Abstract
Inorganic metal oxide nanoparticles are mainly synthesized by sol–gel process. The most important beneficial advantage of mentioned process is facile and the time-consuming route. The resultant meta-stable synthesized inorganic nanoparticles can easily modify different substrates and alter their performance. The presented review investigates the possible applications of nanosols (especially silica and titanium dioxide sols) in the field of textile industry, including the formation of hydrophobic, bioactive or protective metallic oxide coatings on textiles by physical or chemical modifying of nanosols.
Introduction
These days applying nanoparticles especially in the field of chemistry science has many advantages. By using very small compounds under 100 nm scale with high activity (because of their high specific area) bulk or surface properties of various substrates could be changed. Synthesizing metal oxide nanoparticles using conventional methods because of time- and energy-consuming process could not be an appropriate approach. It seems that preparing colloidal metal oxide solutions under ambient conditions would be more beneficial. Nanosols are colloidal solutions of nanometer-sized metal oxide particles in aqueous or organic solvents. Because of very high surface area, the nanosols are metastable, thus, during a coating process a three-dimentional (3D) network would be formed due to aggregation of particles and subsequent evaporation of the solvent. 1 Nanosol coatings are usually prepared by using the sol–gel process. This process can be basically divided into three steps: formation of the nanosol by hydrolysis of the precursor material and subsequent condensation reactions, the coating process, and drying or curing. The precursors are either inorganic metal salts or metal organic compounds such as metal alkoxides or acetylacetonate. Alkoxy derivatives of metals or semimetals are most widely used, whereby hydrolysis transforms them into the corresponding hydroxides. These hydroxides are mostly unstable in higher concentrations and therefore tend to undergo subsequent condensation reactions. The condensation reactions lead to the formation of particles with sizes in the nanometer range. Frequently used precursors are, for example, tetraethoxysilane Si(OC2H5)4 (TEOS), titanium(IV)isopropoxide Ti(OC3H7)4, or Al(OC4H9)3. It should be taken into account that insufficient heat treatment after applying nanosol coatings will result in the formation of an amorphous network that is called “xerogel.” 1
Eqs 1 and 2 show an example of possible reactions during the synthesis of colloidal silica nanoparticles from an alkoxy silane precursor:


The above-mentioned colloidal solution has a low viscosity (in the range of 1–6 mPas) in liquid dispersion containing nanosized particles. Synthesized metal oxide nanoparticles can be easily applied on the surface of a variety of materials, leading to the formation of dense layers once the solvent evaporates. 1
Modification of Nanosols
Prepared sols are modified using two major methods: chemical and physical routes. In chemical modifying method, usually active chemical substances such as polysiloxanes, alkylsilanes, organic domains, and so on are introduced inside the network of nanoparticles by forming covalent or even physical bonds. Ibrahim et al. used hexamethyldisilazane for surface modification of synthesized silica nanoparticles. 2 Alkylsilane chemical modifier resulted in the formation of monodisperse colloidal solution as a result of prevention of silica nanoparticle agglomeration and reduced surface energies. Berendjchi et al by dopping Cu nanoparticles inside inorganic silica network obtained grape-like clusters with the capability of trapping air inside (Figure 1). Due to the potential ability of complex forming copper nanoparticles, the morphology of resultant network was changed. 3

Transmission electron microscopic images of (a) unmodified silica nanoparticles and (b) chemical modified with copper (Cu) nanoparticles. 3
By adding macromolecular compounds such as dyestuffs, proteins, polymers, and so on in the physical modifying route the actual encapsulation procedure occurs, resulting in the formation of the corresponding compound during or after the hydrolysis process of the precursor. 1 Miao et al prepared silica microcapsules containing butyl-stearate by emulsifying butyl-stearate inside silica network and obtained core shell microcapsules that have beneficial applications in energy-saving building materials. 4
One of the most important factors for applying nanosol coatings onto textile fibers is adhesion between substrate and coating material. This is especially the case for most of the synthetic polymeric fibers. Several methods such as cross-linking agents, corona, or plasma treatments, high temperature processing, and so on, are used for activating such surfaces. 1 After preparing the corresponding coatings usually using padding process and appropriate heat treatment, metal oxide nanoparticle networks would be fixed on the textile surfaces. 2 –15 In the presented review, we will investigate the possible applications of nanosols for changing the performance of textile fibers.
Fiber Spinning
It has been reported that glass fibers could be produced using dry spinning of silica sols. By doing so, inorganic composite fibers based on silicon dioxide (SiO2) and also other metal oxides or mixtures of both would be prepared. Spinning of nano fibers from sol–gel-based solutions using electrospinning method is also reported. Electrospinning process has some advantages because of simplicity of producing of fiber sheets based on the inorganic fibers. Preparing the inorganic fleeces could be advantageous for certain applications including preparation of catalytic materials, promising a good solvent and temperature stability, combination of lightweight and high surface area, and so on. Nanofibers are also of interest for the preparation of certain filter materials for filtering very small particles. 1
Fiber Protection
Fire Resistance
As mentioned before, a combination of a lightweighted and flexibility with high tensile strength is one of the advantages of using textile materials. Flame-retardant finishes in textile industry have attracted great interest because every year enormous number of people all around the world die due to fire accidents. For technical textiles employed in high-temperature applications or aggressive zones, there is also a huge demand for modifying them to withstand high temperatures and delay the burning rate. The purely inorganic nanosol networks are themselves inflammable; therefore, they are not expected to increase the inflammability of fiber materials. It is expected that enhanced flame-retardant activity achieved by coating inorganic nanosols would be the result of a barrier effect. On one hand, applying the nanosol coatings as thin as possible will make it more effective; and on the other hand, as xerogel matrices are porous the coatings cannot probably act as a sufficient oxygen barrier because of the above-mentioned limiting factors that reduce the effectiveness of thermal shielding ability. 1
In the field of textile industry, there are two mechanisms for creating the fire-retardancy effect on textile materials. One of them is to prevent the possible reactions of active free hydroxyl or hydrogen radicals generated from burning process. In this area, chlorine- or bromine-containing substances are often used for fire protection. By decomposition of these compounds during a fire, hydrogen chloride and hydrogen bromide will be released. Then these compounds react with highly reactive hydroxyl and hydrogen radicals, forming less energetic chlorine or bromine radicals. Hydrogen and hydroxyl radicals promote the radical processes in the flame, which are mainly responsible for the propagation of a fire. As a result, the initial burn rate will be slowed down because of reduction of these radicals, thereby increasing the time to flashover. Frequently used brominated species as flame retardants are polybrominated diphenyl ether, polybrominated biphenyl, or brominated cyclohydrocarbons. 1
Another major approach is to create a protective char layer having nonporous and dense structure characteristic. For example, using phosphate esters incorporated in a coating (or forming the coating itself) and converting to char by a fire, a protective layer will be formed. The transport of oxygen into the polymer would be hindered by these barrier layers, and simultaneously they will act as a shield against the heat. During decomposition, the expansion of some compounds is observed based on ammonium polyphosphates or dicyandiamide. This action will cause forming a thick char layer that acts as the shield against oxygen and heat. 1
UV Protection
Apart from the well-known perils on human beings due to exposure to the sun’s radiation, there
are enormous synthetic fiber polymers that are decomposed by UV-light, examples are polyester,
Changing Hydrophobicity
Cotton is probably the most common natural fiber in textile industry. For many applications creating a hydrophobic surface on cotton textiles is a major challenge. Most of the synthetic fibers that are usually used in textile industry are fairly hydrophobic. Thus, most of the time, water-repellent finishing is applied on natural fibers. Before revealing the scanning electron microscopes (SEMs) the term “hydrophobicity” was presented based on fundamental wetting theories (Yang, Wenzel and Cassie - Baxter equations). 5 –7
Yang presented a formula for measuring the so-called “static contact angle (CA)” when a droplet
is placed on a solid surface:
Where γSA and γSL are the surface energies of the solid against air and liquid, respectively, and
γLA is the surface energy of liquid against air.
5
θ
Based on the above equation, less surface energy equals to more hydrophobicity, but the static CA will decrease up to a certain value. Actually by chemically modifying a surface, we cannot create a super-repellent surface.
The possibility of producing a superhydrophobic surface was proposed by Wenzel in 1936.
6
Based on eq 4, we
can conclude that beside chemical modification, physical properties of materials (roughness) can
play an important role with respect to the surface hydrophobicity:
Rf (roughness factor) is equal to the ratio of the solid surface area and its flat projected area.
In 1944, another theory was presented by Cassie and Baxter.
7
Based on this theory (eq
5), the hydrophobicity of a surface can be enhanced by trapping air inside the valleys of a
rough surface at the triple contact line (composite interface between air, liquid, and solid
substrates):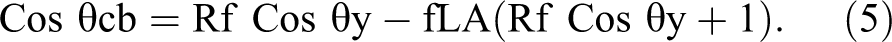
The fraction of trapping air (fLA) plays a major role. With higher air pockets fraction (flA), the amount of cos θcb decreases, enhancing the superhydrophobicity effect on the roughened surfaces.
According to the above-mentioned explanation, for the creation of a superhydrophobic surface two factors are necessary: low surface energy (chemical factor) and roughness (physical factor).
Synthesized metal oxide nanoparticles in sol–gel method have potential applications in creating the roughness on textile surfaces. As shown in Figure 2, they are spherical in shape; and when a surface is coated with nanoparticles, the morphology of surface is altered.

Altering the morphology of cotton fiber before (a) and after (b) treating with silica nanoparticles. 3
As mentioned before, most of the water-repellent finishing processes are applied on natural fibers especially on cotton ones. In order to create a superhydrophobic surface, we can reduce the surface energy of a roughened surface or create roughness on a surface having low free energy. In order to create roughness by the sol–gel method, usually TEOS and its hydrolysis is used under alkaline condition. 8 In this way, nanoparticles created have more stability because of their increasing size; and when placed on the surface, they could create roughness on the substrate. Due to the existence of enormous hydroxyl groups on the surface of nanoparticles, usually through the self-assembly process and by introducing an alkyl silane containing alkyl or flouroalkyl side chains, the free surface energy of the substrate is reduced and a superhydrophobic surface is created. 3,9 –14 Xu et al. created superhydrophobic cotton fabric by fabricating rough surfaces using both SiO2 nanoparticles and ZnO nanorod arrays and also using low surface energy by subsequent modification with n-dodecyltrimetoxysilane (DTMS). In this article, both SiO2 nanoparticles and ZnO nanorods had similar diameter; however, the samples prepared with ZnO nanorods had more air trapping between the adjacent ZnO nanorods because of higher aspect ratio of ZnO nanorods. As a result, slide angle was reduced when compared to the samples treated with SiO2 nanoparticles and DTMS. Wetability of cotton fabric showed superhydrophobic properties with static CA of more than 150° for 5 μL water droplets. In this article, it was concluded that lower sliding angle of cotton fabric treated with ZnO is attributed to discontinuous three-phase contact line that shows better water-repellent properties. 9 Yu et al. created a superhydrophobic complex coating for cotton fabrics based on silica nanoparticles and perfluorooctylated quaternary ammonium silane coupling agent (PFSC). The complex film was prepared via sol–gel process. Silica nanoparticles made the textile surface much rougher, and the PFSC on the top layer lowered the surface free energy. Fabrics coated with this coating showed excellent water-repellent property and the CA increased from 133° for those cotton fabric treated with PFSC without silica sol to up to 145°. Also, the oil repellency was improved and the CA of CH2I2 on the fabric surface treated with SiO2 nanoparticles and PFSC increased from 125° to 131° (in comparison with fabrics treated with PFSC only). 10 Bae et al. created superhydrophobic cotton fabrics by treating with silica nanoparticles and a cost-effective water-repellent agent (WR agent; an emulsion consisting of perfluroacrylate, benzyl methacrylate, water, etc.). In this article, two types of silica nanoparticles were synthesized via the sol–gel process and their shape, size, and composition were characterized; and it was concluded that silica nanoparticles were spherical and had diameters of about 143 and 378 nm. For cotton fabrics treated with WR agent alone in concentrations lower than 0.3% wt, the CAs of water remained on cotton surface were lower than 20°. The hydrophilic silica nanoparticles did not change the properties of cotton, and they showed that water drops are absorbed inside fabric due to the hydroxyl groups existing on the surface of silica nanoparticles. However, for cotton fabrics treated with silica nanoparticles and the WR agent, even at very low concentration of WR agent (about 0.1% wt), the CA of more than 130° was obtained. Thus, it was concluded that superhydrophobic cotton fabric can be prepared by a combination of silica nanoparticles and a WR agent, which is inexpensive compared to fluorosilanes. 11 Erasmus and Barkhuysen created superhydrophobic cotton fabric by modification with flourosilane. In this article, superhydrophobic surface and self-cleaning ability of cotton was created by 1H, 1H, 2H, 2H flouro-octyl tri etoxy silane, and it was recognized that an increased level of treatment increases the CA and causes self-cleaning properties. 12 Li et al. created superhydrophobic cotton fabric via sol–gel method and using water glass (sodium silicate) as the starting material. Such surfaces were produced first by dip coating of silica hydrosols that were prepared via hydrolysis and condensation of water glass onto cotton substrates. Then the silica coating was modified with a nonfluoro compound (hexadecyltrimetoxysilane [HDTMS]) for attaining a thin film and finally superhydrophobicity with a water CA of higher than 151° was obtained. 13 Hoefnagels et al. turned cotton fabrics from hydrophilic to superhydrophobic by in situ introduction of silica particles into cotton fibers in order to generate a dual-size surface roughness, followed by hydrophobization with polydimethylsiloxane. The treated fabrics exhibited a static water CA of 155° for a 10-μL droplet. In this article, it was mentioned that the sliding angle of water droplets depends on the droplet volume and is in the range of 7° for a 50-μL droplet to 20° for a 7-μL droplet. Also, it was mentioned that when perfluoroalkyl chain is introduced to the surface of silica particles, the superhydrophobic fabric becomes highly oleophobic, so that the static CA for a 15-µL sunflower oil droplet becomes 140° and its sliding angle will be 24°. 14
Antimicrobial Coatings
Another major finishing in textile industry refers to elimination of microorganisms from
textiles. Antimicrobial coatings have been intensively studied and probably are the commercially
most important finishes. There are some drawbacks with regard to the settlement of microorganisms on
textiles. Because of specific structure and large surface of textile materials especially in the
natural ones, good adhesion and excellent water storage properties are expected. Such textiles in
contact with the skin (under sufficient temperature and humidity conditions) offer an ideal place
for the growth of pathogenic bacteria, because of their porous and hydrophilic structure. Preventing
the degradation of textile fibers, limiting the prevalence of bacteria, reducing odor formation, and
protecting users by avoiding the transfer and spread of pathogens are some possible advantages of
the above-mentioned finishes. Many approaches to create an antimicrobial textile by nanosol
treatment have been proposed by researchers. There are different methods to destroy germs using
nanosol finishes, which include the employment of photoactive nanosol coatings containing
TiO2 in anatase form or those using the controlled release of embedded biocides or even
using composite coatings containing TiO2 and SiO2 sols.
1
Xing et al. used low temperature and cost-effective process for creating antimicrobial effect
on cotton textiles based on sol–gel method. In this study, they used water glass as precursor and
the antimicrobial treatment was performed by treating silica impregnated cotton textiles with silver
nitrate solution. Samples had excellent antibacterial activity against
Creating both antibacterial and superhydrophobic effects simultaneously on cotton textiles has also been reported. 3 In the previous study, by dopping Cu nanoparticles inside silica sol, we obtained an improved superhydrophobicity (static water CA of 155°) accompanied with excellent antibacterial activity against both of Gram-positive and Gram-negative bacteria. In the first step, silica nanoparticles were synthesized using general Stöber method. Then Cu nanoparticles were dopped inside inorganic nanoparticle networks in two different percentages (wt %). Resultant coating material was applied on cotton fabric, and HDTMS was used for lowering the surface energy of samples. Due to the air-trapping capability of inclined surface and extreme antibacterial activity of Cu nanoparticles, superhydrophobicity and also antibacterial activity of samples were promoted. In Figure 3 and Table 1, an improved porous structure and also enhanced antibacterial activity of cotton textiles are confirmed by scanning probe microscope and American Association of Textile Chemists and Colorists (AATCC) 100 method, respectively.
Percentage Reduction in Bacteria on the Fabricated Silica-Cu Surfaces.

Source: Reprinted from Berendjchi, A.; Khajavi, R.; Yazdanshenas, M. E. Fabrication of
Superhydrophobic and Antibacterial Surface on Cotton Fabric by Doped Silica-Based Sols with
Nanoparticles of Copper.

Scanning electon microscope and scanning probe microscope micrographs of surface fabricated by (a & b; left) untreated (middle) silica sol, (right) 0.5% copper (Cu)-doped silica; and (c) water drops on surface fabricated by 0.5% Cu-doped silica sol and its contact angle. 3
Conclusion
Different possible applications of metal oxide nanoparticles prepared by sol–gel method were discussed. In all cases, subsequent modifying by appropriate chemical compounds is needed. Silica nanoparticles are used more than the other types of inorganic metal oxide nanoparticles because of their special shape and also higher activity.
Footnotes
Acknowledgments
The authors acknowledge Dr. Manafi from the Department of Chemistry of Islamic Azad University–South Tehran Branch for his constructive advice in this review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
