Abstract
Background:
We sought to utilize a noninvasive technology to assess the effects of activity on Achilles tendon stiffness and define baseline Achilles tendon stiffness in female college athletes compared with nonathletes using tendon shear wave velocity as a marker for tendon stiffness.
Hypothesis:
Training status and exercise may affect Achilles tendon stiffness.
Study Design:
Prospective cohort study.
Level of Evidence:
Level 4.
Methods:
A total of 32 college-age female athletes were prospectively enrolled (n = 17 varsity athletes and n = 15 nonathletes). Demographic characteristics, activity level, and previous injuries were recorded. Sonographic shear wave elastography (SWE) was used to assess Achilles tendon shear wave velocity bilaterally for all subjects, both at baseline and after 2 minutes of exercise. Student t tests were used to compare the mean elastography measurements between participants stratified by athlete status and pre/postexercise stimulus. Analysis of variance (ANOVA) was used to compare the mean proximal, middle, and distal Achilles tendon elastography measurements.
Results:
As seen by a greater mean shear wave velocity (8.60 ± 1.58 m/s vs 8.25 ± 1.89 m/s; P = 0.02), athletes had stiffer tendons than nonathletes. Exercise stimulus decreased average tendon shear wave velocity (8.57 ± 1.74 m/s vs 8.28 ± 1.72 m/s; P = 0.05). Tendon shear wave velocity was greatest proximally and least distally with significant differences between each region (P < 0.001). In addition, there was a significant 2-way interaction between weekly training status and foot dominance (P = 0.01). Post hoc analysis showed that this result was due to differences in tendon shear wave velocity between the dominant and nondominant lower extremity in nonathletes (7.73 ± 2.00 m/s vs 8.76 ± 1.62 m/s; P < 0.001).
Conclusion:
Female varsity collegiate athletes have higher baseline Achilles tendon stiffness as measured by SWE compared with nonathletes. Mean tendon stiffness varies based on Achilles measurement location. SWE is a quick, cost-effective, and noninvasive imaging modality that can be used to evaluate tendon stiffness and elasticity.
Clinical Relevance:
SWE is an efficient and noninvasive imaging modality that can evaluate dynamic tendon stiffness and elasticity. SWE may be helpful to assess injuries in female college athletes and may play a role in risk stratification or clinical follow-up. In theory, SWE could be used to identify athletes with increased elasticity as a marker for potential risk for rupture in this population.
Sonographic shear wave elastography (SWE) is an established imaging modality that may prove to be a useful noninvasive tool for evaluating the musculoskeletal system, including tendons, ligaments, and soft tissues.26,32 SWE provides measurements in a targeted region of interest (ROI) under direct sonographic visualization. Elastography measurements are derived by differences in the shear wave velocity, which is dependent on tissue stiffness and elasticity.5,23,27,43,45 The underlying principle is that shear-waves propagate faster in stiffer and contracted tissues, and along the long axis of the tendon. SWE provides both an objective and quantitative evaluation of tissue stiffness and has been reported to have more reliable performance than alternative sonographic elastography techniques, specifically compression elastography. Further, previous studies have validated SWE in quantifying tendon-stiffness via measuring tendon shear wave velocity.5,8,9,15,34,35 SWE measurements can be obtained both quickly and dynamically and include the added portability and diagnostic utility of ultrasonography, features that make it ideal for point of care scenarios. Thus, SWE holds promise as a useful adjunctive functional imaging test in concert with conventional ultrasonography and may have potential utility in guiding tendinopathy treatments.10,15
Previous studies have reported variations in SWE tendon stiffness measurements in the presence or absence of tendinopathy and other risk factors. Several authors have shown that higher activity levels lead to increased tendon stiffness in the Achilles, patellar, and quadriceps tendons.6,33,38,40,45,46 Slane et al 42 found that tendon stiffness, as assessed by SWE, increased with patient age in the stretched position. In contrast, Petrescu et al 36 found no relationship between age, gender, body mass index or sport on Achilles tendon elasticity. Given these mixed results, it is unclear how these factors affect tendon elasticity. Although Achilles tendon injuries have a male predilection, more female athletes have consistently pursued higher level athletic endeavors, resulting in the need for increased female-specific research attention.20,25,29,39 Furthermore, female-specific tendon mean stiffness values with demographic characteristics that predispose them to tendon injury remains largely unknown. Our hypothesis was that training status and exercise may affect Achilles tendon stiffness.
Methods
Patient Selection
This study was approved by the institutional review board, and written informed consent was obtained from all participants. A total of 32 women from ages 18 to 25 years were enrolled (n = 17 varsity athletes and n = 15 nonathletes). Collegiate athletes were recruited with flyers and referral by athletic trainers. Nonathletes were recruited through on campus flyers and through social media. Exclusion criteria were (1) previous surgery on the Achilles’ tendon, (2) current pregnancy, (3) fluoroquinolone use, and (4) immunosuppressive therapy.
Data Collection
All participants completed a standardized questionnaire. Participant height (cm) and body mass (kg) were recorded. Participant foot dominance (right vs left) was recorded, as it has been reported previously that long-term exercise may increase dominant foot Achilles tendon thickness and stiffness. 46 Given the possibility that tendinosis may affect tendon stiffness, a 10-point Likert scale was used to assess baseline Achilles tendon pathology (0, no pain; 10, worst pain imaginable), with pain acting as a surrogate marker for tendinosis. In addition, evaluation of the Achilles tendon was performed with ultrasound (US) and presence of any US grayscale or color Doppler abnormality was documented. The average monthly exercise frequency was also recorded. Athletes participated in running-focused sports (lacrosse, soccer, track).
A validated SWE measurement protocol described by Payne et al 35 was used given its demonstrated reproducibility. Achilles tendon images were recorded with subjects lying prone with both feet hanging off the examination table with the Achilles tendon in a relaxed position; this positioning was used to remove tension from the Achilles tendon, which could falsely elevate its elasticity (Appendix Figure A1, available in the online version of this article). A generous standoff layer of US gel was used to help avoid compression that could alter tendon stiffness. Longitudinal scans were utilized as these have been reported to have higher reproducibility than transverse scans. 14 The optimal scan plane of the tendon was identified using grayscale US and placing the transducer perpendicular to the tendon fibers. A 10L4 linear, high-frequency transducer (Siemens, Sequoia) was utilized to obtain qualitative and quantitative shear wave velocity maps ranging from 0.5 to 10.0 m/s.13,21 SWE measurements were obtained in the Achilles tendon’s proximal, mid, and distal aspects (Appendix Figures A2 and A3, available online). The superior border of the calcaneus was used as a reproducible osseous landmark for the most distal measurement on all patients. Three shear wave velocities were recorded from the proximal aspect, 4 in the mid-aspect, and 3 in the distal third of the Achilles tendon. Thus, 10 ROIs were recorded along the midsubstance of the tendon (at 0.5-0.8 cm depth) on both the right and left Achilles tendon per subject, for a total of 640 measurements (Appendix Figures A2, A3, available online). Measurements were obtained by an experienced sonographer and an attending musculoskeletal radiologist with over 17 years of experience.
Bilateral Achilles tendon elastography measurements at baseline/rest and immediately postexercise were obtained. The exercise stimulus included 1 minute of continuous, methodical heel raises followed immediately by 1 minute of plyometric ankle jumps, similar to previous reports.16,18,24,28 The stimulus was used to place the Achilles under physiologic tension to test whether pretreatment with elastic deformation would alter the tendon shear wave velocity. After the stimulus was completed, postexercise Achilles elastography measurements were obtained immediately.
Data Analysis
As mentioned above, multiple shear wave measurements were obtained in the proximal, mid, and distal aspects of the Achilles tendon to account for minor measurement variation in SWE measurements. Specifically, 3 measurements were obtained in the proximal and distal regions, and 4 measurements were obtained in the midportion of the tendon. The SWE measurements were averaged in these 3 regions (proximal, mid, and distal) to calculate the mean regional tendon shear wave velocity (m/s).
Participant characteristics were compared using either Student t tests or chi-square tests for continuous and categorical variables, respectively. The mean tendon shear wave velocity and SD were calculated for each group: training status (athlete and nonathletes), tendon location (proximal, mid, and distal), exercise stimulus (pre and postexercise), and foot dominance (dominant and nondominant). A 2-way analysis of variance (ANOVA) was performed to assess the main effects of training status, exercise stimulus, foot dominance, and tendon region on the mean Achilles tendon shear wave velocity. In addition, 2-way interactions between training status and all other factors were also assessed. Higher-order analyses were omitted, given the relatively small sample size and diminishing statistical power. A Bonferroni post hoc test was performed to evaluate the mean Achilles tendon shear wave velocity differences based on the tendon location (proximal vs mid vs distal). An additional post hoc Student t test was performed to follow up the 2-way interaction between the mean Achilles tendon shear wave velocity based on foot dominance and training status. Statistical significance was considered if P < 0.05. Data were analyzed using IBM SPSS Statistics, Version 24.
Results
Demographics
Collegiate athletes and nonathletes had similar body mass, height, and foot dominance (P > 0.05). The participants were all nonsmokers. None had pre-existing comorbidities. Resting Achilles tendon shear wave velocity did not vary based on participant body mass (P = 0.19) or height (P = 0.35). There were no differences in pain score based on training status. Compared with the nonathletes, the athlete group exercised more frequently (≥5 times per week versus 3-5 times per week), for a longer duration (60 minutes vs 30-60 minutes), and at greater intensity levels (moderate-high vs light-moderate). The exercise frequency of nonathletes ranged from 1 to 20 days per month (Table 1).
Participant demographics
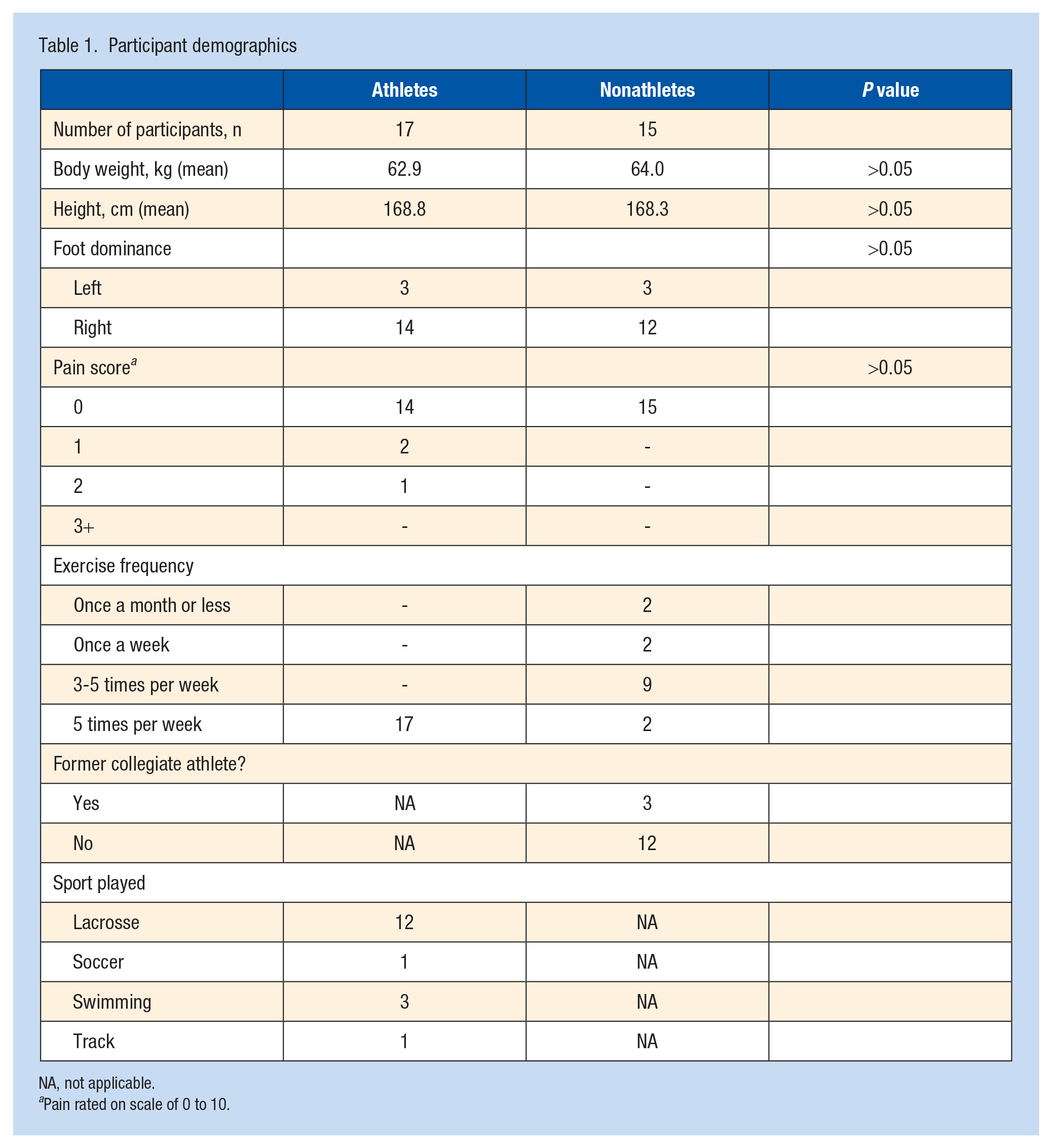
NA, not applicable.
Pain rated on scale of 0 to 10.
US Imaging
In both athlete and nonathlete groups, grayscale and color Doppler US evaluation of bilateral Achilles tendons pre and postexercise demonstrated no evidence of tendon pathology (tendinosis or tear or synovitis/inflammation (Appendix Figure A4, available online).
Two-Way ANOVA
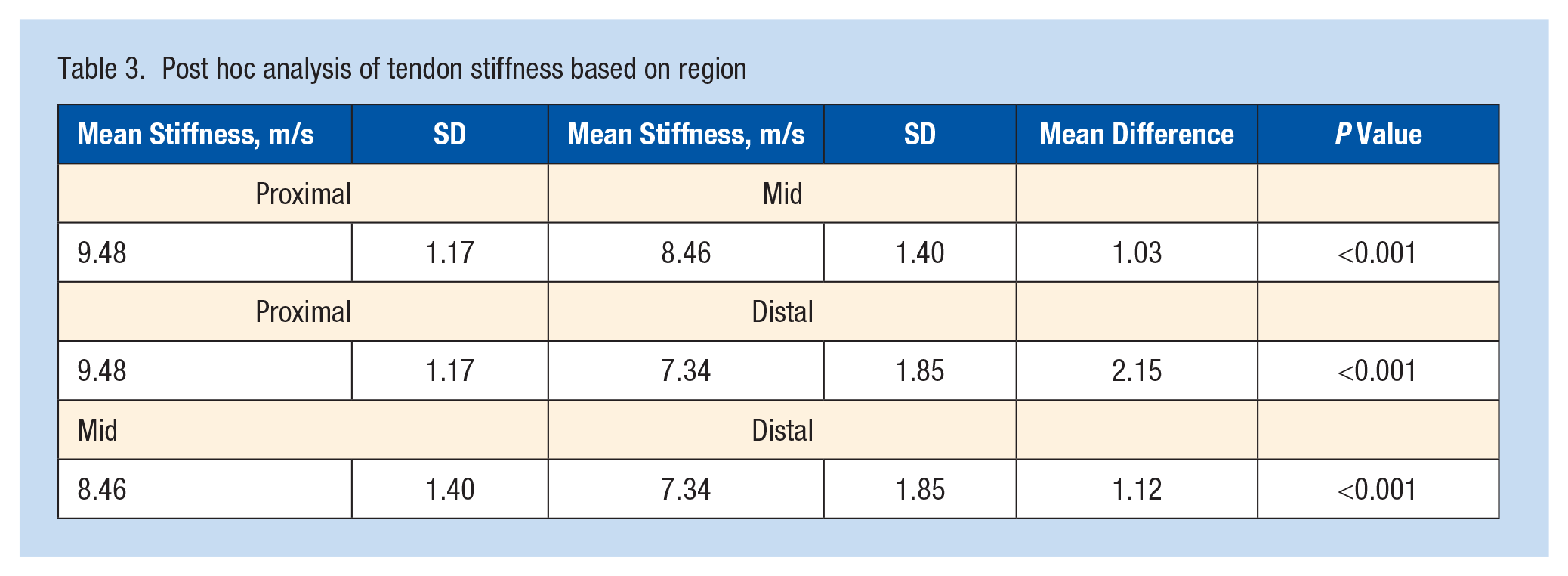
All main effects of training status, exercise stimulus, foot dominance, and tendon region were statistically significant (Table 2). Athletes had a greater tendon shear wave velocity than nonathletes (8.60 ± 1.58 m/s vs 8.25 ± 1.89 m/s; P = 0.02). The exercise stimulus decreased the tendon shear wave velocity (8.57 ± 1.74 m/s vs 8.28 ± 1.72 m/s; P = 0.05). The dominant lower extremity has a lower tendon shear wave velocity than the nondominant extremity (8.10 ± 1.83 m/s vs 8.75 ± 1.58 m/s; P < 0.001). Tendon shear wave velocity was also highest proximally and lowest distally, with significant differences between each region (Table 2 and Figure 1; P < 0.001).
Two-way ANOVA analysis of mean tendon shear wave velocity
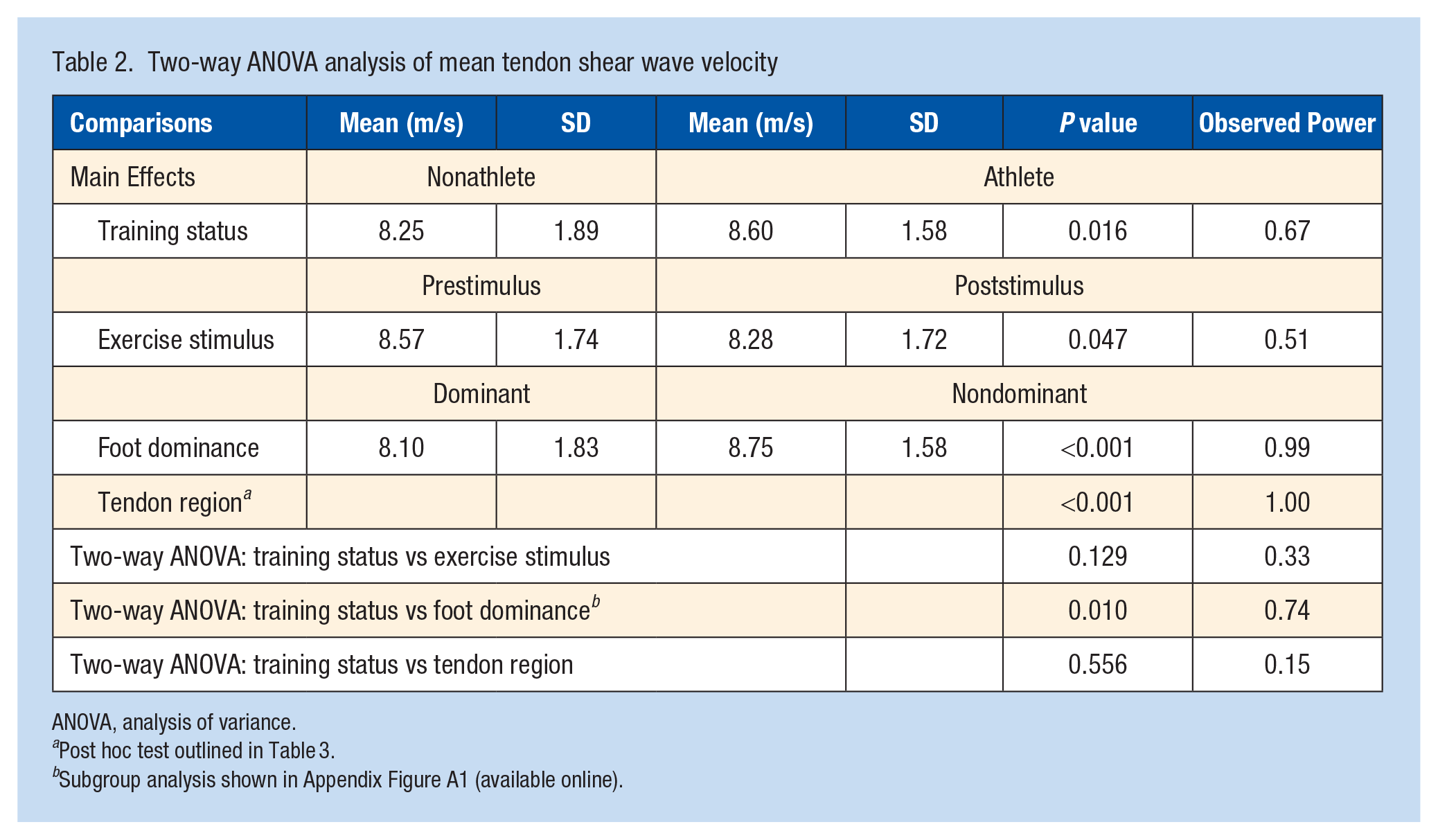
ANOVA, analysis of variance.
Post hoc test outlined in Table 3.
Subgroup analysis shown in Appendix Figure A1 (available online).
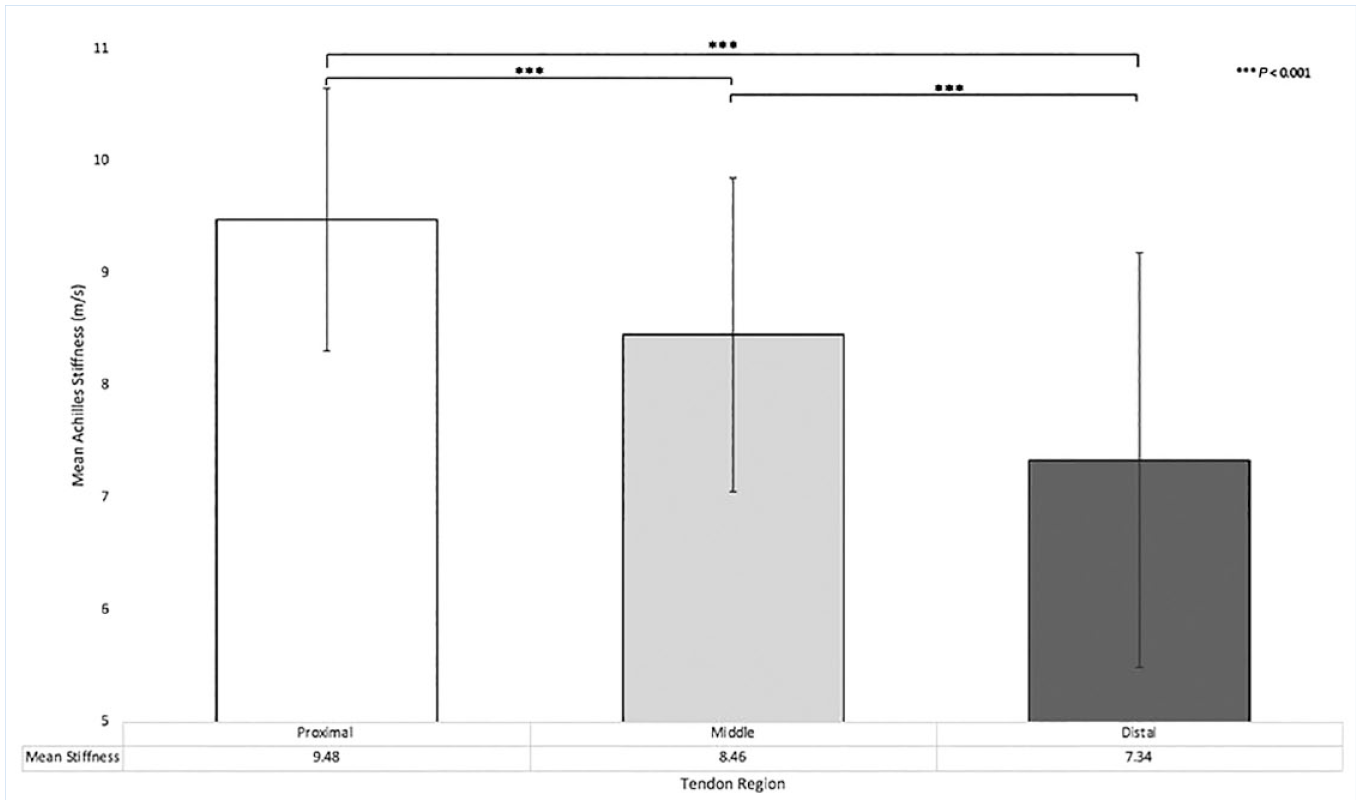
Achilles tendon stiffness based on region.
There was a significant 2-way interaction between training status and foot dominance (Table 1; P = 0.010). Post hoc analysis demonstrated that this result was due to differences in tendon shear wave velocity between the dominant and nondominant lower extremity in nonathletes (Figure 2 and Table 3); 7.73 ± 2.00 m/s vs 8.76 ± 1.62 m/s; P < 0.001), a difference that was not present in the athlete group (8.47 ± 1.60 m/s vs 8.74 ± 1.55 m/s; P = 0.23). No other 2-way interactions were present (Table 2; P ≥ 0.13).
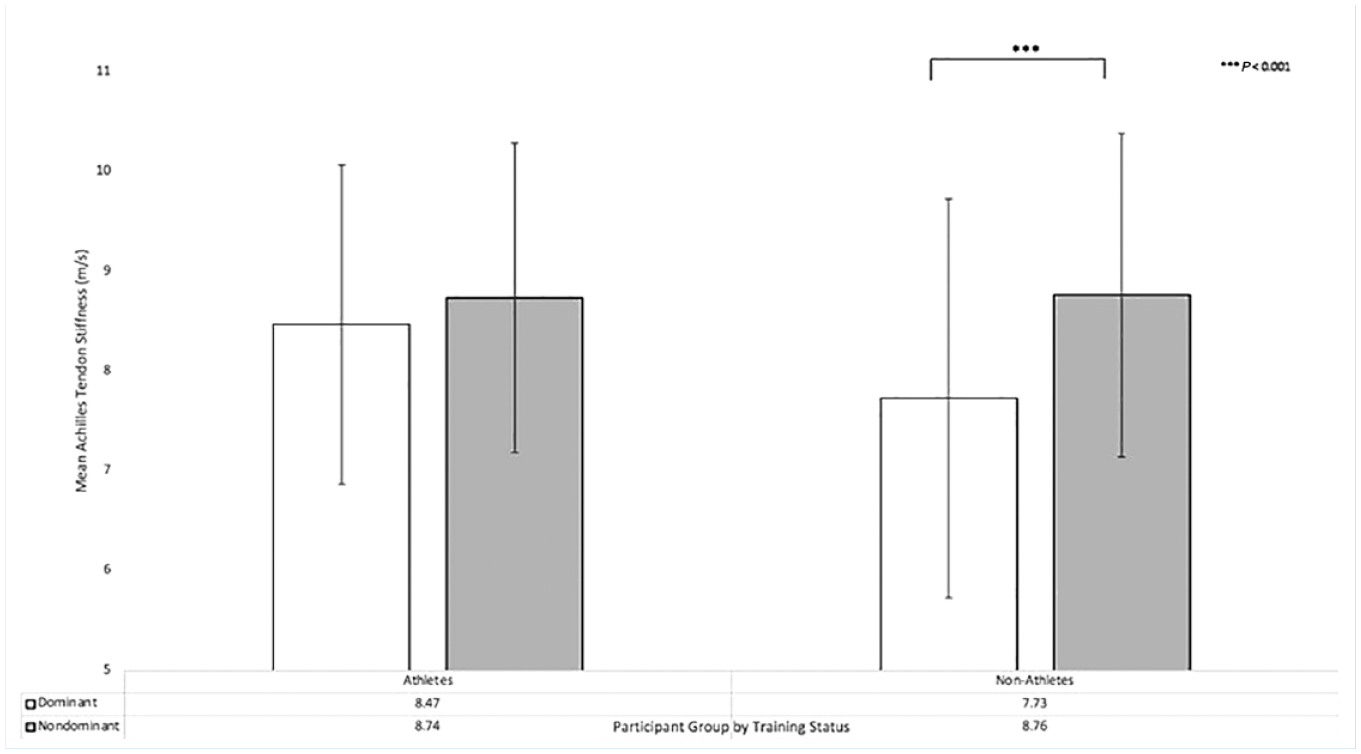
Extremity dominance on tendon stiffness by training status.
Post hoc analysis of tendon stiffness based on region
Discussion
Using tendon shear wave velocity as a marker for tendon stiffness, female varsity collegiate athletes have a higher baseline tendon stiffness than their nonathlete counterparts. Interestingly, mean shear wave velocity varies based on Achilles measurement location, which correlates with the known anatomic variation in the Achilles. Consistent with our hypothesis, training status and exercise affect Achilles stiffness, and tendon stiffness differs between dominant and nondominant lower extremities in nonathletes.
The cohort’s baseline mean Achilles tendon stiffness was slightly higher but comparable with that found in previous studies, which were not sex specific and were obtained using a different US model.9,11 Dirrichs et al, 11 reporting in kPA rather than m/s, cite a similar stiffness trend of 183.8 kPA in athletes and 103.6 ± 30.5 kPa in nonathletes, compared with the current study reporting 230 and 213.4 kPA, respectively. A previous study had an older cohort (mean age, 42 ± 13.4 years) with widely varied activity levels (novice to professional athletes). 11 Both age and activity level can influence mean Achilles tendon stiffness as measured by SWE.6,42 The current study provides a valuable addition to the growing body of literature utilizing SWE for noninvasive tendon evaluation, reporting pre- and postexercise values in female patients. It has been demonstrated previously that tendon stiffness, both at baseline and after stretching, in female athletes differs from that of their male counterparts. 2 In addition, previous studies report similar findings of an increased baseline tendon stiffness in female college athletes compared with controls. 19 As female athletes continue to compete at high competition levels, gender-specific research is valuable due to the known physiologic differences between male and female counterparts.7,28,42
The increase in tendon stiffness in female athletes compared with nonathletes may represent a physiologic response to training. The exercise stimulus subsequently decreased the tendon stiffness, which may be due to a change in tendon metabolic activity in response to mechanical loading.31,41 It is plausible that a decrease in tendon stiffness with exercise may be protective against injury. 22 The change in tendon stiffness based on these 2 variables should be taken into account when utilizing this imaging modality to evaluate Achilles tendon pathology.
Achilles tendon stiffness decreased from proximal to distal. This finding is consistent with previous studies, reporting a higher shear modulus in the proximal region of the Achilles than the distal region.1,21 Anatomically, the tendon varies based on blood supply and myotendinous connections; this may be relevant as the watershed areas are more prone to injury and have a diminished healing potential. 3 It also emphasizes the necessity for consistent measurement locations when utilizing this imaging modality.
There was also a significant 2-way interaction between training status and foot dominance due to differences in tendon stiffness between the dominant and nondominant lower extremity in nonathletes. This result is driven by a difference between foot dominance in athletes versus nonathletes. The observed difference may be secondary to a differential effect on stiffness in athletes, as the dominant foot may see strain in load-bearing exercises, leading to increased fatigue. It is also possible that this observation is secondary to a type 1 statistical error, thus should be reproduced in future studies.
While Achilles and patellar SWE have had multiple research applications, SWE has had limited clinical uptake.11,35 Few pilot studies have attempted to characterize baseline values for normal and torn Achilles tendons but are limited to small sample sizes. 4 Before US imaging, functional tendon capacity was limited to in vitro mechanical testing, 30 or in vivo sensor implantation. 37 More recently, Achilles tendon imaging predominantly involves conventional sonography (grayscale and color Doppler) and magnetic resonance imaging (MRI). 12 Imaging modalities like MRI nearly always provide static images that do not allow a dynamic clinical tendon evaluation. Given previous variable reports, gender- and age-specific reference Achilles tendon elastography values are necessary for SWE to have clinical utility.
There are notable limitations in both this study and this imaging modality. SWE obtains measurements from a ROI that can vary because of user selection and size limitations based on the US manufacturer; these factors add additional variation, making reproducibility challenging. SWE is sensitive to transducer pressure and probe orientation relative to the examined structure, thus may vary based on sonographer and technique. 44 This is notable since shear waves propagate faster along the long axis of healthy tendons than the short axis, thus probe orientation may affect results. The technical variations in SWE measurements is a previously cited concern regarding its clinical application.17,33 In addition, the sonographer and musculoskeletal radiologist were not blinded to participants status as an athlete or nonathlete.
Conclusion
Female varsity collegiate athletes have higher baseline Achilles tendon stiffness as measured by SWE compared with nonathletes. Mean tendon stiffness varies based on Achilles measurement location.
Supplemental Material
sj-docx-1-sph-10.1177_19417381231153657 – Supplemental material for Evaluation of Achilles Tendon Stiffness as Measured by Shear Wave Elastography in Female College Athletes Compared With Nonathletes
Supplemental material, sj-docx-1-sph-10.1177_19417381231153657 for Evaluation of Achilles Tendon Stiffness as Measured by Shear Wave Elastography in Female College Athletes Compared With Nonathletes by Alyssa D. Althoff, Kayleigh Vance, Megan Plain, Russell A. Reeves, Jennifer Pierce, Frank W. Gwathmey and Brian C. Werner in Sports Health: A Multidisciplinary Approach
Footnotes
The following author declared potential conflicts of interest: F.W.G. is employed as a consultant by Stryker, a paid speaker/presenter for Arthrex Inc., and receives publishing royalties, financial, or material support from Sunders/Mosby-Elsevier.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
