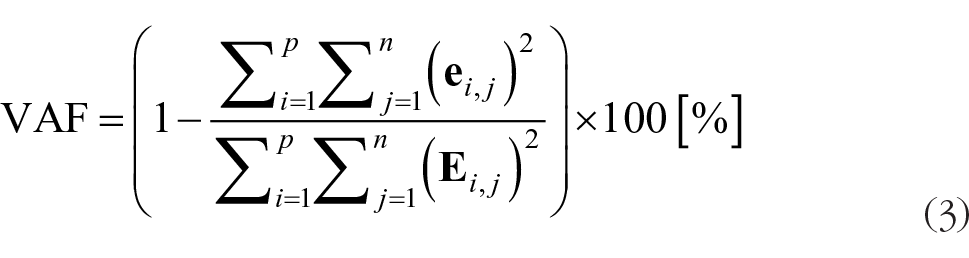Abstract
Background:
Shoulder pain, known as swimmer’s shoulder, is the most common injury for swimmers. Studies that have analyzed muscle activity have focused on the shoulder joint. However, the whole-body muscle coordination of swimmers with swimmer’s shoulder is not clear, although swimming requires movements of the upper limbs, trunk, and lower limbs to obtain propulsive force. This study investigated differences in muscle coordination between swimmers with and without swimmer’s shoulder during the front crawl and backstroke using muscle synergy analysis.
Hypothesis:
Swimmers with swimmer’s shoulder have muscle synergies differing from those without it.
Study Design:
Case-control study.
Level of Evidence:
Level 4.
Methods:
A total of 20 elite swimmers who regularly swam front crawl and backstroke were included (swimmer’s shoulder, n = 8; control, n = 12). Muscle synergy data were analyzed using the nonnegative matrix factorization method and compared between groups.
Results:
For both front crawl and backstroke, there were 2 synergies in the control group and 3 synergies in the swimmer’s shoulder group. During recovery, the control group showed coordinated triceps brachii, serratus anterior, upper trapezius, lower trapezius, internal oblique, and external oblique muscles activities; however, in the swimmer’s shoulder group, the contribution of the upper limbs decreased and only that of the trunk muscles increased.
Conclusion:
A comparison of muscle coordination during the front crawl and backstroke performed by swimmers with and without swimmer’s shoulder revealed that coordination differed during the recovery phase. During both front crawl and backstroke, the swimmer’s shoulder group could not maintain coordination with the upper limb when the trunk rolled, and split synergy was formed between the upper limbs and trunk.
Clinical Relevance:
Because coordination of the upper limbs and trunk is important during the recovery phase of front crawl and backstroke, swimmer’s shoulder rehabilitation should introduce exercises to improve their coordination between the upper limbs and the trunk.
Shoulder pain is the most common injury that occurs in swimming 11 ; shoulder pain that occurs in swimmers is known as swimmer’s shoulder. Elite collegiate swimmers swim 14,000 m daily and must rotate their shoulder joints more than 2500 times, 24 which inposes a chronic and heavy load on the shoulder joint. Most risk factors for shoulder pain experienced by swimmers have been identified by measuring the strength and range of motion of the shoulder joint on land rather than in the water. 31 Studies on factors related to swimmer shoulder pain have frequently been retrospective, reporting high laxity and instability, 29 specific shoulder range of motion, 40 reduced endurance, 32 tendencies of reduced internal rotation strength, 17 lack of scapular stability, 18 greater posterior humeral head position, 18 and lack of core stability. 32 Evaluations on land alone cannot identify and prevent chronic shoulder pain in the water.
Several studies have performed motion analyses of joint shoulder movement during swimming in water. Swimmer’s shoulder is a painful syndrome of the anterior shoulder induced by repetitive impingement of the rotator cuff beneath the coronal acromion arch. 8 Typically, this diagnosis is labeled impingement syndrome but, as the mechanism may vary, other terms have been proposed, including rotator cuff disease, rotator-cuff-related pain, and subacromial pain syndrome.1,2 In swimming, repeated stress from shoulder movements during the butterfly and front crawl techniques may cause impingement of the supraspinatus and biceps tendons, leading to rotator cuff lesions.4,43 Hand positioning and breathing techniques are also known to influence shoulder movements during these events.37,38,42,43 Furthermore, rotation of the body is related to shoulder pain from disciplines requiring body rotation, such as front crawl and backstroke.22,41 The mechanisms described also suggest that asymmetric body rotation may contribute to the development of shoulder pain.37,38
A few studies that have examined swimmers with shoulder pain using electromyography (EMG) were conducted in the 1990s. 28 These studies reported muscle activity around the shoulder joint characteristic of swimmers with shoulder pain.21,25,28 Recently, swimming records have improved; therefore, the factors that cause shoulder pain may have changed. In addition, previous studies have focused on muscles around the shoulder joint23,27,28; however, swimming requires movement of the trunk and upper and lower limbs to generate propulsion. Therefore, muscles surrounding the trunk may also affect the occurrence of shoulder pain. Evaluating the coordination of muscles in the trunk and upper limbs during swimming may help identify risk factors that cause shoulder pain in swimmers.
Motor coordination can be assessed using muscle synergy analysis, a tool that provides insight into basic motor control science and motor coordination strategies while quantifying functional deficits in pathological conditions. Nonnegative matrix factorization (NMF) analysis comprises a group of algorithms often used for muscle synergy analysis. 13 Based on EMG data, NMF can be divided into 2 components: muscle synergy (weighting for each muscle) and temporal activation patterns. Muscle synergies indicate muscle coordination, the correspondence of weight to the contribution of each muscle, whereas temporal activation patterns indicate the activation timing of muscle synergy. 44 The use of muscle synergy analysis for sports studies has increased recently3,9,25,30 and has become mainstream for evaluating muscle coordination. However, in swimming, although muscle synergy analyses have been performed for breaststroke and dolphin kick,16,36 no injury-related studies have been conducted on swimming technic involving body rotation.
During this study, we focused on front crawl, which accounts for 80% of a typical single practice session for elite swimmers, 39 and backstroke, which is associated with a high prevalence of shoulder pain. 15 This study aimed to determine whether the muscle coordination of the upper extremity and trunk during the front crawl and backstroke of elite swimmers differed between those with and without shoulder pain. We hypothesized that swimmers with shoulder pain would have differing muscle synergies to those without shoulder pain, and that swimmers with shoulder pain would have poor upper limb and trunk coordination.
Methods
Participants
A total of 20 male swimmers participated in this study. They were classified into control (n = 12) and swimmer’s shoulder pain (SS) groups (n = 8). All participants were elite swimmers at the national level who practiced with a collegiate swimming team 10 times per week. All were level 2 or 3 swimmers by the definition of Ruiz-Navarro et al. 26 The control group inclusion criterion was no previous shoulder pain or shoulder injury during their athletic career. The control group exclusion criteria were current musculoskeletal injuries of the upper extremity, neurological impairment of the upper extremity, previous surgery of the upper extremity, or any reported pain or discomfort of the shoulder.
The SS group inclusion criteria were a primary clinical diagnosis of swimmer’s shoulder by an orthopaedic surgeon, defined as a primary complaint of shoulder pain, for >3 months, at an intensity of >3 on an 11-point numerical rating scale (with 0 indicating no pain and 10 the worst pain imaginable). The SS group consisted of 8 patients with rotator cuff injuries (6 supraspinatus and 2 subscapularis injuries) and a recent history of shoulder pain. Exclusion criteria were previous upper extremity surgery, pain that interfered with swimming during the experiment, and other musculoskeletal or neurological conditions that could affect swimming. All participants did not miss practice and were at a level to participate in competition.
No significant differences in anthropometric characteristics were observed between the 2 groups. The sample size for this study was set with reference to a previous study that compared muscle synergy during breaststroke swimming in 8 novices and 8 elite swimmers and found significant differences. 36 Participants were fully informed of the risks, benefits, and stresses of the study, and written informed consent was obtained from all participants. The study was conducted in accordance with the Declaration of Helsinki and approved by the University Research Ethics Committee (2016-267).
Experimental Setting
The experiment was conducted in a 50-m indoor pool. After a 15-min warm-up that included low- to moderate-intensity aerobic exercise and drills, including lower limb movements, participants performed 25 m of front crawl and backstroke at the pace of a 100 m race. The trials were conducted randomly, and the participants began each swim with a wall kick. Two cameras (High-Speed Camera 1394; DKH Inc) were placed 10 and 15 m from the wall, captured the motion of swimmers in the sagittal plane, and recorded underwater images at a sampling rate of 200 Hz. Wireless light-emitting diode markers (Kirameki; Nobby Tech Inc) were placed on the styloid process, lateral epicondyle of the humerus, acromion, and greater trochanter on the right side in the control group and on the injured side in the SS group to separate the phases of the swimming cycle. One swimming cycle was defined as the time from when the right or injured side entered the water to when the same hand entered the water again. One swimming cycle was assumed to be 100%, with 3 phases (early pull-through, late pull-through, and recovery phase), and the data intervals per phase were recorded. The underwater phase with the hand in the water were defined for each participant. Early pull-through started when the hand entered the water and ended when the humerus was perpendicular to the water’s surface. Late pull-through started after early pull-through and ended when the hand left the water. The recovery phase began when the hand exited the water and ended when the hand entered the water. These phases were based on previous research. 28
EMG Data Analysis
EMG data were measured using a wireless EMG system (Biolog2; S&ME Inc), which recorded 11 muscles on the right side of the body in the control group and on the side of the injured shoulder in the SS group. The positions of the electrodes were determined using SENIAM as follows 7 : biceps brachii (BB), triceps brachii (TB), latissimus dorsi (LD), serratus anterior (SA), pectoralis major (PM), upper trapezius (UT), lower trapezius (LT), internal oblique (IO), external oblique (EO), rectus abdominal (RA), and erector spinae (ES). The electrodes were placed with reference to previous studies.7,20,21 Before surface electrode attachment, the skin was rubbed with a skin abrasive and alcohol to reduce impedance. The electrodes were attached parallel to the impedance of the muscle fibers to a level of <2 kΩ. Disposable Ag/AgCl surface electrodes were used in pairs (BlueSensor N-00-S; METS Co), and the sampling frequency was set to 1000 Hz. Using the methodology of Martens et al, 14 the electrodes were waterproofed by covering them with water-resistant tape.
To synchronize the video and EMG data, a synchronizer (PTS-110; DKH Inc) was connected to both trigger channels. Video and EMG data were recorded simultaneously. The recorded EMG data were analyzed using biomedical software (BIMUTAS-Video; Kissei Comtec Co). Three swimming cycle measurements were analyzed. The raw data were bandpass filtered (fourth-order Butterworth) between 20 and 450 Hz to reject signal noise. The full wave rectified and linear envelope signals for each muscle were obtained by zero-lag Butterworth low-pass filtering (fourth-order cut-off frequency of 20 Hz) of the fully rectified EMG signals. The EMG data were normalized relative to each muscle’s associated peak value data and interpolated to 201 time points. Muscle synergy studies often interpolate data to 100 or 101 points; however, because sports movements are quick, with multiple changes at a single point, the interpolation of our data was based on a study by Turpin et al, 35 who interpolated muscle synergy data during rowing to 200 points.
Muscle Synergies
EMG data were normalized and interpolated using MATLAB (MATLAB R2020; MathWorks, Inc). For muscle synergy analysis, NMF was performed for the preprocessed EMG dataset using the algorithm described by Lee and Seung.
12
The matrix of EMG signals was factorized into 2 components: muscle synergy vectors (
where
The VAF was defined as 100 × the coefficient of determination from the uncentered Pearson correlation coefficient. 34 We defined the standard as VAF >90%.
VAF was calculated using the following equation:
where i ranged from 1 to p and j ranged from 1 to n. Therefore, in this study, i was set from 1 to 11, and j was set from 1 to 201. These methods followed those used during previous studies. 16
Statistical Analysis
The Shapiro-Wilk normality test with Lilliefors correction was also used to assess the normality of the data for the mean swim velocity and the percentage of each phase. Swim velocity was calculated from the time at the 10- and 15-m passage points. An unpaired t test was performed to compare the mean swim velocity and the percentage of each phase between groups. Effect sizes were calculated using Cohen’s d for the unpaired t test. The evolution of VAF with the number of synergies extracted was compared between the 2 populations using a repeated measures analysis of variance. We performed an unpaired t test for each muscle synergy vector to compare individual muscle weightings between groups. These comparisons were performed using IBM SPSS Statistics for Windows version 24 (IBM Corp). Activation patterns were compared between groups using statistical parametric mapping (MATLAB R2020; MathWorks, Inc). Statistical significance was set at P < 0.05.
Results
Individual Data
All data are presented as mean and standard deviation. The control group had an age of 20 ± 1 years, a height of 1.8 ± 0.1 m, a body mass index (BMI) of 22.5 ± 1.1 kg/m2, swimming experience of 13 ± 2 years, and FINA point score of 764.0 ± 43.5 points, level 3 by the classification of Ruiz-Navarro et al. 26 The SS group had an age of 21±2 years, the height of 1.8 ± 0.1 m, BMI of 23.5 ± 1.3 kg/m2, swimming experience of 13±4 years, and FINA Point Score of 779.0 ± 29.0 points, level 2 or 3 by the classification of Ruiz-Navarro et al. 26
Table 1 presents the kinematic outcomes of front crawl and backstroke swimming. Swimming velocities of front crawl were 1.75 ± 0.09 1.78 ± 0.09 m/s, and of backstroke were 1.53 ± 0.09 and 1.58 ± 0.10 m/s, for the control and SS groups, respectively. The rates of each phase during 1 cycle of front crawl were as follows: during the early pull-through phase, 44.2 ± 7.6% (mean ± SD) and 40.1 ± 5.0%; the late pull-through phase, 22.0 ± 4.0% and 20.2 ± 3.1%; and the recovery phase, 34.1 ± 5.6% and 39.1 ± 4.3% for the control and SS groups, respectively. The recovery phase rate during the front crawl was significantly higher in the SS group than in the control group (P < 0.05) (Table 1).
Kinematic variables measured during the front crawl and backstroke swimming
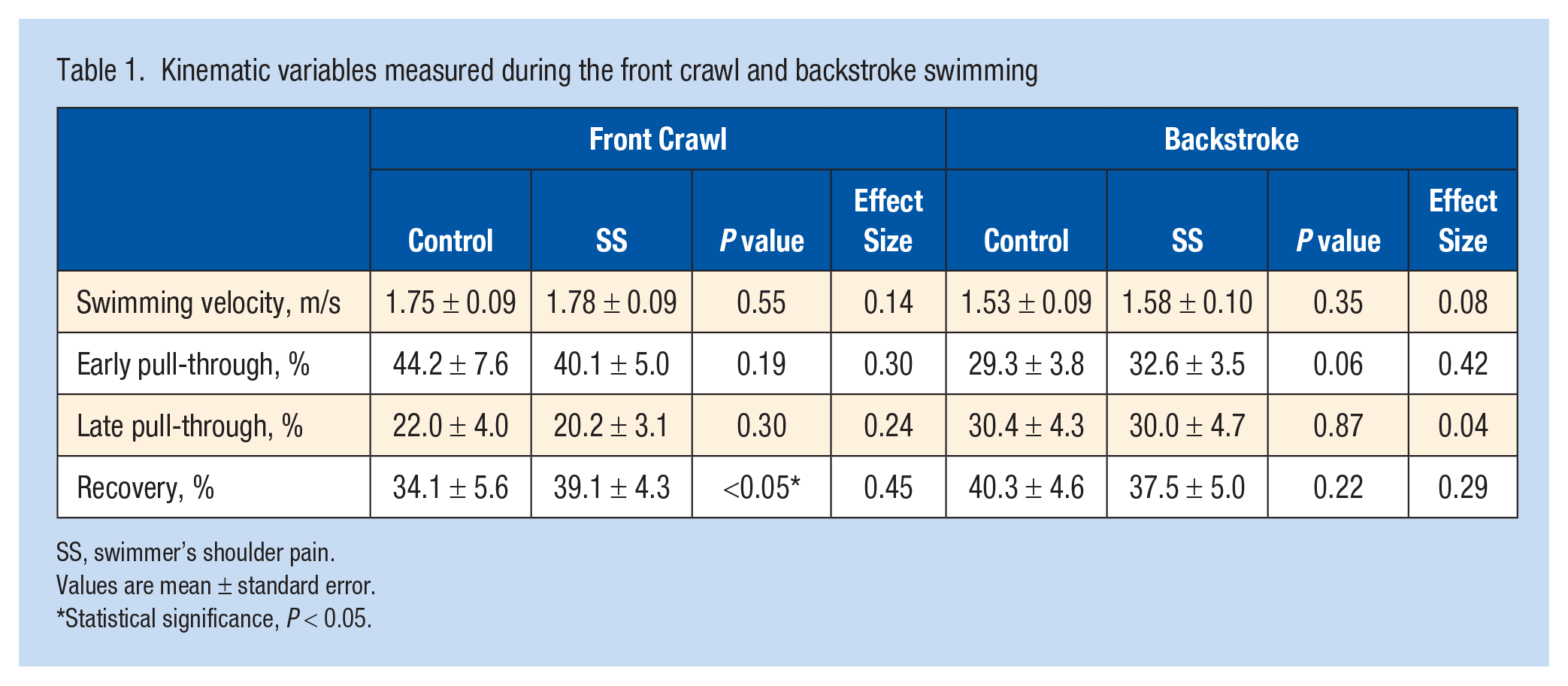
SS, swimmer’s shoulder pain.
Values are mean ± standard error.
Statistical significance, P < 0.05.
During backstroke, the rates of each phase during 1 cycle were as follows: during the early pull-through phase, 29.3 ± 3.8% and 32.6 ± 3.5%; the late pull-through phase, 30.4 ± 4.3% and 30.0 ± 4.7%; and during the recovery phase, 40.3 ± 4.6% and 37.5 ± 5.30% for the control and SS groups, respectively. No significant differences in each phase were observed between groups (Table 1).
The individual EMG patterns for each muscle and each population are depicted in Figures A1 and A2 in the online Appendix (available in the online version of this article), which show representative recordings of the EMG signals of 11 subjects performing front crawl and backstroke. Visual confirmation showed no clear artifacts in the recorded EMG signals for front crawl and backstroke.
Muscle Synergies: Dimensionality
The dimensionality analysis from the concatenation of all participants revealed that 2 to 3 muscle synergies were sufficient to reconstruct muscular activation for both groups. Appendix Figure A3 (available online) describes the relationship between the number of synergies and VAF. The number of synergies was the lowest, at which VAF exceeded 90%. The VAF exceeded 90% in all muscles at 2 synergies in the control group and at 3 synergies in the SS group. Therefore, the number of synergies was 2 in the control group and 3 in the SS group (Figure A3).
Muscle Synergies Describing Intermuscular Coordination During Front Crawl and Backstroke
Front Crawl
Figure 1 shows the synergy of front crawl swimming in the control and SS groups. Synergy 1 was active during 40% of 1 cycle of the swimming motion and acted during the early pull motion. Synergy 2 was active in 65% to 100% of the control group and acted on recovery; in the SS group, it was active in 60% and acted from late pull to early recovery. The activation patterns were smaller in the SS group than in the control group in 0% to 18% (P = 0.09) and 81% to 100% (P = 0.06) of the swimming cycle. Synergy 3 was present only in the SS group and was active during 20% to 80% of 1 cycle, likely acting from late recovery until after hand entry (Figure 1).
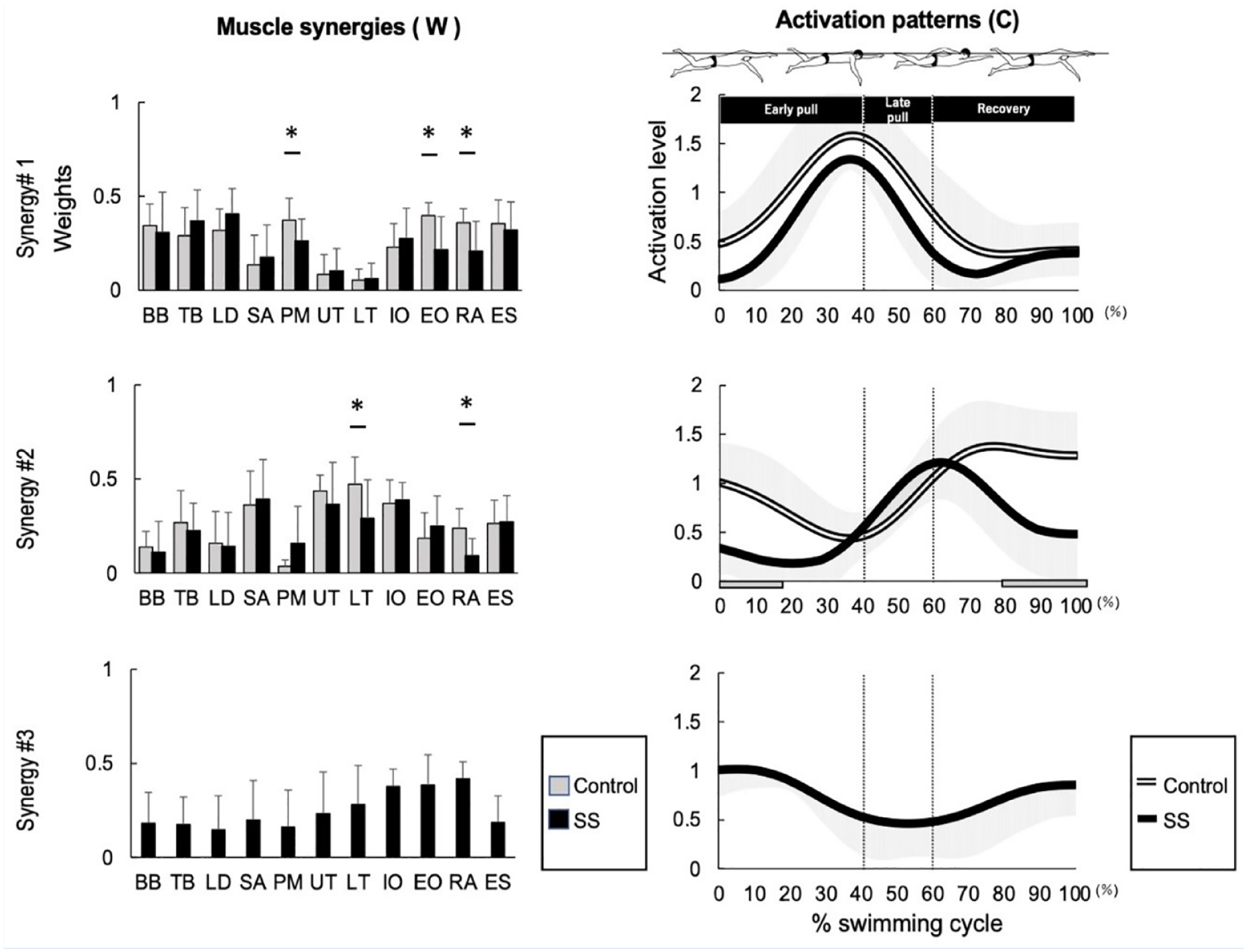
Comparison of muscle synergy during front crawl between the control and SS groups. The muscle synergy vectors are shown on the left side of the figure and are aligned to the corresponding activation coefficient. The synergy activation coefficients are shown on the right side of the figure. BB, biceps brachii; TB, triceps brachii; LD, latissimus dorsi; SA, serratus anterior; PM, pectoralis major; UT, upper trapezius; LT, lower trapezius; IO, internal oblique; EO, external oblique; RA, rectus abdominal; ES, erector spinae.
Synergy 1 was active during the early pull phase, and muscle synergy showed higher contributions from BB, TB, LD, PM, IO, EO, RA, and ES. Significantly different muscle synergy vectors were (for the control and SS groups, respectively) as follows: the PM, 0.37 ± 0.12 and 0.26 ± 0.12 (P = 0.01); the EO, 0.40 ± 0.07 and 0.22 ± 0.18 (P < 0.01); and the RA, 0.36 ± 0.07 and 0.21 ± 0.16 (P = 0.01). Synergy 2 was active from late pull to recovery, and TB, SA, UT, LT, IO, and ES muscle synergies had high contributions. Significantly different muscle synergy vectors were (for the control and SS groups as follows, respectively) as follows: LT, 0.47 ± 0.14 and 0.29 ± 0.21 (P = 0.01); and RA, 0.23 ± 0.10 and 0.09 ± 0.14 (P = 0.01). Synergy 3 was present only in the SS group, was active from late pull to early recovery, and the muscle synergies with higher contributions were IO, EO, and RA. There were no significantly different vectors between groups.
Backstroke
Figure 2 shows the synergy of backstroke swimming in the control and SS groups. Synergy 1 was active during approximately 30% of 1 cycle of the swimming motion and acted as an early pull motion. Synergy 2 was active in approximately 60% to 100% of the control group and acted on recovery. In the SS group, it was active in approximately 50% to 60% and acted on the late pull to early recovery. The activation patterns were smaller in the SS group than in the control group in 0% to 13% (P = 0.01) and 75% to 85% (P = 0.02) of the swimming cycle. Synergy 3 was present only in the SS group and was active during approximately 60% of 1 cycle, likely because of early recovery. Figure 2 depicts the extracted synergies of the backstroke and compares the control and SS groups. Synergy 1 was active during the early pull phase, and muscle synergies showed higher contributions from the BB, TB, LD, PM, IO, EO, RA, and ES. Synergy 2 was active from late pull to recovery, and the muscle synergy of TB, SA, UT, and LT had high contributions. The synergy vectors for the LT were significantly different: 0.40 ± 0.10 and 0.23 ± 0.14 (P = 0.01) for the control group and SS group, respectively. Synergy 3 was present only in the SS group, was active from late pull to early recovery, and had muscle synergies with higher contributions from the IO, EO, and ES.
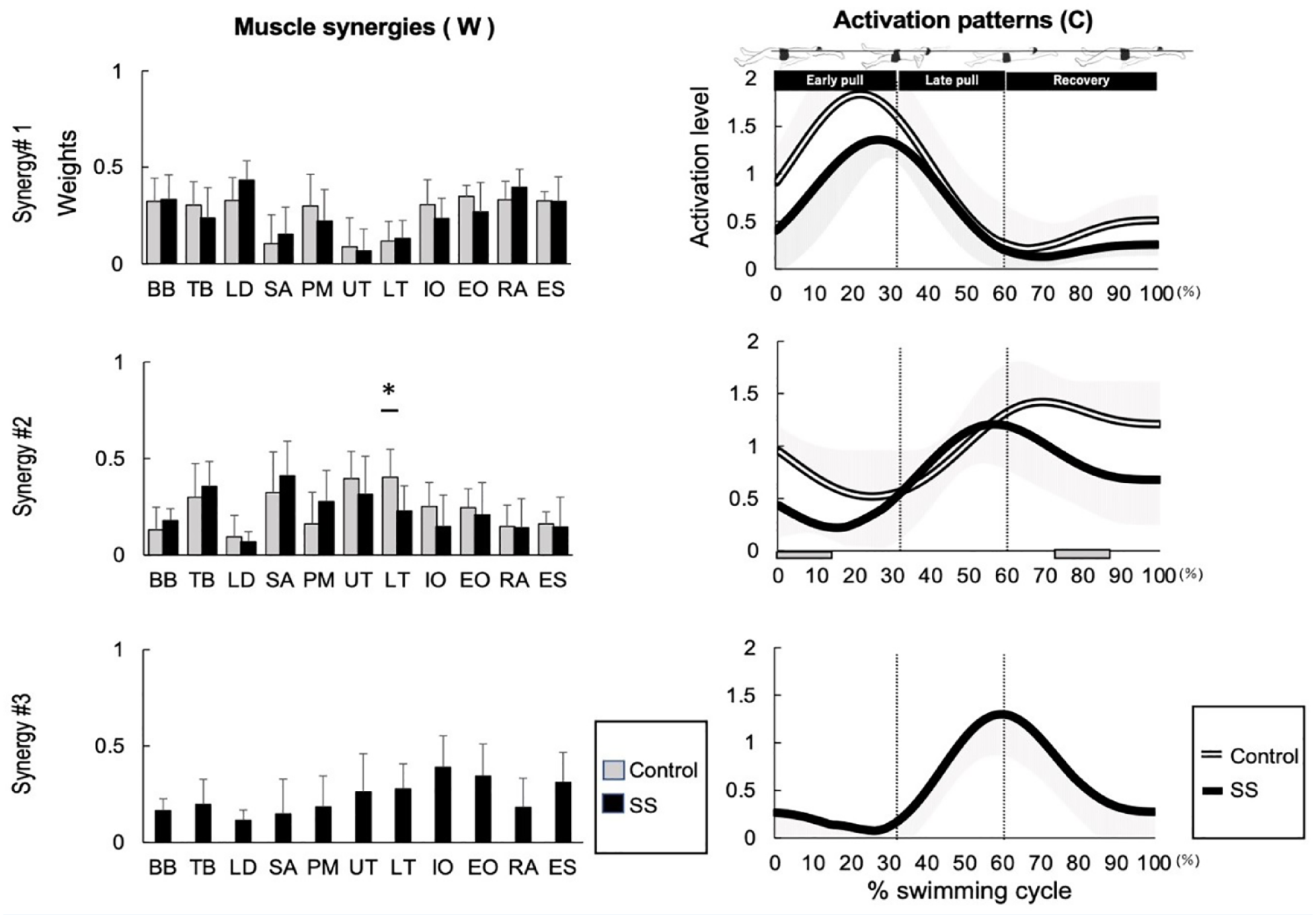
Comparison of muscle synergy during backstroke between the control and SS groups. The muscle synergy vectors are shown on the left side of the figure and are aligned to the corresponding activation coefficient. The synergy activation coefficients are shown on the right side of the figure. BB, biceps brachii; TB, triceps brachii; LD, latissimus dorsi; SA, serratus anterior; PM, pectoralis major; UT, upper trapezius; LT, lower trapezius; IO, internal oblique; EO, external oblique; RA, rectus abdominal; ES, erector spinae.
Discussion
We analyzed muscle synergy during front crawl and backstroke conditions and found that synergy differed between swimmers with and without shoulder pain. Swimmers with shoulder pain had more muscle synergy than those without (Figures 1 and 2).
Muscle Synergies With and Without SS During Front Crawl
Synergy 1 was involved in the early pull motion. During this phase, most propulsion is generated. 6 Moreover, the hydrodynamic leg sinking torque peaks during the first half of the underwater upper limb motion during the front crawl. 5 In the control group, the muscle contributions for PM, EO, and RA were larger than those of the SS group. Trunk flexors such as EO and RA can resist the subsidence torque of the legs by being activated by efferent contraction in the control group, but they may not be able to resist in the SS group. As a result, that burden on the shoulder joint may increase. Synergy 2 was involved during the recovery phase; however, synergy 2 was active throughout the recovery period in the control group. The SS group was active from the late pull phase to the early recovery phase and became less active at approximately 80% during the activation pattern. The muscle contributions of LT and RA were less than those of the control group. Impingement in the case of a swimmer’s shoulder occurs when the scapula is in an elevated position. 43 Because the LT can depress the scapula, we considered that the control group could perform the pull movement through the depression of the scapula by the LT, thereby avoiding the development of SS. The contribution of RA may have been larger in the control group to counter the leg sinking torque that the action of the upper limbs may cause.
Synergy 3 was present only in the SS group, and its activity was enhanced immediately before and after water entry. Synergy 3 had contributions from the IO, EO, and RA muscles, which are the rotators and flexors of the trunk. It is inferred that the SS group failed to maintain coordination with the upper limbs during trunk rotation and formed a split synergy between the upper limbs and trunk. During a kinematics study, we found that there was no difference in the shoulder joint angle during front crawl between swimmers with and without shoulder pain, but there was a difference in the rotation angle of the hip joint, suggesting that trunk movement and muscle activity influence shoulder joint injuries. 38 According to a review of SS, it was concluded that it is necessary to include the stability and kinetic chain of the trunk to prevent SS. 6 The results of the present study suggest that it is important to coordinate the upper limbs and trunk.
Muscle Synergies With and Without SS During Backstroke
During backstroke, synergy 3 was present only in the SS group (Figure 2). Synergy 1 was involved in the early pull, and the muscle synergies were the same in both groups. Synergy 2 was active during recovery. Only in the SS group was synergy 3 present during the timing from late pull to early recovery. In the control group, synergy 2 corresponded to the recovery period, and its activity was enhanced throughout the recovery period, and the muscle synergy for LT was larger than that in the SS group (P = 0.01). Because the recovery movement involves the same flexion of the upper limbs as the front crawl, the low activity of LT in the SS group may prevent the upward rotation of the scapula from being suppressed, leading to impingement. Synergy 3 had high contributions from the IO, EO, and ES and existed only in the SS group. Synergy 3 may have been formed because of insufficient upper limb elevation and trunk rotation coordination.
Practical Applications
To summarize the results of this research, during swimming techniques involving trunk rotation, such as backstroke and crawl, there is muscle synergy during which the contribution of the upper limb muscle group is low, and the trunk muscle group is high when trunk rotation is greatest. Therefore, during the rehabilitation of front crawl and backstroke swimmers, it is important not only to restore the shoulder function but also to enhance the linkage between the trunk and upper extremities. Specifically, it is advisable to introduce exercises that focus on contralateral movements (eg, right upper limb, trunk, and left lower limb) linked with the upper limbs to obtain a smooth rolling motion.
The study results bring to attention the debate on “the optimal swimming technique.” Coaches and therapists should know that swimmers with a shoulder pain history may swim with different muscle activity patterns compared with swimmers without a history of shoulder pain. Although the causal relationship between shoulder pain and different muscle synergy pattern is unclear, swimmers with shoulder pain might change the muscle activity pattern to adapt to the shoulder pain. Further prospective cohort studies are required to clarify the causal relationship between the differed muscle synergy pattern and shoulder pain. Synergy analysis can evaluate muscle coordination and may be useful for evaluating competitive swimming, which requires coordinated whole-body movements. In the future, the muscle synergy of each swimming technique should be clarified to apply it to training and rehabilitation or performance improvement and injury prevention.
Limitations
The major limitation of this study was the small number of participants. Therefore, it is only preliminary or pilot data. During this study, the groups were divided based on shoulder pain. Therefore, it is unclear whether the synergy in the SS group was caused by pain or whether this synergy pattern caused pain. Synergy is altered in the presence of pain. 19 However, there is a possibility that pain occurred because of this synergy. This needs to be clarified by a prospective cohort study. Furthermore, because there were only 2 underwater high-speed cameras, a complete motion analysis was not performed, and a 3-dimensional evaluation of motion was not possible. Therefore, during this study, motion analysis was used only for phase division and the calculation of swimming speed. If a detailed motion analysis could have been performed, then the mechanism of shoulder injury could have been investigated in more detail.
This study included elite participants. Therefore, the results of this study cannot be generalized to other populations, such as swimmers with different ability levels (juniors or masters). Therefore, caution should be exercised when interpreting the results of this and similar studies because the mechanism of shoulder injury may differ depending on ability level.
Conclusion
A comparison of muscle coordination during the front crawl and backstroke by swimmers with and without shoulder pain revealed that coordination differed during the recovery phase. In the SS group, during both front crawl and backstroke, the group with shoulder pain could not maintain coordination with the upper limbs when the trunk rolled, and split synergy was formed between the upper limbs and trunk. In rehabilitation for SS will require training to maintain coordination between the upper limbs and the trunk when the trunk rotates.
Supplemental Material
sj-docx-1-sph-10.1177_19417381231166957 – Supplemental material for Comparison of Muscle Coordination During Front Crawl and Backstroke With and Without Swimmer’s Shoulder Pain
Supplemental material, sj-docx-1-sph-10.1177_19417381231166957 for Comparison of Muscle Coordination During Front Crawl and Backstroke With and Without Swimmer’s Shoulder Pain by Yuiko Matsuura, Naoto Matsunaga, Hiroshi Akuzawa, Tomoki Oshikawa and Koji Kaneoka in Sports Health: A Multidisciplinary Approach
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.