Abstract
Introduction
The concept of blue-carbon was first raised in 2009 to alude coastal ecosystems that capture and store (‘sequester’) organic carbon and include mangrove forests, tidal marshes, and seagrass meadows as the main players (Mcleod et al., 2011; Nellemann et al., 2009). Mangroves and other blue-carbon ecosystems have raised considerable interest for their potential contribution to the mitigation of anthropogenic CO2 emissions (Thomas, 2014). A major service of global impact provided by mangrove ecosystems is their role as carbon sinks (Chmura et al., 2003), allowing carbon to be stored in soil for centuries to millennia, as opposed to the decadal scales typical of carbon stored in plant biomass, which makes up the majority of organic carbon (Corg) stocks in terrestrial systems (Duarte et al., 2013; Mateo et al., 1997; Pedersen et al., 2011).
The evidence available to date suggests that carbon stocks in mangrove forests can be highly variable, and a diversity of factors influence carbon sequestration and storage, such as differences in soil type, infauna, hydrology, forest type, salinity, forest age, and geomorphological setting (Adame et al., 2017; Donato et al., 2011; Ouyang & Lee, 2020; Saintilan et al., 2013). Mangrove carbon stocks also vary between species. Species with higher primary productivity can have greater carbon stocks and higher rates of organic matter input into soil (Gress et al., 2017; Liu et al., 2014; Owers et al., 2016). Several studies have measured increases in soil Corg density and content with increasing age of individual stands (Chen & Twilley, 1999; Kathiresan et al., 2021; Ray et al., 2023; Wang et al., 2021; Wiarta et al., 2019), highlighting large local and regional differences in age-related soil Corg stock patterns (Alongi & Zimmer, 2024).
Several studies have reported positive relationships between aboveground and belowground organic carbon (Donato et al., 2011; Wang et al., 2013). Most carbon in mangrove ecosystems is stored belowground as peat (Donato et al., 2011), which, unlike shorter-lived aboveground biomass (AGB), often remains undecomposed for millennia (Ezcurra et al., 2016; McKee et al., 2007). The belowground biomass (BGB) of mangrove forests is affected by forest age (Ha et al., 2018; McKee, 2001), plant characteristics (Ezcurra et al., 2016; McKee et al., 2007) and environmental conditions (Cahoon et al., 2003; Otero et al., 2009). However, there is high uncertainty in the estimation of BGB and production, as most estimates have been derived primarily from allometric equations (Adame et al., 2017; Li et al., 2018).
Field research to estimate carbon stocks in mangrove ecosystems has focused primarily on study sites located in humid regions in temperate and tropical latitudes (Alongi & Dixon, 2000; Chmura et al., 2003; Donato et al., 2011), with relatively few studies in subtropical or arid regions (Adame et al., 2013; Ezcurra et al., 2016) where low rainfall, high temperatures, evapotranspiration, and soil conditions could affect carbon storage. The high importance of coastal ecosystems for carbon sequestration services in arid environments requires identifying the factors that account for their relatively small soil carbon stocks (Schile et al., 2017). In addition, the correct assessment of the spatial variation of mangrove forest carbon stocks is needed for ecosystem service valuations (Friess et al., 2022). The objectives of this study were 1) to determine the aboveground and belowground carbon stocks of the mangrove ecosystem and 2) to determine how the arid environment influences storage levels.
Methods
Study Area
The La Cruz Lagoon, located in the Gulf of California, in the town of Bahía de Kino, Sonora (111°52′52.65″ W– 28°47′14.76″ N), is a coastal lagoon with hypersaline conditions in summer, with low river water inputs (Glenn et al., 2006). It is of global importance due to its physical characteristics, location, and high species diversity, having been designated as Ramsar site number 2154, with an approximate area of 6,665.15 ha (Ramsar, 2013). The mangrove forest of the La Cruz estuary is dominated by black mangrove (Avicennia germinans L. Stearn), with little coverage of red mangrove (Rhizophora mangle L.) concentrated in the southwest, near the estuary mouth; in addition, there are associated species such as Batis maritima, Salicornia sp., and Distichlis palmeri (Meling-López et al., 1993). The maximum ambient temperature recorded in the locality is 48.5°C in July, and the minimum temperature is 15.0°C in February, with an annual precipitation of 161.3 mm (CONAGUA, 2016). The local climate is warm and dry with summer rainfall, BW (h’) hw according to Köppen’s classification (García, 2004). We delimited two sampling sites in the mangrove forest: Site 1 and Site 2 (Figure 1). Two monitoring units (MU1 and MU2) were established at each site. The study comprised an annual sampling period from May 2021 to April 2022. Sampling Sites in the Mangrove Ecosystem, La Cruz Lagoon, Gulf of California. Climograph.
Field Study
Physicochemical Parameters in Water and Soil
The physicochemical variables of surface and interstitial water were measured using a Hanna HI9828 multiparameter (Moreno-Casasola & Warner, 2009). For interstitial water, 4 piezometers were installed at a depth of 0.5 m, while surface water was collected during flood tide. At each site, we recorded salinity using the classification according to Cowardin et al. (1979) in practical units of salinity, redox potential (mV), temperature (°C), and pH.
Two soil samples were collected in each MU monthly from the upper 20 cm of soil using a 4″ diameter nucleator. The samples were transported to the laboratory, dried, and ground for texture analysis using the Bouyucos method (Klute, 1982), organic matter (% OM) by wet digestion (Walkley & Black, 1934), and total phosphorus content using the method of Bray and Kurtz (1945), and total nitrogen with the Kjeldahl method (SEMARNAT, 2000). In addition, two soil samples of known volume were collected in each MU (96 samples) to determine the bulk density and moisture content of the soil according to Moreno-Casasola and Warner (2009). Soil moisture content is the percentage of water stored in one gram of soil; a value of 100% means that 1 g of soil stores 1 g of water (Infante, 2011).
Mangrove Forest Structure
To estimate the structure of the mangrove forest, a 10 m × 10 m quadrant was established in each sampling unit (4 in total). The species identity, height, and diameter at breast height (DBH) of each tree were recorded at each quadrant; with this data, tree density, coverage, and basal area were calculated (CONABIO, 2013).
Primary Productivity Based on Litterfall
Four litter traps were placed in each MU, for a total of 16 traps (Moreno-Casasola & Warner, 2009). Litter samples were collected each month and transported to the laboratory. These samples were dried at 60°C, and each component was weighed, leaves, stipules, wood, branches, flowers, and fruits. Litter production values are ex-pressed as g·m2·month-1 and Mg·ha·year-1.
Belowground Root Biomass
Soil root samples were extracted at a depth of 45 cm with a stainless-steel corer measuring 0.45 m long by 11 cm in diameter, using the methodology of Adame et al. (2014). Three cores were extracted from each sampling unit (12 cores in total); each nucleus was divided according to depth strata: 0 cm to 15 cm, 15 cm to 30 cm, and 30 to 45 cm (Castañeda-Moya et al., 2011). The samples were refrigerated and transported to the laboratory, where they were immersed in water, separating floating living roots from dead roots (considered necromass) that sank to the bottom. Living roots were classified into three diameter classes: fine (<2 mm), medium (2 mm–5 mm), and large (>5 mm) diametric classes; then, they were dried and weighed to obtain root biomass (gDw·m2) (Castañeda-Moya et al., 2011). An approach to gain a greater understanding of root biomass distribution has been to explore the root/shoot ratios (BRB:AGB), defined as root biomass (including necromass) divided by aboveground biomass (Mokany et al., 2006).
The total pneumatophore sample was collected at ground level in 50 cm × 50 cm quadrants distributed at random within the MU, with two quadrants in each UM (8 quadrants in total). Pneumatophores were kept at 4°C and transported to the laboratory for height measurement; they were then oven-dried at 60°C and weighed. The density (no. of pneumatophores·m2) and biomass (grams of dry weight per square meter of soil; gDw·m2) were determined.
Root Production
In each MU, two 0.5 cm mesh cores filled with peat moss were placed (Adame et al., 2014). One bag per MU was removed six months later, at Time 1 (T1); 12 months later (T2), the second bag was removed. Root production was determined as the difference between the initial biomass (T0) and that observed at T1 and T2. The samples were transported to the laboratory to determine biomass; to this end, the samples were immersed in water to separate the living roots from dead roots as the belowground root biomass procedure. The roots were dried and weighed (Dw·m-2·d-1).
Carbon Estimates
Allometric Equations Used to Determine Mangrove Tree Biomass.

BA= basal area; Ag; Avicennia germinans; B = biomass; DBH = diameter at breast height (cm); ρ = wood density (g·cm3).
The conversion factor used to determine the carbon content from tree biomass was proposed by Kauffman and Donato (2012): Carbon content of each tree (MgC·ha-1) = tree biomass * carbon conversion factor (0.48). For leaf litter and pneumatophores, we used a factor of 0.45 (Howard et al., 2014); organic carbon concentrations between 38% and 49% stored in this component are reported (Kauffman et al., 1995). Carbon stored in living roots stores between 36 % and 42 % of carbon in tropical forests (Jaramillo et al., 2003), so we considered a conversion factor of 0.39. For necromass, a conversion factor of 0.5 was considered (Howard et al., 2014) because the carbon content stored in dead wood in tropical forests is around 50 %. All carbon contents were expressed as MgCorg·ha-1, biomass was expressed in gDw·m-2, and area in ha, and estimated as follows:
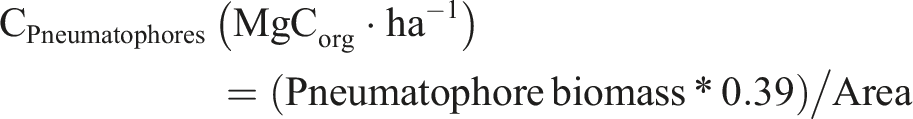


We followed the standardized laboratory and calculation protocols established by Howard et al. (2014). In each monitoring unit, one soil core was extracted at a depth of 1 m (Howard et al., 2014); it was divided into 4 samples according to the depth stratum: 0 cm–15 cm, 15 cm–30 cm, 30 cm–50 cm, and 50 cm–100 cm (Marín-Muñiz et al., 2014). The samples were stored at 4°C and transported to the laboratory for analysis (Howard et al., 2014; Marín-Muñiz et al., 2014). The percentages of total C and organic C were determined by flash combustion on a Flash 2000 Elemental Analyzer (Thermo Fisher Scientific) Waltham, MA, USA (https://www.thermofisher.com, retrieved November 16, 2023). The inorganic carbon content was determined by calcinating a sample in a muffle oven at 550°C for four hours and the organic carbon content was obtained as the difference of the percent of total carbon and the percent inorganic carbon from the calcinated sample.
Statistical Analysis
We analyzed spatial and temporal differences in the physicochemical parameters of water and soil, mangrove forest structure, pneumatophores, litter, living roots, necromass, and root production, using a one-way ANOVA and Tukey’s tests (Steel & Torrie, 1996). Kolmogorov-Smirnov & Levene tests were applied to verify data normality and homoscedasticity. The statistical analysis was performed with the IBM SPSS Statistics software.
Results
Variation of Physicochemical Parameters in Soil
The temporal variation (monthly) of physical and chemical elements in Soil (phosphorus, nitrogen, organic matter and moisture content) can be observed in Figure 2. Spatially, a low Soil sand content was recorded in the monitoring sites, with an average of 18.9 ± 1.1%, 61.5 ± 1.4 % clay, and 19.6 ± 0.6 % silt, with no significant differences between sites (F = 7.6, N = 288, p = 0.9) (Table 2). The mean bulk density was 0.59 ± 0.2 g·m-3, with significant differences between sites (F = 13.2, N = 88, p = 0.002). Monthly Variation of Total Phosphorus, Total Nitrogen, Organic Matter, and Humidity Content in Sediment in the Monitoring Sites of the La Cruz Lagoon. Texture, Bulk Density, and Electrical Conductivity of Soil at Monitoring Sites.

Physicochemical Variation in Surface and Interstitial Water
Variation of Physicochemical Parameters in Surface and Interstitial Water During the Monitoring Months.
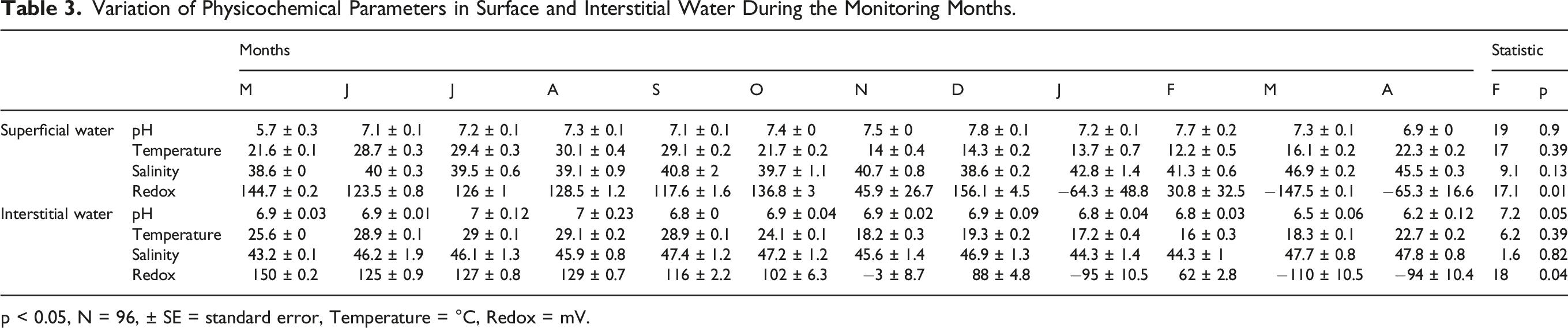
p < 0.05, N = 96, ± SE = standard error, Temperature = °C, Redox = mV.
Mangrove Forest Structure
Structural Attributes of Mangroves.

MU: Monitoring unit (±SE = standard error).
Leaf Litter Production
A decrease in the mean litter content, 2.21 Mg·ha-1·year-1, was recorded; the leaf component recorded the highest value, 15.5 ± 1.5 g·m-2·month-1, with no significant differences (F = 1.56, N = 24, p = 0.34). The largest number of flowers was recorded from June to September, with a cumulative amount of 1.03 ± 0.4 g· g·m-2·month-1, corresponding to 68.2 % of the annual total. Fruit production was only recorded from July to December, with the highest production in September (14.7 ± 1.8 g·m-2·month-1 (Figure 3). Monthly surface water temperature was positively correlated with litter production (r = 0.85, p = 0.04). Leaf Litter Production by Components and Water Temperature.
Belowground Root Biomass (BRB)
Root Biomass at Each Site by Diameter Class and Depth Stratum (gDw·m−2).

(p < 0.05, N = 72) (±SE = standard error). S = Site.

Necromass Content at Sites by Depth Strata.
Root Production at the Monitoring Sites by Diameter Class.

(±SE = standard error), T1= 6 months, T2 = 12 months.
Carbon Stock in Mangrove Biomass
Carbon Stock by Mangrove Component (MgCorg·ha−1).
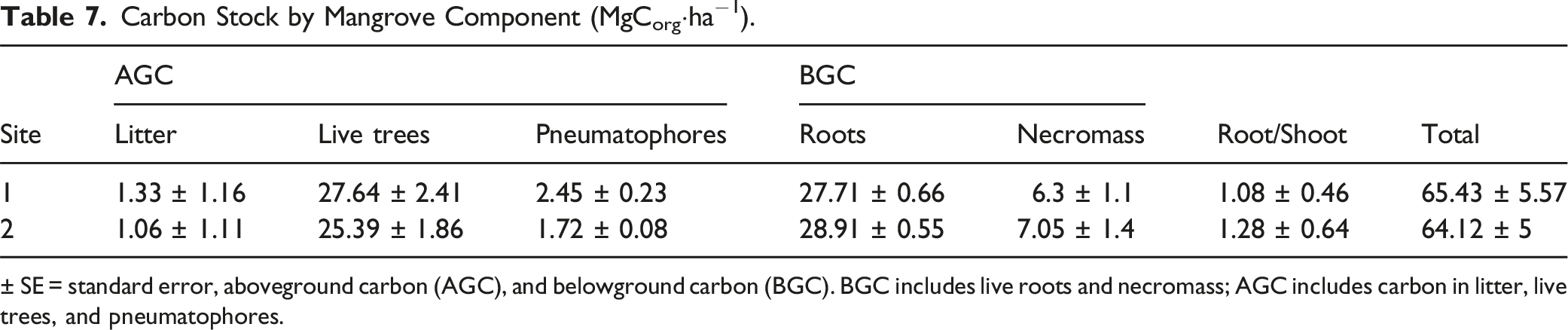
± SE = standard error, aboveground carbon (AGC), and belowground carbon (BGC). BGC includes live roots and necromass; AGC includes carbon in litter, live trees, and pneumatophores.
Carbon in Soil
Organic Carbon Store in Soil.

±SE = standard error.
Discussion
In coastal regions with dry climate and precipitation deficit, freshwater runoff is crucial for mangrove establishment and development (Smith, 1992; Twilley et al., 1986). Thus, precipitation plays a central role in the arid and semi-arid areas of Mexico, and a minimal change in rainfall dynamics can influence productivity in these areas (Mitsch & Gosselink, 1993). In the La Cruz Lagoon, the annual cumulative precipitation is low (130.5 mm); however, it is similar to the precipitation recorded in estuarine areas surrounding the Gulf of California, as El Soldado, Tóbari, and Moroncarit, where annual precipitation was approximately 324.3 mm in 2020, most of the rainfall occurred from June to October (Torres et al., 2021).
In the present study, high levels of salinity and low contents of organic matter in the soil were identified. Mangrove forests located in arid zones face high salinity and nutrient scarcity, growing at their physiological limits (Torres, Frausto-Illescas, et al., 2023). High soil salinity creates stressful conditions for mangroves and, consequently, leads to low structural development and lower primary productivity (Twilley et al., 1986). The results confirm the analysis carried out in a review article on mangroves in arid regions, which showed that they have high densities, limited height, and are dominated mainly by Avicennia spp. (Adame et al., 2021).
In La Cruz Lagoon, the mangrove species Avicennia germinans and Rhizophora mangle are present, with A. germinans being the dominant mangrove species. Our measurements showed a monospecific forest of A. germinans with structural attributes characteristic of the dwarf-scrub mangrove type found in arid zones of the Gulf of California (Lugo & Snedaker, 1974), similar to those reported by Bautista-Olivas et al. (2018) in Bahía del Tóbari and Torres, Sanchez-Mejia, et al. (2023) in the Lobos-Guásimas lagoon system. In the La Cruz Estuary, the low structural development can be attributed to the high surface and interstitial salinity that inhibits mangrove growth, as identified by Adame et al. (2021) and Torres, Sanchez-Mejia, et al. (2023). In addition, Agraz-Hernández et al. (2011) identified A. germinans as a species tolerant to hyperhaline interstitial water, allowing it to reach high densities at highly saline sites.
Several studies have identified high litter production in sites near tropical areas (Morrisey et al., 2010). However, there have been few studies on litter productivity in arid mangroves (Sánchez-Andrés et al., 2010; Torres et al., 2022, 2023b) that transition from tropical to subtropical latitudes (López-Medellín & Ezcurra, 2012) where evaporation (∼3000 mm·year-1) exceeds precipitation (∼300 mm·year-1), and a microtidal regime predominates (Torres et al., 2022). Monospecific Avicennia germinans sites studied in the Gulf of California by Arreola-Lizárraga et al. (2004) identified low litter productivity (1.75 Mg·ha-1·year-1), similar to the level estimated in the present study. However, higher values were reported at sites with three mangrove species (A. germinans, Rhizophora mangle, and Laguncularia racemosa), as identified by Torres, Sanchez-Mejia, et al. (2023), with 4.9 Mg·ha-1·year-1 in the Lobos-Guásimas lagoon system. Félix-Pico et al. (2006) estimated 8.05 Mg·ha-1·year-1 in El Conchalito, and Torres et al. (2022) recorded 4.9 Mg·ha-1·year-1 in Moroncarit, 3.5 Mg·ha-1·year-1 in Tóbari, and 3.2 Mg·ha-1·year-1 in El Soldado.
Low structural development leads to low litter-based productivity (Lovelock & Duarte, 2019). Litter productivity depends on the structure of the tree (i.e., height) (Kathiresan & Bingham, 2001). Woodroffe (1982) characterized the relationship between litter productivity and tree height, observing that the higher the tree, the more leaf-litter it produces. In addition, A. germinans showed the lowest productivity compared to R. mangle and L. racemosa (Bouillon et al., 2008; Torres et al., 2017), which can be attributed to leaf morphology. In dwarf mangroves, litter contribution to soil carbon stocks is important but the belowground root turnover is especially important.
Plants in nutrient-poor soils tend to allocate more biomass to roots, maximizing the efficiency to capture the most limiting resource and increasing the proportion of belowground roots (Gleeson & Tilman, 1992). The low organic matter content in La Cruz Lagoon (8.1 % on average) promotes high root production that results in high belowground biomass, as identified by Torres, Frausto-Illescas, et al. (2023) in the El Vizcaíno biosphere reserve, with a root-biomass value of 8.65 Mg·ha-1 in El Dátil Lagoon and 6.05 Mg·ha-1 in Laguna San Ignacio. Torres, Sanchez-Mejia, et al. (2023) recorded an average of 52.12 ± 9.2 Mg·ha-1 in the Lobos-Algodones-Guásimas lagoon system in the Gulf of California. Torres et al. (2021) studied and documented root biomass in El Soldado (5.12 Mg ha-1), El Tóbari (5.51 Mg·ha-1) and Moroncarit (5.79 Mg·ha-1). However, a study in the temperate zone of the Gulf of Mexico (Torres et al., 2019) recorded a higher soil organic matter content of 16.2 ± 1.8 % with a lower belowground root biomass of 1.91 Mg·ha-1.
The belowground biomass of mangroves may have been affected by forest age (McKee, 2001), plant characteristics (Ezcurra et al., 2016; McKee et al., 2007), environmental conditions (Otero et al., 2009; Sherman et al., 1998) and measurement methods (Muhammad-Nor et al., 2019). In mangroves, the contribution of roots of different size classes to total biomass is variable; it has also been found that root biomass decreases with soil depth (Adame et al., 2017). In La Cruz Lagoon, fine roots showed the greatest contribution to root biomass, with the highest biomass in the upper 15 centimeters; similarly, total root biomass decreased with depth. As reported in Florida by Castañeda-Moya et al. (2011) and Kenya by Tamooh et al. (2008), higher necromass contents have also been observed in the upper depth stratum (0–20 cm) while it decreases with depth. High belowground root biomass may show a greater contribution to soil organic carbon than leaf litter production, due to its faster production rate. The present study recorded a BGB:AGB ratio greater than one, meaning that more than half of the biomass is allocated to the underground stratum. As pointed out by Castañeda-Moya et al. (2011) and Virgulino-Júnior et al. (2020), in dwarf-scrub mangroves, vegetation height and belowground production are negatively correlated.
Several studies have reported higher root productivity and biomass related to high salinity values (Adame et al., 2014; Sherman et al., 2003), and these results have been interpreted as a mechanism to tolerate stress associated with highly saline conditions (Chapin, 1991). This condition appears to vary among mangrove species and according to the environmental conditions (Krauss et al., 2014). However, our estimates of BGB production indicate a pattern that contrasts sharply with the one recorded by Saintilan (1997), who found an increase in the contribution of BGB with increasing salinity. High variability in the values reported in the underground biomass (roots) can be attributed to global and local factors (Clough, 1992) and non-uniformity in the methodologies may also be reflected in these data (Giraldo, 2005), such as harvesting plots (Golley et al., 1962), soil cores (Briggs, 1977), trench (Tabuchi et al., 1983), stratified coring (Saintilan, 1997), and allometric equations (Matsui, 1998). It is recommended to use a combination of methods to reduce uncertainty, increase depth and number of samples for the soil cores method.
Carbon storage in mangroves is a function of the amount of biomass, which varies according to tree age and growth efficiency (Banerjee et al., 2020). Most of the carbon in mangrove ecosystems is stored underground as peat (Donato et al., 2011), which, unlike shorter-lived aboveground biomass, often remains undecomposed for millennia (Ezcurra et al., 2016; McKee et al., 2007). Herrera-Silveira et al. (2020) mention that the North Pacific recorded the lowest aboveground organic carbon stock (58.9 ± 12 MgCorg·ha-1), similar to the values recorded in La Cruz Lagoon, with a carbon stock related to aboveground and belowground biomass of 64.8 ± 5.3 MgCorg·ha-1, which accounts for 142.41 ± 37.1 MgCorg·ha-1 of the carbon stored in soil, as reported by Alongi (2020), who mentioned that aboveground and underground C biomass accounted for 14.8 % and 8.7 % of the ecosystem total C stocks. Herrera-Silveira et al. (2020) mentioned that the mean belowground Corg consisted of approximately 77 % of the total Corg stock for Mexico.
Mangroves have the highest C reserves (386 Mg·ha-1) compared to other coastal ecosystems such as salt marshes (255 Mg·ha-1) or seagrasses (108 Mg·ha-1) (Howard et al., 2014). Managing coastal ecosystems for carbon sequestration services in arid environments requires identifying the factors that explain their relatively small soil carbon stocks (Schile et al., 2017). The La Cruz Lagoon shows aboveground and belowground carbon stocks of 207.21 ± 42.4 Mg·ha-1, which can be considered low related to the low structural development and, consequently, a low primary productivity based on leaf litter, being belowground roots the component that represented the greatest contribution to belowground Corg. These data indicate that mangrove Corg levels increase in soils as roots grow, die, and accumulate, suggesting that mangrove C is best preserved if mature mangrove forests are maintained undisturbed (Alongi, 2020). Herrera-Silveira et al. (2020), in their review of carbon stocks and stores in Mexico, identified that the mean aboveground tree biomass was 113.6 ± 5.2 MgCorg·ha-1, while the mean belowground Corg (soils and roots) was 385.2 ± 22 MgCorg·ha-1, for a combined total of 498.8 MgCorg·ha-1.
This study showed the blue-carbon stock related to the biomass and soil stores in the mangrove ecosystem in an arid ecosystem of the Gulf of California. Results recorded the La Cruz Lagoon showed a low structural development with a litter productivity of 2.21 Mg·ha·year-1, with aboveground carbon biomass of 29.8 ± 3.4 MgCorg·ha-1 and a belowground biomass value of 35.1 ± 88 MgCorg·ha-1, with a root:shoot ratio slightly greater than 1. These results suggest that belowground carbon related to mangrove biomass is higher than aboveground biomass. The estimated total carbon was 207.21 ± 42.4 MgCorg·ha-1. The mangrove ecosystem in the La Cruz Lagoon has high belowground biomass values that represent a greater belowground carbon storage, which helps justifying promoting conservation and restoration strategies to improve or recover the ecosystem services it provides. It is recommended to evaluate other components that store carbon, such as dead wood, mulch, and seedlings, among others, which were not considered in the present study. Furthermore, the methods for studying dwarf-scrub mangroves should be standardized, especially with respect to their structural attributes and litter production.
Implications for Conservation
Recently, the role of mangrove forests in carbon storage has raised interest as they store four times more carbon than any other tropical forest, including rainforests (Donato et al., 2011). The information provided in this study reveals the blue carbon storage capacity of the mangrove ecosystem both in structural biomass and in soil. Managing coastal ecosystems for carbon sequestration services in arid environments requires characterizing the diverse components where carbon is stored (Schile et al., 2017).
The implications for conservation of our findings on mangrove ecosystems in tropical and/or subtropical regions lies in their importance in maintaining a high capacity to capture atmospheric CO2, to serve as a carbon sink and storage facility, thus providing a nature-based ecosystem service. This information also serves as baseline knowledge to justify the restoration of areas degraded by anthropogenic activities.
Footnotes
Statements and Declarations
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially funded by the Department of Scientific and Technological Research (DICTUS), Universidad de Sonora. PROFAPI_2023_CA_008 Instituto Tecnológico de Sonora.
Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
