Abstract
Background
Monitoring stroke patients in critical-care units for 24 h after thrombolysis or endovascular thrombectomy is considered standard of care in current guidelines but is not evidence-based. Due to the COVID-19 pandemic, our center adopted a targeted protocol in April 2021 with 24-h critical-care monitoring no longer being guaranteed for stroke patients receiving reperfusion treatment. We aim to compare the incidence and timing of complications during the year under the targeted approach compared to prior years when the standard of care was followed.
Methods
We conducted a single-center retrospective cohort study. We analyzed data from stroke patients treated with thrombolysis and/or endovascular thrombectomy in 2019 (pre-COVID-19, standard of care), 2020 (during COVID-19, standard of care) and 2021 (during COVID-19, targeted protocol). Data extracted included demographics, the nature and timing of complications within the first 24 h, and the unit at the time of complication.
Results
Three hundred forty-nine patients were included in our study: 78 patients in 2019, 115 patients in 2020, and 156 patients in 2021. In 2021, 32% of patients experienced at least 1 complication within the first 24 h compared to 34% in 2020 and 27% in 2019. In 2021, 33% of patients admitted to critical-care units had a complication compared to 29% in non-critical care units. In 2021, 70% of complications had occurred by hour 8 compared to 49% in 2020 and 29% in 2019.
Conclusions
The incidence and timing of complications did not significantly worsen under the targeted approach compared to prior years and were not associated with hospital location.
Introduction
Since the initiation of intravenous thrombolysis with alteplase (tPA) for the treatment of acute ischemic stroke in the mid-1990s, 1 it has been considered standard of care for all patients receiving thrombolysis with or without endovascular thrombectomy to be monitored in critical-care units (such as Level 1 or Level 2 intensive-care units) for 24 h post-treatment.2-4 This practice is resource-intensive and lacks empirical support, reflecting 30-year old concerns about lingering hypofibrinogenemia induced by tPA and the risk of intracranial hemorrhage post-thrombolysis. 5 While this risk is not negligible and is well-documented at 3%–7%,6-8 the half-life of alteplase is measured on the order of minutes, 9 and it is not clear whether post-thrombolysis complications are evenly distributed among stroke patients and over the subsequent 24 h. Similarly, there is no rationale other than tradition for 24 h of monitoring for patients undergoing thrombectomy without thrombolysis, whose risk of hemorrhagic complications is unlikely to be present for 24 h. 10 A handful of recent observational studies suggest that certain subgroups of patients (eg, lower initial stroke severity or negative MRI findings post-thrombolysis) may not require admission to a critical-care unit11-13 or may require a shorter period of intensive monitoring.14,15 To our knowledge, no randomized trials have been published supporting 24 h of monitoring for patients post-thrombolysis or post-thrombectomy.
Aims
The third wave of the COVID-19 pandemic in early 2021 caused significant resource limitations in hospitals – especially critical care units – across Canada. 16 In response, The Ottawa Hospital adopted temporarily a targeted stroke monitoring protocol in mid-April 2021 to help alleviate the burden on critical care units (see S5). This targeted approach allowed the stroke neurologist to admit certain stroke patients receiving thrombolysis to other units in the hospital including general neurology or internal medicine wards. In other words, stroke patients were no longer guaranteed by protocol access to 24 h of critical-care monitoring. This pragmatic reorganization of care created the opportunity to study non-standard of care monitoring practices for acute stroke patients. This study sought to evaluate the incidence and timing of complications among stroke patients during the first 24 h following the administration of thrombolysis and/or endovascular thrombectomy (EVT), and their association with the hospital unit to which they are admitted. Crucially, we are interested in determining if these outcomes varied significantly from past years when standard of care practices were followed. We hypothesized that, under the targeted stroke monitoring protocol, the incidence of major complications would be low (less than 20%), that the incidence of symptomatic intracranial hemorrhage would not exceed 7% (which is the higher-bound rate of post-thrombolysis intracranial hemorrhage in the published literature), that the majority of complications (>50%) would occur in the first 8 h post admission, that the incidence and timing of complications would not be associated with the hospital unit to which patients are admitted, and that these findings would not vary significantly from past years.
Patients and Methods
We conducted a single-center retrospective cohort study that analyzed clinical and imaging data gathered from a high-volume comprehensive stroke center. The study was approved by the local institutional review board. All relevant data were extracted from electronic medical records and were reviewed by 2 members of the research team (TLT and CD). The data supporting the findings of this study are available from the corresponding author upon reasonable request by a qualified investigator. We followed the STROBE guidelines for conducting and reporting observational research (the complete STROBE checklist can be found in Supplemental document S6).
Patient Population
The Ottawa Hospital uses a QI infrastructure that tracks all stroke codes called to the CIVIC Campus Emergency Department (ED). This database was used to identify all patients presenting as stroke codes who received tPA and/or EVT and who were admitted to the hospital between April 15 (date The Ottawa Hospital’s new stroke monitoring protocol was issued) and September 30, 2021. Additionally, we collected the same data on patients fulfilling inclusion criteria from April 1 to September 30, 2020 (first wave of COVID-19) and June 1 to September 30, 2019 (pre-COVID-19). This shorter data collection period in 2019 is explained by the fact that the hospital underwent a change in electronic medical records in June 2019 that prevents our access to records before that time. This resulted in unequal sample sizes between our 3 years. Patients presenting as stroke codes as admitted in-patients, patients presenting as stroke codes with a diagnosis other than ischemic stroke, and patients presenting with an ischemic stroke who were not candidates for reperfusion treatment were excluded from this study. No informed consent was required as this study did not influence the care of patients.
Patient baseline characteristics (age, sex), COVID-19 status, National Institutes of Health Stroke Scale (NIHSS) score at presentation and at 24 h, time of symptom onset (or last seen well), reperfusion treatment received (intravenous thrombolysis administration [tPA or tenecteplase], EVT, or both), timing of reperfusion treatment, and antiplatelets therapy received were collected.
Outcomes
We recorded the incidence and timing of complications occurring in the first 24 h after the stroke code was initiated. Complications were divided as major (Neurological deterioration [≥4 points on NIHSS], new or larger ischemic stroke on neuroimaging, hemorrhagic transformation, symptomatic intracranial hemorrhage, myocardial infarction, systemic bleeding, and death) and minor (angioedema, fever [temperature >37.9°C], seizure, groin hematoma, rapid assessments of critical events (RACE) call, and move to higher unit). The incidence of hemorrhagic transformation was collected from the non contrast head computed tomography (CT) performed for all patients 24 h after reperfusion treatment (according per our institution’s stroke protocol) or earlier if there was a concern about symptomatic intracranial hemorrhage. Symptomatic intracranial hemorrhage was defined as any hemorrhagic transformation with neurological deterioration as indicated by a change in NIHSS ≥4 points compared to baseline. 17 If a patient had multiple complications, the timing of the first complication was used in the analyses. The hospital units to which patients were admitted right after reperfusion treatment, in which they were at the time of the complication, and at 24 h were also collected. These were categorized as critical care units (medical intensive care units [ICU], Neurological Acute Care Unit [NACU], Post-anesthesia Acute Care Unit [PACU]) and non-critical care units (neurology ward, medicine ward, or emergency departments [ED-Resuscitation, ED-emergent, and ED-Observation]), with the main difference being that nurses in critical care units have more training to identify neurological deterioration and have less patients under their care. Patients in non-critical care units would technically still undergo frequent vitals and neurological status monitoring, as per standard post-stroke guidelines (see S5). This is in line with the concept of “step-down” monitoring, but in non-critical care setting.
Statistical Analysis
Data were analyzed using descriptive statistics with chi-squared analysis as well as Kaplan-Meier curves with log-rank tests in accordance with the outlined questions and hypotheses. An interim analysis took place after data from the first 100 patients were collected to see if an unexpectedly high rate of complications (arbitrarily set at 40%) occurred, in which case the pandemic-related changes in practice would have been reassessed. Preplanned subgroup analysis included analysis by treatment group (thrombolysis, EVT, and thrombolysis + EVT) and by NIHSS on presentation (under or equal to 10, and over 10).
Results
Demographics
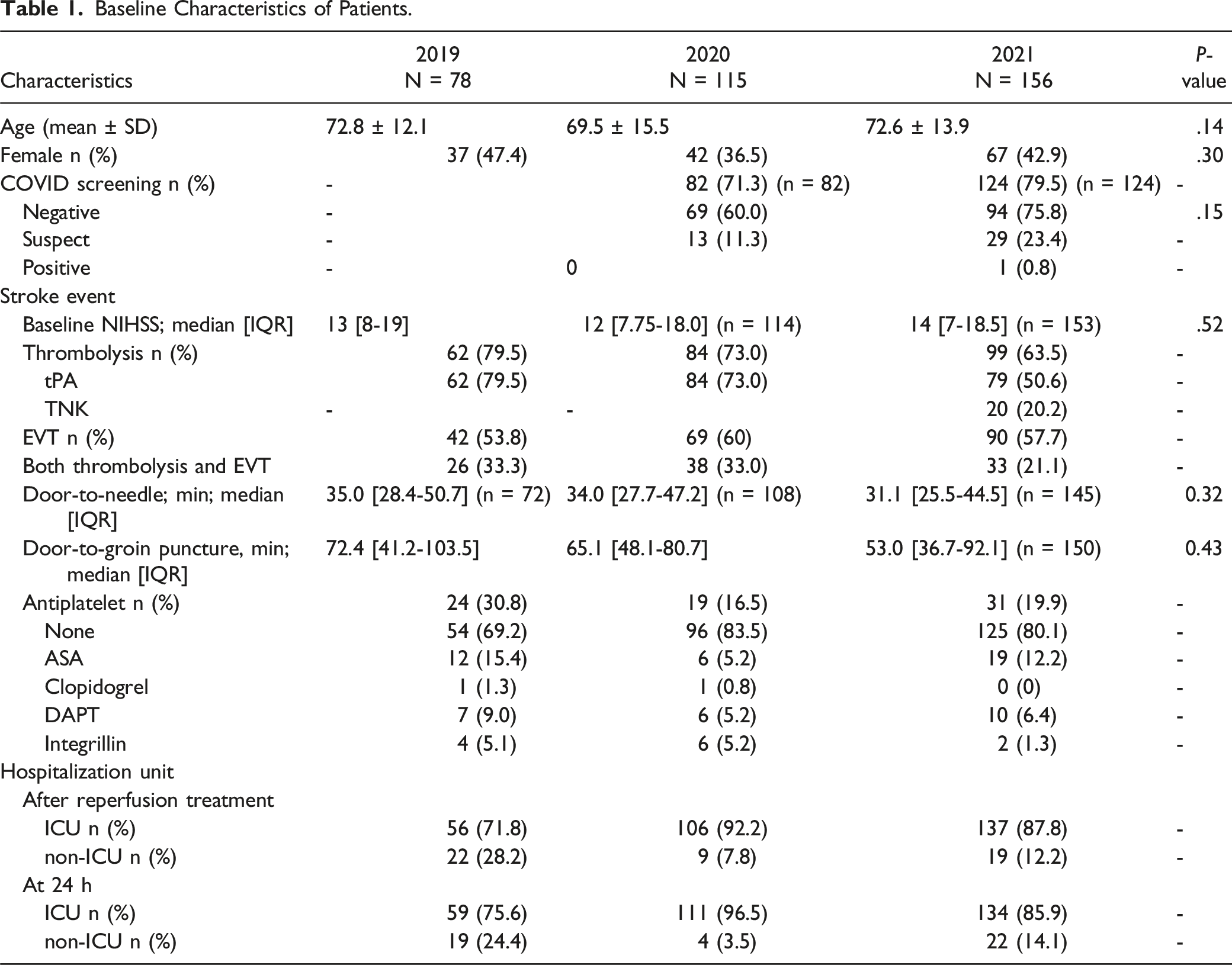
Baseline Characteristics of Patients.
Complications
Complications After Reperfusion Therapy Within 24 h in 2019, 2020 and 2021.
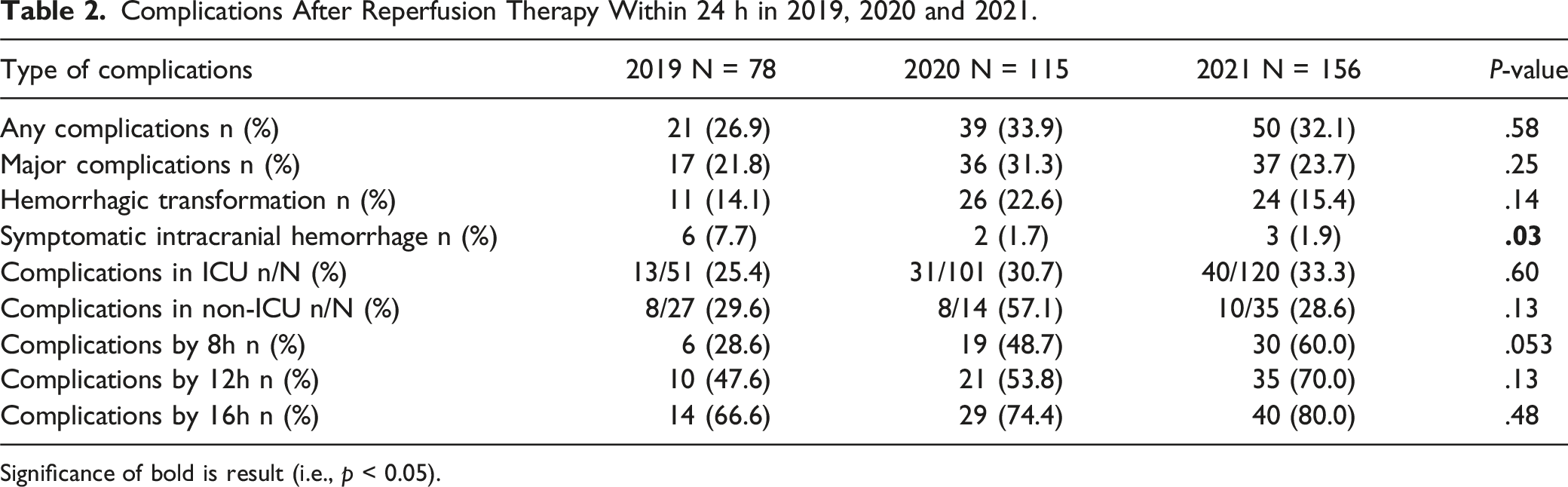
Significance of bold is result (i.e., p < 0.05).
Complications by Time
Figure 1 compares the timing of complication over the first 24 h of care for all 3 years and Figure 2 compares it based on the unit to which patients were admitted. Across all three years, 60% of complications occurred within 12 h. Twenty-nine per cent of complications occurred in the first 8 h in 2019 compared to 49% in 2020 and 60% in 2021, P = .053, which trended towards statistical significance. This trend disappeared with time. Figure 2 shows no significant difference (log-rank P > .05) in 2019 or 2021 for how early complications were detected in critical care vs non-critical care units. There was a statistical difference in 2020 for any complications (log-rank P = .011) and major complications (log-rank P = .050), but favoring an earlier detection for complications in non-critical care units. Rate of complications in the first 24 h following stroke code initiation in acute stroke patients treated with reperfusion therapies between 2019 (red), 2020 (green) and 2021 (blue). (A) Rate of any complications (log-rank P = .3); (B) Rate of major complications (log-rank P = .2); (C) Rate of hemorrhagic transformation (log-rank P = .2); (D) Rate of symptomatic intracranial hemorrhage (log-rank P = .034). Rate of complications over the first 24 h following stroke code initiation in acute stroke patients based on unit of admission (critical care units, red; non-critical care units, blue) in 2019, 2020 and 2021. (A) Rates of any complications in 2019 (log-rank P = .7); (B) Rates of any complications in 2020 (logrank P = .011); (C) Rates of any complications in 2021 (log-rank P = .5); (D) Rates of major complications in 2019 (log-rank P = 0.9); (E) Rates of major complications in 2020 (log-rank P = .05); (F) Rates of major complications in 2021 (log-rank P = .7).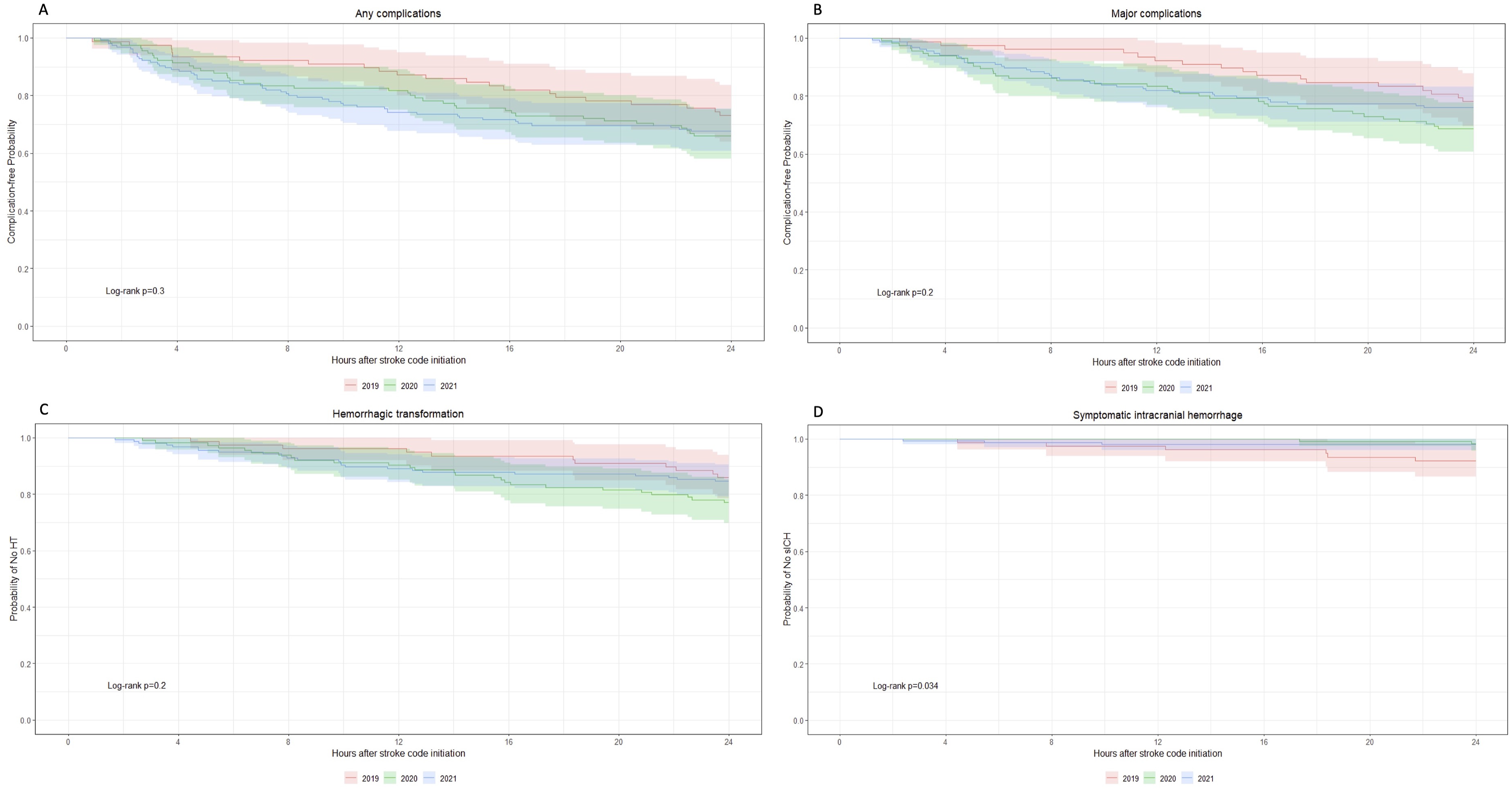
Complications by Unit
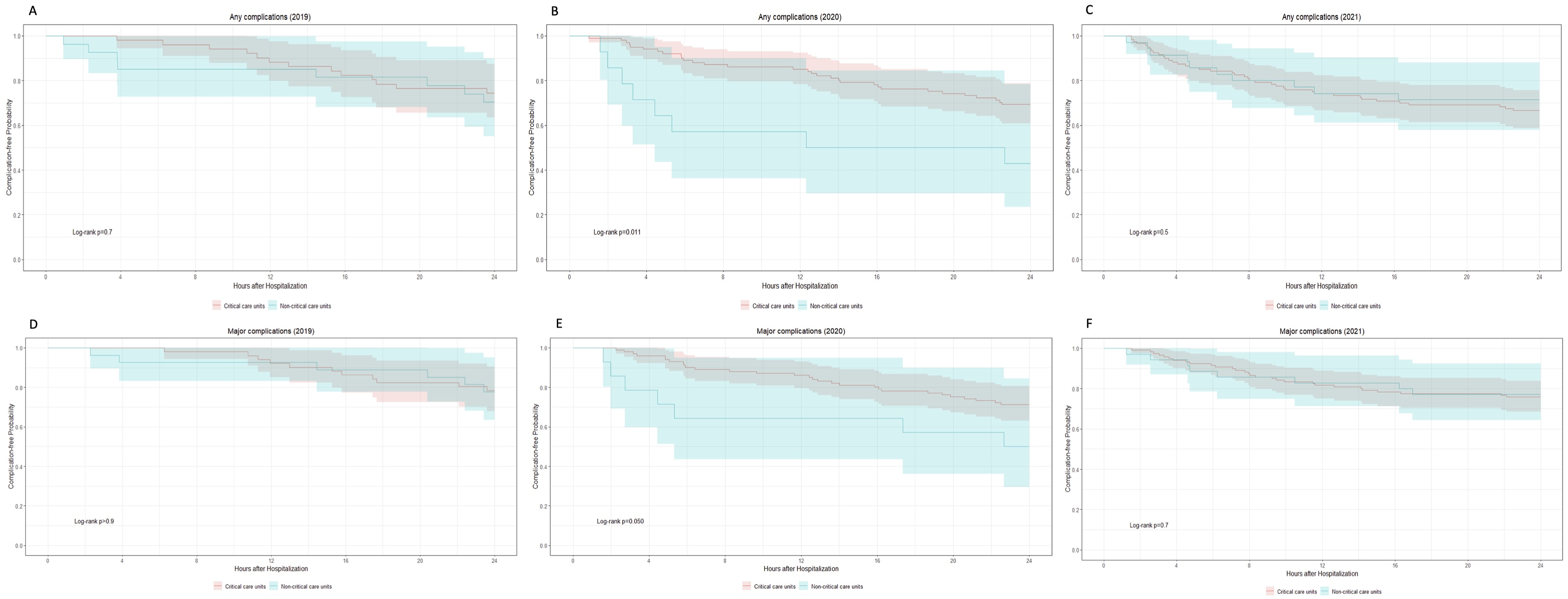
In 2021, 137 patients (88%) were initially hospitalized in critical care units compared to 106 patients (92%) in 2020 and 56 patients (72%) in 2019. At 24 h, the proportion of patients in ICU was reduced in 2021 (86%) while it increased in 2019 (76%) and 2020 (97%). Forty of the complications (80%) in 2021 occurred in patients admitted to critical care units compared to 36 (80%) in 2020 and 16 (62%) in 2019. The incidence of all complications that occurred in patients admitted to critical care units was 33% in 2021, 31% in 2020 and 25% in 2019, P = .60. The incidence of all complications that occurred in non-critical care units was 29% in 2021, 57% in 2020 and 30% in 2019, P = .13. There was a statistical association in 2020 between the unit of admission and the incidence of any complication (log-rank P = .01) and major complications (log-rank P = .05). However, this association was not present in 2021 or 2019 (Figure 2). There was no statistical difference in the rate of complications between all 3 years in critical care units (log-rank P = .2), but a trend towards statistical significance in non-critical care units with a higher rate of complications in 2020 compared to 2021 and 2019, log-rank P = .08 (Figure 3). Rates of any complications over the first 24 h following stroke code initiation in acute stroke patients based on unit of admission in 2019 (red), 2020 (green), and 2021 (blue). (A) Rates of any complications in critical-care units (log-rank P = .2); (B) Rates of any complications in non-critical care units (log-rank P = .08).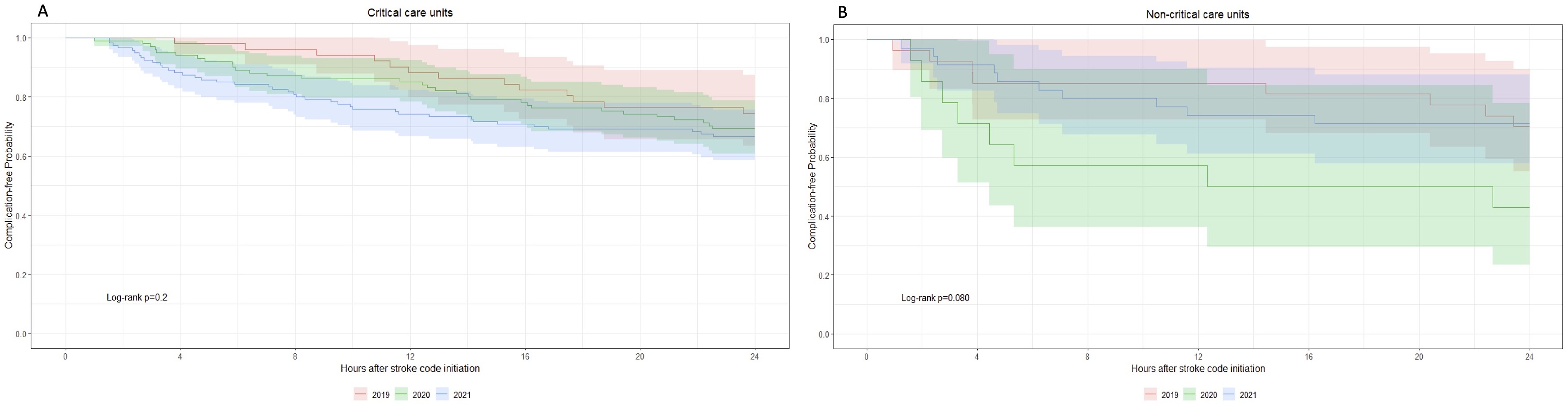
Complications by Treatment Groups
The incidence of any complications, of major complications, and of hemorrhagic transformation was lower in the thrombolysis group while the incidence of symptomatic intracranial hemorrhage was lower in the EVT group (Figure S1-S2). Over all 3 years, the incidence of any complication was 23% in the thrombolysis group, 34% in the EVT group and 41% in the combined therapy, log-rank P = .01. The incidence of major complications followed a similar pattern with 20% for the thrombolysis group, 28% for the EVT group and 32% for the combined therapy, but the difference was not significant, log-rank P = .08. Lastly, the incidence of symptomatic intracranial hemorrhage was not significantly different between the groups with 3% in the EVT group, 4% in the thrombolysis group, and 3% in the combined therapy group, log-rank P > .9.
Complications by NIHSS
When patients were analyzed by dichotomized NIHSS (above and below 10), several statistically significant differences were identified (see Figure S3-S4). Of note, the median NIHSS for the patients with NIHSS ≤10 was 7, compared to 18 for the group of patients with NIHSS >10. The incidence of any complication was 24% for patients presenting with a NIHSS ≤10 compared to 43% for NIHSS >10, log-rank P = .002. The incidence of major complications was 21% for NIHSS ≤10 and 37% for NIHSS >10, log-rank P = .006. Lastly, the incidence of symptomatic intracranial hemorrhage was higher for NIHSS >10 with 5% compared to 2% for NIHSS ≤10, but this difference was not significant, log-rank P = .15.
Discussion
This study took advantage of a pandemic-driven change in our center’s stroke monitoring protocol to interrogate the standard of care by which stroke patients undergoing reperfusion therapies require 24 h of critical-care monitoring. We compared the incidence and timing of complications in the initial 24 h of care among stroke patients receiving thrombolysis and/or EVT in relation to the hospital unit to which they were admitted, before (2019 and 2020) and after (2021) the protocol change. Our main hypothesis was that the change in monitoring protocol would not impact the incidence or timing of complications, or their association to hospital location.
The results presented seem overall supportive of our hypothesis: we saw no significant difference either in incidence, severity or timing of complications. The incidence of any complications and major complications was similar across all 3 years. The only notable difference was a higher rate of symptomatic intracranial hemorrhage in 2019 (7.7%), prior to any change in protocol. There was also no significant difference in the timing of complications: if anything, complications occurred or were identified earlier in 2021 than in previous years. For instance, by 8 h after the stroke code, 60% of complications had occurred in 2021 compared to 49% in 2020 and 29% in 2019, which trended towards significance (P = .053). Finally, no association was found between the incidence of complications and the unit to which patients were admitted in 2021.
Yet, in order to draw the appropriate conclusions from these findings, we have to discuss an unexpected result that questions a premise to our hypothesis. While we expected the change in protocol – essentially not guaranteeing a critical care bed for stroke patients receiving reperfusion therapy – to lead to a higher proportion of patients being admitted to non-critical care units compared to previous years, this was not the case: most (88%) patients were admitted to critical care units in 2021, which was comparable to 2020 (92%) and higher than 2019 (72%). This is unexpected in 2 ways: we expected less patients to be admitted to critical care units under the targeted protocol, but also more patients to be admitted to critical care units when the standard of care was in place. Regarding the former unexpected finding, this could be explained by the fact that there was no specific new criteria under the targeted protocol to send patients to an critical care units; it was left to the clinical judgement of the stroke neurologist on duty to make the best informed decision for the patient. They could have continued to send patients to critical care units simply by habit, or even by concern of not following the standard of care if a critical care bed was in fact available. Interestingly, however, less patients were admitted to critical care units by the end of the 24 h of monitoring in 2021 while there were more patients admitted to critical care units by the end of the 24 h in 2020 and 2019. This could suggest that neurologists would consider downgrading patients outside the critical care units more rapidly if able to. Regarding the latter unexpected finding, this probably testifies to the significant bed pressure our center’s critical care units have to deal with at all times. No matter the possible explanations behind this unexpected result, this raises the possibility of a type II error: maybe we did not find differences in outcomes because there were no significant differences in the intervention studied in the first place.
While we recognize how this unexpected result limits the testing of our initial hypothesis, we believe that the findings of our study can bypass this limitation and that key takeaways can still be drawn for 2 reasons. First, we can interpret differently what the new targeted protocol represent: instead of directly assessing the safety of admitting a higher proportion of patients to non-critical care units, our study evaluates the safety of a targeted protocol in which more clinical judgement is left to the stroke neurologist who can decide where the patient goes based on patient’s characteristics and clinical context compared to the standard of care protocol which is more rigid and ethically entails a “first come first serve” approach. Second, the very unexpected result that questions the premise of our study actually supports our hypothesis differently. While it was unexpected that close to 30% of patients would be admitted to non-critical care units in 2019 under the standard of care, this subpopulation is what we aimed to study in the first place. We thought the pandemic pressures represented a unique occasion to study patients admitted to non-critical care units, but as it turns out, a non-negligible proportion of patients are at all times in our center. And our findings show that there was no difference in incidence of complications in patients admitted to critical care units (25.4%) or non-critical care units (29.6%) in 2019 (see Table 2 and Figure 2(A)–(D)). A statistical difference was found in 2020, but this finding is limited by a small sample size of 14 patients admitted to non-critical care units. Taken all together, we believe these findings suggest that monitoring patients in non-critical units could be a safe alternative for some patients.
Overall, the findings in our study align with the literature.18,19 The incidence of symptomatic intracranial hemorrhage in our study at 3% is within the documented risk of 3%–7% in the literature.6-8 Patients with a NIHSS ≤10 were generally half as likely to suffer a complication compared to patients with a NIHSS >10. Similarly, the rate of symptomatic intracranial hemorrhage jumped from 2% for NIHSS≤ 10 to 5% for NIHSS >10. This is similar to other studies that support the proposition that patients with higher NIHSS are more likely to experience neurological deterioration or to require critical care resources.13,15 The incidence of any complication was highest in the combined therapy groups and lowest in the thrombolysis only group. The incidence of symptomatic intracranial hemorrhage was similar in the thrombolysis and combined therapy groups, and are comparable to the findings of the major trials comparing EVT alone to EVT plus thrombolysis.20,21 However, the incidence of symptomatic intracranial hemorrhage was lower in the EVT group (2%) compared to the other two groups (3%–5%). This differs from the published literature showing that the rate of symptomatic intracranial hemorrhage is not different when EVT is used alone or in combination to thrombolysis.22,23 Recent observational studies have investigated the timing of complications following thrombolysis and showed that over 80% of symptomatic intracranial hemorrhage and neurological deterioration occurs before 12 h post-treatment.14,15 Our study showed similar findings in 2021 with 80% of any complications and all 3 symptomatic intracranial hemorrhage occurring within 12 h post-treatment. A randomized controlled trial (RCT) led by Dr Craig Anderson in Australia is exploring the question of 8 h of post-tPA monitoring vs the standard 24 h monitoring but is currently only at the recruitment phase.
We acknowledge several limitations to our study. First, the sample sizes across the 3 years are unequal, as explained in our methods section, and overall quite small, limiting the power of our study. Second, as alluded to in the paragraphs above, the selection bias in our study given its observational design limit the conclusions we can draw. Third, the single-center nature of the study does limit the generalizability of our results. Finally, the modified Rankin Score (mRS) at 3 months was not collected for most patients, which limits the interpretation of the long-term clinical impact of our study. For instance, even if complications were potentially detected earlier in 2021 compared to prior years, it is unclear whether this led to a change in long-term outcomes.
In conclusion, the targeted protocol – not guaranteeing critical care monitoring – in April 2021 does not appear to have impacted negatively the care of stroke patients at our center in the first 24 h following thrombolysis or thrombectomy: the overall incidence of complications did not increase compared to previous years, there was no association between the incidence of complications and the unit of admission in 2021, and complications were not detected later in 2021 compared to previous years. Moreover, even in years where the standard of care was followed, an unexpectedly high proportion of patients were admitted to non-critical care without seeing a difference in incidence of complications or a delay in their recognition. These findings, combined with the results that most complications occurred within 12 h and that lower NIHSS were associated with less complications, could signify that monitoring stroke patients for 24 h in a critical care setting following reperfusion therapy may not be required in all cases. We hope this study will contribute to a broader discussion and motivate further research on the necessity of 24 h of critical care level monitoring for all stroke patients receiving reperfusion therapies.
Supplemental Material
Supplemental Material - Stroke Hospitalization Administration & Monitoring: Routine or COVID-19 Care (SHAMROCC)
Supplemental Material for Stroke Hospitalization Administration & Monitoring: Routine or COVID-19 Care (SHAMROCC) by Timothé Langlois-Thérien, Michel Shamy, Brian Dewar, Tim Ramsay, Ronda Lun, Dylan Blacquière, Robert Fahed, Dar Dowlatshahi, Grant Stotts, and Célina Ducroux in The Neurohospitalist
Footnotes
Acknowledgements
I would like to thank co-authors Dr Ducroux and Dr Shamy for their ongoing support as well as Dr Ramsay and his team for their help with the statistical analysis.
Author Contributions
MS and CD researched literature and conceived the study. MS, CD, BD, TR and TLT were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. TLT wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
TLT
Ethical Statement
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request by a qualified investigator.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
