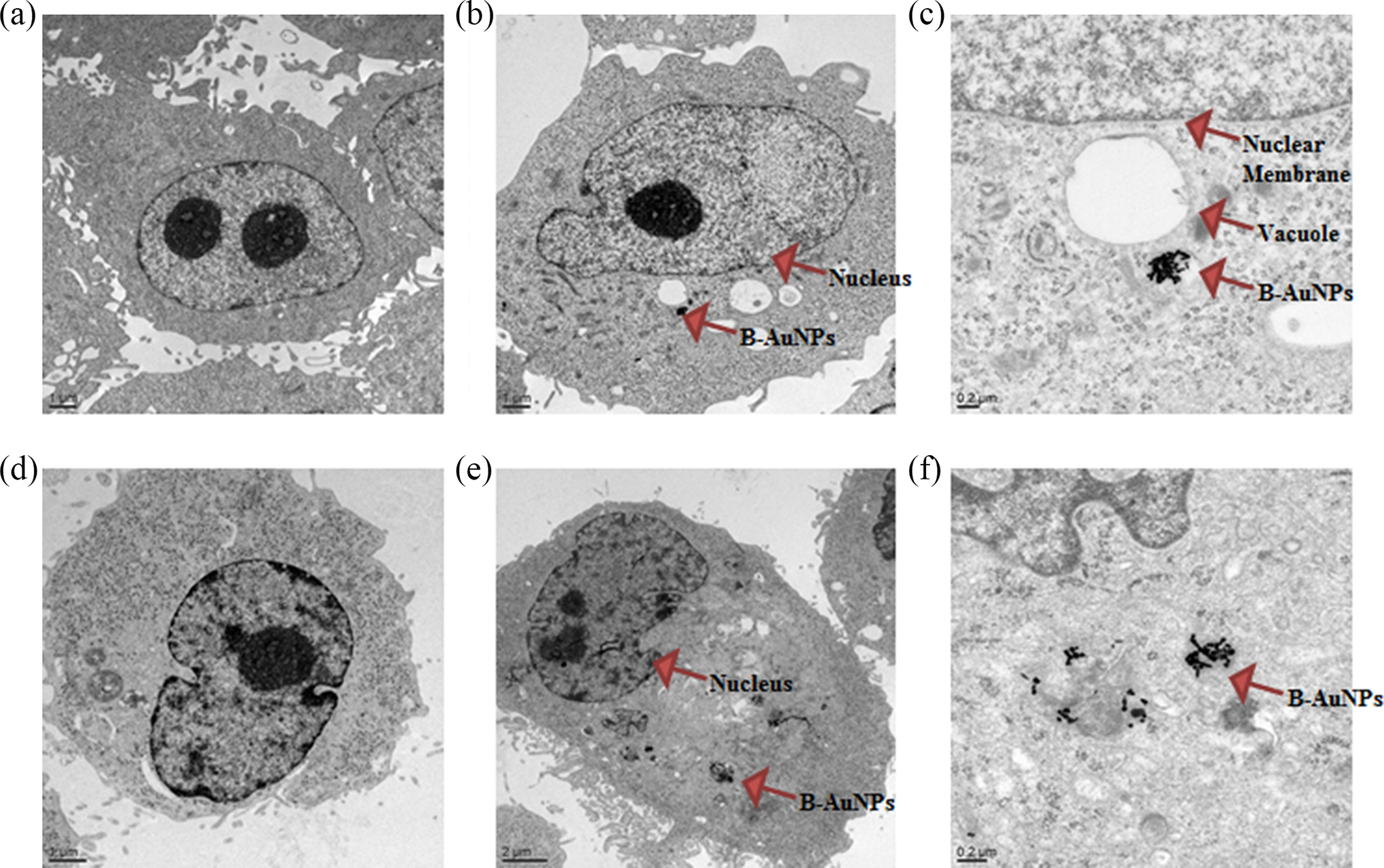
Cellular internalization of B-AuNPs via TEM against (a) PC-3 cell line control; (b, c) PC-3 cell line treated; (d) MDA-MB-231 cell line control; (e, f) MDA-MB-231 cell line treated. TEM, transmission electron microscopy.
Analytical Profile of Broccoli Extract
GC–MS Analysis
In order to characterize and quantify the hydrophobic and moderate-polar volatile phytochemicals in broccoli aqueous extract, we have exploited the volatility of most ITCs by Varian 3400cx GC with a Hewlett Packard cross-linked methylsiloxane DB-5 capillary column coupled with a Varian Saturn 2000 ion-trap mass selective detector. The quantitative ions were selected by injecting working standards of allyl ITC, phenyl ethyl ITC, and sulforaphane. Selection of diagnostic and quantitative ions was optimized by a procedure described by Lin et al. 25 The most predominant product ions generated during electron ionization were recorded. The background spectra subtraction was subsequently performed to identify the combination of the ions, so the selectivity and sensitivity can be optimized. The ions providing the highest signal-to-noise ratio for quantification was selected. The calibration curves were developed using five concentrations of the standard samples to relate known concentrations to the ion intensities (Figure 8). The perusal of Table 2 represents the concentration of various compounds present in broccoli extract.
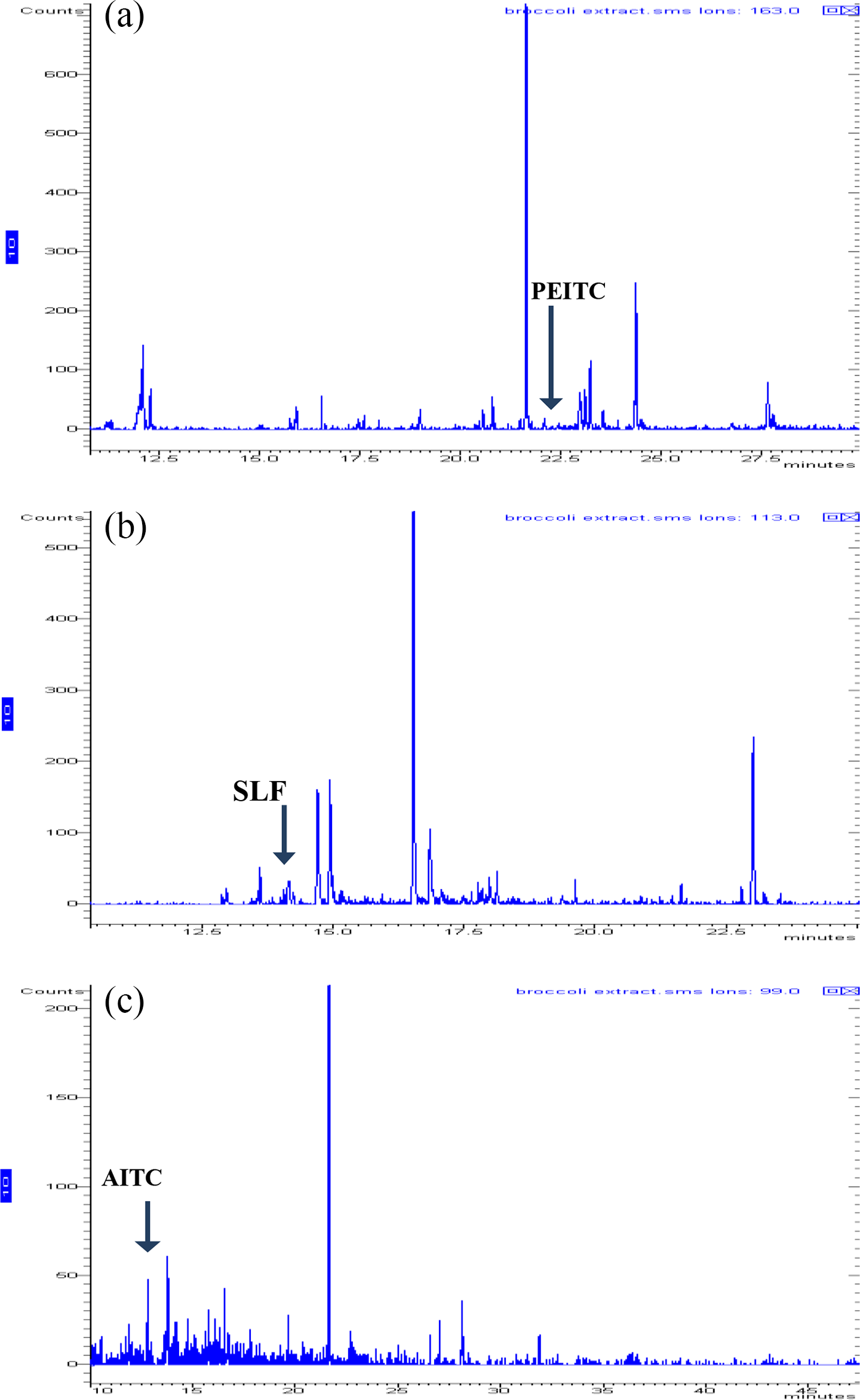
The product ion chromatograms for (a) PEITC (m/z 99), (b) SLF (m/z 113), and (c) AITC (m/z 99). AITC, allyl isothiocyanate; PEITC, phenyl ethyl isothiocyanate; SLF, sulforaphane.
GC–MS Data of Various Phytochemicals in Broccoli Water Extract.

Note. RT, room temperature; MW, molecular weight.
LC–MS–MS Analysis
The detailed data-dependent LC–MS–MS screening based on broad-range library searches (NIST-05 library) and fragmentation trees was able to identify several bioactive phytochemicals belonging to main classes: phenolic (flavonoids) and GLs. Determination of the molecular parent ion to be screened and the product ion to be used for quantification were determined from spectra obtained by injecting 20 µL of a standard solution containing 1000 µg/L on a Waters Alliance 2695 high-performance liquid chromatography (HPLC) system coupled with Waters Acquity TQ triple quadrupole mass spectrometer (MS/MS). Full spectrum of the deprotonated molecule ion [M - H]- and the product ions was generated, and the predominant fragmented ion was selected as the product ion for quantification. The example of the optimization process for the analysis of phenethyl glucosinolate is illustrated in Figures 9 and 10. The results of the optimization for each compound were summarized in Table 3.
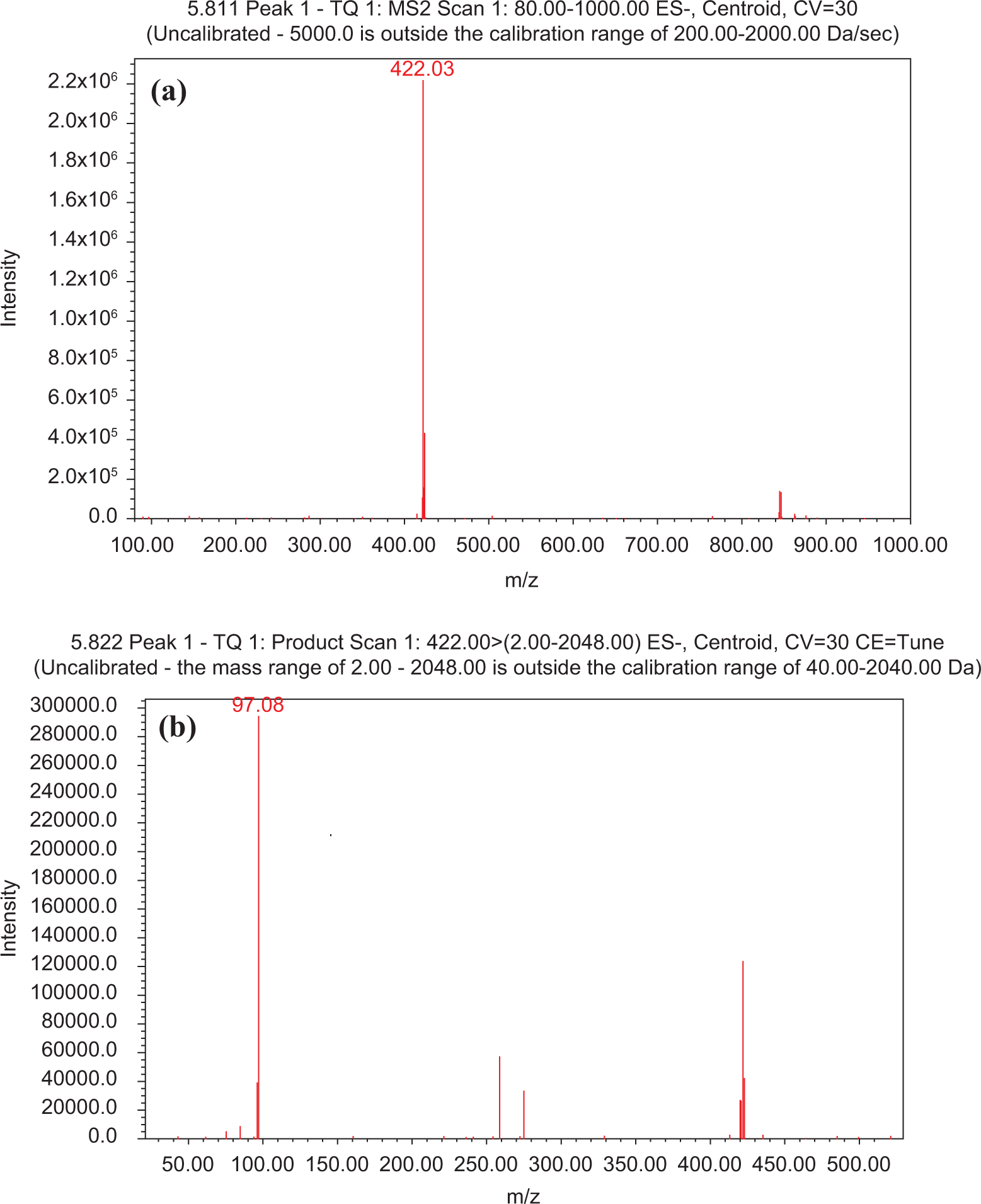
Full-scan mass spectra of molecular (a) and product (b) ions of phenethyl glucosinolate (PHG).

Ion chromatogram for quantitative estimation of product ion m/z 97 corresponding to phenethyl glucosinolate (PHG) in the extract.
LC–MS–MS Data of Various Phytochemicals in Broccoli Water Extract.

Note. RT, room temperature; MW, molecular weight.
Quantitative Estimation of Broccoli Phytochemicals in AuNPs
The spectrophotometric analysis of broccoli extract in 0.01 M NaCN solutions gave a λmax at 280 nm. The corresponding λmax for B-AuNPs is at 535 nm. B-AuNPs were titrated with various concentrations of NaCN in order to digest AuNPs and thereby calculate the amount of broccoli phytochemicals bound to AuNPs. Through spectrophotometric analysis, we monitored the disappearance of the peak at 535 nm (due to B-AuNPs) and followed increase in the extinction coefficient of the peak at 280 nm (due to released phytochemicals: flavonoids, GLs, and ITCs). Figure S4 demonstrates that the digestion of B-AuNPs with 0.01 M NaCN releases the broccoli phytochemicals bound to AuNPs, as the peak at 535 nm continued to decrease in intensity while the intensity of the peak at 280 nm increased until all the bound broccoli phytochemicals were completely removed from the surface of AuNPs. The plotting of various concentrations of broccoli extract in 0.01 M NaCN versus optical density provides quantitative estimation of bound broccoli phytochemicals on AuNPs. From these studies, one can infer that over 85% of the free broccoli used in the preparation of AuNPs remains bound to AuNPs to produce B-AuNPs. It may be noted that the above calculations have an excellent accuracy because spectrophotometric analysis using known concentration of broccoli extract through extinction coefficient measurements gave an error of 1 to 2%.
Generation of AuNPs by Individual Broccoli Phytoconstituents
The chemical analysis of broccoli extract revealed that it showed the presence of major phytochemicals including polyphenols (flavonoids), GLs, and ITCs derivatives. Attempted synthesis of AuNPs using the commercially available individual constituents of broccoli was performed in order to determine the critical chemical reduction roles to convert gold salt into the corresponding AuNPs. The results of these experiments have unambiguously confirmed that glucosinolate and flavonoids provide dual roles of reduction of the gold salt and serve as “surface-capped” stabilizers of AuNPs produced in this green nanotechnology process. It is important to note that AuNPs produced through broccoli extract resulted in the formation of large size nanoparticles (core size > 40 nm). In order to produce smaller and uniformly sized nanoparticles, we have optimized synthetic protocols that involved using increased concentrations of specific phytochemicals from broccoli. We have discovered that cocktails of flavonoid and GLs, when deliberately introduced within the reaction mixtures, produced excellent quality monodispersed B-AuNPs. Therefore, the addition of excess flavonoid and GLs phytochemicals into the naturally prepared broccoli extract (from broccoli) resulted in the production of monodispersed and smaller sized nanoparticles (core size < 20 ± 2 nm).
In additional experiments, we have utilized gum arabic (GA), a naturally occurring glycoprotein, as a stabilizer to increase the in vitro and in vivo stability of B-AuNPs. In our earlier studies, we have demonstrated that encapsulation of AuNPs with GA glycoprotein affords optimum in vitro and in vivo stabilities. 26 Addition of GA during the preparation of B-AuNPs resulted in the production of highly robust B-AuNPs. The progress of all the individual reactions was monitored by ultraviolet (UV) visible absorption spectrophotometry, and the representative spectra of individual phytochemicals are shown in Figure S5.
Discussion
A number of investigations in recent years have focused on the antioxidant, detoxifying, and cancer-preventive potential of Brassicaceae. These properties have been largely attributed to their GLs content, and their ability to release bioactive ITCs upon myrosinase hydrolysis. 7,27 Broccoli embodies a rich group of phytochemicals including vitamin C, folic acid, gallic acid, flavonoids, and GLs and their derivative ITCs. Through our detailed GC–MS and LC–MS–MS studies, we have confirmed the presence of all these phytochemicals in the aqueous extracts of broccoli used in our studies. These phytochemicals possess combination of active functional groups, such as hydroxyl, aldehyde, and carboxyl units—that synergistically result in the efficient chemical reduction of NaAuCl4 into the corresponding AuNPs. The strong antioxidant capacity of vitamin C, folic acid, flavonoids, and GLs and their derivative ITCs, therefore, allows transfer of electrons from the broccoli phytochemicals to the Au3+ ion leading to the formation of Au0.
The assessment of in vitro stability and bioefficacy of the newly fabricated B-AuNPs will play an important role in their potential applications in nanomedicine. Our detailed studies on in vitro stability of B-AuNPs have clearly shown that phytochemicals in broccoli, when coated onto AuNPs, provide exceptional in vitro stability. The size distribution analysis of B-AuNPs has confirmed that these biocompatible nanoparticles are well dispersed with no agglomeration. The phytochemical coatings on AuNPs surface are expected to cause substantial changes in the hydrodynamic size of B-AuNPs. The hydrodynamic diameter of B-AuNPs as determined from DLS measurements is 90 ± 5 nm, suggesting that broccoli phytochemicals (vitamin C, folic acid, gallic acid, flavonoids, and GLs and their derivatives ITCs) are capped on AuNPs. The measurement of charge and ζ provides crucial information on the stability of nanoparticle dispersion. The magnitude of measured ζ is an indication of repulsive forces and can be used to predict the long-term stability of the nanoparticulate dispersion. The stability of nanoparticulate dispersion depends upon the balance of the repulsive and attractive forces that exist between nanoparticles as they approach one another. If all the particles have a mutual repulsion, then the dispersion will remain stable. However, little or no repulsion between particles lead to aggregation. The negative ζ of −29.0 mV for B-AuNPs indicates that the particles repel each other strongly, thus minimizing aggregation of nanoparticulates (Table 1 and Figure 1). These results suggest that the green nanotechnological process reported herein provides both the production and the stabilization processes under mild conditions without the intervention of any man-made toxic chemicals.
The cytotoxicity of B-AuNPs under in vitro conditions against human cancer cells was examined by MTT assay. The B-AuNPs showed cytotoxic effects with the increasing concentrations at different time points against various cancer cells. B-AuNPs showed significant (p < .05) cytotoxic effects against T47D (breast cancer cells) as compared to MDA-MB-231, SkBr3, PC-3, and U266 cancer cell lines. The order of cytotoxic activity of B-AuNPs in our experiments is as follows: MDA-MB-231 < PC-3 < U266 < SkBr3 < T47D. On the basis of these findings, we infer that the cytotoxic effects of broccoli phytochemicals are significantly higher when they are coated onto gold nanoparticulate surfaces. Therefore, our green nanotechnology approach, which allows embedding broccoli phytochemical cocktail onto AuNPs, is a highly attractive pathway for the delivery with consequent enhancement of the antitumor effects of broccoli phytochemicals.
The in vitro anticancer effects of B-AuNPs have been further confirmed by apoptotic and necrotic cells measurements through flow cytometry assay. In this assay, the mode of cell death can be clearly distinguished by annexin-V-FITC and 7-AAD absorption. During the early events of apoptosis, the plasma membrane phospholipid phosphatidylserine is translocated from the inner side of membrane leaflet to the outer side, which has a very high affinity for annexin-V antibody. Thus, annexin V-FITC-positive and 7-AAD negative cell populations are indicative of apoptosis and annexin V-FITC-positive and 7-AAD positive cells indicate necrosis. Our results have provided compelling evidence that B-AuNPs induce apoptosis in PC-3 cancer cells as compared to control untreated and broccoli extract–treated samples. These results taken together suggest that coating of broccoli phytochemicals on to AuNPs plays a significant role in the retention of biological activities as well as the delivery of phytochemicals within the cellular matrix.
In order to explore the interaction of B-AuNPs with cancer cells, we have investigated the effect of cellular internalization of B-AuNPs in prostate and triple negative breast cancer cells. It is established that the internalization of nanoparticles strongly depends on their physical characteristics including size, shape, and charge. Cellular internalization studies of AuNPs provide insights into cellular uptake, and such information will enhance the scope of AuNPs in biomedical applications. Selective cell and nuclear targeting of AuNPs will provide new pathways for the site-specific delivery of AuNPs. A number of studies have demonstrated that phytochemicals conjugated AuNPs have increased ability to penetrate cellular membranes with subsequent internalization within the cellular matrix. 18,24 Cancer cells are highly metabolic and porous in nature and are known to internalize solutes rapidly compared to normal cells through enhanced permeability and enhanced permeability retention time (EPR) effect. 28,29 Therefore, we hypothesized that B-AuNPs will also show internalization within cancer cells. Dark field microscopy images showed the internalization of B-AuNPs into the triple negative breast (MDA-MB-231) and prostate (PC-3) cancer cell lines at various concentrations with different time points (Figure 6). We have also confirmed the cellular internalization by TEM images analysis. TEM images of breast and prostate cancer cells treated with B-AuNPs showed significant internalization of nanoparticles within the cells (Figure 7), thus unequivocally validating our hypothesis. The internalization of nanoparticles within cells occurs via several processes, including phagocytosis, fluid-phase endocytosis, and receptor-mediated endocytosis. It is reported that cellular internalization of nanoparticles is dependent on the surface charge and size of the nanopartilces. 30 Numerous studies have shown that surface charge has a significant impact on cellular internalization of a variety of nanocarriers. 31 Positively charged nanoparticles were internalized into cells within 1 h incubation period by endocytosis, whereas negatively charged particles internalize into the cells by phagocytosis. B-AuNPs have a negative potential of −29.0 mV. However, significant internalization of B-AuNPs in breast and prostate cancer cells were observed in our studies. The high propensity of B-AuNPs within cancer cells is presumably occurring through phagocytosis, involving the vesicular internalization of AuNPs. It is important to note that the cellular membrane showed the formation of phagocytic cups, which are usually present in the cell wall if phagocytosis has occurred. In addition, B-AuNPs were found in endosomal vesicles as individual probes in the form of dense mass of nanoparticles (Figure 7). These observations indicate that the B-AuNPs showed synergistic effect and excellent internalization in prostate and breast cancer cells. This unique synergistic cocktail effect of B-AuNPs may provide new opportunities for generating biocompatible AuNPs for applications in in vitro and in vivo nanoparticulate-mediated imaging and therapy.
The analytical profile of the broccoli aqueous extracts revealed the presence of GLs, derivatives of ITCs, and flavonoids moieties in abundance. The aqueous extract contained glucoraphanin (6.72 μg/mL), phenethyl GLs (0.32 μg/mL), quercetin (0.08 μg/mL), folic acid (10.55 μg/mL), vitamin C (12.85 mg/mL), allyl-ITC (0.05 μg/mL), 2-phenylethyl-ITC (1.39 μg/mL), and sulforaphane (12.68 μg/mL). The structures of chemical constituents identified in the broccoli extract are summarized in Table S1. The observed in vitro anticancer bioefficacy exhibited by B-AuNPs may be attributed to the presence of GLs, ITCs, and flavonoid contents.
Role of Broccoli Phytochemicals
The chemical roles of different phytochemicals in broccoli responsible for the production of B-AuNPs are still not fully understood, but we believe that water-soluble strong antioxidant constituents of broccoli may be playing a major role in the overall reduction process of NaAuCl4. Results from experiments using individual phytochemicals (GLs, ITCs, flavonoids, vitamin C, gallic acid, and folic acid) have shown that GLs and flavonoids serve as primary reducing agents to reduce gold salt into AuNPs. The ITCs did not form any nanoparticles, suggesting limited or no chemical reduction capability of this class of phytochemicals. However, the sulfur functionality in ITCs aids in the formation of strong gold-thiol bonds, thus rendering optimum in vitro or in vivo stability to B-AuNPs. Therefore, the cocktail effects of phytochemicals in broccoli serve dual roles of reduction of gold salt and consequent stabilization of the resulting nanoparticles through surface capping by thiol compounds from broccoli. It is important to note that our experiments have revealed that the synergistic effects of all the constituents present in broccoli extract allow encapsulation of AuNPs, thus affording excellent in vitro stability against agglomeration.
Materials and Methods
All chemicals used in the synthesis of AuNPs and cell culture, for example, Roswell Park Memorial Institute Medium (RPMI) and minimum Eagle’s essential medium (MEM), streptomycin, penicillin-G, Trypan blue, and MTT, DAPI (4′,6-diamidino-2-phenylindole) dyes were obtained from Sigma (St. Louis, MO). Fetal calf serum and TryplE were obtained from Gibco BRL (Grand Island, NY). Apoptosis kit was obtained from Cayman Chemical Company, USA. PC-3 (prostate cancer) and SkBr3, MDA-MB-231, T47D (breast cancer), and U266 (multiple myeloma) cell lines were obtained from Cytology Core facilities of the University of Missouri, Columbia. Analytical standards were purchased from Fluka-Sigma-Aldrich. Double distilled water was used throughout the experiment.
Preparation of Broccoli Extract
Organic broccoli (B. oleracea) purchased from a local grocery shop were used in all the experiments. Broccoli (100 g) was washed with doubly ionized water to remove any contaminants or dust particles and incubated at 50 °C for 12 h to get dry powder. The extract was prepared by adding 60 mg of broccoli powder into 6 mL of doubly DI water and sonicated for 4 h in sonication bath. The extract was separated from the residual broccoli powder by means of standard filter paper and used for subsequent synthesis of AuNPs.
Measurements
TEM images were obtained on a JEOL 1400 TEM (JEOL, LTE, Tokyo, Japan). The absorption measurements were done using a Varian Cary 50 UV–vis spectrophotometer. The core size of the nanoparticles was measured on DC 24000, Differential Centrifugal Sedimentation (DCS), CPS Instruments Inc., USA. The hydrodynamic diameter and ζ were obtained using Zetasizer Nano S90 (Malvern Instruments Ltd., USA). The concentration of gold metal was calculated by Atomic absorption spectrometry.
Synthesis of B-AuNPs
The broccoli aqueous extract (60 mg/6 mL) was added to a 20 mL vial and stirred for 10 min at 50 °C. Then, 90 μL of 0.1 M NaAuCl4 solution was added, and the color of the solution turned to ruby-red within 5 min, indicating the formation of AuNPs. The solution was filtered through a 0.5 μ filter to use for biological applications. The AuNPs were characterized by UV–vis absorption spectrophotometry, DCS, and TEM analysis.
Generation of Gold Nanoparticles (AuNPs) by Individual Broccoli Phyto-constituents
We have utilized individual phytochemicals from broccoli in our attempts to investigate the actual chemical species of the vegetable that is responsible for converting NaAuCl4 to the corresponding AuNPs. The procedure for making AuNPs by broccoli components (GLs, ITCs, and flavonoids) is as follows: To a 20 mL vial, 6 mL of doubly DI water was added, followed by the addition of these phytochemical in a separate vial. The mixture was stirred continuously at 50 °C for 10 min. One hundred microliters of 0.1 M NaAuCl4 solution was added to the reaction mixture. The color of the glucosinolate and flavonoids reaction mixture turned to ruby-red within 5 min. However, we did not notice any color change in similar reactions when ITC was used. The change in color of Gls and flavonoids reaction mixture indicated the formation of nanoparticles. The nanoparticles formation was confirmed by UV–vis absorption spectroscopy.
Sample Preparation and Analysis for fAAS
Gold metal content of B-AuNPs was estimated by fAAS using a standard curve spanning 0 to 100 µg/L. 32 B-AuNPs sample were digested with HNO3 and HCl (1:2 ratios) and kept in an oven at 85 °C overnight. After cooling, the digest was diluted in 1:10 ratio with ultrapure water for analysis. The furnace parameters were as specified in the user’s manual for the Perkin–Elmer Analyst 800 ThGA graphite furnace. Quality-control materials (duplicates, spikes, and instrument-calibration verification) were within appropriate ranges.
In Vitro Stability Study
The stability of B-AuNPs was confirmed by mixing AuNPs to aqueous solutions of 1% NaCl, 0.5% cysteine, 0.2 M histidine, 0.5% HSA, 0.5% BSA, pH 7, and pH9. The stability of the conjugates was measured by monitoring the UV absorbance over a period of 1 h, 4 h, 24 h, 48 h, and 1 week. A negligible change in UV–vis plasmon band confirmed the retention of nanoparticulate composition in all mixtures. We also measured the stability of B-AuNPs at different dilution by UV–vis spectrophotometry, which is very important for biomedical application.
Cell Viability Assay
The in vitro cytotoxicity evaluation of B-AuNPs was performed as described by the supplier Promega, USA. 33 Briefly, in vitro cytotoxicity against human cancer cell lines (PC-3, MDA-MB-231, SkBr3, T47D, and U266) was determined using 96-well tissue culture plates. One hundred microliters of cell suspension (1 × 105/mL) were added to each well of the tissue culture plate. The cells were allowed to grow in a CO2 incubator (37 °C, 5% CO2, and 90% relative humidity [RH]) for 24 h. The medium was replaced with the medium containing different dilution of test samples (25–200 µg/mL) separately again for 24 and 48 h in CO2 incubator at 37 °C. Tetrazolium bromide salt solution (10 µL/well) was added to the cell suspension and incubated for 4 h; the formazan crystals formed were dissolved in 100 µl detergent. The plates were kept for 4 h in dark at 25 °C to dissolve all crystals, and the intensity of developed color was measured by micro plate reader (Dynastic MR 5000, USA) operating at 570 nm wavelength. Percentage of cell viability was calculated by using the formula: [(T/C) × 100], where C = absorbance of control, T = absorbance of treatment. The IC50 values of test samples were also calculated.
Apoptosis Assay
This experiment was performed according to the manufacture’s protocol (Cayman Chemical Company, USA). 34 The PC-3 cancer cells were incubated with or without B-AuNPs and broccoli extract with different dilution series for 12 and 24 h. Briefly, cells were collected, washed twice with cold PBS, centrifuged at 400 g for 5 min, and resuspended in 1× binding buffer. Then cells were transferred to a 2 mL culture tube; 250 µL of Annexin V-FITC and 7-AAD in 1× binding buffer were added. Cells were gently vortex-mixed, and incubated for 10 min at room temperature in the dark. Furthermore, 250 µL of 1× binding buffer were added to each tube, and samples were analyzed by FACScan flow cytometry (FACSort, Becton Dickinson, USA). For each sample, 30,000 ungated events were acquired.
Cellular Uptake Study
The PC-3 and MDA-MB-231 cells (5 × 105 cells) were seeded into 6-well plates in RPMI/DMEM medium separately and allowed to adhere for 24 h in CO2 incubator at 37 °C. B-AuNPs (25 and 50 μg/mL) were added to cells followed by 12 h of incubation at 37 °C. The cells were washed three times with PBS, centrifuged into small pellets, and fixed with 2% glutaraldehyde 2% paraformaldehyde (PFA) in sodium cacodylate buffer (0.1 M). The cells were further fixed with 1% buffered osmium tetraoxide in 2-Mercaptoethanol buffer and dehydrated in graded acetone series and embedded in Epon-Spurr epoxy resin. Sections were cut at 85 nm using a diamond knife (Diatome, Hatfield PA). The sections were stained with Sato’s triple lead stain and 5% aqueous uranyl acetate for organelle visualization. 35 The prepared samples were examined on JEOL 1400 TEM microscope (JEOL, Peabody, MA) operated at 80 kV at the University of Missouri’s Electron Microscopy Core Facility.
Cytoviva Study
The in vitro cellular internalization analysis of B-AuNPs was performed using dark field cytoviva microscopic techniques. Ultraclean and sterile cover slips were kept in 6-well plates. The PC-3 and MDA-MB-231 (5 × 105 cells) were seeded into 6-well plate in RPMI/DMEM medium separately and incubated for 24 h in CO2 incubator at 37 °C. B-AuNPs (25 and 50 μg/mL) were added to cells followed by 4, 8, and 12 h of incubation at 37 °C. The cells were washed 10 to 12 times with 1× PBS, and fixed with 4% PFA. Cells were further washed 2 to 3 times with cold 1× PBS. Slides were prepared by using DAPI nuclear dye and observed with CytoViva dark field microscope coupled with dual mode fluorescence. Cell morphology was initially observed, followed by uptake of nanoparticles. Images were captured via Dage Imaging Software.
Chemical Characterization of Broccoli Extract
GC–MS Analysis
The qualitative and quantitative analyses of various volatile phytochemicals in broccoli extract were performed using a Varian 3400cx GC with a Hewlett Packard cross-linked methylsiloxane DB-5 capillary column (30 m × 0.25 mm i.d.) coupled with a Varian Saturn 2000 ion-trap mass selective detector (Varian Inc., Walnut Creek, CA). The GC temperature program was 35 °C for 10 min; ramp to 200 °C at 10 °C/min; and finally ramp to 260 °C at 3 °C/min and hold for 6 min. Injector temperature was held at 260 °C for 5 min. Split injection mode with split ratio of 1:100 was used. Helium was used as the carrier gas at a flow rate of 1 mL/min. The transfer line between the GC and mass spectrometer was held at 280 °C, and the ion trap manifold was set to 250 °C. For quantitative analysis, chromatograms were reconstructed at the base peak and calibration was obtained with authentic standards.
LC–MS–MS Analysis
The nonvolatile phytochemicals in broccoli extract were determined by a Waters Alliance 2695 HPLC system coupled with Waters Acquity TQ triple quadrupole mass spectrometer (MS/MS). The GLs, flavonoids, folic acid, gallic acid, and vitamin C were separated by a Phenomenex (Torrance, CA) Kinetex C18 (100 mm × 4.6 mm; 2.6 µm particle size) reverse-phase column. The ion source in the MS/MS system was electrospray ionization (EI) operated in the negative ion mode with capillary voltage of 1.5 kV. The ionization sources were programmed at 150 °C and the desolvation temperature was programmed at 450 °C. The MS/MS system was operated in the multireaction monitoring (MRM) mode with the collision energy of 30 V.
Quantitative Estimation of Broccoli Phytochemicals in B-AuNPs
The coating of broccoli phytochemicals on the surface of AuNPs is important to understand the overall stability, size, charge, and therapeutic efficacy. The amount of broccoli phytochemicals on the surface of AuNPs was quantified by sodium cyanide digestion experiment. B-AuNPs (50 μL) was digested with 0.01 M NaCN solution (2 mL final volume) for 60 min followed by UV–vis spectral analysis.
Statistical Analysis
All experimental data were given as mean ± SE. Statistical analysis was carried out using the one-way analysis of variance (ANOVA) using Graph Pad Prism software. Probability values were found to be ≤ 0.05).
Conclusion
Cocktail of broccoli phytochemicals serve as efficient reducing agents to reduce gold salt into biocompatible AuNPs capped by nontoxic phytochemicals affording excellent in vitro stability. Our studies indicate that AuNPs conjugated with broccoli phytochemicals show excellent internalization within prostate (PC-3) and breast (MDA-MB-231) cancer cells with consequent in vitro anticancer effects against various cancer cells. The in vitro anticancer activity is fully corroborated by the efficient delivery of ITCs, flavonoids, and cocktails of broccoli phytochemicals, coated on AuNPs, through various cellular internalization pathways mediated by AuNPs. This unique synergistic cocktail effect of B-AuNPs may provide new opportunities for generating AuNPs for applications in nanoparticulate-mediated imaging and therapy.
Footnotes
Acknowledgment
The studies reported in this publication have been made possible through intramural funding of the University of Missouri through the Interdisciplinary Intercampus Research Program.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
