Abstract
Spinal cord hemorrhage remains an uncommon vascular pathology with no standard approach to treatment. Intramedullary hematomyelia is the rarest subtype and has only been described in case reports, making it a diagnosis easier to miss. A 55-year-old male with history of polysubstance abuse presented for abrupt onset non-traumatic back pain that progressed over several days evolving into paraplegia, sensory loss up to the T3 dermatome, and incontinence of bowel and bladder. His MRI imaging of the cervical and thoracic spine was concerning for a T1 and T2 hyperintense lesion extending from the internal medulla to the upper thoracic spine causing edema. The initial differential diagnosis, based on history and progressive neurologic decline, included a longitudinally extensive transverse mellitus of unclear etiology. He received high dose steroids and plasma exchange without any clinical improvement. An extensive autoimmune, inflammatory, infectious, and demyelinating workup via serum and CSF analysis was unremarkable. He was transferred to a tertiary facility, where repeat MRI spinal imaging included GRE sequences that was significant for increased susceptibility. MRA imaging of the spine was completed showing no vascular malformation. Overall, the diagnosis of hematomyelia was felt to be most likely. Despite aggressive inpatient rehabilitation, the patient remains paraplegic and requires long term management consistent with severe spinal cord injury. This case highlights the importance of recognizing vascular syndromes as a cause of acute myelopathy, understanding the imaging findings of blood breakdown products on MRI, and the desperate need for successful therapeutic strategies to combat a potentially devastating neurologic process.
Introduction
Vascular disorders of the spinal cord require urgent evaluation, recognition, and treatment to provide the greatest chance of avoiding poor outcomes. Spinal cord hemorrhages are uncommon, with some papers suggesting the incidence is one per 100 000 population annually, representing .3%–.9% of all spinal cord lesions. 1 Intramedullary hematoma, or hematomyelia, is the rarest type of spinal cord hemorrhage, typically presenting with acute back pain, radicular pain, or sudden headache with associated progression of neurologic deficits. 2 Trauma is the most common cause, followed by vascular malformations, malignancy, bleeding disorders, use of anticoagulation, and several rarer etiologies. 3 Diagnosis may be challenging, not only because the clinical course depends on the location and severity of the lesion, but because the presentation can either be acute, subacute, or chronic in nature. 4 Two large retrospective studies previously showed that patients initially diagnosed with idiopathic transverse myelitis frequently had alternative diagnoses, which is worrisome with this condition given frequently poor outcomes. Neuroimaging is important to confirm the diagnosis, as magnetic resonance imaging (MRI) can reveal flow voids suggesting a vascular malformation, alterations in signal intensity on T1 and T2 signals due to breakdown of blood products, and increased susceptibility on GRE sequences. 5 Several treatment options have been reported in the literature, including both pharmacologic and surgical, however there is no consensus treatment protocol. The indication and proper timing for surgery, and even prognosis, remain controversial. 6
Case
A 55-year-old-male with history of chronic alcohol and tobacco use, intramuscular testosterone use, and prior cocaine abuse, presented to a community hospital for sudden onset back pain with numbness, tingling, and weakness of the right foot, which spread into the right groin and abdomen over a few days. ED service at that time suspected radiculopathy with exacerbated chronic back pain. X-ray imaging of the thoracic and lumbar spine did not reveal any acute process. He was discharged home with a short course of steroids and muscle relaxers. A week later, he returned to the hospital with persistent symptoms involving bilateral lower extremities, a sensory level in the abdomen, bowel/bladder incontinence, and difficulty ambulating. Although he exercised twice daily and was a construction worker, he denied any recent trauma or injury. He denied taking any blood thinners and had no family history of similar symptoms. Our neurology service was consulted for further evaluation more than 10 days after symptom onset.
Neurologic examination showed intact mental status and cranial nerves. There was physiologic tone in the upper extremities, with normal strength and sensation. He did have decreased sensation in all modalities from the lower extremities up to the T3 dermatome. Medical Research Council (MRC) strength was 2/5 in proximal and distal muscle groups of the bilateral legs. He had hyporeflexia in the bilateral patella and achilles. MRI brain showed no acute process, T2 hyperintensity, or enhancing lesion, and MRI lumbar spine was unremarkable. MRI thoracic spine revealed abnormal T2 signal hyperintensity within the central spinal cord, extending from T2-T9. There was also a small T1 hyperintense lesion within the posterior aspect of the spinal cord at the T1-T2 level. The MRI cervical spine showed a large area of T2 hyperintense signal within the cervical cord extending from the inferior medulla down to the level of C7 with associated edema and minor enhancement. T1 hyperintensity was also present in the same territory, most appreciated at the C4-C6 level (Figure 1 and 2). GRE sequences were not completed for the initial MRI studies. Magnetic resonance imaging of the cervical spine, sagittal plane. T1 sequence (left) shows hyperintense signal extending caudally from the brainstem to the T2 level. T1 post contrast (middle) shows patchy enhancement most appreciated at the C6 level. T2 sequence (right) shows more obvious extensive hyperintensity in the same location, with some hypointensity at the C6 level. Magnetic resonance imaging of the cervical spine, axial plane. T1 sequence (left) shows hyperintense signal in the central cord at C4-C5 level. T2 sequence (right) shows hyperintense signal with possible hypointense rim in the central cord.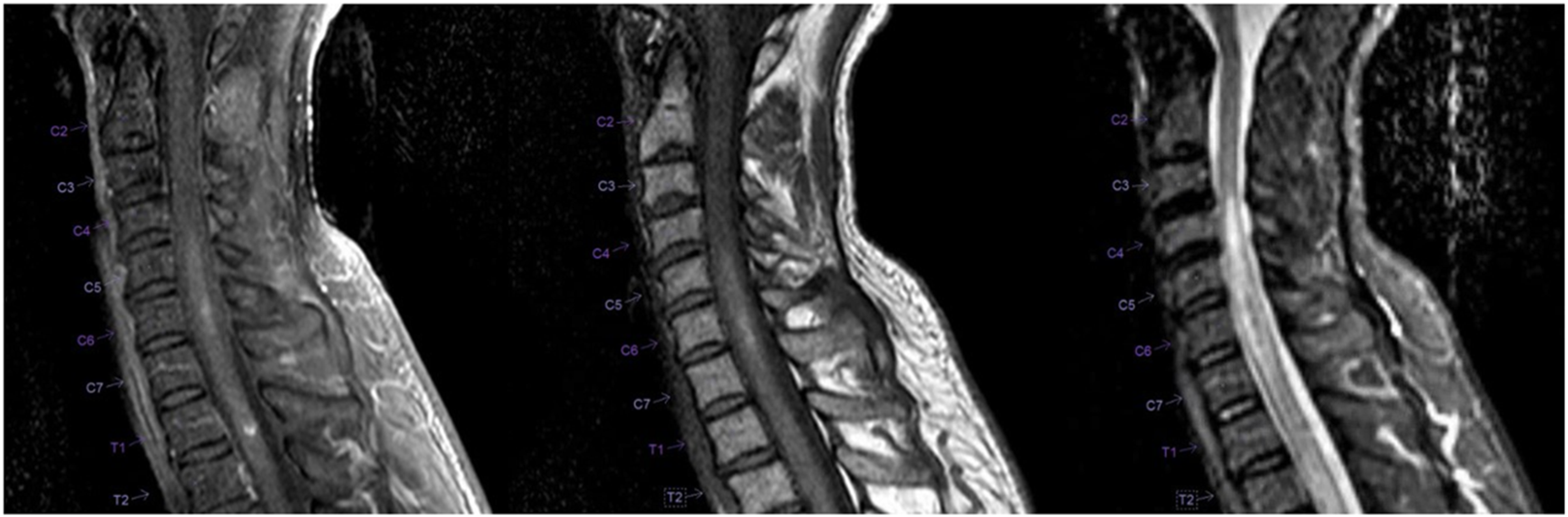
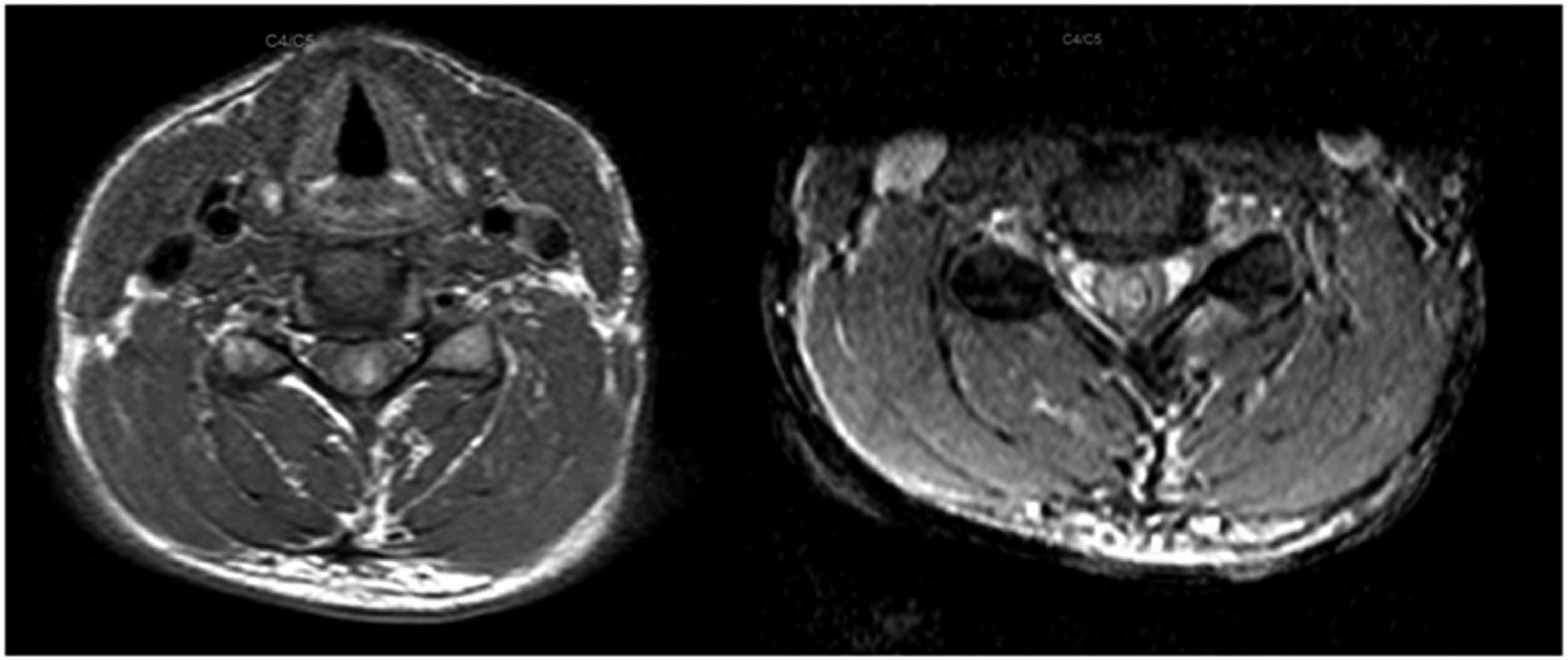
Initial working diagnosis was a longitudinally extensive transverse myelitis of unknown cause, with consideration for autoimmune, infectious, demyelinating, or inflammatory process being possible etiologies. Lumbar puncture showed protein of 53 mg/dL (normal 15-45 mg/dL), glucose 96 mg/dL (normal 40-80 mg/dL), 11 white blood cells/mm³, and 11 red blood cells/mm³. Oligoclonal bands were not present. MOG-IgG and Aquaporin-IgG antibodies, as well as ANCA panel, were negative. Testing was negative for HIV, HSV, Quantiferon TB, RPR, West Nile, Histoplasma, Cryptococcus, Aspergillus, Hepatitis B and C. Vitamin B12, Vitamin B6, Copper, and Zinc were also normal. Urine drug screen and ethanol levels were negative. Liver enzymes (AST, ALT), PT, and INR were normal. Hematologic workup showed mixed coagulant labs, with hemoglobin 19.8 gm/dL, hematocrit 59.6%, elevated activity of antithrombin 151%, factor IX 183 units/dL, factor VIII 261 units/dL, vWF 291 IU/dL, and protein C 166%. In attempts to provide some clinical improvement, high dose steroids for 5 days was initiated. Unfortunately, the patient’s weakness continued to progress until he had 0/5 strength in the bilateral lower extremities. More aggressive treatment with plasma exchange was started, and fibrinogen level was decreased to 89 mg/dL following therapy.
Due to the complex nature of the case, and no available neurosurgery service at the hospital, the patient was transferred to a tertiary center for further management. Repeat MRI cervical and thoracic spine imaging reaffirmed intrinsic T1 and T2 hyperintensities in the spinal cord with edema. GRE sequences were included and revealed correlating increased susceptibility. MRA of the spine, followed by a diagnostic spinal angiogram, did not reveal any obvious vascular malformation. Given the clinical history, imaging findings, and otherwise negative workup, a diagnosis of spontaneous, nontraumatic, intramedullary hematomyelia was felt to be most likely, with potential origin in the thoracic cord. The patient underwent long term rehabilitation without any meaningful improvement in his lower extremity weakness or ability to ambulate. Follow up MRI imaging of the cervical and thoracic spine 3 months after his initial presentation showed intramedullary hypointense signals on T1, T2, and GRE sequences with resolution of the prior associated edema (Figure 3). Since 2019, he has required a mobilized wheelchair and faces challenges of coping with a catastrophic spinal cord injury, including recurrent UTIs, chronic pain, lower extremity DVT (in 2021, treated previously with Xarelto), and severe depression with suicidal thoughts. Magnetic resonance imaging of the cervical spine approximately 3 months after initial studies. (A) T1 sagittal plane showing an extensive hypointense signal in the central cord extending from the brainstem to at least T2 level with resolution of previous edema. (B) T1 post contrast sagittal plane does not show any residual enhancement. (C) T2 sagittal plane again shows a linear hypointense lesion correlating with the T1 sequence. (D) GRE sequence has increased susceptibility in the central cord from the brainstem to at least T2 level, consistent with hemosiderin deposition.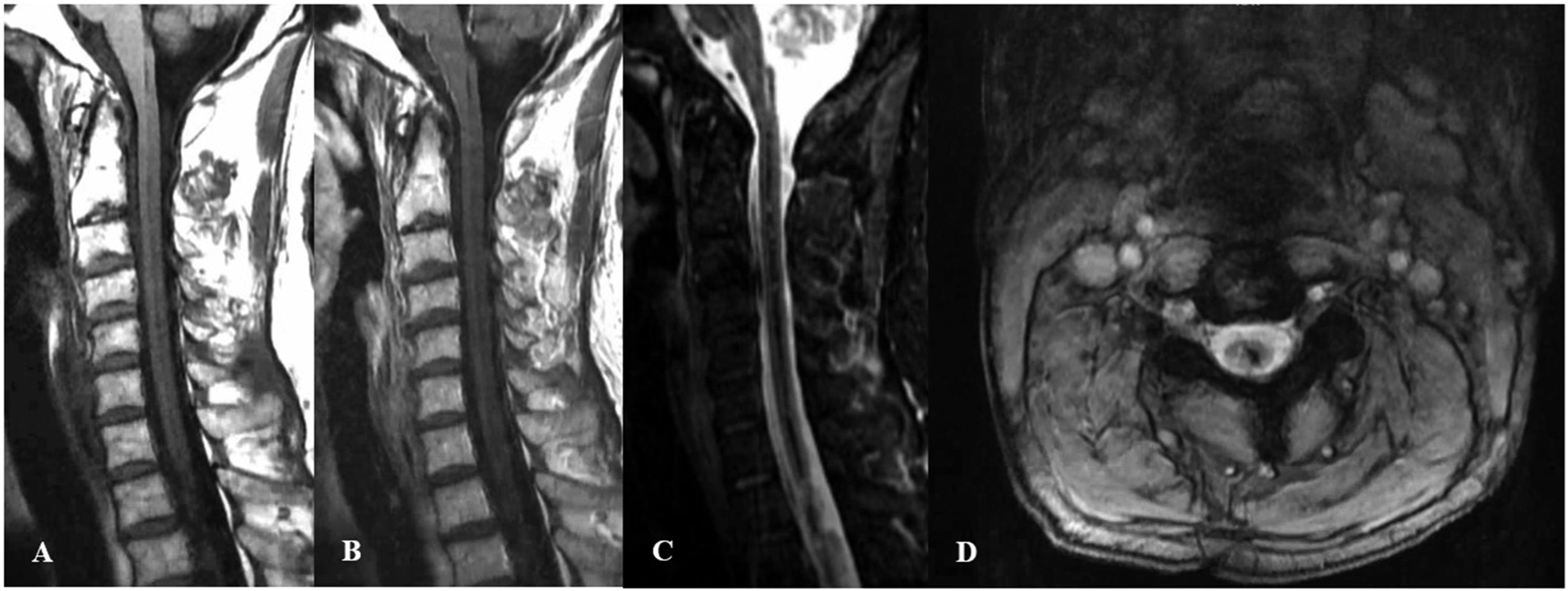
Discussion
Several important concepts are appreciated from this case that providers should be aware of when evaluating spinal cord vascular pathology such as hematomyelia. Considering the subacute progression of symptoms, longitudinally extensive T1 and T2 hyperintensities, edema, and mild enhancement, more focus was on a possible inflammatory, demyelinating, infectious, or autoimmune myelopathy. Unavailability of GRE sequences initially to determine presence of hemorrhage likely impacted the team’s differential. Follow up imaging weeks later included GRE sequences which showed increased susceptibility, and this finding persisted on MRI imaging 3 months later. Hemorrhage on MRI transitions through various intensity signals on T1 and T2 sequences as hemoglobin is altered from an oxygenated state to hemosiderin; 7 a progression typically not found in other more common causes of non-hemorrhagic myelopathies. Our neurology service likely evaluated the patient in the late subacute phase of hemorrhage and follow up imaging months later confirmed evolution to the chronic phase as T1 and T2 showed hypointensity. The significance of the hematologic labs here is uncertain as there appeared to be a mix of both prothrombotic and antithrombotic markers. The patient was using exogenous testosterone, which may have contributed to his thrombocytosis and increased antithrombin levels. Chronic alcohol use can contribute to coagulopathy and increase risk of hemorrhage, yet his liver function, platelets, PT, and INR were nonrevealing. Illicit drug use such as cocaine may lead to vasospasm and vascular pathologies, but the urine drug screen was negative. CSF analysis did reveal a mild pleocytosis with 11 white blood cells/mm³, but this was felt to be a nonspecific finding. From a clinical perspective, acute discomfort is not only common in spinal cord hemorrhage, but in other conditions such as spinal cord infarction or even disc herniation. 5 Due to progressive symptoms over several days with asymmetric features, a vascular process initially was felt to be less likely, leading to a delay in diagnosis. A clear cause of our patient’s hematomyelia is still relatively unknown, as there was no associated trauma. MRI spinal imaging did not show any flow voids, and spinal angiogram did not reveal a clear vascular malformation. The workup in its entirety eventually seemed to support a vascular myelopathy more than the other previously mentioned possibilities.
In conclusion, spinal cord hemorrhage is a rare but severe form of acute myelopathy often misdiagnosed with no guideline driven recommendations for management. This case details a 55-year-old male presenting for back pain with focal neurologic deficits in the lower extremities, progressing to severe paraplegia with a thoracic sensory level and bowel/bladder dysfunction. His presentation and testing were concerning for a severe idiopathic transverse myelitis, with diagnostic workup focused mainly on a possible inflammatory, infectious, demyelinating, or autoimmune etiology. He was treated with steroids and plasma exchange, and with no clinical improvement, the patient was transferred to a tertiary facility, where additional testing (repeat MRI imaging followed by spinal angiogram) failed to find a source for his hemorrhage. This lead to an eventual diagnosis of hematomyelia. Neurologists need to be mindful that vascular disease, although rare, can also impact the spinal cord, and oftentimes leads to devastating morbidity. Understanding the differential diagnosis and workup (especially spinal imaging findings) for the development of acute/subacute myelopathy can significantly alter a patient’s treatment and clinical course.
Footnotes
Author’s Note
This manuscript has not been submitted for publication elsewhere.
Author Contributions
The author has contributed substantively to the conception, design, analysis, and interpretation of data, contributed substantively to the drafting of the manuscript, gave final approval of the version to be published, and agrees to be accountable for all aspects of the work in ensuring that questions related to accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Informed consent was obtained from the patient for completion of the manuscript, and self-identifying information was excluded. An informed consent for publication of this report was provided by the patient.
