Abstract
Recombinant zoster vaccine (RZV) was recommended in 2020 in the US for adults aged ≥50 years. This retrospective study used cross-sectional data from 2020 National Health Interview Survey respondents aged ≥50 years. Sample weights were applied to allow results to be representative of the non-institutionalized US population. Associations between respondent characteristics and coverage were explored using logistic regression. An estimated 14.1% of US adults aged ≥50 years had received ≥1 dose of RZV by 2020. Coverage varied by race and ethnicity (6.3% Hispanic, 6.9% Black/African American, 13.7% Asian, 16.6% White), education (6.9% for grade 1–11 to 24.1% with master’s degrees), household income (8.8% for <$35,000 to 18.8% for ≥$100,000), age (7.3%, 14.6%, 19.9%, and 18.1% for ages 50–59, 60–64, 65–74, and ≥75 years, respectively), health insurance (2.2% without, 14.8% with), recency of last health visit (1.8% if >3 years prior to 15.1% if <1 year), and receipt of influenza vaccine in the past year (3.9% without, 21.1% with) (all P < 0.001). In multivariable analysis, factors associated with lower RZV coverage included Black/African American race, Hispanic ethnicity, age 50–59 years, lower household income, less recent last health visit, and no influenza vaccination. In conclusion, only 1-in-7 non-institutionalized Americans aged ≥50 years reported RZV coverage by 2020, with significant disparities among subgroups defined by race and ethnicity, age, and levels of educational attainment, income, and insurance. These results highlight an opportunity to improve herpes zoster protection through increased vaccination, pursued in a more equitable manner.
Herpes zoster (HZ) is caused by reactivation of latent varicella zoster virus. 1 It is characterized by a painful, itchy rash and skin lesions, most commonly on the trunk. 1 HZ can have a profound impact on functional status and quality of life, 2 and can result in complications, most commonly postherpetic neuralgia (ie, chronic pain at the rash site). 1
Approximately one in three individuals in the United States (US) will develop HZ during their life. 1 HZ risk increases with age due to a natural age-related decline in cell-mediated immune function, 3 with incidence rates in the US during 2011 ranging from 6.7 to 12.8 cases per 1000 person-years among individuals aged 50–59 to ≥80 years, respectively. 4 HZ risk is also increased among immunosuppressed individuals (eg, human immunodeficiency virus, cancer) and those with certain comorbidities (eg, chronic obstructive pulmonary disease, diabetes). 5 HZ has been associated with considerable health care costs among immunocompetent and immunocompromised individuals.6,7
Since 2018, the US. Advisory Committee on Immunization Practices (ACIP) has recommended 2 doses of recombinant zoster vaccine (RZV) for adults aged ≥50 years. 8 RZV is recommended regardless of prior receipt of zoster vaccine live (ZVL), 8 which was first recommended by ACIP in 2008 for adults aged ≥60 years, but was discontinued in the United States in 2020. 9 In 2022, ACIP recommendations for RZV expanded to include all immunocompromised/immunosuppressed adults aged ≥19 years. 9
National Health Interview Survey (NHIS) results from 2020 indicate that only 14.1% of adults aged ≥50 years had reported receipt of ≥1 dose of RZV. 10 More respondents (29.4%) reported receipt of any HZ vaccine (≥1 dose of RZV or ZVL), with estimates varying widely by race and ethnicity (15.6% [Hispanic], 17.4% [non-Hispanic Black], 30.5% [non-Hispanic Asian], and 33.4% [non-Hispanic White]), 10 suggesting a need for more equitable HZ prevention. However, disparities in RZV coverage by race and ethnicity as reported by NHIS participants have not yet been published. Accordingly, the current study used NHIS data to estimate RZV coverage in 2020 among various subgroups of US adults aged ≥50 years; to explore associations between selected characteristics and 2020 RZV coverage; and to compare RZV coverage levels over time (2019–2021).
Materials and Methods
Study design and data source
This retrospective study was based on data from the NHIS, which is an annual cross-sectional household interview survey that targets the civilian non-institutionalized US population using geographically clustered sampling techniques. 11 The questionnaire includes items related to demographics; health insurance; chronic conditions; access to, and use of, health care services; health-related behaviors; and level of functioning and disability. Items within the NHIS related to vaccination are detailed in Supplementary Table S1. Supplementary Data S1 contains additional information about how the NHIS was conducted.
Study population and outcomes
All NHIS respondents aged ≥50 years at the time of their participation in the 2020 NHIS survey were selected for inclusion; however, only respondents with informative responses were included in analyses (further details below). Outcomes included 2020 RZV coverage (≥1 dose), overall and among subgroups defined by various social determinants of health, sociodemographic parameters, access to health care, and clinical characteristics (see Supplementary Table S2); 2020 RZV coverage (≥1 dose) among individuals who had previously received ZVL, overall and among various subgroups; associations between selected characteristics and 2020 RZV coverage (≥1 dose); and 2020 RZV coverage by number of doses, overall and among various subgroups. Additionally, trends in RZV coverage between 2019 and 2021 were assessed and compared with selected vaccines (ie, ZVL, influenza, and pneumococcal). This objective included all respondents aged ≥50 years with informative responses from the 2019, 2020, and 2021 NHIS surveys.
Statistical analyses
All analyses were performed using SAS version 9.4, and leveraged its specialized procedures for survey data that consider the complexity of study design in estimations of variance (see Supplementary Data S2). All results were weighted by applying NHIS weights to give results considered representative of the US non-institutionalized population. Two-sided 95% confidence intervals (CIs) were calculated using the Clopper–Pearson method adapted for complex surveys. 12
Preliminary analyses
The impact of including or excluding missing and non-informative responses (“don’t know,” “refused,” “not ascertained,” “unknown”) was tested. Based on similarities of outcomes, only informative responses (“yes” or “no”) were considered. Further details of all preliminary analyses are provided in Supplementary Data S3.
Main analyses
Vaccine coverage was mainly estimated as the number of respondents who reported receipt of a given vaccine divided by the corresponding number of respondents with informative responses. As receipt of RZV was only asked to respondents who reported receiving an HZ vaccine, RZV coverage was estimated as the number of respondents who reported receipt of RZV divided by the sum of those who reported “yes” or “no” to RZV and those who reported “no” to any HZ vaccine. RZV coverage was estimated overall and among various subgroups (see Supplementary Table S2), among respondents who reported receipt of ZVL, and by number of RZV doses. Differences in RZV coverage by subgroup were analyzed using the Rao–Scott Chi-square test.
Odds of RZV coverage by selected characteristics (Supplementary Tables S2 and S3) were explored using logistic regression models (Supplementary Data S4) and are presented as odds ratios (ORs) with accompanying 95% CIs.
The percentages of individuals who reported receipt of any vaccine for HZ (≥1 dose of RZV or ZVL ever, ≥1 dose RZV ever, ≥1 dose ZVL ever), influenza (past 12 months), and pneumococcal (ever) during 2019, 2020, and 2021, respectively, are presented. Analysis of variance tests were performed for trend analysis at a global alpha level of 5%.
Results
Among 37,358 respondents to the 2020 NHIS, 18,384 were aged ≥50 years and 17,668 provided informative responses to receipt of RZV. All results presented in the text and figures are weighted, while unweighted and additional weighted results (including 95% CIs) can be found in the Supplementary Tables.
RZV coverage (≥1 dose; 2020)
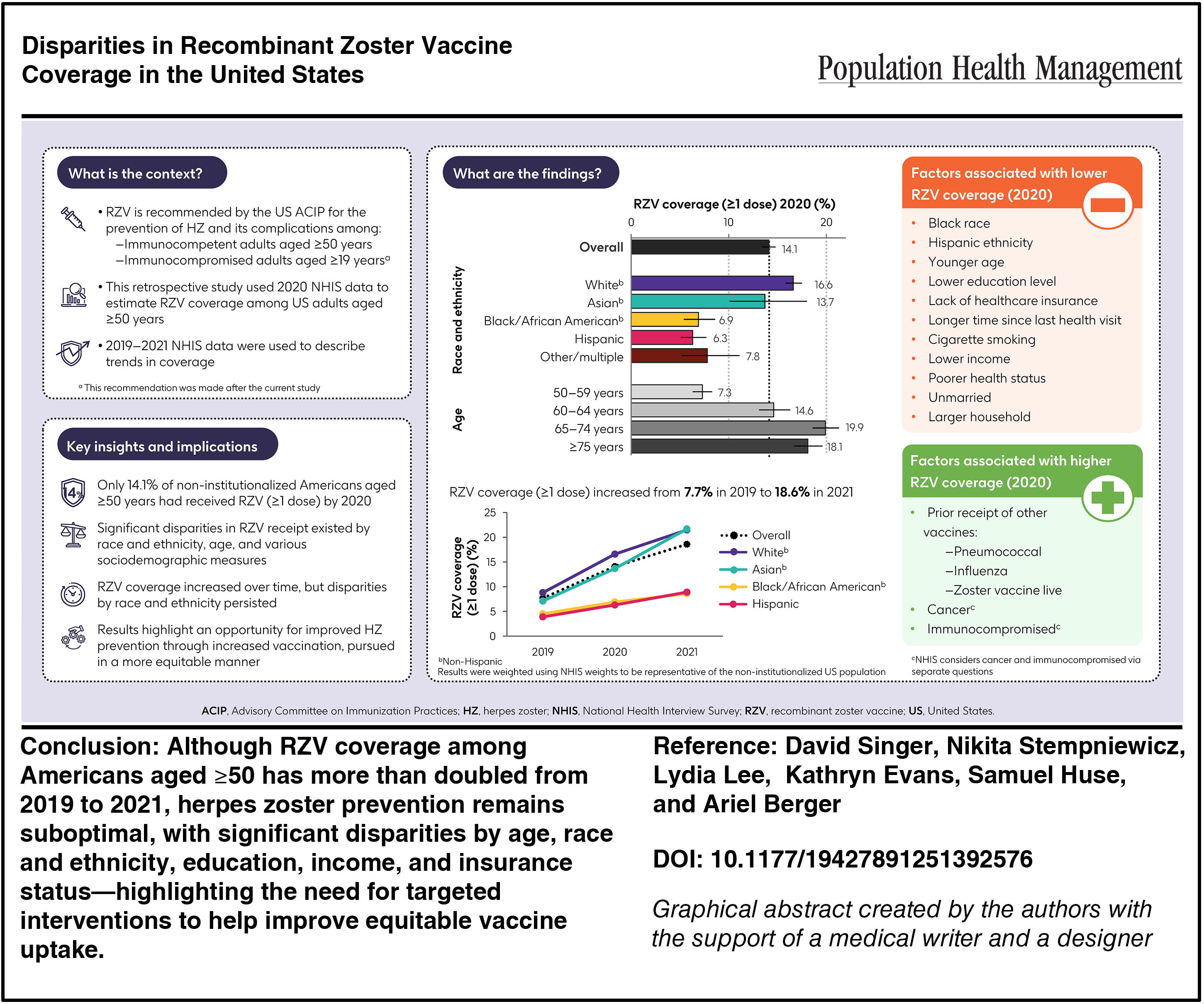
RZV coverage in 2020 was estimated to be 14.1% (Fig. 1; Supplementary Table S4). Coverage varied significantly by race and ethnicity, from 6.3% among non-Hispanic Black/African American to 16.6% among non-Hispanic White individuals (P < 0.001) (Fig. 1; Supplementary Table S5). It also varied significantly by marital status (8.8% separated to 15.5% married), household members (8.4% if ≥5 to 17.8% if 2), household children (6.7% if 1 to 15.1% if 0), and education level (6.9% if grade 1–11 to 24.1% if master’s degree) (all P < 0.001). RZV coverage was significantly lower among individuals with lower household incomes, lower income-to-poverty ratios, 13 social security payments, and lower food security. It varied significantly by age (7.3% for age 50–59 years to 19.9% for 65–74 years; P < 0.001), but not sex.
RZV coverage (≥1 dose) among individuals aged ≥50 years (based on informative responses to the 2020 NHIS), by selected sociodemographic characteristics and social determinants of health. aNon-Hispanic. bOnly based on 42 respondents. cOccupational, technical, or vocational. dAcademic program. eHousehold income divided by poverty threshold (defined by the US Census Bureau according to family size and number of children). 13 fIncome from public assistance. GED, General Educational Development; NHIS, National Health Interview Survey; RZV, recombinant zoster vaccine; SSDI, social Security Disability Insurance; SSI, supplemental security income; US, United States.
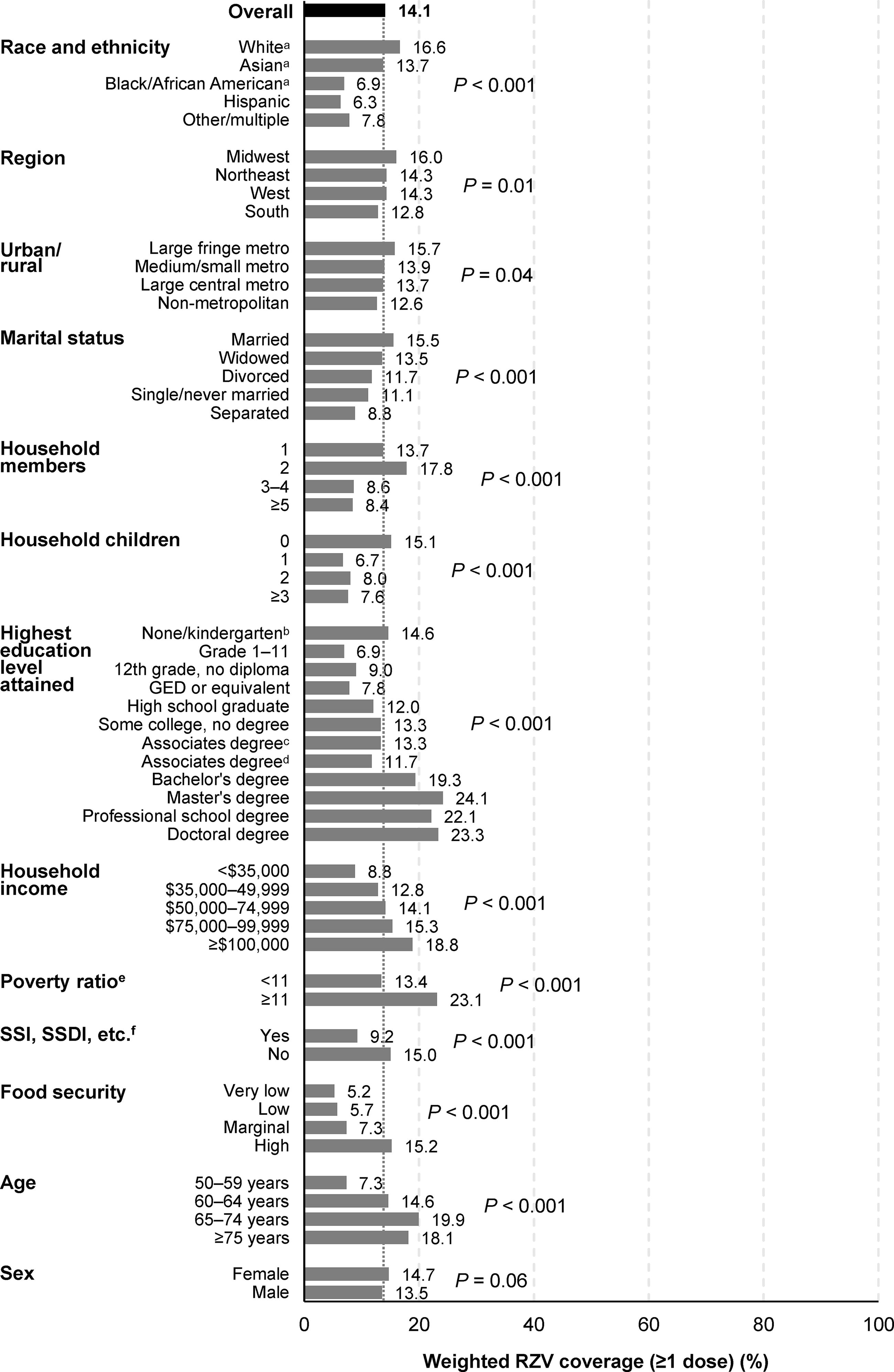
RZV coverage was significantly lower among individuals reporting no health insurance (2.2% vs. 14.8% with insurance; P < 0.001) and varied significantly by type of insurance (Fig. 2; Supplementary Table S6). It was also lower among individuals with no available place for care, worry/difficulty paying for health care, delayed/no care due to cost, a longer time since their last health visit, and less frequent urgent care visits; however, coverage was lower among individuals with more frequent emergency room visits.
RZV coverage (≥1 dose) among individuals aged ≥50 years (based on informative responses to the 2020 NHIS), by selected characteristics related to access to health care. aExcluding medicare advantage. ER, emergency room; NHIS, National Health Interview Survey; RZV, recombinant zoster vaccine.
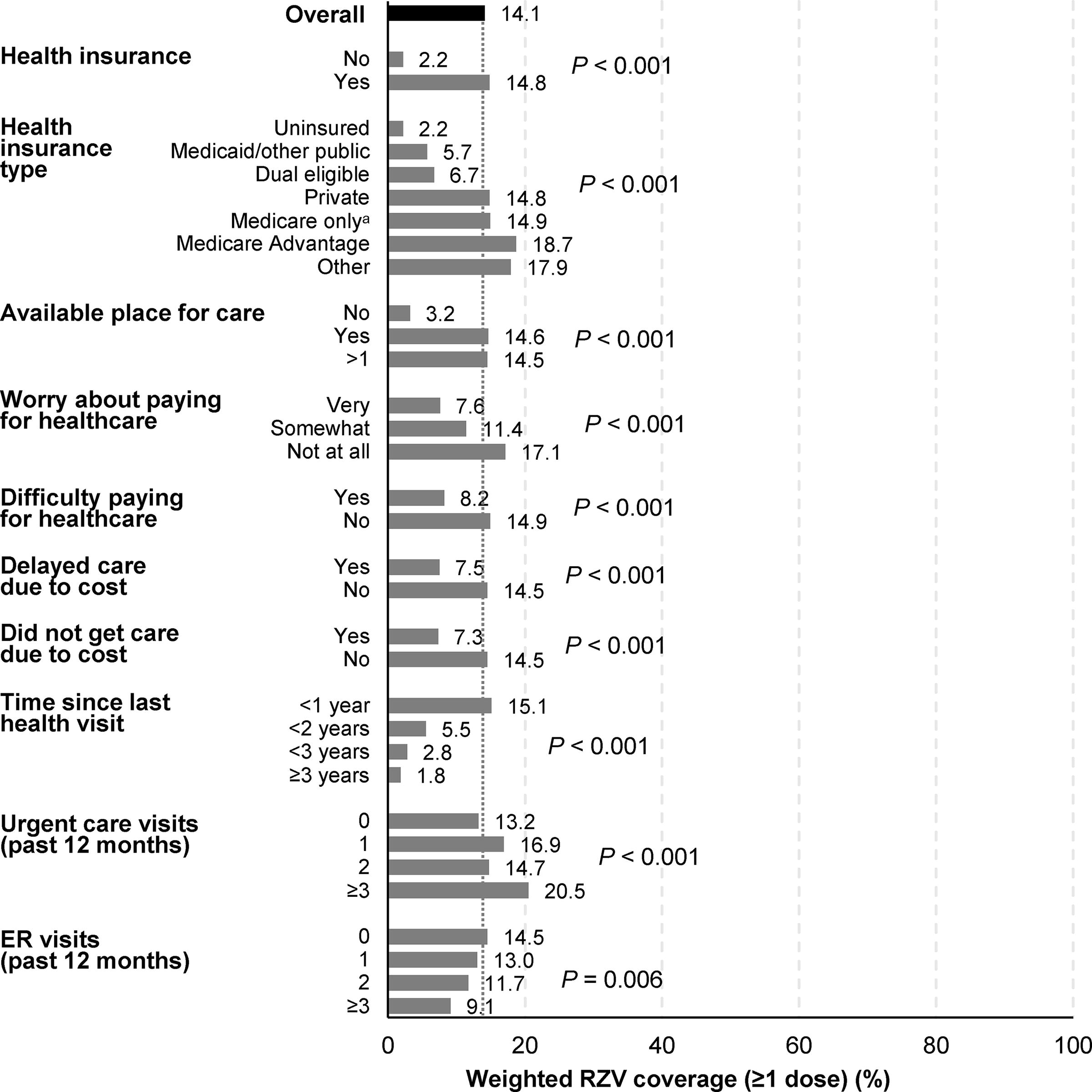
RZV coverage was significantly lower among respondents with worse reported general health status (8.7% for poor to 16.6% for very good health; P < 0.001) (Fig. 3; Supplementary Table S7). It was also significantly lower among people who smoke (5.6% current some days to 16.7% former; P < 0.001). Coverage was significantly higher among respondents who had ever been immunocompromised (19.1% vs. 15.2%; P = 0.009) (including cancer [19.2% vs. 13.0%; P < 0.001; ascertained in a separate item]). Coverage did not vary significantly by presence (vs. absence) of weak/failing kidneys, hypertension, coronary artery disease, anxiety/depression, asthma, diabetes, chronic lung disease, stroke, or dementia. However, coverage was significantly higher among individuals who had received influenza vaccine in the past 12 months (21.1% vs. 3.9%) or pneumococcal vaccine ever (25.8% vs. 6.1% of those aged ≥65 years) (both P < 0.001).
RZV coverage (≥1 dose) among individuals aged ≥50 years (based on informative responses to the 2020 NHIS), by selected clinical characteristics. aChronic obstructive pulmonary disease, emphysema, chronic bronchitis. NHIS, National Health Interview Survey; RZV, recombinant zoster vaccine.
RZV coverage was higher among individuals who reported having received ZVL (25.1% vs. 14.1%; significance not tested). Coverage varied significantly by race and ethnicity (13.1% for non-Hispanic Asian to 27.0% for non-Hispanic White individuals; P = 0.001) and age (13.9% for 50–59 years to 27.9% for 60–64 years; P = 0.001) but not sex (Supplementary Fig. S1; Supplementary Table S8).
Univariable and multivariable analyses (≥1 dose; 2020)
Univariable analysis results are detailed in Supplementary Table S9. In multivariable analyses, factors that were significantly associated with lower self-reported RZV coverage included non-Hispanic Black/African American race (OR 0.55; 95% CI 0.43–0.71) and Hispanic ethnicity (0.61; 0.47–0.79) (both vs. non-Hispanic White), lower education level (0.64; CI 0.47–0.86 for ≤12th grade vs. masters/professional/doctorate), lower household income (eg, 0.55; 0.47–0.65 for <$35,000 vs. ≥$100,000), age 50–59 years (0.47; 0.39–0.58 vs. ≥75 years), and current vs. never smoking (0.67; 0.52–0.85) (Fig. 4; Supplementary Table S9). Conversely, factors that were significantly associated with higher coverage included receipt of influenza vaccine in the past 12 months (4.83; 4.07–5.74), more recent last health visit (2.46; 1.41–4.27 for <1 vs. ≥3 years), and having an available place for care (1.91; 1.24–2.95).
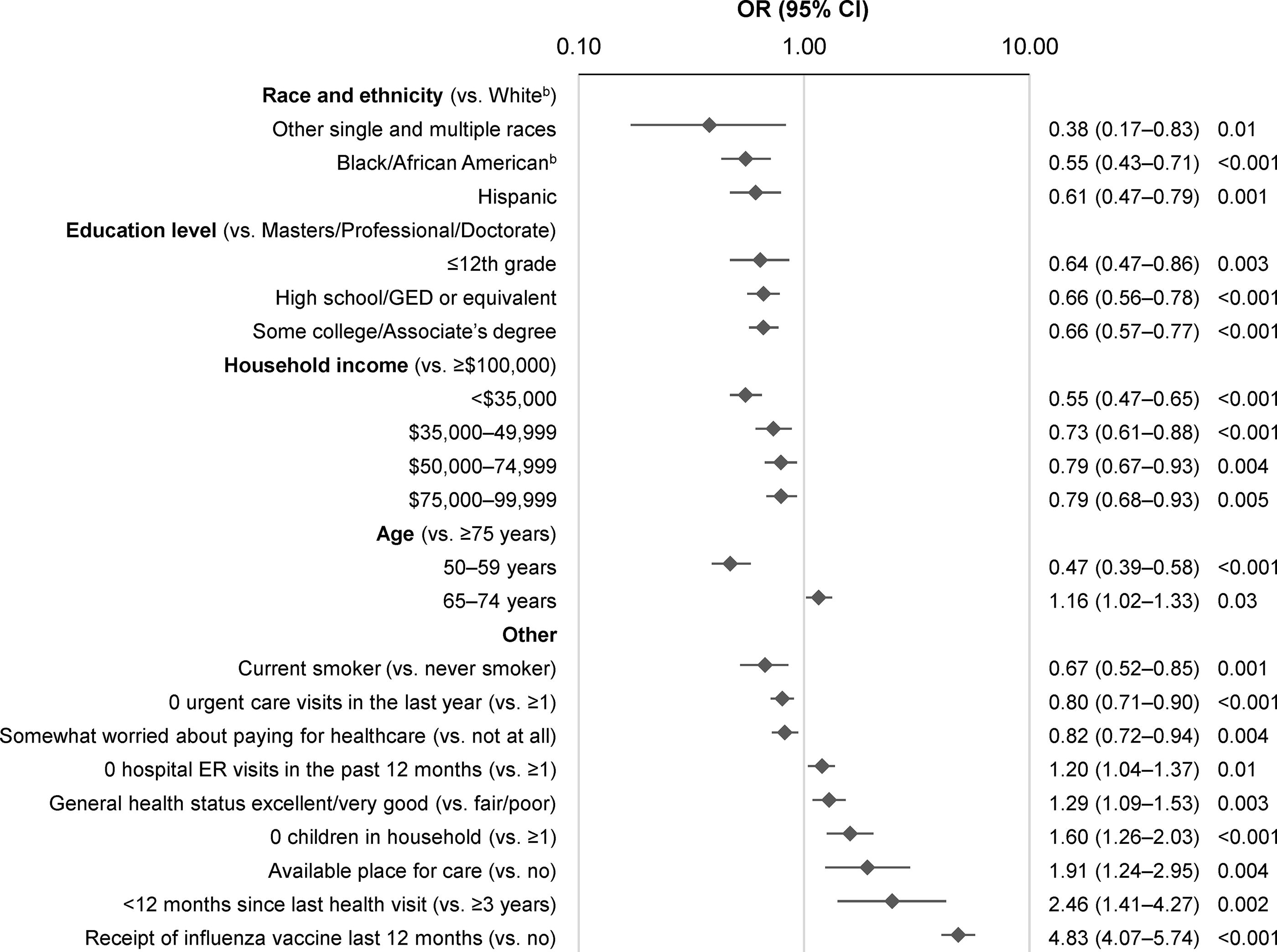
Significant associations between characteristics and RZV coverage (≥1 dose) among individuals aged ≥50 years (based on informative responses to the 2020 NHIS), by multivariable logistic regressiona (N = 17,226). aThe model did not include variables that were only available in the second half of the year (ie, immunocompromised status, weak/failing kidneys, delayed/no medical care due to COVID-19). bNon-Hispanic. CI, confidence interval; ER, emergency room; GED, General Educational Development; NHIS, National Health Interview Survey; OR, odds ratio; RZV, recombinant zoster vaccine.
RZV coverage by number of doses (2020)
Overall, 10.8% of respondents indicated receipt of 2 doses of RZV, 3.2% reported 1 dose, and 0.1% reported an unknown number of doses (Supplementary Fig. S2; Supplementary Table S10). Patterns of two-dose coverage were similar to those of ≥1-dose coverage (ie, higher among non-Hispanic White and Asian individuals and older age groups).
Vaccine coverage over time (2019–2021)
RZV coverage (≥1 dose) increased from 7.7% in 2019 to 18.6% in 2021 (P < 0.001; Fig. 5; Supplementary Table S11). Although coverage increased in all subgroups defined by race/ethnicity, differences in coverage persisted over time (Fig. 6; Supplementary Table S11).
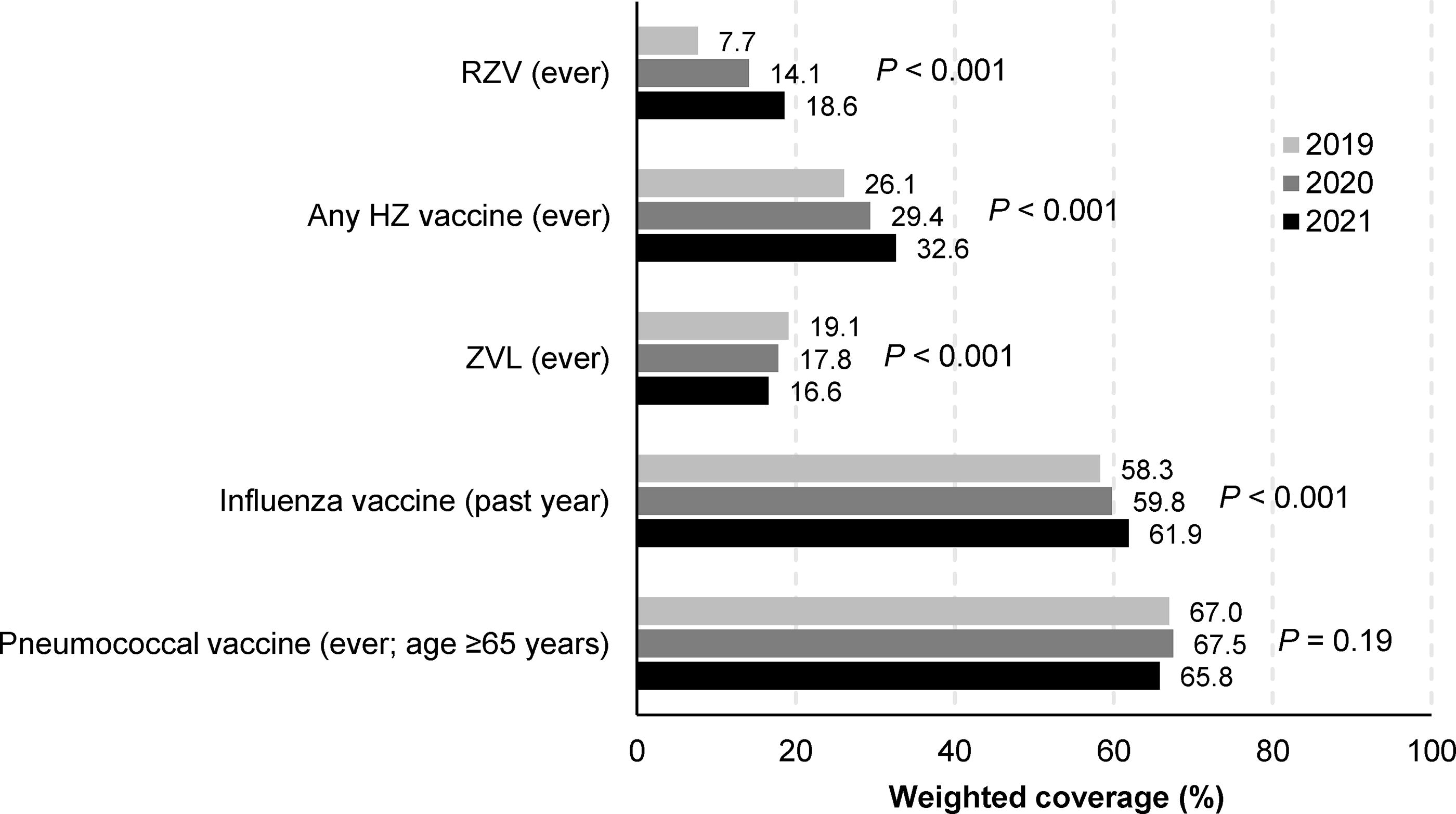
Vaccine coverage among individuals aged ≥50 years, by survey year. HZ, herpes zoster; RZV, recombinant zoster vaccine; ZVL, zoster vaccine live.
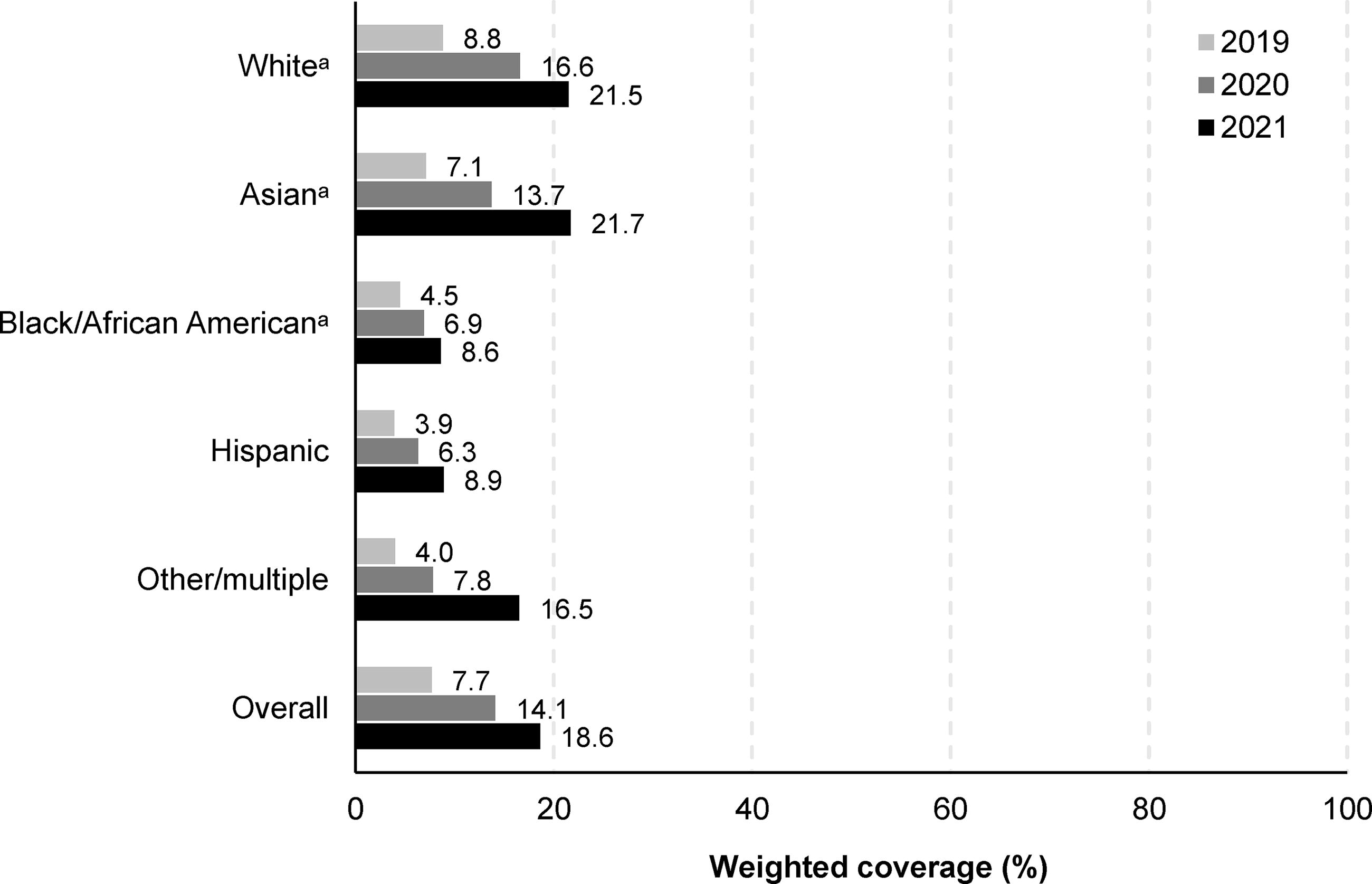
RZV coverage (≥1 dose) among individuals aged ≥50 years (based on informative responses to the 2020 NHIS), by race and ethnicity and survey year. aNon-Hispanic. NHIS, National Health Interview Survey; RZV, recombinant zoster vaccine.
Coverage with any HZ vaccine increased from 26.1% in 2019 to 32.6% in 2021, while ZVL coverage decreased from 19.1% to 16.6% (both P < 0.001; Fig. 5; Supplementary Table S11). Reported receipt of influenza and pneumococcal vaccines were considerably higher than for HZ vaccines (approximately 60%–70%) in all 3 years.
Discussion
Based on NHIS data, only 14.1% of non-institutionalized Americans aged ≥50 years reported receipt of RZV by 2020. Only 10.8% reported two doses, which are required to attain full protection. Significant disparities were identified, with Hispanic and non-Hispanic Black/African American individuals reporting coverage levels approximately one-half those of non-Hispanic Asian and White individuals. Individuals aged 50–59 years were less than half as likely as older age groups to have received RZV. After adjusting for various characteristics in multivariable analyses, odds of RZV vaccination remained lower among Hispanic and non-Hispanic Black/African American individuals and younger age groups.
Our findings are consistent with previous analyses of NHIS data that have shown numerically lower coverage with HZ vaccine (any) among racial and ethnic minority individuals (vs. non-Hispanic White)10,14 and younger individuals (among those aged ≥50 years). 10 Additionally, they align with an analysis based on claims data that found lower RZV coverage among non-Hispanic Black and Hispanic vs. Asian and non-Hispanic White individuals, as well as increasing coverage with increasing age. 15 Other analyses of NHIS data have also revealed lower levels of coverage against other diseases (eg, influenza, pneumococcal disease) among Hispanic and non-Hispanic Black individuals.10,14 Other studies have reported lower COVID-19 vaccination coverage among non-Hispanic Black and Hispanic vs. non-Hispanic White (and Asian) adults.16,17
RZV coverage was generally higher among adults with higher levels of education: 7%–15% among subgroups who reported up to an associates degree; 19%–24% for those with higher degrees. Other studies have also reported increased vaccine coverage among adults who have attained higher levels of education.14,18
RZV coverage was significantly lower among subgroups defined by various socioeconomic measures associated with lower financial and health care security. Individuals with private health insurance or Medicare had significantly higher RZV coverage estimates than those with Medicaid or other public insurance. Although significant in univariable analysis, health care insurance (yes vs. no; type), income-to-poverty ratio, receipt of public assistance, level of food security, difficulty paying for health care, and delayed/no care due to cost were not retained in the final multivariable model, likely due to overlap with variables related to household income and degree of worry about paying for health care. Other studies have reported lower RZV coverage among “probable renters” versus “probable homeowners” 15 and other vaccines among adults with lower income levels and/or no health insurance.14,18 Previously, many insured individuals who had received RZV had to pay at least part of the cost. 19 However, this potential barrier has likely been eliminated, as most adult vaccines, including RZV, are now covered without out-of-pocket costs under private insurance, Medicare, and Medicaid for most individuals. 20
RZV coverage among immunocompromised adults aged ≥50 years, although higher than among immunocompetent individuals, was only 19.1%. Currently, such individuals are recommended to receive RZV based on both their age 8 and immunocompromised status, 9 both of which increase the risk of HZ3–5 (and complications such as postherpetic neuralgia).21,22 However, as the latter recommendation had not yet been made as of 2020, further studies with more recent data are needed to better understand its impact on coverage levels in the US.
Not surprisingly, RZV coverage was approximately four to five times higher among individuals who had versus not received other vaccines recommended for adults. Although influenza vaccine was retained in the multivariable model, the pneumococcal vaccine was not, likely due to overlap with influenza vaccination. RZV coverage was around twice as high among individuals who had previously received ZVL as in the overall population. This is not surprising, as receipt of prior vaccinations is strongly associated with receipt of subsequent vaccines.15,23 However, despite the better coverage among those who had previously received ZVL, almost 75% of those who had received ZVL had not received RZV despite the current ACIP recommendations to administer RZV to adults who previously received ZVL. 8 Other studies24,25 have indicated that some health care providers may not be aware of this recommendation and may be less likely to recommend RZV to such individuals. 26
Although RZV coverage (ever) among adults aged ≥50 years increased from 7.7% in 2019 to 18.6% in 2021, this remained much lower than corresponding self-reported receipt of influenza vaccine (past year; approximately 60%) and pneumococcal vaccine (ever; age ≥65 years; approximately 65%). These higher vaccine coverages suggest an opportunity to further improve RZV coverage among adults aged ≥50 years. However, these findings also suggest there is room for improvement in the coverages of other vaccines recommended for adults. Disparities in RZV coverage by race and ethnicity did not improve from 2019 to 2021, and given the other disparities identified in this study, suggest a need for interventions to improve the equitable implementation of HZ prevention recommendations.
With the goal of increasing vaccine equity, the Centers for Disease Control and Prevention’s “Partnering for Vaccine Equity” program aims to improve vaccine awareness, access, and confidence among racial and ethnic minority individuals. 27 The National Vaccine Advisory Committee also recently highlighted the longstanding inequities in vaccination in the United States and the need for improvement. 28 They identified multiple disadvantaged groups, including racial and ethnic minority individuals, individuals without health insurance, individuals with disabilities, and people experiencing homelessness, as well as highlighting inequalities by geographic location. 28 A recent review on strategies to improve vaccination coverage among older adults found that the use of various interventions (eg, telephone calls, text messages, emails, telehealth monitoring) could be beneficial. 29 Providing individuals with information on HZ vaccination and mailing them a prescription significantly improved uptake in one study, 30 as did development and implementation of a computerized clinical support system in another. 31 However, automated telephone calls from pharmacies were only beneficial in one of two studies.32,33 A recent US study reported that automated outreach, pop-up clinics, and mobile vans could be used to improve vaccination rates among underrepresented groups. 34 Another approach may be to co-administer adult vaccines, as is routinely done in children, 35 although this may require provider education. Clinical trials involving RZV co-administration demonstrated no evidence of interference with the immune response to either vaccine.36–39 In addition, ACIP guidelines state that RZV can be coadministered with other adult vaccines at different anatomical sites, although some coadministration studies are ongoing.8,9
As noted above, results from the current study could help inform strategies regarding such interventions by allowing them to be targeted to the various population subgroups with the lowest RZV coverage.
Strengths and limitations
A key strength of this study is that the NHIS uses geographically clustered sampling techniques to identify and enroll a nationally representative sample of the non-institutionalized US population. 11 This allows findings to be extrapolated to approximate the entire non-institutionalized US population, thus increasing the generalizability of the findings. Sampling and selection bias are also minimized using these sampling techniques. This level of generalizability is unlikely to be present in other data sources such as health care claims or electronic health records in which the population represented is often restricted to a single health network, region, and/or type of insurer.
However, information is only available for individuals who agreed to participate, thereby introducing the possibility of selection bias. Although self-reported race and ethnicity data are considered the gold standard, 40 subject recall about vaccinations may be imperfect, resulting in possible recall bias. All vaccinations were self-reported and could not be verified, so actual coverage levels may be lower or higher than estimated in the current study. The exclusion of non-informative responses could also have resulted in slight over- or under-estimations of RZV coverage (see Supplementary Data S3). As only 2.6% of responses were non-informative, this likely had a minimal impact on the results, although this could have varied between subgroups.
Lastly, an important consideration is the use of NHIS data from 2020, which coincided with the start of the COVID-19 pandemic. Data from the Centers for Disease Control and Prevention showed that weekly vaccination rates (including RZV) were substantially lower during March to July 2020 compared with these months during 2019 among Medicare beneficiaries aged ≥65 years. 41 As a result, estimates of RZV uptake in 2020 may be lower than what would have occurred in the absence of the pandemic. However, NHIS data from 2017 to 2022 show a steady annual increase in overall RZV coverage (reported as ever having received the two-dose series) between 2018 and 2022. 42 While the findings from this study reflect disparities observed in 2020, they may partially reflect COVID-19 pandemic-related disruptions. In addition, the impact of the COVID-19 pandemic on RZV uptake may have varied across sub-populations. Hence, future studies with more recent data on RZV uptake will be important to assess whether the disparities observed in this study persisted.
Conclusions
Although estimated RZV coverage among non-institutionalized Americans aged ≥50 years more than doubled from 7.7% in 2019 to 18.6% in 2021, HZ prevention remained suboptimal. Coverage levels varied across individuals of different races and ethnicity, and were lower among adults aged 50–59 years and those with lower educational attainment, lower household income, and no health insurance. The deeper understanding of RZV coverage provided from this study may help to inform the design, implementation, and evaluation of interventions and policy changes focused on improving the equitable implementation of HZ prevention recommendations. Interventions and targeted outreach to subgroups of greatest disparity may help to improve RZV coverage, which would protect these otherwise vulnerable populations against HZ and its unwanted sequelae and improve health equity.
Authors’ Contributions
Mr. Stempniewicz.: Conceptualization and methodology. Mr. Berger, Ms. Evans, and Mr. Huse.: Formal analysis, investigation, validation. Dr. Singer and Ms. Lee.: Formal analysis. All authors.: Writing—reviewing and editing. All authors approved the final version of the article and followed ICMJE recommendations.
Footnotes
Acknowledgments
The authors thank Véronique Pagé for her contributions (statistical support and consultation) to the study. The authors also thank Enovalife Medical Communications Service Center for editorial assistance and article coordination, on behalf of GSK. Jenny Lloyd (on behalf of GSK) provided writing support.
Author Disclosure Statement
Dr. Singer and Mr. Stempniewicz are employed by and hold financial equities in GSK. Ms. Lee is a university worker hired by GSK. Mr. Berger, Ms. Evans, and Mr. Huse are employees of Evidera, an independent consultancy that was paid by GSK to conduct this study. All authors declare no other financial and nonfinancial relationships and activities. Dr. Singer, Ms. Evans K., Mr. Stempniewicz N., Ms. Page V., Mr. Huse S., and Mr. Berger A. Disparities in recombinant zoster vaccine coverage among adults in the United States by race and ethnicity and SDOH. J Gen Intern Med 2023;38(Suppl 2):S182.
Funding Information
GSK funded this study (GSK study identifier: VEO-000352) and was involved in all stages of study conduct, including analysis of the data. GSK also took in charge of all costs associated with the development and publication of this article.
Data Sharing
Data could be shared upon reasonable request.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
