Abstract
Background
To this date, whether to administer intravenous thrombolysis (IVT) prior to endovascular thrombectomy (EVT) for stroke patients still stirs some debate. We aimed to systematically update the evidence from randomized trials comparing EVT alone vs EVT with bridging IVT.
Methods
We searched MEDLINE, EMBASE, and the Cochrane Library to identify randomized controlled trials (RCTs) comparing EVT with or without IVT in patients presenting with stroke secondary to a large vessel occlusion. We conducted meta-analyses using random-effects models to compare functional independence, mortality, and symptomatic intracranial hemorrhage (sICH), between EVT and EVT with IVT. We assessed risk of bias using the Cochrane risk-of-bias tool and certainty of evidence for each outcome using the GRADE approach.
Results
Of 11,111 citations, we included 6 studies with a total of 2336 participants. We found low-certainty evidence of possibly a small decrease in the proportion of patients with functional independence (risk difference [RD] −2.0%, 95% CI −5.9% to 2.0%), low-certainty evidence that there is possibly a small increase in mortality (RD 1.0%, 95% CI −2.2% to 4.7%), and moderate-certainty evidence that there is probably a decrease in sICH (RD −1.0%, 95% CI −1.6% to .7%) for patients with EVT alone compared to EVT plus IVT, respectively.
Conclusion
Low-certainty evidence shows that there is possibly a small decrease in functional independence, low-certainty evidence shows that there is possibly a small increase in mortality, and moderate-certainty evidence that there is probably a decrease in sICH for patients with EVT alone compared to EVT plus IVT.
Introduction
Intravenous thrombolysis (IVT) has been a long-standing, evidence-based treatment approach for acute ischemic stroke.1,2 However, IVT typically needs to be delivered within 3-4.5 hours in most patients, has certain contraindications, may not provide adequate reperfusion, especially in patients with large vessel occlusions, and may even increase the risk of intracranial hemorrhage. 3 As such, trials were conducted to evaluate the use of endovascular therapy in patients with acute ischemic stroke. Endovascular therapy includes a variety of techniques, such as aspiration thrombectomy, stent-retriever technology, or intra-arterial thrombolysis. 4
Given the robust evidence supporting the use of endovascular therapy in patients with acute ischemic stroke secondary to a large vessel occlusion,5-14 several key factors need to be considered in the utilization of IVT and endovascular thrombectomy (EVT) in patients with large vessel occlusions: inadequate recanalization where IVT may not be effective enough in reopening large or proximal intracranial vessel occlusions – approximately 11% of patients with large vessel occlusions achieved successful recanalization with IVT; 15 the significant treatment effect of EVT for both IVT-eligible and IVT-ineligible patients with large vessel occlusions; potential delaying effect of IVT in initiating EVT; potential increased risk of bleeding events, including symptomatic intracranial hemorrhage (sICH); and risk of clot disintegration and subsequent embolization of clot fragments to distal areas. Additionally, IVT may not be ideal in conditions that require acute stenting and anti-platelet therapy. 16 Bypassing IVT may also potentially be more cost-effective in certain scenarios. 17 Considering these factors, many RCTs and post-hoc analyses of RCTs have investigated the safety and efficacy of EVT alone compared to bridging IVT prior to EVT. However, the findings from these studies have presented conflicting evidence regarding whether EVT alone is non-inferior to EVT with pre-treatment IVT in patients with acute ischemic stroke caused by a large vessel occlusion.18-23 More recently, the results from SWIFT-DIRECT and DIRECT-SAFE both suggested that EVT alone was not shown to be non-inferior to EVT with pre-treatment IVT.24,25 In SWIFT-DIRECT, final reperfusion rates were significantly lower among the EVT group, whereas in DIRECT-SAFE, the high reperfusion rates were similar across both treatment groups.24,25 The 2019 European Stroke Organisation (ESO) – European Society for Minimally Invasive Neurological Therapy (ESMINT) guidelines found high quality of evidence recommending the use of EVT and best medical management within 6 hours after symptom onset, and moderate quality of evidence for use of EVT and best medical management in patients presenting within the 6 hour to 24 hour window. 26 The ESO-ESMINT guidelines published an expedited recommendation strongly recommending IVT plus EVT over EVT alone. 27 Similarly, the 2019 American Heart Association guidelines provided a Class I recommendation for the use of IVT prior to EVT in IVT-eligible patients. 28
There are several published systematic reviews exploring the efficacy of bridging IVT prior to EVT compared to EVT alone;15,29-41 however, they do not consistently assess risk of bias or the certainty of evidence in each outcome, and vary in their study inclusion criteria and methodology. Due to the controversy of this topic and the addition of 2 recently published RCTs,24,25 we sought to conduct an updated systematic review and meta-analysis to compare the outcomes of patients who underwent EVT alone vs EVT plus IVT.
Methods
Standardized Reporting and Registration
We reported the review following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guideline (Supplement 1). 42 We registered the systematic review protocol on PROSPERO (CRD42022315608), and the protocol has been published. 43
Eligibility Criteria
We used the following eligibility criteria for study selection:
Population
Adult patients ≥18 years old presenting with an acute ischemic stroke secondary to a large vessel occlusion. We excluded any in vitro, animal or cadaveric/post-mortem studies.
Intervention
Intravenous thrombolysis with a fibrinolytic agent (ie, alteplase or tenecteplase) and 1 or more of the following techniques for thrombectomy: stent retrievers, aspiration catheters, or a combination of the above. We excluded those who underwent intra-arterial thrombolysis alone (eg, urokinase, tissue plasminogen activator), and/or systemic administration of agents other than fibrinolytic agents alone (eg, glycoprotein 2b/3a inhibitors).
Comparison
Thrombectomy without the use of IVT
Outcomes
We included studies that reported on functional independence or independence, mortality, and sICH. We defined functional independence or independence as a modified Rankin scale (mRS) score of zero to 2 at 90 days. We further defined mortality as death due to any causes at 90 days. After expert consensus, we defined sICH as any intracranial hemorrhage contributing to worsening neurologic exam, clinical deterioration, or death, adapted from the Heidelberg Bleeding Classification. 44
Setting
We included studies in any setting (ie, emergency department, hospital ward, intensive care unit (ICU), designated site, etc.).
Type of Study
For quantitative assessment, we included RCTs only. All other study designs, such as conference abstracts, editorials, commentaries, guidelines, literature reviews, news articles, systematic reviews, and meta-analyses were excluded from our study.
Search Strategy
With the help of an information specialist, we searched the following general databases: MEDLINE (through Ovid), EMBASE, and the Cochrane Library. All searches covered the time period from inception to December 14, 2021. We did not restrict searches to language. The detailed search strategies are described in Supplement 2. We also searched the reference lists of included studies and hand-searched grey literature with the aid of content experts on the review team.
Study Selection
Prior to the screening process, reviewers participated in calibration exercises during which they pilot tested a standardized screening form. Next, each citation (title and abstract) was screened by 1 reviewer, and then verified by a second reviewer. For citations that were judged potentially eligible, we retrieved their full texts and included them. Each full-text was screened by 1 reviewer and then verified by a second reviewer. A third reviewer resolved disagreements when necessary.
Data Collection
We developed and piloted a standardized data abstraction form. The reviewers used the form to extract data independently and in duplicate for the following characteristics: study characteristics (eg, author information, country of origin, study design), patient characteristics (eg, sample size, age and sex of patients, past medical history, National Institute of Health Stroke Scale (NIHSS) score, the proportion for each vessel involved as well as determined stroke etiology), characteristics of interventions and comparators (eg, intravenous thrombolytic agent used and respective dose, thrombectomy device used), and treatment outcomes, including mortality, functional independence, successful reperfusion, intracranial hemorrhage (including sICH), and procedural complications and other adverse events. Data was extracted into an Excel spreadsheet using Microsoft Excel 2021.
Risk of Bias Assessment
We assessed the risk of bias of all included studies using revised Cochrane risk-of-bias tool (RoB 2). The RoB 2 tool assesses bias related to randomization, deviations from intended interventions, missing outcome data, outcome measurement methods, and selection of the reported outcome. 45 This tool was previously validated on a sample of 70 RCTs, and found low interrater reliability and usability as barriers to its implementation. 46 We addressed concerns for low interrater reliability and other challenges, such as difficult terminology, by training the reviewers to improve the reliability of RoB 2. 46 We resolved disagreements between reviewers through consensus and/or with the assistance of an adjudicator.
Data Synthesis
We grouped each outcome reported by at least 2 studies for direct comparison. We standardized the definitions and measurements of each outcome via consensus. We conducted a meta-analysis of the outcomes of interest using the generic inverse variance method and random effects analysis model via Review Manager (RevMan) 5.4. For dichotomous outcomes reported by at least 1 RCT, we calculated the relative risk (RR) using the crude event rate and the associated 95% confidence intervals (CI) to inform relative effectiveness. We calculated risk difference (RD) based on the RRs from our study and the baseline risks from a well-designed, high-quality multi-center observational study of 6.350 ischemic stroke patients. 47 When only median and range values are reported, or when standard deviations are not reported, we used methods outlined in Cochrane handbook for systematic reviews of interventions and by Wan et al to estimate means and standard deviations.48,49 We reported our synthesized findings as funnel plots to assess for asymmetry per outcome of interest, and also estimated the Egger test of the intercept to assess for publication bias. 50 We considered a significance threshold of P ≤ .05 for two-tailed comparisons.
Subgroup Analysis
We evaluated statistical heterogeneity using inconsistency measures, Cochran’s Q test and I2. We used a priori hypothesis to explain heterogeneity between studies. We performed subgroup analyses irrespective of the heterogeneity estimates if there are at least 2 studies representing each subgroup.
Certainty of Evidence Assessment
For each outcome, we assessed the certainty of the evidence (as high, moderate, low, or very low) using the GRADE approach, and developed GRADE evidence profiles.51,52 The GRADE approach is important used to rate the certainty of evidence per outcome (ie, certainty that the true effect resides beyond a specific threshold or within a defined range) for systematic reviews. 53 We narratively described the strength of comparison for each outcome using the following terms: “there is” for high-certainty of evidence, “there probably is” for moderate-certainty of evidence, and “there possibly is” for low- or very low-certainty of evidence. We used previously derived thresholds for the minimally important difference for functional independence, mortality, and sICH, adapted from Wang and colleagues. 54
Results
Figure 1 shows the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flowchart. Of 11,111 citations, we identified 53 eligible studies, and included 6 studies with a total of 2336 participants. PRISMA flow diagram.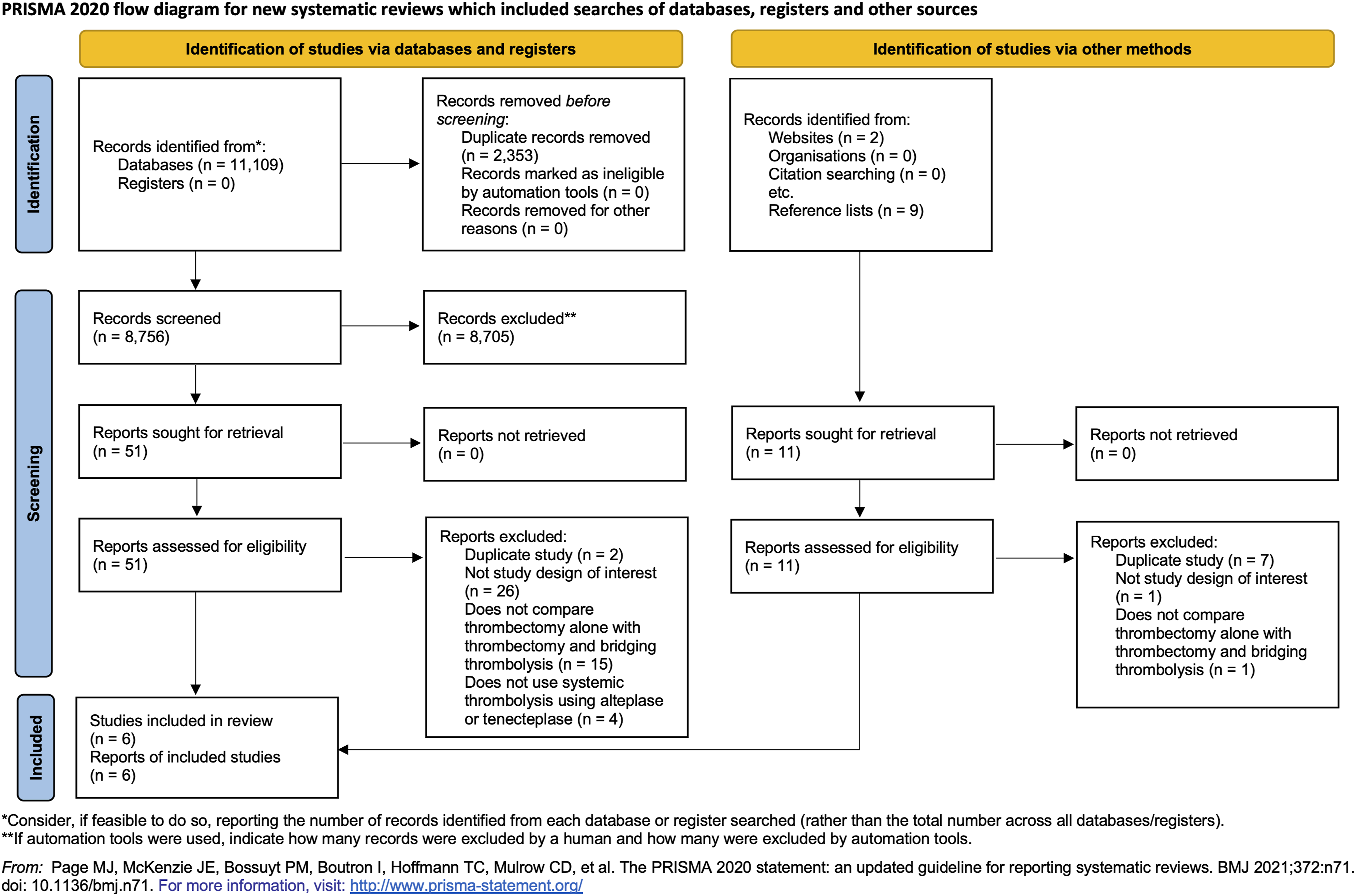
Characteristics of Included Studies
Characteristics of Included Studies.
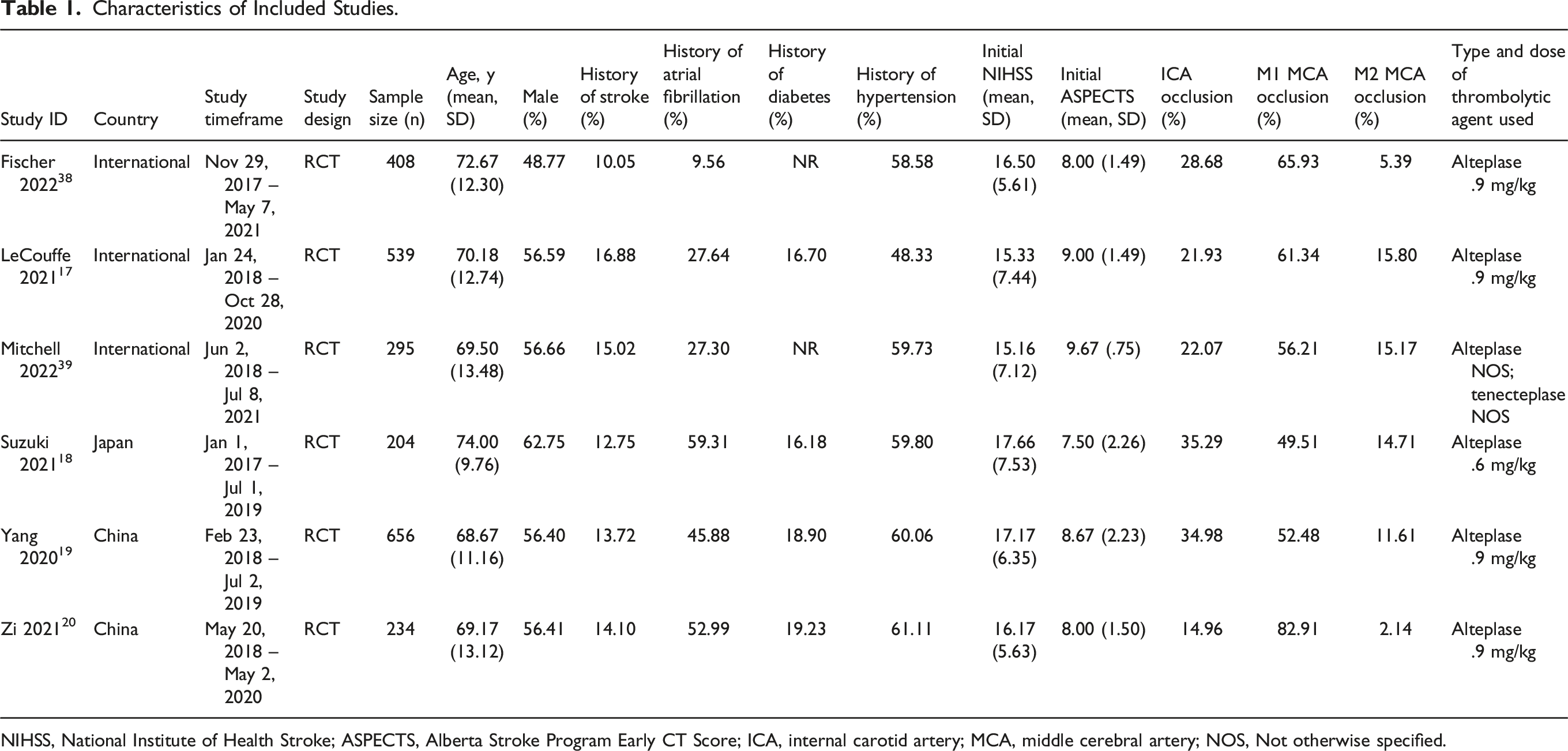
NIHSS, National Institute of Health Stroke; ASPECTS, Alberta Stroke Program Early CT Score; ICA, internal carotid artery; MCA, middle cerebral artery; NOS, Not otherwise specified.
Risk of Bias Assessment
All RCTs blinded outcome assessors but did not blind participants or providers due to the nature of the intervention. When specified, most studies randomized allocation sequence, and reported <10% missing outcome data. More specifically, while the nature of the intervention precluded adequate blinding of patients or providers, most studies were rated as moderate to high risk of bias due to inadequacy or variation in outcome measurements (eg, variability in the definitions of sICH used), and due to lack of description about allocation concealment (ie, no information on how the allocation sequence was generated). Additionally, cointerventions may have affected study outcomes given ineffective blinding of participants or providers. We did not detect any publication bias as illustrated in the funnel plots (Supplement 3-6).
Outcomes for EVT with IVT vs EVT Alone
Functional Independence (mRS Score zero to 2)
GRADE Evidence Profile for all RCTs Comparing Outcomes for Patients Who Underwent EVT Alone Versus EVT Plus IVT
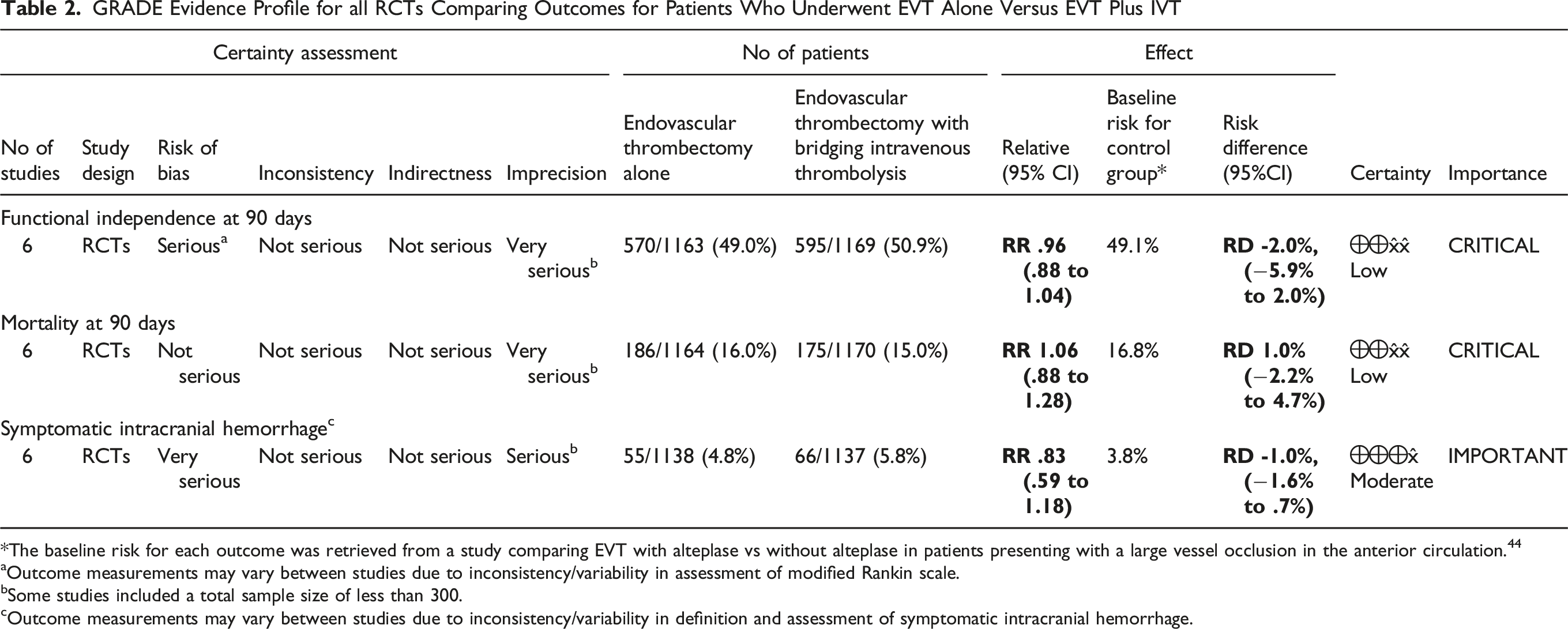
*The baseline risk for each outcome was retrieved from a study comparing EVT with alteplase vs without alteplase in patients presenting with a large vessel occlusion in the anterior circulation. 44
aOutcome measurements may vary between studies due to inconsistency/variability in assessment of modified Rankin scale.
bSome studies included a total sample size of less than 300.
cOutcome measurements may vary between studies due to inconsistency/variability in definition and assessment of symptomatic intracranial hemorrhage.
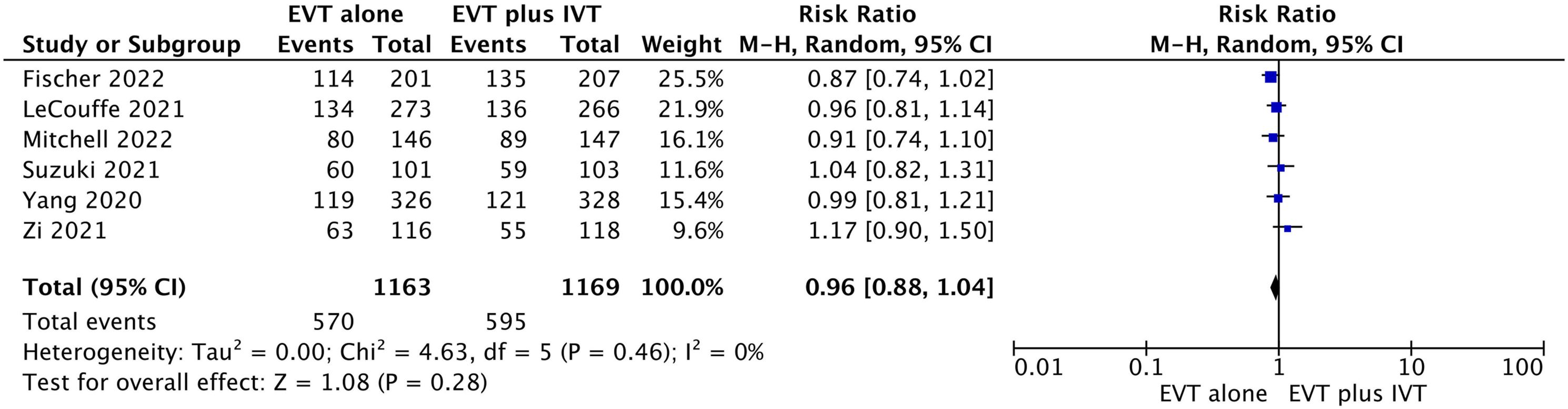
Forest plot for endovascular thrombectomy (EVT) alone vs EVT with bridging thrombolytics for functional independence defined as modified Rankin scale (mRS) score of zero to 2 at 90 days.
Mortality at 90 Days
All studies reported mortality recorded at 90 days.20-25 We found low-certainty evidence that there is possibly a small increase in mortality at 90 days for patients with EVT alone compared to EVT plus IVT (RR 1.06, 95% CI 0.88 to 1.28; RD 1.0%, 95% CI −2.2% to 4.7%) (Table 2, Figure 3). Forest plot for endovascular thrombectomy (EVT) alone vs EVT with bridging thrombolytics for mortality at 90 days.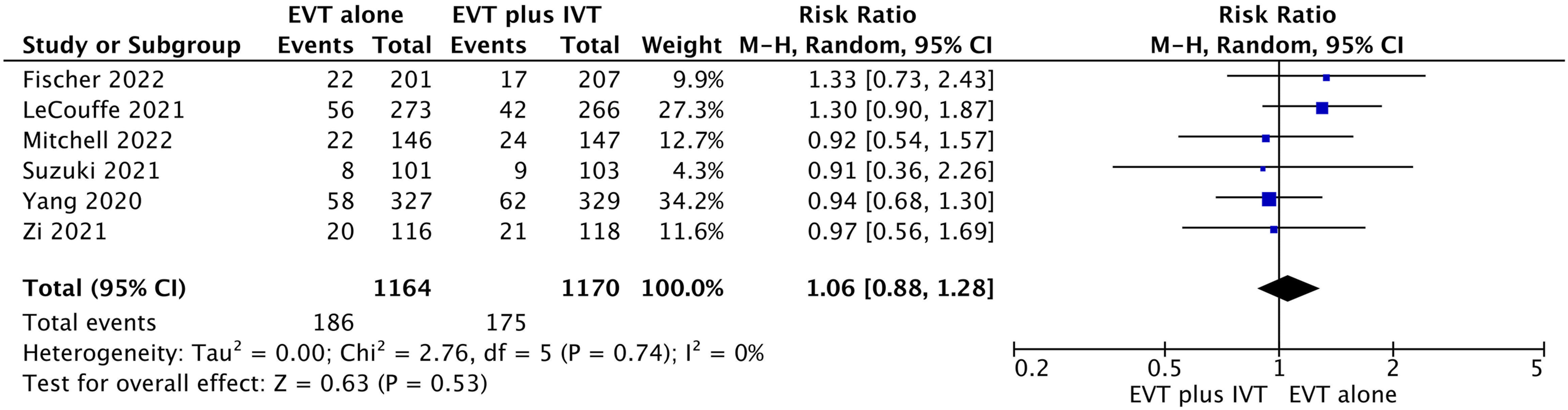
Symptomatic Intracranial Hemorrhage
All studies reported sICH despite variability in the definitions between studies.20-25 We found moderate-certainty evidence that there is probably a small decrease in sICH for patients with EVT alone compared to EVT plus IVT (RR .83, 95% CI 0.59 to 1.18; RD −1.0%, 95% CI −1.6% to .7%) (Table 2, Figure 4). Forest plot for endovascular thrombectomy (EVT) alone vs EVT with bridging thrombolytics for symptomatic intracranial hemorrhage.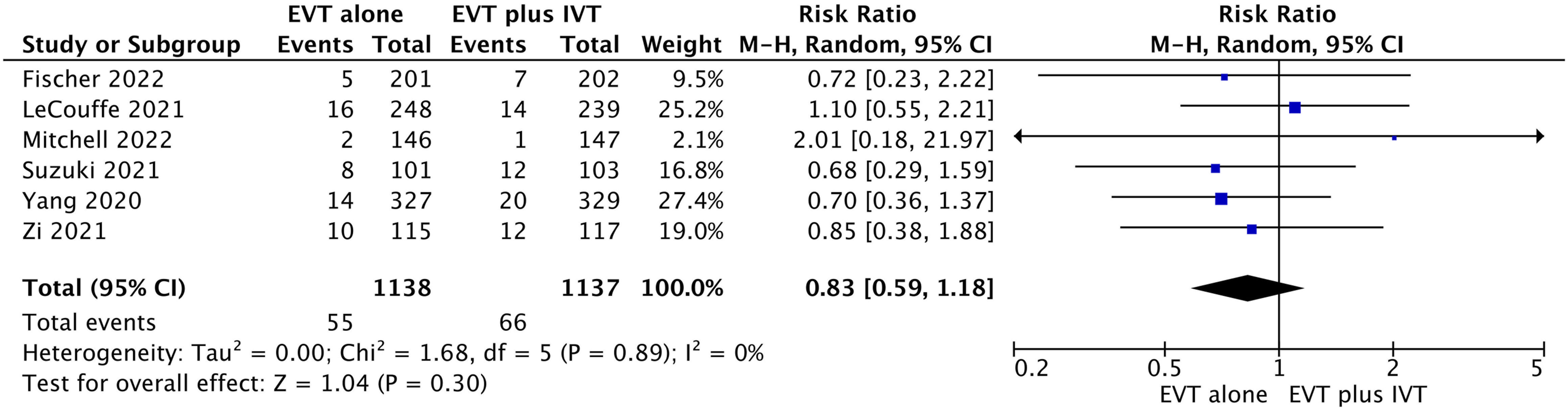
Procedure-Related Complications
In total, 4 studies reported procedure-related complications.20,22,23,25 There was no significant difference in procedure-related complications for patients with EVT alone vs those with EVT plus IVT (RR .93, 95% CI 0.77 to 1.13) (Supplement 7).
Discussion
In this study, we compared the outcomes of patients who underwent EVT alone vs EVT plus IVT. We found that there are differences in absolute risk for functional independence, defined as mRS value of zero to 2, at 90 days, mortality at 90 days, and sICH between those patients who underwent EVT alone vs EVT plus IVT. The quality of evidence ranged from low-certainty to moderate-certainty evidence.
To our knowledge, this is the first systematic review conducted to compare outcomes between EVT alone vs EVT plus IVT using all RCTs published in peer-reviewed journals as compared to previous systematic reviews that used conference data for 2 RCTs,54,55 incorporating the most updated data from 6 RCTs. Moreover, this is the only systematic review on this subject that compared the procedural complications between the 2 groups for 4 RCTs. Our systematic review used rigorous methods in title and abstract screening, full-text evaluation, and risk of bias and certainty of evidence assessment using the RoB 2 tool and GRADE structured framework, respectively. We used a comprehensive search strategy without any language restrictions. We also included several research methodologists and content experts in our decision-making process.
The main limitation in our study is that we could not perform a meta-regression analysis, because there were not enough studies or power to account for potential confounding variables. Another limitation included inconsistency in the methods of measurement of outcomes. We found that the definition of outcomes, especially that pertaining to sICH, was not standardized across the studies. After gathering input from our clinical experts, we analyzed the data for sICH in the following manner for the included RCTs: the Heidelberg bleeding criteria for 3 RCTs,20,22,23 specific author-defined criteria for 2 trials,24,25 and the Safe Implementation of Thrombolysis in Stroke-Monitoring Study (SITS-MOST) criteria for 1 trial. 21 Mitchell and colleagues defined sICH as evidence of ICH with an associated increase of ≥4 NIHSS points within the first 24 hours post-stroke on computed tomography imaging. 25 Fischer and colleagues defined sICH as core laboratory-adjudicated parenchymal hematoma type 1 or 2, subarachnoid hemorrhage, or intraventricular hemorrhage within 24 hours (±6 hours) associated with an increase of ≥4 NIHSS points compared to baseline. 24 Given this variability in outcome definitions, we cautiously meta-analyzed the data and found that our pooled effect estimates did not have any statistical heterogeneity (I2 = 0%); thus, our selected outcome definitions were justifiable. Another limitation pertaining to the included RCTs was the inability to meta-analyze the ordinal change in mRS for the patients, which serves as a better estimate of long-term outcomes compared to dichotomized mRS. 56 Additionally, only 1 RCT used both alteplase and tenecteplase; therefore, it was difficult to interpret whether tenecteplase may have had an alternative treatment effect without any direct comparisons. 25 This same RCT also included patients with a basilar artery occlusion contrary to other RCTs which only included patients with internal carotid artery or middle cerebral artery occlusions 25 ; as such, we cannot extrapolate our findings to other vascular territories. Another limitation in our study is that we used the RoB 2 tool to assess risk of bias, which may have contributed to low interrater reliability 46 ; however, to mitigate this issue, we ensured the reviewers were adequately trained before using the tool.
In our systematic review, we assessed the certainty of the evidence when comparing outcomes between patients with underwent EVT alone vs those who underwent EVT plus IVT. Our systematic review and meta-analysis found that low-certainty evidence, low-certainty evidence, and moderate-certainty evidence, in possibly a small decrease in functional independence with an mRS of zero to 2 at 90 days, possibly a small increase in mortality at 90 days, and probably a decrease in sICH, between patients who underwent EVT alone vs EVT plus IVT, respectively. We found 2 systematic reviews that used the GRADE approach to assess the certainty of evidence when comparing outcomes between EVT alone vs EVT plus IVT.54,55 Trifan and colleagues found low quality of the evidence when comparing functional independence between the 2 groups, favoring EVT plus IVT over EVT. However, when the analysis was restricted to RCTs only, there was moderate quality of the evidence with comparable functional independence between the 2 groups. 55 They also found low quality of evidence suggesting decreased mortality at 90 days among the EVT plus IVT group compared to the EVT alone group, and moderate quality of evidence showing comparable differences in sICH between the 2 groups. 55 In contrast, Wang and colleagues found low certainty of evidence for a non-significant association when comparing functional independence at 90 days between the 2 groups, which is a similar finding in our systematic review. 54 Wang and colleagues also found low-certainty evidence suggesting EVT alone possibly results in a small increase in mortality compared to EVT plus IVT, and moderate-certainty evidence suggesting that EVT alone probably results in a small decrease in sICH compared to EVT plus IVT. The discrepancies in the certainty of evidence are attributed to multiple factors, including weighting of different biases, imprecision bias, as well as the number and type of included studies. Trifan and colleagues included longitudinal randomized or non-randomized studies in their analysis, which may have contributed to an overestimation of the effect estimates. 55 However, both systematic reviews included data from 2 unpublished RCTs, SWIFT-DIRECT and DIRECT-SAFE, which may have slightly affected their outcomes.54,55 There are also multiple systematic reviews comparing EVT plus IVT to EVT alone, but they do not consistently assess risk of bias or the certainty of evidence for each outcome and do not include the finalized data from each trial.15,29-41
The extent to which IVT plays a role in the treatment of acute stroke prior to EVT is subject to multiple variables in real-world practice. While IVT can potentially result in distal migration of clots and hemorrhagic transformation, it may result in recanalization, 57 especially for distal vessel occlusions,58-60 posterior circulation strokes,61,62 and smaller clot burdens. 63 A recent subgroup analysis of DIRECT-MT found that there was no significant treatment-by-occlusion site interaction of IVT for internal carotid artery, or M1/M2 branches of the middle cerebral artery when compared to no IVT prior to EVT. 64 However, the small sample size, especially for M2 branch occlusions, and a non-significant increase in sICH for M1 and M2 branch occlusions in the IVT plus EVT group may have potentially contributed to worse outcomes compared to the EVT only group. 61 IVT can prove beneficial when it is the only means of reperfusion therapy in the case of technical difficulties in accessing intracranial vessels or in cases where time to endovascular therapy is delayed. 65
The ESO-ESMINT guidelines made a strong expedited recommendation for EVT plus IVT over EVT alone. 27 Similarly, the 2019 American Heart Association guidelines provided a Class I recommendation for the use of IVT prior to EVT in IVT-eligible patients. 28 Wang and colleagues also engaged a guideline panel to contextualize their findings and found that the available evidence supports a weak recommendation in favor of EVT alone. 66
In certain conditions, the future of IVT before EVT is still controversial. While results from our systematic review showed that clinical outcomes among EVT alone and EVT plus IVT treatment groups are comparably similar, it is important to interpret these results with caution as our results are based on a study-level meta-analysis. Our meta-analysis results should be used to inform providers' case-by-case management decisions, rather than being interpreted as stand-alone. Findings from an individual participant data meta-analysis may be more useful in estimating intervention effects based on data from subgroup analyses. A recent post-hoc analysis of SWIFT-DIRECT highlighted the need for an individual participant data meta-analysis in order to detect an interaction effect for time to treatment, mainly stroke symptom onset to time of IVT administration, with clinical outcomes. 67 Expanding on the importance of exploring the potential heterogeneity of the IVT effect size prior to EVT, as highlighted in the post-hoc analysis, it is crucial to delve into the transition to tenecteplase. 67 The EXTEND-IA TNK trial compared tenecteplase vs alteplase before thrombectomy and found that tenecteplase administration was associated with higher reperfusion rates and better functional outcomes. 68 In light of the results from the EXTEND-IA TNK trial, these findings provide compelling evidence supporting the potential benefits of utilizing tenecteplase as a valuable agent in clinical decision-making for patients undergoing EVT. This transition may offer valuable insights into whether bridging with tenecteplase can enhance outcomes compared to bridging with alteplase, as emphasized by Nguyen and Fischer. 16
Conclusion
We conducted a systematic review and meta-analysis to compare the outcomes of patients who underwent EVT alone vs EVT plus IVT. Our findings showed that there is possibly a small decrease in functional independence at 90 days with low certainty of evidence, possibly a small increase in mortality at 90 days with low certainty of evidence, and probably a decrease in sICH with moderate certainty of evidence, between those patients who underwent EVT alone vs EVT plus IVT.
Supplemental Material
Supplemental Material - Endovascular Thrombectomy With or Without Thrombolysis for Stroke: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Supplemental Material for Endovascular Thrombectomy With or Without Thrombolysis for Stroke: A Systematic Review and Meta-Analysis of Randomized Controlled Trials by Rami Z. Morsi, Yuan Zhang, Julián Carrión-Penagos, Harsh Desai, Elie Tannous, Sachin Kothari, Assem Khamis, Andrea J. Darzi, Ammar Tarabichi, Reena Bastin, Layal Hneiny, Sonam Thind, Elisheva Coleman, James R. Brorson, Scott Mendelson, Ali Mansour, Shyam Prabhakaran and Tareq Kass-Hout in The Neurohospitalist
Footnotes
Acknowledgements
None.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Clinical Trial Registration
Systematic review protocol registration PROSPERO (CRD42022315608).
Data Availability Statement
The ![]() used to support the findings of this study are available from the corresponding authors upon request by email.
43
used to support the findings of this study are available from the corresponding authors upon request by email.
43
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
