Abstract
In 2015, multiple randomized clinical trials showed an unparalleled treatment benefit of stent-retriever thrombectomy as compared to standard medical therapy for the treatment of a large artery occlusion causing acute ischemic stroke. A short time later, the HERMES collaborators presented the patient-level pooled analysis of five randomized clinical trials, establishing class 1, level of evidence A for stent-retriever thrombectomy, in combination with intravenous thrombolysis when indicated to treat ischemic stroke. In the years following, evidence continues to mount for expanded use of this therapy for a broader category of patients. The enabling technology that changed the tide to support endovascular treatment of acute ischemic stroke is the stent-retriever. This review summarizes the history of intra-arterial treatment of stroke, introduces the biomechanics of embolus extraction with stent-retrievers, describes technical aspects of the intervention, provides a description of hemodynamic implications of stent-retriever embolectomy, and proposes future directions for a more comprehensive, multi-modal endovascular approach for the treatment of acute ischemic stroke.
A brief history of endovascular treatment of acute ischemic stroke
Endovascular intervention to treat large vessel occlusions (LVO) causing an acute ischemic stroke began in 1983, with the report from Aachen, Germany, describing five patients suffering posterior circulation LVOs who were treated with microcatheter directed, local fibrinolysis using streptokinase. 1 In this early experience, three of five patients realized successful recanalization with clinical improvement. Using neurointerventional techniques to deliver local fibrinolytic agents culminated with the successful Prolyse in Acute Cerebral Thromboembolism II (PROACT) trial, where patients with an angiographically confirmed middle cerebral artery (MCA) occlusion were randomized to receive intra-arterial recombinant prourokinase (r-proUK) with systemic heparinization or only heparin. Patients receiving local fibrinolysis with r-proUK were more likely than the control group to recanalize (66% vs. 18%, P < 0.001) and have good clinical outcome after 90-days (40% vs. 25%, P < 0.04) with no difference in mortality. 2 The risk of symptomatic intracranial hemorrhage within the first 24 h was 10% in the patients treated with intra-arterial fibrinolysis, versus only 2% in the control group. Nearly a decade later, the Middle Cerebral Artery Embolism Local Fibrinolytic Intervention Trial (MELT) in Japan showed that intra-arterial delivery of urokinase (UK) failed to meet the primary endpoint of improved functional independence (modified Rankin Scale (mRS) ≤ 2) after 90 days. 3 However, the planned secondary endpoint, namely excellent outcome at 90 days (mRS≤ 1), was achieved in more patients with catheter-directed UK as compared with the control group (OR:2.46; 95%CI: 1.09–5.54).
These data taken together pointed to the potential advantage of early restoration of blood flow to the ischemic brain by local administration of fibrinolytic agents leading to improved clinical outcomes, often at the increased risk of brain hemorrhage. A solution therefore emerged to mechanically remove the offending embolus without using fibrinolytic agents that may increase hemorrhage risk. A number of devices attempted to realize this goal, with varying levels of early success (reviewed in Nesbit et al. 4 ). Professor Pierre Gobin, at the time working at UCLA, designed the first FDA-cleared retriever, the Mechanical Embolus Removal in Cerebral Ischemia (MERCI, Concentric Medical, Mountainview, CA)) device. 5 This corkscrew-shaped retriever was delivered via microcatheter into the embolus with the goal to remove the retriever with entrapped clot (Figure 1). This was a landmark concept, not only in the mechanical extraction of the embolus but also in achieving FDA 510-K clearance by arguing substantial equivalence with well-established snares designed to remove foreign bodies (and thus the embolus was a foreign body since it did not form in situ) (reviewed in Smith and Furlan 6 ). The pooled analysis of the MERCI and Multi MERCI studies showed that in patients in whom successful recanalization was achieved, nearly half achieved independent outcome after 90 days as compared to only 6% of non-recanalized patients. 7 Moreover, successful recanalization led to a 50% reduction in mortality.
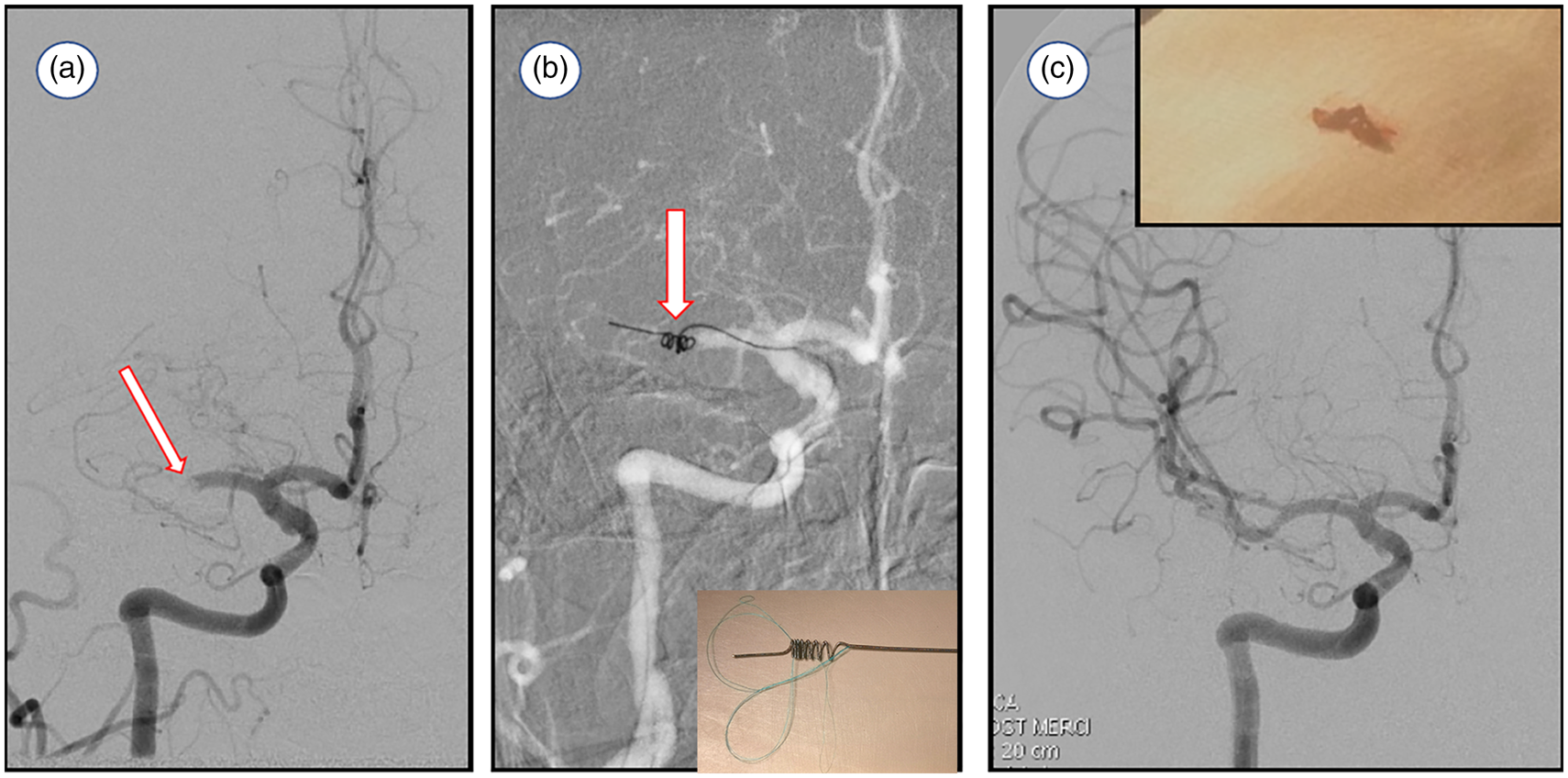
(a) Digital subtraction angiography (DSA, frontal projection) of the right internal carotid artery demonstrates complete occlusion of the mid-M1 segment of the right middle cerebral artery (arrow). (b) Thrombectomy using a Merci retriever with the device deployed at the occlusion site (arrow, inset shows the device). (c) Control DSA after device retrieval shows complete reperfusion of the right middle cerebral artery territory. The retrieved embolus is shown in the inset.
In 2008, aspiration thrombectomy was cleared by the FDA. Originally, this procedure entailed placing a catheter proximal to the clot and using an attached vacuum pump to aspirate the clot. 8 The original system included a separator, a small wire device delivered through the aspiration catheter that would debulk the embolus and unclog the catheter allowing removal of the clot in small pieces. The Penumbra Pivotal trial 9 showed that this technology led to high rates of recanalization at the site of primary occlusion; however, functional independence was only achieved in 25% of patients, which was similar to the control group in the PROACT study. It is important to note that the imaging outcome measure used in this study was a primary target thrombolysis in myocardial infarction score which did not evaluate downstream perfusion. Clot maceration with the separator device and subsequent distal emboli10,11 raised concerns regarding successful primary lesion recanalization as it may not lead to parenchymal reperfusion. Regrettably, in 2013, multiple randomized controlled trials that included retrievers and aspiration thrombectomy systems failed to demonstrate a therapeutic effect over standard medical therapy.12–14
Concurrently, a new generation of closed-cell, retrievable stents had entered the US market around 2006 with the intention to support detachable coils in wide-necked aneurysms. Unlike the previous generation of open-cell stents, these could be deployed and resheathed within the microcatheter; and, as experience grew, operators noted that these stents could be deployed distal to the aneurysm neck and pulled back in the expanded state for accurate placement and without clinical sequelae. These devices were introduced in Europe many years earlier and Henkes et al.15 theorized as early as 2003 that self-expanding stents “might permit thrombus or foreign body retrieval.” During an animal experiment, Wakhloo and Gounis16 noted thrombosis of a pig common carotid artery during the coiling of a surgically created sidewall aneurysm. At that time, a new closed-cell stent had just been introduced and was used to extract the clot from the animal by withdrawing the expanded stent along with entrapped clot, resulting in complete restoration of blood flow. A group at the Cleveland Clinic used the same intracranial stent as a temporary endovascular bypass to create a flow channel within the embolus and performed thrombolysis in a patient with an MCA occlusion. 17 Around the same time in March, 2008, the Solitaire stent approved in Europe as an aneurysm bridging device, was used as a thrombectomy device to recanalize an occluded right MCA since there were no other retrieval devices available to the operator. 18 The first case series using the Solitaire device for thrombectomy came shortly thereafter, and demonstrated in 20 patients that stent-retriever thrombectomy was technically successful in recanalization of LVOs in 90% of the cases with 45% of patients achieving functional independence after 90 days. 19
Two randomized clinical trials (stent-retriever versus the MERCI retriever) showed that stent-retrievers, the Solitaire (Medtronic, Irvine CA) and Trevo (Stryker, Fremont CA) were safe and effective to perform thrombectomy in LVOs.20,21 Finally in 2015, landmark randomized clinical trials concluded that stent-retriever thrombectomy with or without intravenous thrombolysis (administered when indicated) was better than standard medical treatment with or without intravenous thrombolysis.22–25 Stent-retrievers were the first medical devices to be indicated as a treatment of acute ischemic stroke to reduce disability in patients, rather than just being cleared as a tool for embolectomy. The pooled analysis of these trials included more than 1200 patients, and showed the powerful treatment effect of stent-retriever thrombectomy as compared to standard medical therapy in patients with a stroke caused by an LVO treated within 12 h of symptom onset (adjusted cOR 2.49, 95%CI 1.76–3.53). 26 The number needed to treat with stent-retriever thrombectomy to reduce patient disability by at least one point on the primary outcome measure, mRS, was only 2.6, that compares favorably to the most effective treatments in medicine overall. For instance, intravenous recombinant tissue plasminogen activator (r-tPA) administration between 91 and 180 min from stroke onset has a number needed to treat for benefit of 4.3 patients, that increases to 19.3 with treatment between 271 and 360 min from onset. 27 Recently, treatment benefit of stent-retriever thrombectomy in selected patients presenting within 24 h of time last known well was confirmed.28,29 With the remarkable treatment effect of stent-retriever thrombectomy now established, the question of over selection is being tested in a number of on-going trials to explore if patients with large infarct cores at time of baseline imaging, or those presenting with low National Institute Stroke Scale but with a confirmed LVO may also benefit from thrombectomy. This review seeks to explain the biomechanics of stent-retriever thrombectomy with a focus on the ultimate goal of these devices, which is restoration of normal intracranial hemodynamics.
Stent-retrievers
Stent-retrievers are constructed from nickel-titanium alloys (Nitinol), a common material for medical devices particularly in neurovascular and peripheral vascular applications. 30 This alloy has several important features that benefit devices intended for deployment within the cerebrovascular anatomy, and specifically for the application of stent-retriever thrombectomy. The key advantage of Nitinol is superelasticity, meaning nearly an order of magnitude increase in strain as compared to conventional metal alloys while remaining elastic (the material can return to the original shape when the stress is removed). A simple example of a material that is not superelastic is a wire coat hanger, typically made of steel, which when slightly bent can no longer return to its original shape (e.g. plastic deformation). This is the case for balloon-mounted coronary stents, where the balloon is used to plastically deform the stent into its expanded state propping up the coronary artery. On the other hand, a nitinol wire can endure large bending, but still return to the original shape when the stress is removed. Superelasticity of Nitinol exists due to phase transformations that take place in the crystal structure induced by stress, allowing the material to change its shape during an applied stress and yet recover it when the stress is removed. In addition, this phase transformation can be thermally induced, known as the shape-memory effect. The ability to restore the original structure when heated above the transformation temperature is very useful in biomedical applications since the body core temperature is known and the transformation temperature can be set below body temperature and yet above room temperature. These properties allow a porous, laser cut Nitinol tube (self-expanding stent) to be delivered through a small microcatheter and yet expand to its original structure which is many times larger in diameter than the delivery system (Figure 2). These material properties are leveraged for self-expanding Nitinol stents to be used in the intracranial circulation to bridge the neck of aneurysms31,32 or to resist elastic recoil when treating intracranial atherosclerosis.33,34
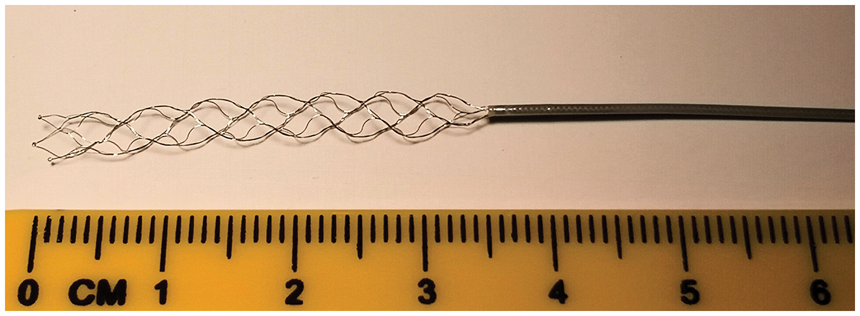
A stent-retriever is a self-expanding device made of nitinol. After deployment via microcatheter, the superelastic stent-retriever can expand to the unconstrained size.
Another unique property of nitinol is stress hysteresis (Figure 3), 30 which again differentiates it from most engineering materials that follow the same linear stress–strain response during loading and unloading. However, the stress–strain curve of nitinol is non-linear, and the stress response is directionally dependent on the strain, meaning the loading curve is different than that of unloading. When the stent is released from its delivery system into an artery which is smaller than the unconstrained shape of the device, it exerts a small chronic outward force trying to return to its stress-free state. 35 This is the force that is exerted outward against the vessel wall (e.g. the Solitaire stent exerts approximately 0.01 N/mm, force per unit length) 36 . The stent’s resistance against elastic recoil of the artery, or an external force such as embolic coils the stent holds into an aneurysm, is termed the radial resistive force, which is much stiffer than the chronic outward force. In other words, the stent exerts a gentle outward force against the artery but is notably stiffer to resist compressive forces.
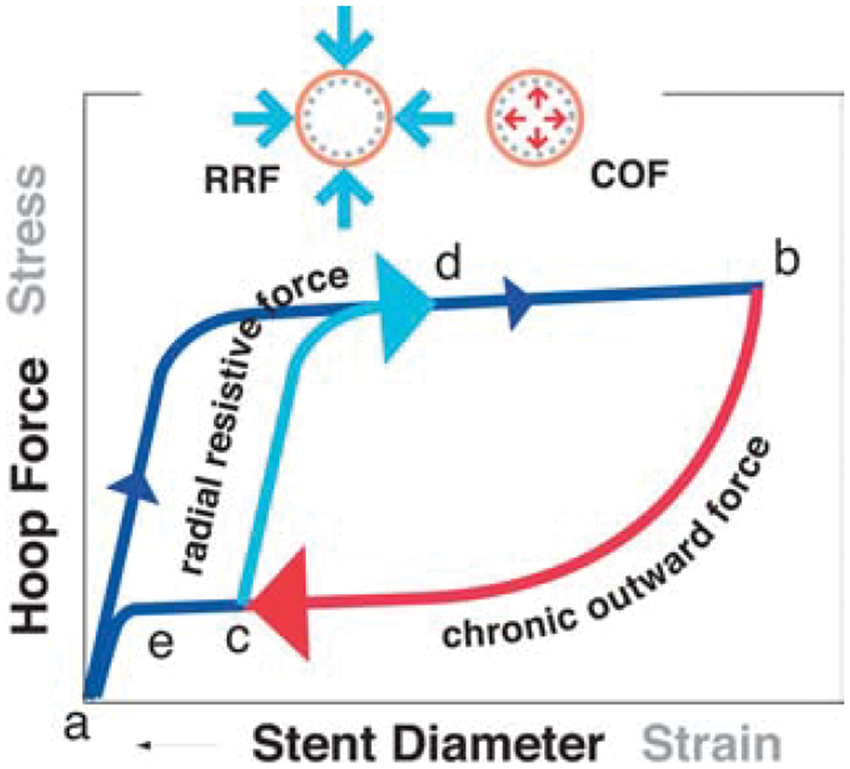
The stress–strain curve resembling a nitinol stent-retriever has many unique features including hysteresis where the loading component (a to b) is different than the unloading (b to c). During loading, for example crimping the stent into a microcatheter, the force resisting this load is called the radial resistive force (RRI). However, during unloading where the stent is deployed into the clot, it exerts a continuous outward pressure called chronic outward force (COF). Reprinted with permission from Springer Nature, European Radiology. 30
These properties taken together offer significant advantages for optimal thrombectomy systems. The embolus is first passed with a microcatheter, with an inner diameter as small as 0.4 mm. Through this small tube, the stent-retriever is delivered into the thrombus by unsheathing it from the microcatheter and allowing it to expand into the clot. Due to both thermal and stress-induced phase transformation, the device attempts to return to its original shape (quite a common device selection is 4 mm unconstrained outer diameter). As it expands, it engages the clot by exerting the chronic outward force, with the struts slicing through the clot much like a cheese grater. 37 Most stent-retrievers include in the instructions for use that the device should be deployed for 3 to 5 min prior to retrieval, since during that time more clot becomes integrated into the device due to the chronic outward force. 37 During this time, a central flow channel is created through the obstruction, 38 restoring between a third and half of the normal blood flow through the MCA. 39 The therapeutic benefit of this early partial flow restoration as compared to the overall treatment effect of the thrombectomy is not known. After the waiting period, the device is withdrawn in its expanded state and, while under tension, the device elongates leading to conformational changes in the cells (the empty spaces between the struts) of the stent. 40 This effectively anchors the embolus to the surface of the device, since there are numerous cells each grabbing hold to the penetrating clot that has entered the body of the stent. The stent-retriever along with ensnared clot is ultimately withdrawn into a large catheter proximally, typically while aspiration is applied, and then withdrawn completely from the body (Figure 4, Supplemental Movie 1). Recent data in animal models 41 and patients 42 show that removal of the embolus from the MCA leads to improvement in blood flow of the affected hemisphere, meaning that recanalization with a stent-retriever leads to parenchymal reperfusion.
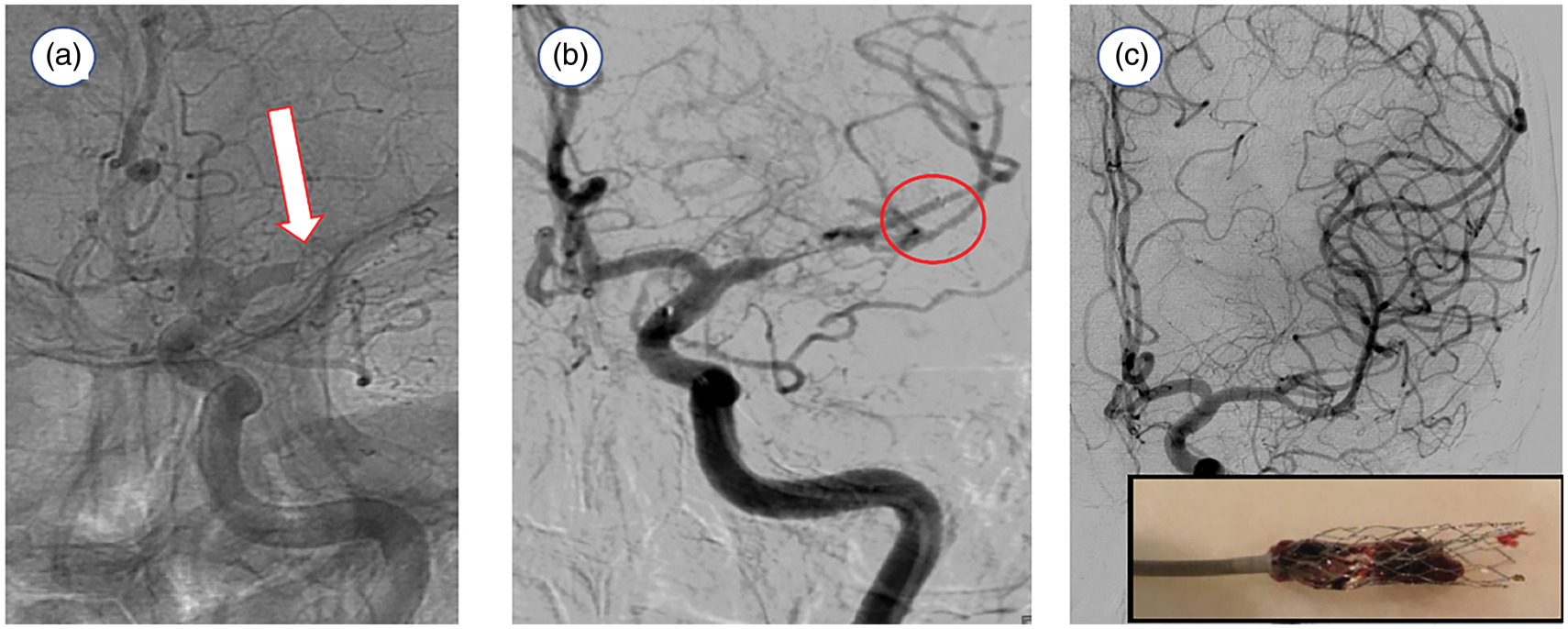
(a) Cerebral angiogram (frontal projection) shows a left middle cerebral artery occlusion (arrow). (b) The stent-retriever was deployed within the left middle cerebral artery (circle=distal end of device), that immediately restores partial blood flow to the ischemic tissue. (c) Control angiography demonstrates complete recanalization of the left middle cerebral artery. The captured thrombus is shown in the inset.
Current thrombectomy techniques
Current thrombectomy techniques can be divided into three main groups: (1) use of a large bore aspiration catheter, (2) use of a stent-retriever or (3) a combined approach using an aspiration catheter together with a stent-retriever.
Aspiration thrombectomy is a simple concept that today entails delivering the largest possible catheter to the face of the embolus and applying suction, either with a syringe or a vacuum pump, to remove the clot from the artery. There are two concepts for embolus extraction using aspiration, the first being complete ingestion of the clot into the catheter or engagement of the proximal aspect of the clot that is corked into the tip of the catheter and then extracted by removal of the aspiration catheter (Figure 5). First approaches using manual aspiration through large bore catheters following failed mechanical thrombectomy were described in 2012 and 2013.43,44 The key concept presented was to use limited number of passes with a retriever device before switching to aspiration thrombectomy, thereby reducing the overall number of passes to have a successful recanalization. The authors concluded that manual aspiration thrombectomy was associated with favorable recanalization rates and clinical outcomes. Similarly, Kang et al.44,45 showed that combining two mechanical thrombectomy techniques, stent-retriever and forced arterial suction, was associated with a trend of higher successful vessel recanalization. Neither of the groups observed an increase in procedure-related complications, including incidence of intraparenchymal hemorrhage.

An aspiration catheter partially ingests a clot, with a large portion of the clot remaining outside the catheter. When the operator extracts the device-clot ensemble under continuous aspiration, fragmentation can occur if the force exerted exceeds the cohesive internal forces of the clot.
The enabling technology for aspiration thrombectomy today is the manufacturing of large bore aspiration catheters of extremely complex design that enable safe navigation to the middle cerebral artery.46,47 A direct aspiration first pass technique (ADAPT) 48 uses a large bore aspiration catheter which is positioned at the face of the thrombus. Aspiration is then applied until the system becomes occlusive, indicating that the proximal portion of the clot has been successfully sucked into the catheter tip. The aspiration catheter is subsequently removed while aspiration is maintained. This ensures that the thrombus remains engaged in the tip of the catheter during removal of the system. Recent randomized trials of the aspiration approach as compared to gold-standard stent-retriever thrombectomy show comparable results in terms of successful recanalization 49 and good clinical outcomes. 50 The key advantage of the ADAPT technique is relative simplicity of the procedure and less cost; however, between one-fifth and one-third of cases required the use of a stent-retriever to achieve recanalization.
Mechanical thrombectomy with a stent-retriever is used to engage the clot by deploying the stent-retriever at the location of the thrombus in the occluded artery. The device is then allowed to intercalate with the clot for a few minutes, attempting to engage the thrombus in the stent struts, before the system is removed. There are numerous variations of technique when deploying a stent-retriever, including the combined use of a large bore aspiration catheter proximally and a stent-retriever deployed distally. In the Solumbra technique,51,52 the stent-retriever is positioned centrally within the clot. The aspiration catheter is slowly advanced to the face of the clot and the stent-retriever is then completely retracted back into the catheter under continuous aspiration. However, this technique can lead to large amounts of distal emboli due to shearing from the outer surface of the stent-retriever during entry into the aspiration catheter. Alternatively, in the Aspiration-Retriever Technique for Stroke (ARTS), 53 the stent-retriever is placed across the thrombus while the aspiration catheter is slightly advanced until resistance is felt, essentially pinching the clot between the stent and the aspiration device. The entire assembly is then locked and carefully removed under continuous aspiration (Figure 6). Other key concepts to this technique is the use of a balloon guide catheter in the cervical internal carotid artery that temporarily arrests antegrade flow during the thrombectomy, as well as microcatheter removal after stent deployment to augment the lumen available in the aspiration catheter for suction. 54 Small variations in relative placement of the devices may impact the procedural outcome. In the SAVE technique, the thrombectomy device is deployed slightly beyond the thrombus with only the proximal 1/3rd of the stent-retriever engaging with the clot. 55 The aspiration catheter is then advanced to the face of the thrombus and aspiration is started. In a next step, the stent-retriever is slightly pulled back, while the aspiration catheter is slowly advanced until a wedge position is reached. The system is then locked and slowly removed.
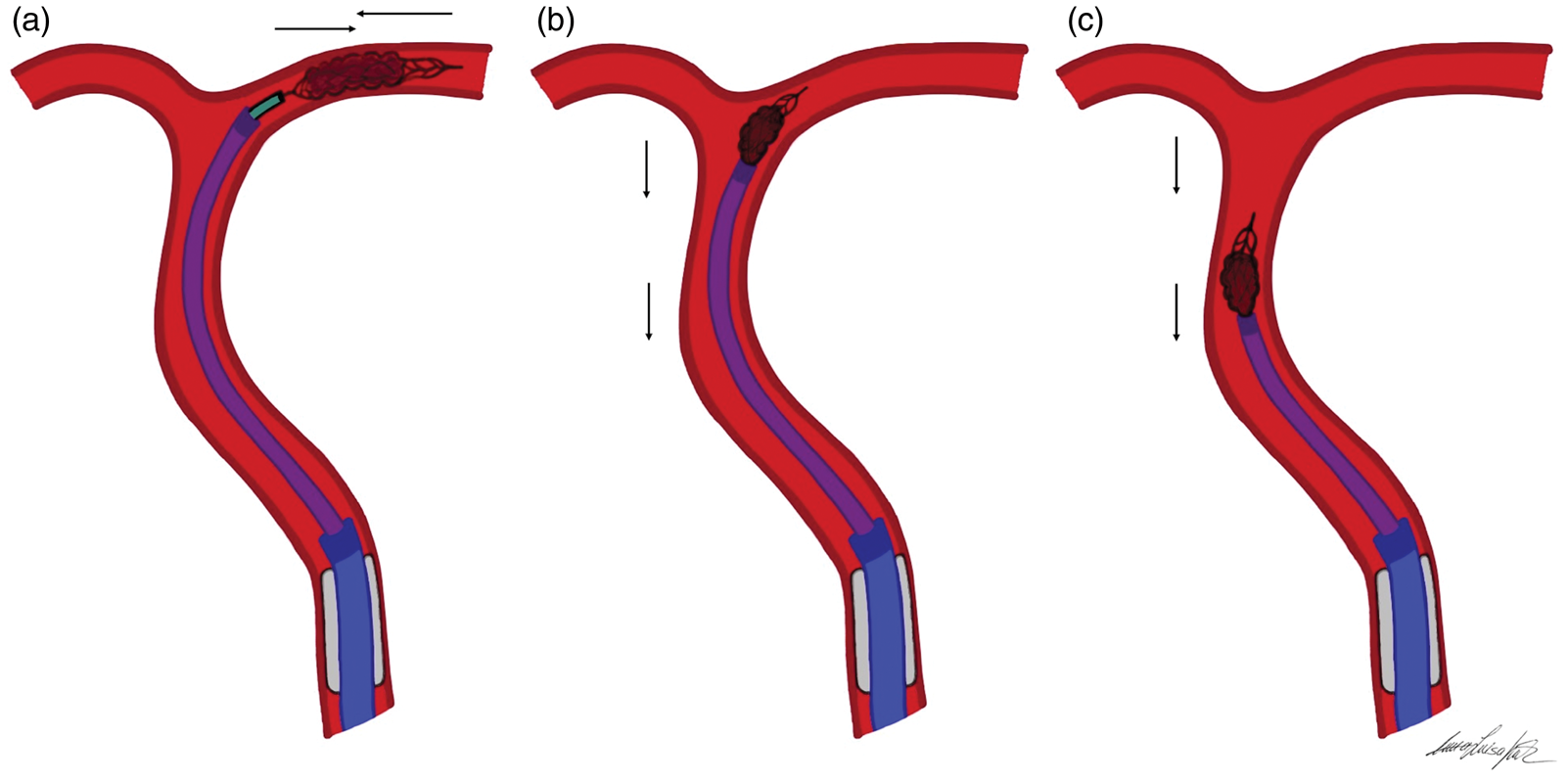
Illustration of the Aspiration-Retriever-Technique for ischemic Stroke (ARTS) in an M1/middle cerebral artery (MCA) occlusion. (a) The aspiration catheter is placed to the face of the clot in the MCA. (b and c) A stent-retriever is then navigated across the clot and allowed to intercalate within the thrombus. Before activating the aspiration pump apparatus, the balloon guide catheter is inflated. Once resistance is felt upon retraction of the stent-retriever, the entire assembly of aspiration catheter and the partially resheathed stent-retriever is locked and carefully withdrawn under continues aspiration with additional flow arrest.
Distal emboli and vascular trauma
An early proposed hypothesis for initial failures in mechanical thrombectomy trials was the concept that recanalization of the MCA does not necessarily lead to reperfusion of the brain parenchyma, also known as the no re-flow phenomenon.
56
Although originally described in the setting of a global cerebral ischemia model,
56
this has been well-documented in the cardiac literature where despite successful percutaneous coronary artery intervention, blood flow to the microvasculature of the myocardium remains impeded (reviewed in Rezkalla and Kloner).
57
One of the mechanisms for the no-reflow phenomenon is atherosclerotic debris, blood clots, or platelet plugs released into the microcirculation. This is a concern also for clot extraction from the brain arteries, where any endovascular manipulation of an embolus threatens periprocedural release of distal emboli. This has been extensively studied in simulated use experiments that deploy patient-specific vascular phantoms and clinically relevant embolus analogs.
10
During these experiments, we learned that clot fragments are lost during discrete processes throughout the procedures:
Gaining access to the clot, which for most current thrombectomy technologies requires crossing the embolus with wires and microcatheters.
58
Initial activation of the retraction, which is even more common for aspiration thrombectomy when the clot is corked in the tip of the catheter and all force is applied to the proximal aspect of the embolus relying solely on adhesion of the embolus to resist fracture (Supplementary Movie 2).
59
As the embolus is extracted from the MCA, as the device-clot ensemble turns around the tip of the internal carotid artery, clot can be snowplowed into the anterior cerebral artery causing distal emboli to a new territory. This is likely more common with stent-retrievers, since most of the clot resides outside of the device.37,60 Mobilization of the device through the tortuous path of the internal carotid artery, and in particular the siphon, can lead to rapid accelerations of the device despite constant and gentle retraction by the operator, leading to loss of clot fragments from the device. The most important cause of clot fragmentation is the eventual entrance into a receiving catheter, where clots corked at the end of an aspiration catheter or caught in the cells of a stent-retriever (but with most of the clot volume on the outside of the device) can be sheared off by the receiving conduit.
Balloon guide catheters (BGC) offer the opportunity to dramatically reduce distal emboli by addressing the largest cause of clot fragmentation and release distally. These are large catheters used outside of the brain, typically in the cervical internal carotid artery. During the thrombectomy, a temporary occlusion balloon just proximal to the tip of the catheter is inflated to arrest antegrade flow (Figure 7). When the thrombectomy device enters the BGC, clot fragments stripped off the device do not migrate due to local flow obstruction by the balloon and backflow through the BGC allows capture of these particles and prevention of distal emboli. Quantitative reduction of distal emboli for BGC-protected thrombectomy has been shown on the bench. 39 Consequently, numerous clinical studies have confirmed that BGC-protected thrombectomy leads to higher first-pass success, reduced procedure time, increased rates of successful thrombectomy, and, most importantly, improvement in clinical outcomes (Table 1).61–65 Despite deep evidence, BGCs are not routinely used during thrombectomy likely due to the navigation performance that is simplified with other access catheters. Other technologies that simplify access yet achieve either embolus capture 66 or local flow control 67 are being investigated to replace BGCs, but still offer the benefit of embolic protection.
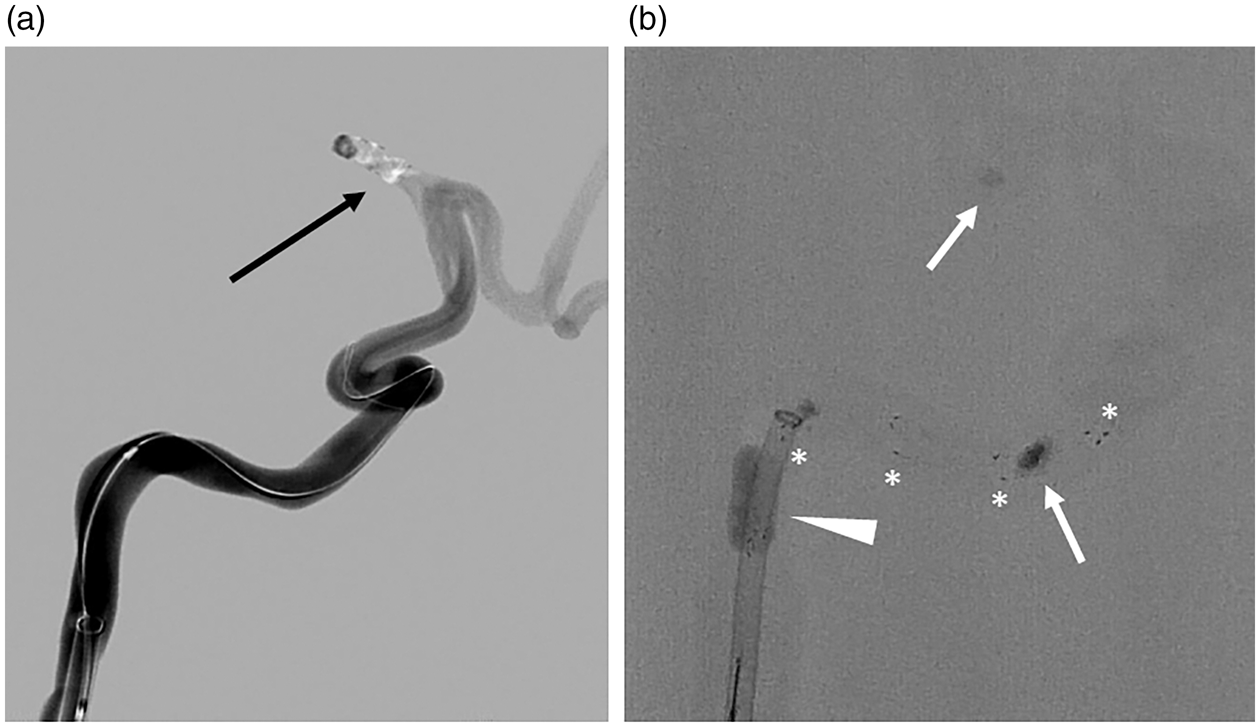
(a) Angiogram in a patient-specific in vitro model shows the site of a middle cerebral artery occlusion (arrow). (b) Simulated stent-retriever thrombectomy shows embolus extraction under proximal balloon protection (arrowhead) where afferent flow is arrested for the removal of the clot (arrows). The stent-retriever has radiopaque markers (*) along the stent for accurate placement.
Clinical impact of balloon guide catheter (BGC) protected thrombectomy.
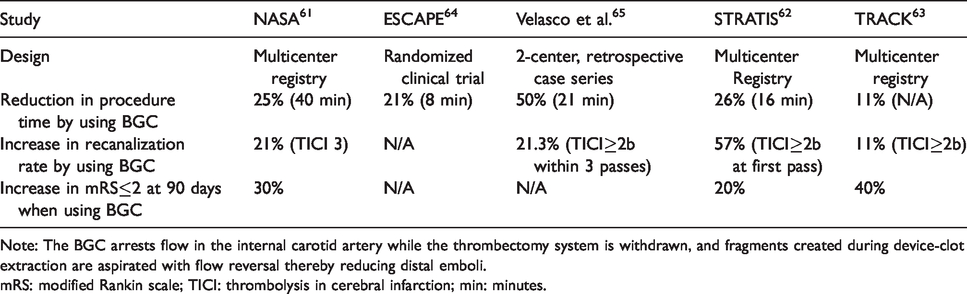
Note: The BGC arrests flow in the internal carotid artery while the thrombectomy system is withdrawn, and fragments created during device-clot extraction are aspirated with flow reversal thereby reducing distal emboli.
mRS: modified Rankin scale; TICI: thrombolysis in cerebral infarction; min: minutes.
Preclinical models of vascular obstruction removed by mechanical thrombectomy have shown acute vascular trauma that includes endothelial cell denudation, tears to the internal elastic lamina, medial edema and mural thrombosis.68–70 The extent of injury increases with the number of thrombectomy passes. 71 However, over time the vessels heal and show only minimal intimal thickening. 72 One study shows that even microcatheterization within the cerebral arteries leads to intimal trauma along the path the microcatheter takes. 73 Even before the results of the overwhelming positive trials of stent-retriever thrombectomy were available, there were clinical reports about post-thrombectomy arterial wall thickening and vessel-wall enhancement on MRI performed within two weeks of thrombectomy, both of which were larger in patients with stent-retriever thrombectomy as compared to those receiving only medical therapy. 74 Vessel wall imaging and permeability imaging within two days of treatment showed that patients receiving both stent-retriever thrombectomy and intravenous r-tPA were more likely to have vessel wall contrast enhancement and blood–brain barrier disruption as compared to patients only receiving r-tPA. 75 Poor clinical outcomes were associated with vessel wall enhancement, and both wall enhancement and increased permeability were correlated to final infarct volume. Stent-retriever thrombectomy shows that patients achieve high rates of good clinical outcome after 90-days 76 and there is evidence that this benefit over standard medical therapy persists for up to one year. 77 This may suggest, as do preclinical models, that the artery remodels and the injury is resolved. However, vascular imaging is not routinely acquired during long-term follow-up and vascular remodeling process in patients following stent-retriever thrombectomy remains largely unknown.
Hemodynamics and mechanical thrombectomy
The debate to determine which imaging surrogate biomarkers should be used to define patients that benefit from intervention is the source of lively discussion and perennial controversy. Of course, accurate perfusion imaging with PET or MRI would likely be preferred, but these imaging techniques are not ubiquitously available and, depending on local infrastructure, could lead to delays in a hyper acute emergency such as stroke. These limitations have led to a widespread adoption of CT-based techniques in acute stroke imaging. 78
The presence or absence of a robust collateral circulation, a prognostic factor in patients with acute ischemic stroke, is relatively quickly and simply assessed using CT angiography. The ability to quickly recruit blood flow via a collateral network to the affected brain area is crucial to delay and, in some cases, even prevent permanent injury. Multiphase CT angiography (CTA) has emerged as a quick and easy-to-use imaging tool that helps make clinical decisions for patients with acute ischemic stroke. 79 Multiphase CTA acquires images at three different time points after the administration of contrast: peak arterial phase, equilibrium/venous phase and delayed venous phase.
One of the most commonly used CT-angiography-based collateral vessel scoring systems is the mCTA Collateral Score, which ranges from 0 (no collateral vessels) to 5 (symmetric appearance of collateral vessels). 80 Other scoring systems include the American Society of Interventional and Therapeutic Neuroradiology/Society of Interventional Radiology (ASITN/SIR) grading system, 81 the Tan collateral scoring system, 82 and the Christoforidis score. 83 The latter has been shown to correlate poorly with the final infarct volume. The ASPECTS and ASITN/SIR grading systems have shown good correlation with early infarct core and mismatch as well as excellent cross correlation. 84
Success of mechanical thrombectomy and size of infarct volume depend on the patients’ collateral network.85,86 Patients with better collaterals showed lower NIHSS and DWI lesion volume on admission, better response to intravenous tPA administration, larger volumes of salvageable tissue at risk, a smaller final infarct core volume, and better functional neurologic outcome.79,87–91 Patients with good collaterals had larger areas of milder perfusion delay than those with poor collaterals (P = 0.005). 87 On the other hand, patients with a poor collateral network showed infarct growth, larger final infarct volume and associated worse functional outcome.87,92,93 Kimmel et al.92 found that higher infarct volume was associated with lower odds of achieving good functional outcome, defined as mRS 0–2 at 90 days (P = 0.049; OR: 0.96, 95% CI: 0.918–0.999).
Therefore, mechanical thrombectomy for eligible patients with poor collaterals should not be delayed as these patients have no vascular reserve and develop larger infarcts faster. Of note, about 36–47% of stroke patients with intermediate or good collaterals who were successfully treated in a 3 to 6-h time window in recent stroke landmark trials did not achieve functional independence. 76 This highlights the fact that the simple presence of a collateral network is not sufficient to predict good outcome. The quality of a collateral network lies with its ability to maintain adequate perfusion to the affected brain parenchyma over a prolonged period. Patients whose collaterals are rapidly failing are considered ‘fast progressors:’ these patients are very sensitive to the duration of ischemia and benefit from fast reperfusion in an early time window. 94 On the other hand, stroke patients whose collateral network is exceptionally robust and well developed may benefit from endovascular treatment well beyond 8 h after symptom onset.28,29,95
Prognostication of patient outcome after thrombectomy may also be feasible using a novel 3 T time-of-flight (TOF) MRA vascular imaging finding. Postprocedural visibility and dilation of the ipsilateral lenticulostriate arteries (LSAs) based on a 0 to 3 grading scale suggest an overall more favorable neurologic and functional outcome in patients with prominent to markedly enlarged LSAs (grades 2 and 3). The authors consider the observed vasodilation of the LSAs a beneficial response to the ischemic insult that may indicate increased metabolic demands needed for tissue repair. 96
Success of a mechanical thrombectomy procedure can be measured using the Thrombolysis in Cerebral Infarction (TICI) scale, assessed using DSA imaging and considered the most widely accepted scoring system to grade recanalizing therapies’ technical success.81,97,98 This system grades postprocedural tissue reperfusion in the territory of the vessel in question according to a five-step scale, with TICI 0 meaning no reperfusion and TICI 3 meaning complete reperfusion. TICI 2 describes incomplete reperfusion, most likely due to persistent occlusion of peripheral branches and residual perfusion deficit in ≤1/3 (TICI 2b) or >1/3 of the territory (TICI 2a). By convention, grades 2b and 3 are considered reliable predictors of good functional outcome and, by default, successful recanalization. 99 However, a retrospective study has shown that while the TICI 3 versus TICI 2b nuances on imaging are negligible, there is a statistically significant improvement in TICI 3 patients versus TICI 2b patients in terms of neurological outcome and hospital stay in a sample size of 262 patients (10 days vs. 12 days, P = 0.014). 98 In another retrospective study by Chamorro et al.,100 a TICI 3 score not only resulted in a better mRS score than TICI 2b (OR 2.018, P = 0.04), but also displayed reduced infarct growth as interrogated by multimodal MRI (P = 0.002). These studies illustrate the fact that a “successful recanalization” does not necessarily mean optimal results for the patient. Since optimizing functional outcome of patients should be the ultimate goal of any neurointerventional procedure, the first pass effect (FPE) can also be introduced into the picture as a predictor of favorable neurological outcome.101,102 FPE is defined as “achieving a complete recanalization with a single thrombectomy device pass.”101,102 Data from the North American Solitaire Acute Stroke Registry database show that FPE was achieved in 89/354 acute ischemic stroke patients and was an independent predictor of good clinical outcome as defined by mRS ≤2 (P = 0.013) after 90 days. 102 In brief, complete embolectomy with no distal clot fragmentation leading to full flow restoration achieved quickly (first pass) is associated with the best clinical outcomes.
Studies identifying patients eligible for thrombectomy in the late time window (up to 24 h since last known well), did use CT perfusion imaging to establish surrogate imaging biomarkers of infarct core.28,29 It remains to be determined if using a mCTA – Alberta Stroke Program Early CT Score (ASPECTS) will be sufficient to identify candidates for thrombectomy after 12 h from stroke onset.
Hemodynamic targets are different before and after mechanical thrombectomy. The current blood pressure goal in patients with acute ischemic stroke is based on the exclusion criteria in the pivotal Phase III trials of recombinant human tissue plasminogen activator 103 with systolic blood pressure (SBP) below 185 mmHg and diastolic blood pressure below 110 mmHg. An overall higher pre-procedural blood pressure is likely a physiologic response and is necessary to maintain penumbra viability. Animal models have shown that blood flow in moderately under perfused tissue is dependent on the SBP. 104 Studies on blood pressure levels during the acute phase of ischemic stroke have shown that any relevant drop in blood pressure is associated with poor prognosis. 105 The same was observed for patients undergoing endovascular therapy with either conscious sedation 106 or general anesthesia.107,108 Lowhagen et al.107 observed that a fall in mean arterial blood pressure of more than 40% was an independent predictor for poor neurological outcome (P = 0.032).
During the post-procedure recovery period, it is crucial to maintain a steady blood pressure as fluctuations in SBP values or overall increased blood pressure variability have been found to result in a higher rate of hemorrhagic complications and worse patient outcome.109–111 Goyal et al.110 found that a 10 mmHg increase in maximum SBP during the first 24 h post endovascular therapy was independently associated with a lower likelihood of three-month functional independence (P = 0.001; OR: 0.70; 95% CI: 0.56–0.87) and a higher odds ratio for three-month mortality (OR: 1.49; 95% CI: 1.18–1.88). In addition, a blood pressure goal of less than 160/90 mmHg during the first 24 h after endovascular therapy was independently associated with a lower likelihood of three-month mortality (P = 0.010; OR: 0.08; 95% CI; 0.01–0.54) when compared to permissive hypertension. A recent multicenter study 112 from 10 comprehensive stroke centers evaluating 1245 stroke patients showed that a higher mean SBP, maximum SBP, and SBP range were associated with lower odds of achieving good functional outcome (P < 0.001, OR: 0.86; P < 0.001. OR: 0.9; and P = 0.003, OR: 0.91). When compared with the reference group with blood pressure between 121 and 140 mmHg, patients in the blood pressure groups 141 to 160 mmHg and greater than 160 mmHg had 43% and 66% lower odds of achieving good outcome at 90 days, respectively. Higher blood pressure post mechanical thrombectomy with successful revascularization was also associated with a higher risk of symptomatic intracranial hemorrhage, patient mortality and need for hemicraniectomy.
The future
Mechanical thrombectomy has revolutionized acute ischemic stroke treatment. The first-generation stent-retrievers are easy to use and achieve high rates of reperfusion. Nevertheless, there is still a subgroup of patients in whom the clot cannot be completely removed. About 20% of emboli are resistant to modern retrieval approaches, either requiring multiple attempts of thrombectomy or completely intractable to recanalization. 113 Current research into identifying thrombus compositions by histopathology and/or imaging is being conducted in hopes of developing a more holistic and personalized approach to stroke treatment.114–119 These data may provide insights to predict which patients will benefit from endovascular therapy, but it does not solve the need for newer technologies and devices to tackle these hard-to-remove, resistant clots. Future research will focus on the interactions of the clot with the device and the vessel wall. It is essential for the next generation technology to be simple and reliable to use, leading to successful clot extraction within minutes of arterial access. One example is even larger bore catheters that can be navigated to the MCA for complete clot ingestion (Supplemental Movie 3), for hard-to-remove clots, 120 or adjunctive devices for stent-retrievers that encapsulate the clot for fragment free removal.66,67 Failed mechanical thrombectomy may also be linked to in situ thrombosis with intracranial atherosclerosis or underlying dissection. Regrettably, emergent large vessel occlusion caused by in situ thrombosis due to intracranial artery disease is not known prior to stent-retriever thrombectomy and may not be the best treatment option, as these lesions typically require bailout stenting due to persistent re-occlusion following thrombectomy. 121 Recently, a new intravascular imaging high-frequency optical coherence tomography (OCT) device has been described for specific application in the neurovasculature. 122 OCT is well known to characterize the details of coronary plaques, 123 and the ability to image the underlying pathology prior to performing thrombectomy or after the first-pass failure may allow optimized treatment in cases of atherosclerosis or dissection.
Also, in vivo and in vitro studies have shown that the use of aspiration catheters and stent-retrievers results in different degrees of vascular injuries.68,69,70,124–126 Some injuries can result in acute platelet activation with potential re-occlusion of the vessel and the possibilities for distal emboli. Another potential vascular injury described after stent-retriever thrombectomy is endothelial denudation 126 and endothelial cells have been found on the surface of stent-retrievers used in patients. 127 Neutrophil infiltration after endothelial denudation has been previously described and may offer a possible explanation for an inhibitory effect of anti-inflammatory therapies on neointimal growth. 128 Interestingly, stroke patients with a higher neutrophil to lymphocyte ratio were more likely to have a severe neurological deficit at discharge and a poorer functional outcome.129,130 With arterial access to perform the procedure, the potential of multi-modal stroke therapy exists; namely, to perform thrombectomy in concert with other locally delivered therapies that may further improve rates of good clinical outcomes. Examples include use of shear-driven nanoparticle release of thrombolytics in combination with stent-retrievers to reduce arterial trauma of thrombectomy, since the stent-retriever is re-sheathed rather than removed in its expanded state. 71 Intra-arterially administered medications already used in neurointerventional surgery may have neuroprotective affects, and potentially could further reduce vessel wall injury. 131 Periprocedural local hypothermia can be realized with arterial access to the affected brain territory: 132 a nonrandomized cohort study of 113 acute stroke patients showed a statistically significant decrease in final infarct volume (P = 0.038), as assessed by non-contrast CT, when comparing MT with intraarterial selective cooling infusion versus MT alone. 133 This cooling method involves infusing a 4°C aliquot of 0.9% saline by way of a microcatheter that would have been advanced beyond the culprit clot. After MT, the cold saline is infused again through the catheter. This procedure is safe and does not contribute in delaying the intervention.133,134
Perhaps the largest opportunity to improve clinical outcomes in stroke is to combine neuroprotection with mechanical thrombectomy. 135 The first randomized clinical trial combining neuroprotection with mechanical thrombectomy has recently been completed. 136 Over 1000 patients were randomized to receive placebo or nerinetide, an eicosapeptide that inhibits post-synaptic density protein 95, prior to receiving mechanical embolectomy. Although the trial failed to achieve its primary endpoint, namely good functional recovery after 90 days, subgroup analysis showed a benefit in patients that received nerinetide without prior intravenous thrombolysis. This unexpected result suggests in humans a drug–drug interaction with nerinetide and alteplase, where plasmin cleaves the amino acid sequences of nerinetide. Future studies of multimodal stroke therapy that combines neuroprotective or neuroreparative strategies with mechanical thrombectomy hold enormous promise for the optimal outcomes of stroke patients. Additionally, peri-procedural adjunctive therapies delivered locally represent a significant opportunity. Preclinical models can help in interrogating neuroprotectant toxicity profiles prior to human exposure, developing a comprehensive understanding of the precise mechanism of action of neuroprotectants, and the opportunity to study long-term neuroprotection, such as neurogenetic structural changes, in addition to short-term neuroprotective outcomes. 137 Over the last decades, numerous pharmacologic agents have shown promising results in pre-clinical studies. However, the success of the experimental findings has failed to translate into clinical benefit. Studies about citicoline, 138 cerebrolysin, 139 calcium channel blockers, 140 N-methyl-d-aspartate antagonists, 141 magnesium, 142 granulocyte stimulating factor (G-CSF), 143 human albumin, 144 NYX-059145 and more have failed to show improved clinical outcomes. A meta-analysis showed in-hospital statin use was associated with good functional outcome (1.31 [1.12–1.53]; P = 0.001), and decreased mortality (0.41 [0.29–0.58]; P < 0.001). In patients treated with thrombolysis, statins were associated with good functional outcome (1.44 [1.10–1.89]; P = 0.001), albeit with an increased risk of symptomatic hemorrhagic transformation (1.63 [1.04–2.56]; P = 0.035). 146 The possibility of selective delivery of these agents either before or after thrombectomy remains an exciting area of research.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X20916002 - Supplemental material for Biomechanics and hemodynamics of stent-retrievers
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X20916002 for Biomechanics and hemodynamics of stent-retrievers by Anna Luisa Kühn, Zeynep Vardar, Afif Kraitem, Robert M King, Vania Anagnostakou, Ajit S Puri and Matthew J Gounis in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: ASP has research grants from Cerenovus, Medtronic Neurovascular, and Stryker Neurovascular, holds stock in InNeuroCo and NTI. MJG has received research support from the National Institutes of Health (NIH), the United States – Israel Binational Science Foundation, Anaconda, Apic Bio, Arsenal Medical, Axovant, Cerenovus, Ceretrieve, Cook Medical, Galaxy LLC, Gentuity, Imperative Care, InNeuroCo, Insera, Magneto, Microvention, Medtronic Neurovascular, MIVI Neurosciences, Neuravi, Neurogami, Philips Healthcare, Progressive Neuro, Rapid Medical, Route 92 Medical, Stryker Neurovascular, Syntheon, and the Wyss Institute.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ASP has been a proctor on a fee-per-hour basis for Stryker Neurovascular, and Cerenovus. MJG is a consultant on a fee-per-hour basis for Cerenovus, Imperative Care, Medtronic Neurovascular, Mivi Neurosciences, Phenox, Route 92 Medical, and Stryker Neurovascular; and holds stock in Imperative Care, InNeuroCo and Neurogami.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
