Abstract
Background and Research Aims
Lesser flamingo (Phoeniconaias minor) is an intra-African migratory bird species recorded mainly in alkaline lakes of the Great Rift Valley. Temporal patterns of Lesser flamingo visits may be related to the abundance and availability of their primary phytoplankton food items, and phytoplankton growth and composition may also be influenced by water quality parameters. This study aims to investigate the relationship between Lesser flamingo abundance and the physico-chemical characteristics of the wetland and its phytoplankton community dynamics.
Methods
We measured the abundance of Lesser flamingos using the ground vantage point counting method. We assessed water physico-chemical characteristics using field measurements of temperature, dissolved oxygen, pH, electrical conductivity and turbidity, and laboratory sample analyses for phosphorus, ammonia, nitrate, silica, alkalinity and chlorophyll a. We also measured phytoplankton diversity and abundance in the laboratory.
Results
The physico-chemical characteristics of the Chelekleka wetland showed a favorable environment for biological productivity and physiological activities of aquatic organisms. High phytoplankton richness (83 taxa) and abundance were recorded in the wetland. Lesser flamingo abundance varied significantly in response to the abundance of preferred phytoplankton prey items, which could be attributed to the effect of aquatic grazers and changes in the physico-chemical characteristics of the wetland.
Conclusion
Water physico-chemical characteristics influence the abundance and availability of phytoplankton in the seasonally flooded wetland, which in turn influences the seasonal visitation and abundance of Lesser flamingos foraging on phytoplankton prey.
Implications for Conservation
We recommend that the local government and other relevant stakeholders implement integrated wetland conservation practices to alleviate wetland degradation and conserve the ecosystem and its associated wildlife.
Introduction
Food availability is among the most important determinant factors affecting bird distribution (Kumssa & Bekele, 2014a). Filter-feeding wading birds such as Lesser flamingos (Phoeniconaias minor) show unpredictable, spontaneous, and sporadic nomadic movements between alkaline saline lakes in search for preferred food items which mainly consist of phytoplankton (Kaggwa et al., 2013). The net primary productivity of phytoplankton which Lesser flamingos rely on is affected by limnological parameters such as nutrient availability, light, and mixing (Kaggwa et al., 2013; Kihwele et al., 2014; Tuite 2000). However, the increasing trend of anthropogenic pressure could shrink the size and reduce the water volume of wetlands which in turn increase nutrient concentration triggering the dominance of potentially toxic algae. Thus, the seasonal abundance of Lesser flamingo varies in relation to the ecohydrological dynamics of wetlands (Kihwele et al., 2014). Among the six extant flamingo species in the world, Lesser flamingo is the most abundant (Childress et al., 2008; Zaccara et al., 2011; Hill, 2016). The largest population of Lesser flamingos are found in the East African saline lakes while smaller populations exist in southern and West Africa and also India (Childress et al., 2008; Schagerl, 2016). The African Lesser flamingo population is estimated to be between 1.57–2.58 million individuals. However, a decline in their population is reported in southern Africa (McCulloch et al., 2003) and raising concerns in eastern Africa (Kihwele et al., 2014).
The decline in the Lesser flamingo’s population is mainly attributed to anthropogenic factors such as soda-ash mining and hydroelectric power schemes and water pollution. As a result, the species is listed as Near Threatened in the 2008 IUCN Red List of Threatened Species (BirdLife International, 2018; Childress et al., 2008; Parasharya et al., 2015) and also species of conservation concern due to the potential vulnerability of the species resulted from loss of wetland feeding site, low number of breeding sites and low reproductive rate (Childress et al., 2008; Parasharya et al., 2015). Since the Lesser flamingo is a highly specialized species that depends on a limited number of saline lakes (Zaccara et al., 2011), strict protection of such habitat ranges that act as an important shelter, feeding site and breeding ground is mandatory (Krienitz et al., 2016; Schagerl, 2016).
Ethiopia is a primary home range of the Lesser flamingo where these birds inhabit the country’s Rift Valley Saline lakes such as Abijatta, Shalla, Metehara, Chitu and Aranguade Bahir (Kumssa & Bekele, 2014b). Chelekleka wetland, a seasonally inundated flood plain, is also another habitat that supports a considerable number of Lesser flamingos (EWNHS, 1996) shared with Greater flamingos (Phoenicopterus roseus).
Chelekleka wetland is home to over 246 bird species. Of which, 39% are migratory (Teklemariam et al., 2023). However, several anthropogenic factors including agricultural expansion and water abstraction threaten Lesser flamingo habitats urging the need for protection of the habitat, management of key sites and increasing public awareness (Childress et al., 2008). Furthermore, understanding of the population dynamics of the species in relation to the biological and chemical status of the lakes they inhabit is critically important for an effective conservation action (Kihwele et al., 2014; Schagerl, 2016; Zaccara et al., 2011). In this regard, this study aimed to investigate changes in Lesser flamingo abundance in relation to physico-chemical parameters and plankton community in Chelekleka Wetland. We hypothesized that variations in the temporal water physico-chemical characteristics and phytoplankton community affect the abundance of seasonally visiting Lesser flamingos.
Methods
Study Area
Chelekleka wetland is a seasonally inundated pan located along the western and southwestern sides of Bishoftu town about 51 kms southeast of Addis Ababa (BirdLife International, 2022; EWNHS, 1996). It is an Important Bird Area located at 8o 39′ 0” - 8o 53′ 0” N and 38o 49′ 0” - 39o 3′ 0” E (Figure 1) encompassing an area of 24,000 ha (BirdLife International, 2022). The wetland is part of the Ethiopian Rift Valley ecosystem with diverse flora and fauna, including many migratory waterbirds (EWNHS, 2010). The study area map; Lesser flamingo counting vantage points (red dot) and water quality sample sites (yellow dot). The wetland is disconnected by the railway and highway constructions.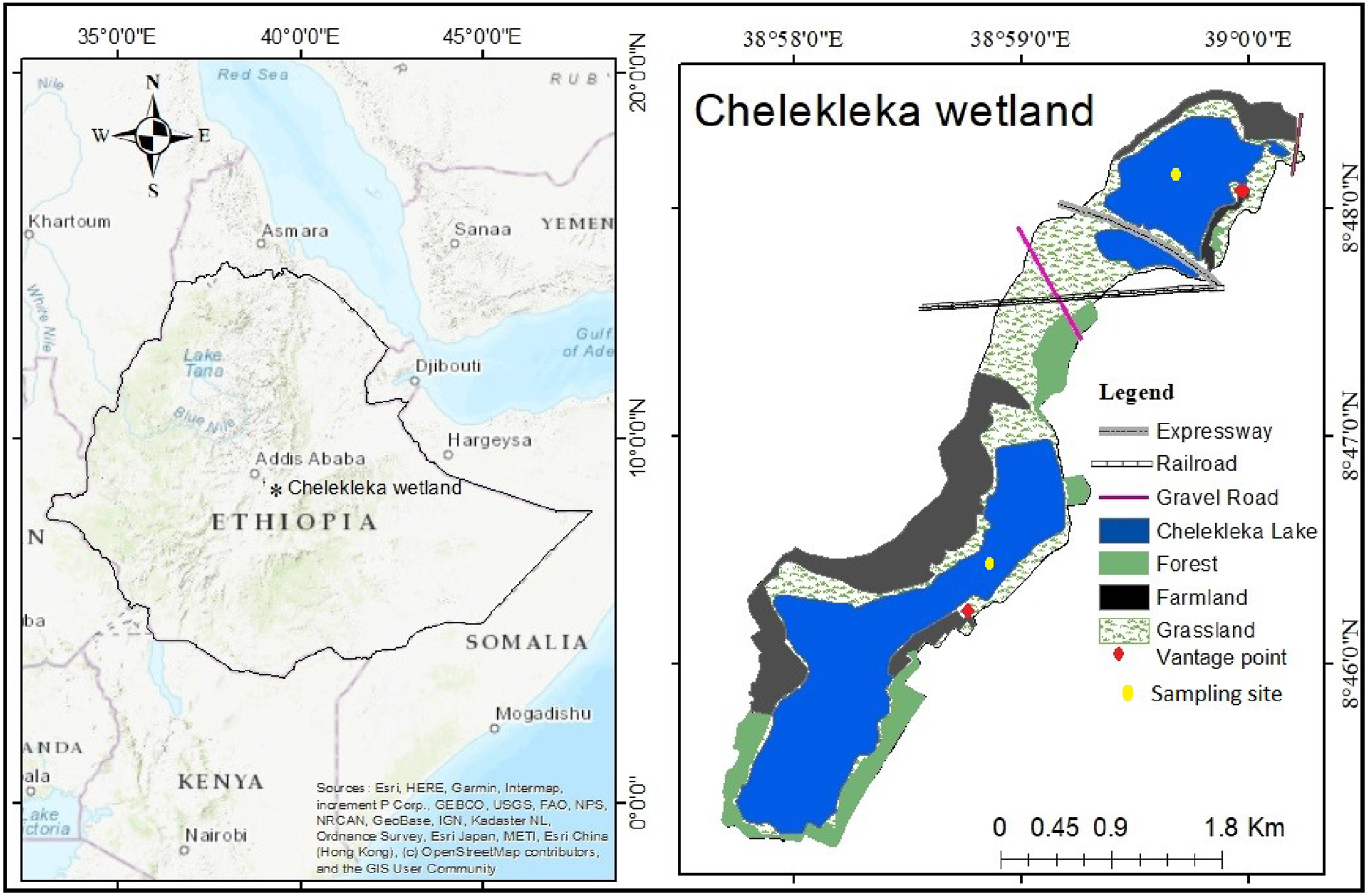
Three administrative Kebeles (i.e. the lowest administrative unit in Ethiopia) of Bishoftu town surround the Chelekleka wetland (Gebreegziabher et al., 2017). The livelihood of communities in these Kebeles depends on the wetland where irrigation cultivation is intensively practiced (BirdLife International, 2022). The wetland is highly polluted from town waste disposal activities posing a serious threat (Gebreegziabher et al., 2017).
Data Collection
Sampling on physico-chemical parameters, phytoplankton, and flamingoes census were proposed to be conducted for 12 months. However, following the complete dryness of the wetland during March and April 2022, the data were only collected for 10 months from May 2021 to February 2022.
Physico-chemical Parameters
Physico-chemical parameters of the wetland were collected once per month from May 2021 to February 2022 at two sites (Figure 1). In situ measurements of physico-chemical parameters (water temperature, dissolved oxygen (DO), electric conductivity (EC), and pH) were carried out using a multi-parameter probe (HQ40d model, Hach company, Germany). Turbidity was measured with a turbidity meter (T-100 Oakton model, Cole-Parmer, Germany). Due to the small and variable size of the study sites (EWNHS, 1996), duplicate subsurface water samples (at 0.10 m depth) were collected and the samples were analyzed in pseudo-triplication at the Addis Ababa University Limnology laboratory to determine the wetland phosphorus, ammonia, nitrate, silica, alkalinity and Chlorophyll a (Chl a) content using JENWAY 6405 UV/Vis Spectrophotometer. For identification, phytoplankton samples were collected by filtering 11 to 16 liters of water via a 15 µm mesh size plankton net and preserved with Lugol’s solution. However, for quantitative analysis, one-liter subsurface water samples were settled for 72 hours using a graduated measuring cylinder after preserving with Lugol’s solution, and 90 % of the supernatant was siphoned off without disturbing the settled algae (Hotzel & Croome, 1999; Liu et al., 2021).
Phenolphthalein alkalinity (PA) and total alkalinity (TA) were determined using phenolphthalein indicator and bromocresol green-methyl red mixed indicator solution, respectively (Wetzel & Likens, 2001). A 100 ml of water samples were titrated with 0.2 N HCl to a pH of 8.3 for phenolphthalein alkalinity (PA) and 4.5 for TA. Concentrations of inorganic nutrients were determined following the procedures outlined in the American Public Health Association (APHA) (2005). The samples used for the analyses of all nutrients except Total phosphorus (TP) were filtered through a glass fiber filter (GF/F). Nitrate (NO3-) was analyzed using the sodium salicylate method and ammonia by the phenate method. Molybdate-reactive silica (SiO2) was determined using the molybdosilicate method (APHA, 1999, 2005). The concentration of Chl a was determined using a spectrophotometric method using 90 % acetone as an extraction solvent.
Phytoplankton Composition
The phytoplankton samples were collected once monthly from May 2021 to February 2022 at two sampling sites (Figure 1). The samples were examined at different magnification levels under an inverted microscope and identified to genus and species level using various taxonomic keys and literature (e.g. Arguelles et al., 2014; Cronberg & Annadotter, 2006; František, 2006; Komárek & Komárková-Legnerová, 2003; Krammer et al., 2002; Lone, 2014). Algal division was carried out following Guiry & Guiry (2013) and enumeration of phytoplankton using the settling technique (Hotzel & Croome, 1999).
A subsample of 1 ml was taken from the sedimented algae and poured onto the Sedgewick-Rafter chamber placed under an inverted microscope (APHA, 1999, 2005). After allowing the aliquot to stand for 30 seconds, cell counts were conducted at 400X magnification with a counting error of 20.33% at a 95% confidence limit (Lund et al., 1958). At least 30 randomly selected grids were counted within the chamber so that 90 to 95% of the taxa present are included in the count (Hotzel & Croome, 1999). For filamentous algae, the numbers of cells per filament were determined for 30 filaments and then the mean number of cells per filament was calculated. The same method was used for colonial algae where the number of cells per colony was first determined for 30 colonies and then the mean number of cells per colony was calculated (Brierley et al., 2007). The average number of cells per filament or colony were multiplied by the number of filaments or colonies to estimate the abundance of filamentous or colonial taxa.
Lesser Flamingo Abundance
The abundance of Lesser flamingos was recorded early morning between 6:30 - 9:00 hrs. and late afternoon between 15:30 - 18:00 hrs. biweekly from May 2021 to February 2022. A total of 80 days of counts were collected using the ground vantage point counting method (Kihwele et al., 2014) at two vantage points that was selected on relatively higher ground along the shore (Figure 1). Two trained observers stopped at each of the established vantage points and recorded the number of birds for 20-25 minutes using binoculars (10 × 42) and/or naked eyes from these vantage points on the same day following Kihwele et al. (2014) once per counting periods. Observers moved between vantage points using vehicles. Both observers counted Lesser flamingos independently at the same vantage points and the number of birds at every vantage point was obtained as the mean value from the observers. At the end of each counting periods, the total number of Lesser flamingos was obtained as the total sum of the mean values obtained from all vantage points. The daily abundance of Lesser flamingos was attained as the mean value from the morning and evening counts. To minimize double counting, birds that left the flock before counting was finished were not considered.
Data Analysis
All analyses were done in R version 4.3.2 (R Core Team, 2023). Species diversity of phytoplankton was computed using the Shannon–Weiner diversity index (H0). Margalef species richness index (D) and Peilou evenness index (J) were also used to evaluate phytoplankton species diversity (Liu et al., 2021) using a Vegan package (Oksanen et al., 2018). Mean spatial readings of physico-chemical variables were compared between seasons using a one-tailed Student’s t-Test (Mendoza et al., 2020). The Hellinger transformation of community data was employed to minimize the influence of dominant species in the analysis (Kindt and Coe, 2005) and the explanatory variables (physico-chemical parameters) were standardized. The significant explanatory variables of the physico-chemical parameters were selected by stepwise selection to understand the drivers of the variation in abundances of the Lesser flamingos and their prey items. The correlations among the environmental variables were evaluated by vif.cca function in Vegan package. Redundancy analysis (RDA) was employed to determine the relationship between the physico-chemical parameters of the wetland and the abundances of Lesser flamingos and their potential prey items from the ten-month sampling. Monthly average numbers of Lesser flamingos were considered for the RDA. Analysis of variance (ANOVA) was used for the statistical evaluation of the RDA model, the effects of explanatory variables retained after the stepwise selection on the response variables. Adjusted R2 which also measures the strength of the relationship between the abundances of Lesser flamingos and their prey items and the physico-chemical parameters of the wetland were employed.
Results
Physico-chemical Parameters
Chelekleka wetland water temperature ranged from 20°C to 25°C with a pH range from 7.8 to 8.5. In the present investigation, DO was found to be in the range of 4.4 – 9.57 mg/l. The minimum phenolphthalein alkalinity and total alkalinity of all the water samples were zero, their maximum values were 10 and 100 mg/l respectively. The wetland’s water electric conductivity ranged from 195 – 447 µS/cm containing 17 to 433 mg/l of Silica. The wetland’s Nitrate, Ammonia, and Total phosphorus concentrations ranged from 19 – 1494, 28 – 1919, and 213 – 4183 respectively.
Seasonal Levels of Physicochemical Parameters of Chelekleka Wetland.

*Significant difference was observed (p < 0.05).
Despite the disconnection caused by railway and expressway constructions in Chelekleka wetland, the two sites exhibited similar physico-chemical properties (p > 0.05) (Figure 2). Physicochemical characteristics of Chelekleka wetland in the southern and northern sites (DO = dissolved oxygen, EC=electrical conductivity, PA = phenolphthalein alkalinity, TA = total alkalinity, TP = total phosphorus, Chl a= Chlorophyll a). Non-overlapping boxes suggest a significant difference between the medians of the different groups (P < 0.05). Conversely, overlapping boxes suggest that there is no significant difference between the groups (P > 0.05).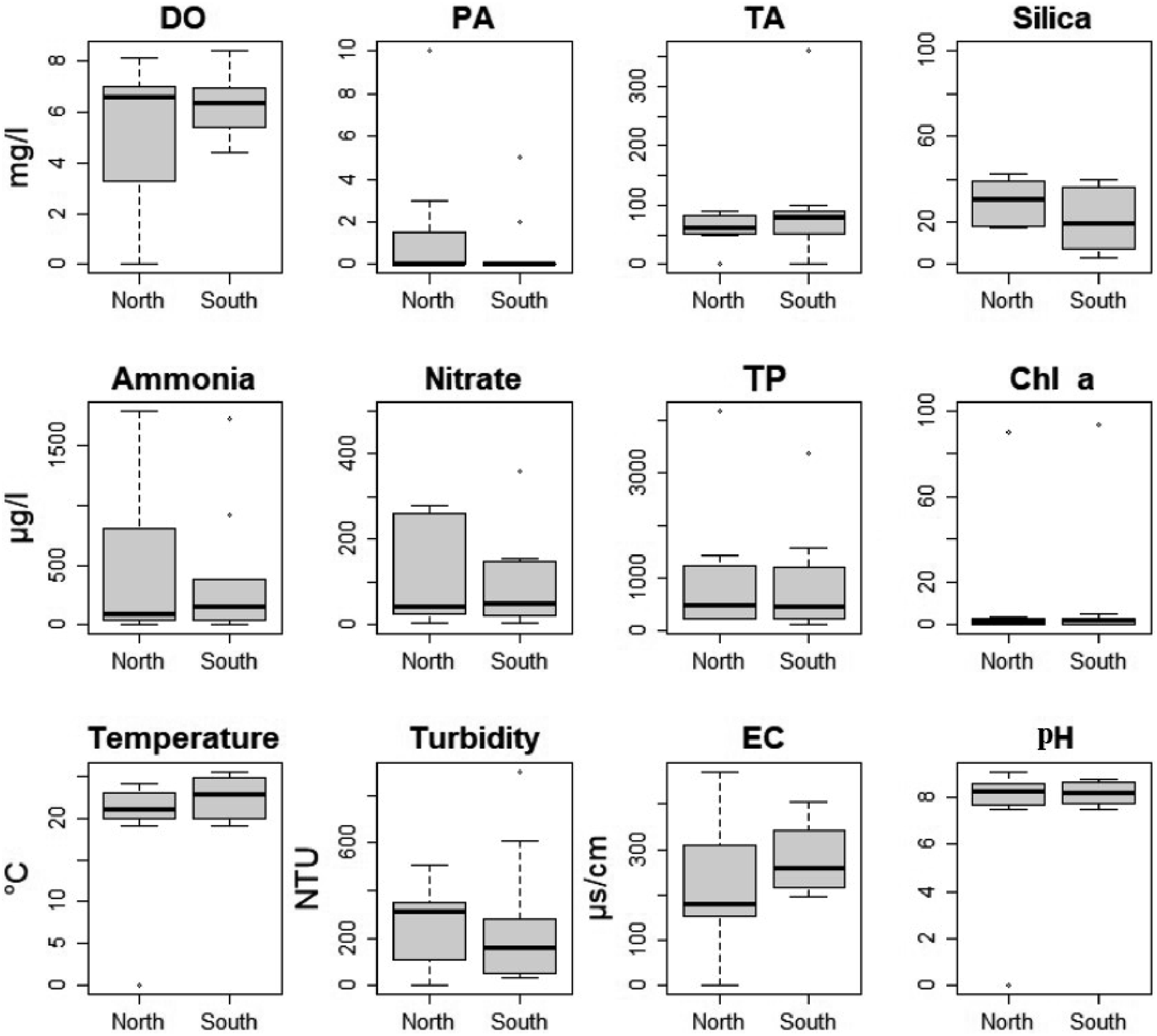
Phytoplankton Community Composition
A total of 83 phytoplankton taxa grouped under 32 families and five phyla namely: Bascilariophyta (diatom), Chlorophyta (green algae), Chrysophyta (Golden algae), Cyanophyta (blue green algae) and Euglenophyta (Euglena) were recorded (Appendix 1). Among the five phyla, Chlorophyta constituted the highest number of taxa, 27(32.5%) followed by Euglenophyta 21(25.3%), Cyanophyta 19 (22.9%), and Bacillariophyta, 15 (18.1%). However, Chrysophyta was represented by one taxon (1.2%). Family Euglenaceae had the highest taxa (11; 13.3 %), while 12 different families were represented by a single taxon (1.2 %). Although a similar phytoplankton community was recorded in the Northern and Southern sampling sites, Family Sphaerocystidaceae was found only in the North site while Aulacoseiraceae, Chromulinaceae, Catenulaceae, Oscillatoriaceae, Chroococcaceae and Pseudanabaenaceae were recorded only in the Southern site (Appendix 1).
Phytoplankton Abundance
The abundance (ind/l) of the dominant phytoplankton community in the wetland (Appendix 1) showed that Botryococcus braunii had the highest abundance (4,002,881 ind/l) followed by Coelastrum cambricum (1,853,675 ind/l). On the other hand, Amphora lybica (615 ind/l) and Uroglena (567 ind/l) recorded the lowest abundance.
Among the recorded phytoplankton communities in Chelekleka, nine of them are reported as a prey item of Lesser flamingos (Krienitz et al., 2016; Kumssa and Bekele 2014b). Chroococcus minor was among the most abundant prey species (293,499 ind/l) and Navicula ventralis was the least abundant prey (540 individuals ind/l).
Amongst the potential prey phytoplankton species of Lesser flamingo, the highest (293,499) and lowest (540), number of individuals per liter of water per sampling months were recorded for Chroococcus minor and Navicula ventralis species respectively. Navicula ventralis is the single representative of diatom phytoplankton with a maximum abundance of 26064 individuals/l in December. In addition, with a maximum of 251,383 individuals/l Oocystis lacustris was the only green algae species recorded as potential food for Lesser flamingo in the wetland during February. A maximum (293,499 individuals/L) of Chroococcus minor was the dominant species among the Cyanobacteria in June and a maximum of 248,726 Euglena sp. from Euglenophyte during July (Figure 3). Monthly abundance of potential phytoplankton prey of Lesser Flamingo.
The highest total abundance of potential prey items of Lesser flamingo was recorded during June 2021 (811243 individuals/l) over dominated by Chroococcus minor and Euglena sp. Though in January 2022 the total abundance of prey items was the second abundant (537385 individuals/l), it is worthwhile to mention that in this month, all types of prey species were observed.
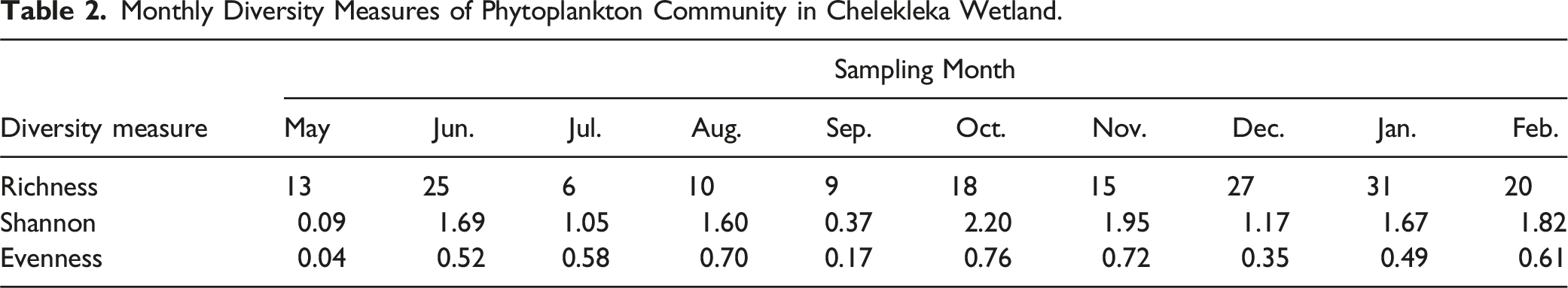
Phytoplankton Richness and Diversity
Monthly Diversity Measures of Phytoplankton Community in Chelekleka Wetland.
Lesser Flamingo Abundance
Lesser flamingos visited the Chelekleka wetland mainly from December to February with a mean of 639 ± 308 and very few numbers during August (9 individuals) and November (three individuals). However, no Lesser Flamingos were observed at the wetland in June, July, September, and October 2021. The maximum number of Lesser flamingos counted during the study was 963 during December 2021 and the lowest, (three individuals) during November 2021. There was a fluctuation in the abundance of Lesser flamingos in different months and between study sites. Though the northern site dried two months before the dryness of the southern site, it supported the relatively highest number of flamingos (604 individuals) compared to the southern site (604 individuals) during December 2021. Generally, following flamingo arrival in December 2021, their abundance gradually increased with a drastic drop in January and February 2022.
Physico-chemical Characteristics and Phytoplankton Abundances Influence on Lesser Flamingo Abundance
The RDA results indicate that the PA, TP, Nitrite, Ammonia, and Chl a are the variables that have more influence on the abundance of phytoplankton prey and Lesser flamingo with the strongest effect from Chl a followed by similar effects of Ammonia and Nitrate (Figure 4). These five explanatory variables explained 23.5% (adjusted R2 = 0.235) of the variance in the Lesser flamingos and their prey abundances. The analysis of variance (ANOVA) showed that the entire model of the RDA is statistically significant (F5= 2.05, p = 0.001), and RDA1 of the canonical axis resulting from the RDA is also statistically significant (F1= 4.07, p = 0.022). Except for PA, all variables included in the RDA model are significant (p < 0.05) as well (Table 3). Lesser flamingos standing out from the cluster of their prey items near the middle (Figure 4) implies that they were not present in many of the months. RDA showing the effects of selected environmental variables (PA = phenolphthalein alkalinity; TP = total phosphorus; Nitrite, Ammonia and turbidity) on the abundances of Lesser flamingo and their potential prey items (Nv = Navicula ventralis; Ol = Oocystis lacustris Ac = Anabaena circinalis ; Ps =Pseudoanabaena sp. Cm = Chroococcus minor; Ct = Chroococcus turgidus; Os = Oscillatoria sp; Sp = Spirulina plathensis; Ea = Euglena acus; Es = Euglena sp.) in a shallow wetland in Central Ethiopia from May 2021 to February 2022. Longer arrows indicate variable strongly drives the variation in the community matrix, and arrows pointing in opposite directions have a negative relationship, whereas arrows pointing in the same direction have a positive relationship. Variance Explained by Individual Physicochemical Parameters on the Abundances of Lesser Flamingos and their Prey Items in Chelekleka Wetland.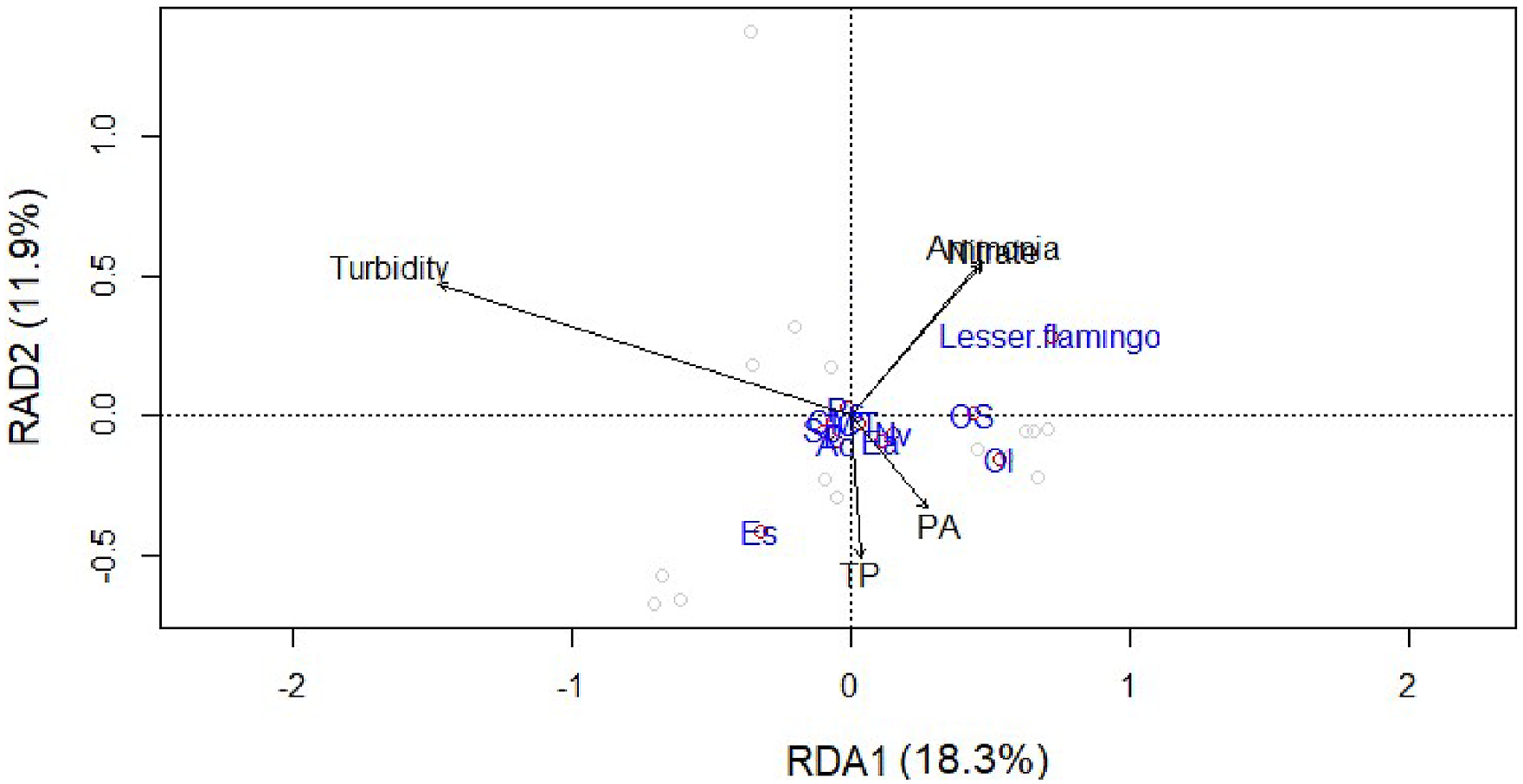

The abundances of Lesser flamingos positively correlated to Nitrate and Ammonia concentrations and negatively correlated to turbidity, TP and PA (Figure 4). Their abundance also indicated a weak negative correlation with abundances of Navicula ventralis, Oocystis lacustris, Chroococcus turgidus, and Euglena acus and highly negatively correlated with abundances of Chroococcus minor, Anabaena circinalis, Spirulina plathensis, Pseudoanabaena sp. and Euglena sp. (Figure 4).
Discussion
Chelekleka wetland experiences monthly fluctuations of physico-chemical characteristics, phytoplankton composition and Lesser flamingo abundance. The wetland supported different compositions of phytoplankton community that attract Lesser flamingos and other bird species suggesting the habitat suitability for various aquatic life.
The wetland water has an alkaline pH range that makes it suitable for aquatic life which is in line with previous findings (Degefu et al., 2014; Enawgaw & Lemma, 2018; Prakash et al., 2020). It was reported that water with a pH range of 6.5-8.5 pH is productive and optimal for the survival of most aquatic life (Mendoza et al., 2020; Prakash et al., 2020; Verma, 2020).
Chelekleka wetland had an optimum water temperature range of 20– 25°C. This range of water temperature is highly favorable for biological productivity and physiological activities of aquatic organisms (Verma, 2020) and phytoplankton in particular (Mendoza et al., 2020; Liu et al., 2021). The dissolved oxygen concentration in Chelekleka wetland fulfills the WHO standard for the survival of aquatic life (Enawgaw & Lemma, 2018; Verma, 2020) in all the months except June. The lowest dissolved oxygen concentration in this month might be related to factors such as cloudy weather, decaying of the aquatic plant, and respiration level of the aquatic life. Mendoza et al. (2020) noted that lower mean dissolved oxygen concentration during the wet season is associated with a limited amount of energy supply from the sun during precipitation time. Low Chl a concentration was recorded in Chelekleka wetland indicating low eutrophication as was reported by Liu et al. (2021).
Wet months recorded high turbidity of water in Chelekleka wetland compared to the dry months. This might be due to the addition of suspended sediments from surface run-off from the terrestrial environment during the rainy season. Similar findings were reported in different previous studies (Enawgaw & Lemma, 2018; Flipos & Subramanian, 2019; Mendoza et al., 2020; Verma, 2020). On the other hand, the dry months recorded a relatively higher concentration of nitrates, ammonia and total phosphorus compared to the wet months. Agricultural runoff from the wetland surroundings where irrigation activities are common, a discharge of sewage effluents from Bishoftu town, and decomposed organic matter and high temperature may have contributed to the increasing concentration of nutrients. This was in agreement with Prakash et al. (2020), Verma (2020) and Enawgaw & Lemma (2018) who reported that changes in nutrient concentration of water bodies depend on the introduction of nutrients from external sources.
The phytoplankton community in Chelekleka wetland was diverse (83 taxa) compared to previous studies in Ethiopian Rift Valley lakes (Enawgaw & Lemma, 2018; Kumssa and Bekele, 2014a) and other areas (Dalal et al., 2013; Flipos & Subramanian, 2019; Liu et al., 2021; Mendoza et al., 2020). This higher composition might be due to the dynamic water depth and content providing a range of conditions supporting different phytoplankton taxa. Chlorophyta recorded the highest species richness that might result from the suitability of the wetland for different green algae to proliferate. Similarly, a relatively high proportion of Chlorophyta in different alkaline lakes was reported in various studies (Dalal et al., 2013; Degefu et al., 2014; Flipos & Subramanian, 2019; Liu et al., 2021; Mendoza et al., 2020). Among the Chlorophyta, Botryococcus braunii is found to be the most abundant species in the wetland and Lake Ziqualla (Degefu et al., 2014) and in Lake Tana (Flipos & Subramanian, 2019) in Ethiopia reported similar findings.
In the study area, a monthly fluctuation in the abundance of phytoplankton community was reported as in other studies in Ethiopia (Enawgaw & Lemma, 2018; Flipos & Subramanian, 2019; Kumssa and Bekele, 2014a). This fluctuation might be associated with the monthly variations in the physico-chemical properties of the water (Liu et al. (2021). In addition, the adaptability of the different types of phytoplankton and grazing pressures from various life forms in the aquatic system can also influence phytoplankton abundance. Variouus scholars (Enawgaw & Lemma, 2018; Flipos & Subramanian, 2019; Kihwele et al., 2014; Liu et al., 2021) also reported the influence of light, water temperature, salinity, water movement, availability of nutrients, and grazing pressure as a major factor affecting phytoplankton community structures.
Lesser flamingo’s monthly abundance was also positively associated with the concentration of Nitrate, Ammonia and their correlated physico-chemical parameters of the wetland. A similar finding was reported by Kihwele et al. (2014) indicating that ammonium and nitrate were positively correlated to the abundance of lesser flamingos in shallow lakes of East Africa. These findings are in agreement with previous studies where the community structure of phytoplankton is significantly affected by nutrient concentration (Mendoza et al., 2020) and other environmental factors (Flipos & Subramanian, 2019; Kihwele et al., 2014; Liu et al., 2021). This suggests that the monthly fluctuations in the number of Lesser flamingos in Chelekleka wetland seem to be controlled by the function of physical and chemical characteristics of the wetland on the availability of their prey items. In addition, the significant positive correlation between the number of Lesser Flamingos and the concentration of ammonia, and nitrate in the wetland is most likely to be due to the presence of a relatively high number of the Lesser flamingo and other waterbirds as their droppings in water contribute to the nutrient enrichment (Kihwele et al., 2014).
There were no records of Lesser flamingos’ Anabaena visitation to the Chelekleka wetland when the abundance of Chroococcus minor and Euglena sp. were highest during the wet season. This might be due to the water’s high inundation and turbidity levels hindering flamingos from accessing those food items. Environmental variables such as water level fluctuation, turbidity nutrient concentrations, and trophic interactions could disturb aquatic biodiversity including the filter-feeding wading (Gownaris et al., 2018). However, Lesser flamingos' visits to the wetland were the highest from December to February during which potential phytoplankton prey consumed by Lesser flamingos, Navicula ventralis, Chroococcus turgidus, Anabaena circinalis, Spirulina plathensis and Oscillatoria sp. recorded the highest in abundance. The negative correlation between the Lesser flamingo abundance with their potential prey items might be due to the depredation effect by flamingo and other grazers. The differences in correlation among the preys might be related to the variation in preference of those preys by Lesser flamingo. Previous reports also showed that variation in the abundance of Lesser flamingos is influenced by changes in abundance and availability of their preferred foods (Kihwele et al., 2014) that in turn are influenced by the physico-chemical characteristics of the wetland (BirdLife International, 2018; Kaggwa et al., 2013; Kihwele et al., 2014; Krienitz et al., 2016; Kumssa and Bekele, 2014a; Tuite, 2000). This suggests the importance of the physico-chemical features of a wetland in regulating wetland biodiversity including seasonal visitor birds such as Lesser flamingos.
Implications for Conservation
Chelekleka wetland is a reasonably seasonally flooded alkaline habitat in the Ethiopian Rift Valley system that supports a diverse array of aquatic life forms including different phytoplankton taxa and the seasonally migratory species Lesser flamingo. The wetland is nationally recognized as an Important Bird Area (EWNHS, 1996) and internationally registered as a Key Biodiversity Area of international significance (Key Biodiversity Areas Partnership, 2024). Furthermore, the wetland is home to 246 species of resident and migratory bird species (Teklemariam et al., 2023) and diversified zooplankton and macroinvertebrate communities (Gebrekidan et al., 2024). Most importantly, the wetland is ecologically important for the near-threatened Lesser flamingo that utilize the area as an important foraging ground. The wetland physico-chemical characteristics vary across different months affecting phytoplankton diversity and hence Lesser flamingo abundance. Habitat degradation due to various anthropogenic factors such as irrigation agriculture, diverting feeder stream flow, pollution from nearby household sewage, wastewater from urban areas, and industrial effluents pose a serious threat to the sustainability of the wetland and its biodiversity (Gebrekidan et al., 2024; Teklemariam et al., 2023). Hence, urgent conservation interventions such as integrated wetland management may minimize anthropogenic threats and protect the ecosystem and its wildlife.
Footnotes
Acknowledgments
The authors thank Rufford Foundation, Addis Ababa University and the Ethiopian Wildlife Conservation Authority/KFW Conservation and Sustainable Use of Biodiversity in Priority Protected Areas of Ethiopia Project for funding this study as well as IDEA WILD and Wolkite University for supporting research equipment. The research permit from the Bishoftu City Environment, Forest, and Climate Change Authority is also well-acknowledged.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Rufford Foundation grant number 35814-1, Addis Ababa University Thematic fund 2019/20-22 and the Ethiopian Wildlife Conservation Authority/KFW Conservation and Sustainable Use of Biodiversity in Priority Protected areas of Ethiopia Project Grant number BMZ ID 2014671333. Mebrat Teklemariam has received all research supports from Rufford Foundation, Addis Ababa University and Ethiopian Wildlife Conservation Authority.
Appendix
Note *too rare to count. APhytoplankton identified only along the North site. BPhytoplankton identified only along the South site.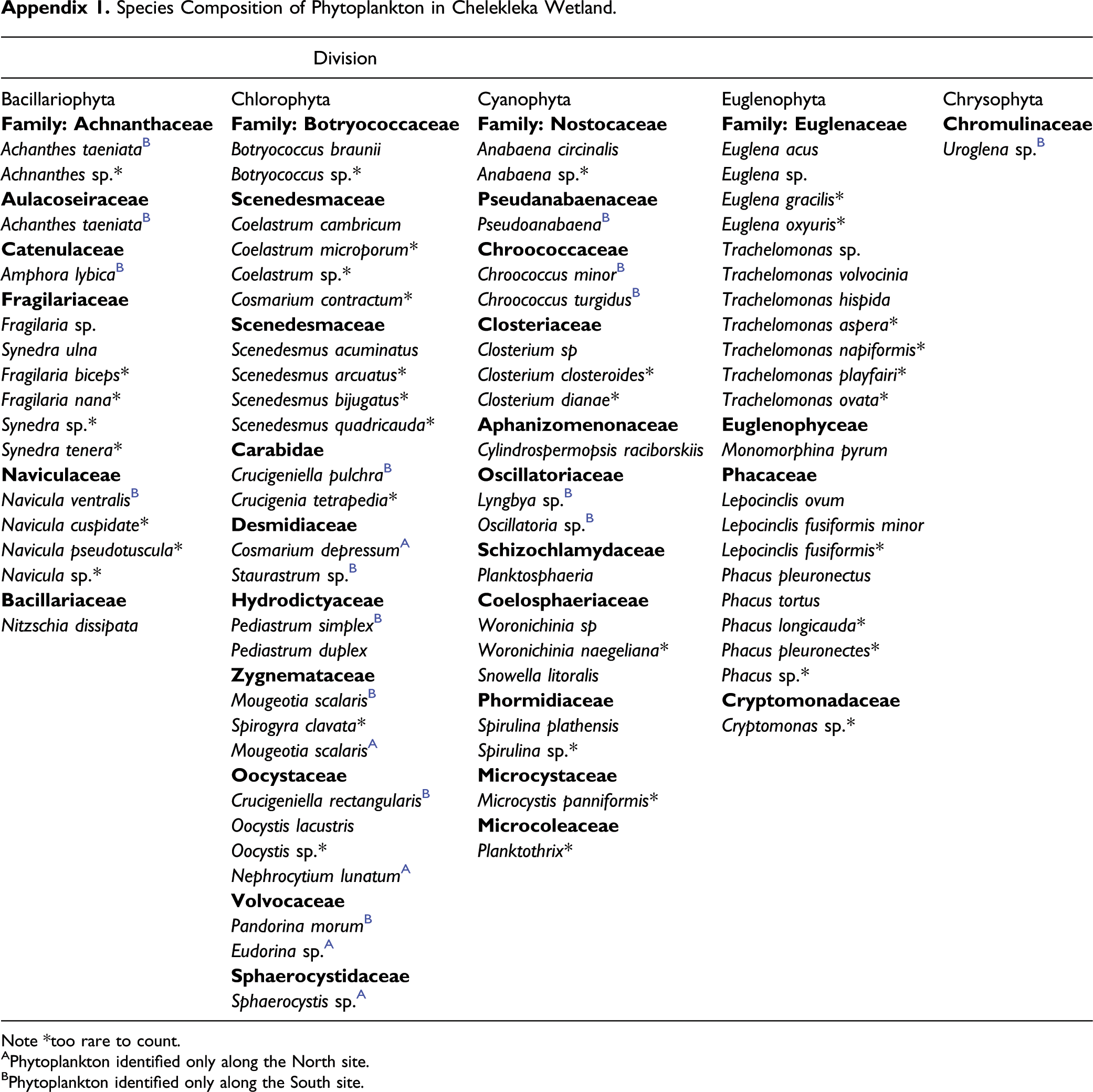
Division
Bacillariophyta
Chlorophyta
Cyanophyta
Euglenophyta
Chrysophyta
Achanthes taeniata
B
Botryococcus braunii
Anabaena circinalis
Euglena acus
Uroglena sp.
B
Achnanthes sp.
*
Botryococcus sp.
*
Anabaena sp.
*
Euglena sp.
Euglena gracilis
*
Achanthes taeniata
B
Coelastrum cambricum
Pseudoanabaena
B
Euglena oxyuris
*
Coelastrum microporum
*
Trachelomonas sp.
Amphora lybica
B
Coelastrum sp.
*
Chroococcus minor
B
Trachelomonas volvocinia
Cosmarium contractum
*
Chroococcus turgidus
B
Trachelomonas hispida
Fragilaria sp.
Trachelomonas aspera
*
Synedra ulna
Scenedesmus acuminatus
Closterium sp
Trachelomonas napiformis
*
Fragilaria biceps
*
Scenedesmus arcuatus
*
Closterium closteroides
*
Trachelomonas playfairi
*
Fragilaria nana
*
Scenedesmus bijugatus
*
Closterium dianae
*
Trachelomonas ovata
*
Synedra sp.
*
Scenedesmus quadricauda
*
Synedra tenera
*
Cylindrospermopsis raciborskiis
Monomorphina pyrum
Crucigeniella pulchra
B
Navicula ventralis
B
Crucigenia tetrapedia
*
Lyngbya sp.
B
Lepocinclis ovum
Navicula cuspidate
*
Oscillatoria sp.
B
Lepocinclis fusiformis minor
Navicula pseudotuscula
*
Cosmarium depressum
A
Lepocinclis fusiformis
*
Navicula sp.
*
Staurastrum sp.
B
Planktosphaeria
Phacus pleuronectus
Phacus tortus
Nitzschia dissipata
Pediastrum simplex
B
Woronichinia sp
Phacus longicauda
*
Pediastrum duplex
Woronichinia naegeliana
*
Phacus pleuronectes
*
Snowella litoralis
Phacus sp.
*
Mougeotia scalaris
B
Spirogyra clavata
*
Spirulina plathensis
Cryptomonas sp.
*
Mougeotia scalaris
A
Spirulina sp.
*
Crucigeniella rectangularis
B
Microcystis panniformis
*
Oocystis lacustris
Oocystis sp.
*
Planktothrix
*
Nephrocytium lunatum
A
Pandorina morum
B
Eudorina sp.
A
Sphaerocystis sp.
A
