Abstract
Chimeric antigen receptor (CAR-T) cell therapy is highly effective against hematological cancers but is associated with immune mediated side effects, including neurotoxicity. The most commonly described presentations of immune cell mediated neurotoxicity syndrome (ICANS) include cortical symptoms and generally localize to the central nervous system. In this report, we present a patient with acute onset of bilateral facial nerve palsy following CAR-T cell therapy, followed by a complete clinical recovery. Aside from a temporary anisocoria, he had no other neurologic symptoms and no encephalopathy or seizures. MRI Brain was non-contributory and cerebrospinal fluid revealed a modest increase in lymphocytes without systemic leukocytosis and viral studies were all negative. He was diagnosed with bilateral facial nerve palsy secondary to CAR-T cell therapy and subsequently treated with a course of steroids. Several weeks after presentation he returned to his neurological baseline. The presentation of CAR-T cell mediated facial nerve palsy is both clinically and scientifically relevant for physicians, patients, and researchers.
Keywords
Introduction
Chimeric antigen receptor T (CAR-T) cell therapy is a novel highly effective immunotherapy for hematological malignancies. Documented side effects include cytokine release syndrome (CRS), typically occurring within 1 week, and ICANS (Immune Cell Associated Neurotoxicity Syndrome). 1 ICANS has been documented in 48% of CAR-T therapy patients, typically between weeks 1-3 post infusion and is associated with cortical symptoms, including cognitive dysfunction, aphasia, tremor, seizures, and lethargy. 1
Unilateral facial nerve palsy is a common neurological phenomenon with an incidence of 20 per 10 000 per year that is frequently idiopathic. 2 Bilateral facial nerve palsy is rare, occurring in 0.3-2% of cases and is more frequently associated with systemic causes. 3
In this report, we describe a unique case of acute onset bilateral simultaneous facial nerve palsy occurring 2 weeks after CAR-T cell infusion for multiple myeloma (MM). Facial nerve palsy should be considered as an early presentation of neurotoxicity in MM-directed CAR-T cell therapy.
Patient Information
A 76-year-old man was diagnosed with MM with a serum IgG kappa concentration of 40.5 g/L and bone marrow demonstrated 60% plasma cell composition. Fluorescence in situ hybridization (FISH) did not reveal a driving genetic cause. Skeletal survey demonstrated no lytic lesions. Medical profile included hypertension but was otherwise noncontributory: specifically, no obesity, diabetes, or respiratory illness. Seven months prior to presentation, the patient received 7 cycles of bortezomib, lenalidomide, dexamethasone, and palliative radiotherapy to the cervical spine, achieving partial remission. At this time, the only noted neurological side effect was grade 1 peripheral neuropathy.
Prior to presentation, he received lymphoid depleting therapy (cyclophosphamide 300 mg PO daily and fludarabine 24 mg PO daily). Anti-BCMA CAR-T cell (Ciltacabtagene autoleucel; Cilta-cell) infusion took place 2 weeks before re-presentation.
One-week post-infusion, the patient had a grade 1 Cytokine Reaction Syndrome (CRS)—with CRP climbing to 28. Piperacillin/tazobactam was administered post-infusion for 10 days. At that time, he remained neurologically asymptomatic and did not meet ICANS criteria.
The patient presented for follow up 1-day post hospital discharge with a one-day history of rapidly progressive bilateral facial paresis. He first noticed inability to close his eyes, and over the course of the following morning, he was no longer able to eat food without spilling out the sides of his mouth. He endorsed dysarthria and reported ongoing myalgias. He denied loss of taste and change in muscle bulk. His oncologist recommended urgent presentation to the Emergency Room.
Clinical Findings
On initial examination, this gentleman had normal vitals and was fully alert and oriented. His immune effector cell encephalopathy (ICE) score on admission was 10, indicating no deficits. Speech was mildly dysarthric secondary to facial palsy, but with intact content, fluency, and repetition. Cranial nerve exam was significant for anisocoria with R pupil > L pupil (5 mm, 3 mm), more evident in light conditions. Both pupils were reactive to light, with no relative afferent defect. Visual acuity and fields were intact, with no ophthalmoplegia. There was mild ptosis equal bilaterally, which the patient was able to overcome. He was unable to raise his eyebrows bilaterally, nor close his eyes fully, with a 2 mm gap between eyelids at maximum effort. Bell’s phenomenon was observed. He was unable to smile bilaterally, in a symmetrical fashion, with bilateral buccinator weakness. Facial sensation was normal except mild subjective numbness of lower lip bilaterally. There was no peripheral weakness, large fiber sensory deficits, or coordination difficulties. Reflexes were grade 2 throughout with down-going plantar responses.
Diagnostic Assessment
MRI/MRA showed mild cranial nerve VII enhancement and notably demonstrated no cranial nerve III enhancement (Figure 1). Of note, MRA did remark an incidental right 1-2mm ICA aneurysm. Cerebrospinal fluid analysis for cytopathology and infectious workup was performed, as shown in Table 1. Aside from a mild lymphocytic CSF pleocytosis which resolved by day 8, no infectious indicators were found. Bilateral facial nerve palsy after CAR-T therapy. T1 pre- (left) and post- (right) gadolinium enhancement demonstrating mild bilateral cranial nerve VII enhancement. Diagnostic Evaluation Performed during Hospital Stay to Investigate Cause of Bilateral Facial Nerve Palsy. Abbreviations: CMV = cytomegalovirus; CN = cranial nerve; CRP = C reactive protein; CSF = cerebrospinal fluid; EBV = Epstein barr virus; HHV = human herpes virus; HSV = herpes simplex virus; NAT = nucleic antigen test; NK = natural killer; VZV = varicella zoster virus; WBC = white blood cells.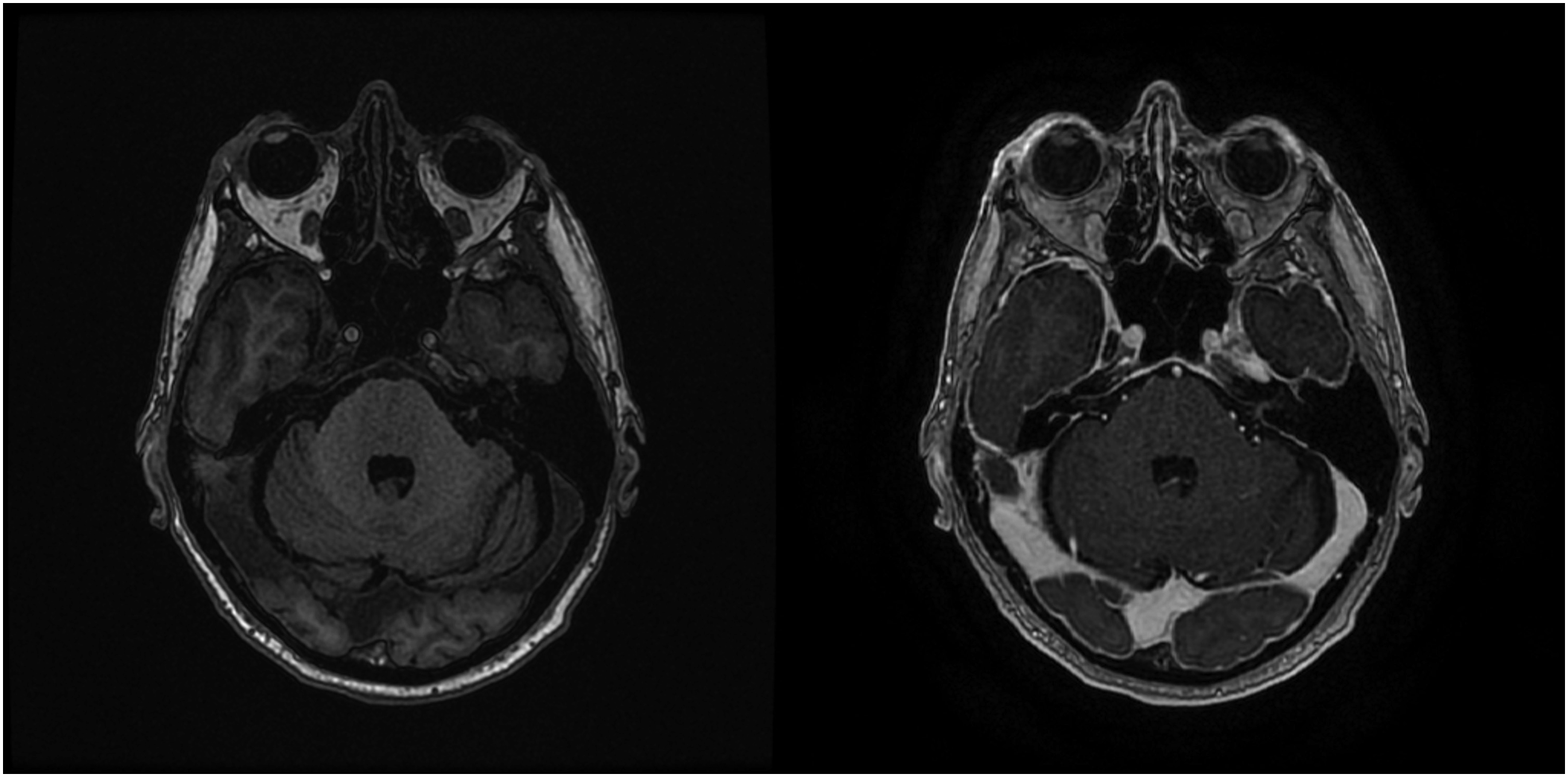

Based on the clinical course and absence of alternative diagnoses, the working diagnosis was bilateral facial nerve palsy secondary to CAR-T cell therapy. Isolated right pupillary mydriasis was not associated with other evidence of cranial nerve III dysfunction.
Therapeutic Intervention
Due to the initial broad differential, the patient was empirically treated with acyclovir, dexamethasone, fluconazole, meropenem, and vancomycin. Levetiracetam was given per institutional protocol for suspected CNS involvement with CAR-T therapy. Once severe pathology was ruled out, and clinical symptoms remained stable, medications were discontinued, and he was discharged on Day +8.
The patient was monitored in clinic after discharge for response to the CAR-T cell therapy. On Day +21, the patient had begun to note improvement in their bilateral facial palsy symptoms. Namely, he was able to raise his eyebrows bilaterally, puff out his cheeks, and subjectively noted more movement in his face. He continued to improve and as of Day +42, the patient was at neurological baseline without further facial weakness or anisocoria.
Discussion
In the current case we present a novel case of acute bilateral facial nerve palsy following CAR-T therapy, broadening the scope of reported neurologic side effects of the treatment.
Several features are supportive of a CAR-T related cause: CSF showed an increased WBC count which was lymphocyte predominant and suggestive of a T cell mediated mechanism. Given that there is currently no diagnostic test to prove CAR-T cell mediated neuropathy, other plausible causes of facial nerve palsy were ruled out, including viral infection and direct MM involvement. The onset of disease was not associated with any viral prodrome, and the patient was on prophylactic valacyclovir, decreasing the likelihood of HSV infection. Although all viral infections cannot be conclusively ruled out, a thorough and timely infectious workup was performed on blood and CSF and was entirely negative for tested viral DNA. Structural and malignant causes of facial nerve palsy are unlikely to present acutely and bilaterally, especially with clinical resolution over weeks. MM infiltration of the nervous system was a possibility in this patient. However, this was not apparent on CSF flow cytometry and would be unlikely develop in association with escalating therapy. It is unclear whether the isolated right pupillary mydriasis was evidence of additional cranial nerve involvement or may have been due to other causes. Given the lack of prescribed anticholinergic medications, an immune process seems most likely although cannot be confirmed in the present cases. A presentation of acute inflammatory demyelinating polyneuropathy with cranial nerve involvement is felt less likely given the presence and persistence of deep tendon reflexes on bedside testing. Given the above, CAR-T cell related facial nerve palsy remains the most plausible diagnosis.
The patient was treated with a course of dexamethasone from day +1 to day +8, wherein there was no change in presentation. They then saw complete remission of symptoms by Day +42. Steroids have been an effective measure in treating ICANS.4,5 Corticosteroids are also a critical early treatment of idiopathic facial nerve palsy, resulting commonly in complete resolution within 6 months. 6 In our case, it is unclear whether the use of steroids led to reversal of symptoms. Given that corticosteroids are effective in treating ICANS and idiopathic facial nerve palsy, we believe that the steroids were an appropriate treatment avenue and did contribute to complete reversal of symptoms.
Proposed mechanisms of CAR-T neurotoxicity are an ongoing focus of the immunotherapy community. There has been suggestion of a breakdown of the blood-brain-barrier secondary to gross endothelial activation, permitting CNS infiltration of circulating lymphocytes. 7 ICANS may be due to an uncontrolled cytokine response, like CRS. Increased relative CNS-serum cytokine ratio has been demonstrated in primate models following infusion of B-cell directed CAR-T cell therapy. 8 Patients with more extreme CRS response, and by association a known increased cytokine response, have been associated with increased rates of ICANS. 5 The workup of our patient did not include CSF cytokine levels, however several publications have noted increased cytokine and immune cells present in CSF of ICANS patients.5,9 However, others report results inconsistent with a neuro-inflammatory mechanism. Pathology reported from an autopsy conducted on a patient with CAR-T therapy mediated encephalopathy, few T cells and no CAR-T cells were found in brain parenchymal tissue. 10 Though the exact mechanism is unclear, it is evident that this patient had a higher relative concentration of T cells in the spinal fluid, which is consistent with the immune-mediated mechanisms proposed. By these proposed mechanisms, CAR-T cell therapy results in a complex neuro-inflammatory environment that may cause neuropathy.
This case is a detailed documentation of the development of facial nerve palsy in the temporal context of CAR-T cell therapy without concomitant ICANS. This case is limited in that current diagnostic tools are unable to exhaustively exclude a viral or idiopathic cause. Facial nerve palsy has not to date been documented in CAR-T therapy, however, does impact patient quality of life. Additionally, neurotoxicity is uncommonly seen in CAR-T therapy targeting MM, emphasizing the importance of this presentation. The occurrence of cranial neuropathy in the context of CAR-T cell therapy is important when considering neurotoxicity in immunotherapy and may imply a similar mechanism in idiopathic facial nerve palsies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
