Abstract
Soil respiration represents the largest carbon (C) flux from terrestrial ecosystems to the atmosphere. We created a study site in tropical lowland rainforest and used static chamber method to measure the temporal variations of soil respiration and their relationship with environmental factors at monthly time scale. The temporal variations of soil respiration showed a seasonal pattern related to soil temperature (
Global warming caused by the increase of carbon dioxide (CO2) concentrations is currently one of the most important issues to be addressed at a global scale (IPCC Climate Change, 2013). Soil respiration is the largest source of CO2 emissions from terrestrial ecosystems to the atmosphere (Bond-Lamberty & Thomson, 2010; Wu et al., 2011). Climatic warming is hypothesized to increase rates of soil respiration, potentially fueling further increases in global temperatures (Carey et al., 2016). Accurate estimation of soil respiration efflux and accurate identification of the factors controlling soil respiration are very important to understand the ecosystem C cycle under global climate change scenarios (Bond-Lamberty & Thomson, 2010).
Tropical forests contain 40% of the total C in global terrestrial ecosystems (Beer et al., 2010; Jobbágy & Jackson, 2000; Pan et al., 2011), of which, 56% is found in aboveground biomass and 32% in soils (Ngo et al., 2013; Pan et al., 2011). They exchange more CO2 with the atmosphere than any other biome on Earth (Cavaleri et al., 2015) and, thus, play a key role in the global C cycle (Goodrick et al., 2016). In the coming decades, the tropics will experience unprecedented changes in temperature, rapid increases in atmospheric CO2 concentrations, and significant alterations in the timing and amount of rainfall (Anderson, 2012; Diffenbaugh & Scherer, 2011; Mora et al., 2013).
In principle, soil respiration is the combination of autotrophic respiration by plant roots and associated microorganisms (i.e., rhizosphere respiration) and heterotrophic respiration by microbes decomposing soil organic matter (Hanson et al., 2000; Högberg & Read, 2006; Ryan & Law, 2005). Soil moisture and temperature are two primary abiotic drivers of root and microbial activities (Davidson et al., 1998; Liu et al., 2016; Lloyd & Taylor, 1994). Understanding their potential moderating effects on soil respiration is essential for predicting the responses of soil respiration to climate changes.
Soil respiration rates have been studied in many of the world’s ecosystems to explain the relationship between soil respiration and environmental factors. In boreal ecosystems and temperate regions, soil temperature is the most important determinant of soil respiration rate (Shibistova et al., 2002; Vargas et al., 2010; Xu & Qi, 2001). Compared with other factors, temperature can directly affect root and microbial metabolic rates (Kuzyakov & Gavrichkova, 2010; Lükewille & Wright, 1997) as well as temporal variations in soil respiration. The C efflux from soil in these regions can be estimated using an empirical function and soil temperature data (Adachi et al., 2009). Different types of models have been used: exponential or Arrhenius equations (Lloyd & Taylor, 1994; MacDonald et al., 1995; Thierron & Laudelout, 1996), linear models (Rochette et al., 1991), and quadratic models (Holthausen & Caldwell, 1980). Although these models have been reported to be successful in fitting data obtained from an individual experiment under some specific circumstances, they suggest different explanations for the response of soil respiration to temperature. The
In tropical regions, soil temperatures do not strongly influence the soil respiration rates; instead, most previous studies have highlighted the control of soil moisture on soil respiration (Davidson et al., 2000; Schwendenmann et al., 2003; Sotta et al., 2006). Soil CO2 efflux can be suppressed in both low and high soil water content (Davidson et al., 2012; Liptzin et al., 2011). Several nonlinear relationships have been proposed to link soil respiration rate and soil water content (Cook & Orchard, 2008; Davidson et al., 2000), indicating optimal conditions for microbial decomposition and root respiration at intermediate moisture conditions. However, these relationships remain empirical, and it is unknown how they vary with soil, climate, and forest type (Rubio & Detto, 2017). Sotta et al. (2004) claimed that short-term variation in soil respiration rates depend on soil temperature, but the soil water contents might be a limiting factor of long-term variation in soil respiration rates in central Amazonian tropical forests.
Therefore, the effects of soil temperature and moisture on soil respiration still need to be expanded. We investigated soil respiration by measuring soil CO2 flux, soil temperature, and soil moisture at monthly time scale in Diaoluoshan tropical lowland rainforest in Hainan Island. We aimed to address two issues in this study: (a) the relationship between soil respiration and key environmental factors (soil temperature and moisture) in Diaoluoshan tropical lowland rainforest in Hainan Island and (b) the performance of different regression models with the soil respiration data of Diaoluoshan tropical lowland forest and how C efflux was affected.
Methods
Site Description
This study was conducted in Diaoluoshan National Nature Reserve (DNNR) located in the south-eastern part of Hainan Island, China (18°40′ N, 109°54′ E, elevation 255 m, Figure 1). The climate shows strong seasonality because of the tropical marine monsoon climate. The dry and rainy seasons are clearly divided, and more than 80% of the precipitation is concentrated in the rainy season extending from late April to October. Annual rainfall is relatively high and may reach up to 2,160 mm (typhoons might bring some rainfall). The multiyear mean annual temperature is 24.6°C, with the highest value occurring in July and the lowest in January (Zhao et al., 2019).

Geographic Location of Study Site. DNNR = Diaoluoshan National Nature Reserve.
DNNR has typical zonal forests, including lowland rainforests, mountain rainforests, and hilltop forests. Our study site was categorized as a lowland tropical forest (Yang et al., 1994). The mean canopy height of the forests is approximately 20 to 25 m. The canopy height is lower than that of the inland tropical forests at the same latitude, which might partly be a consequence of typhoons. Before the establishment of the National Nature Reserve, the forest was extensively cut in the 1980s. The dominant species is
Experiment Design
The field campaign was conducted in a 1-ha permanent plot. Three quadrats were selected in the permanent plot to install soil chambers. The chamber consisted of polyvinyl chloride collar of 20 cm in diameter and 10 cm in height, which embedded seamlessly and inserted 3 to 5 cm into the soil, followed by the removal of aboveground biomass. Five chambers were randomly installed in each quadrat, keeping their same position for minimizing interference with the soil environment.
From September 2016 to January 2018, we performed field measurements at monthly intervals and generally completed them before noon. Diurnal variation measurements (7:00–19:00) were conducted with the frequency of each hour on March 18, 2019.
Field Measurements
We used a portable greenhouse gas analyzer, purchased from Los Gatos Research Company (LGR Inc., San Jose, CA, USA), to measure soil respiration. It could store full absorption spectra at a frequency of 1 Hz for further processing or corrections on fluxes. Soil temperature and moisture at 5 cm depth were measured simultaneously with Decagon 5 TM sensor (Decagon Devices Inc., USA). We performed at least three measurements in each chamber, allowing enough time for the analyzer to warm up each time.
Statistics
The one-way analysis of variance was used to determine the differences in mean soil respiration rates during the rainy and dry seasons. A Pearson’s correlation analysis was used to determine whether there was a correlation between soil respiration and soil temperature and moisture and correlated at
Regression Models
Single-Factor Models of Soil Respiration and Moisture
As shown in Equations (1) to (3), three models that only consider the relationship between soil respiration and moisture were used to fit the relations. Their functions are as follows:
Linear model:

Power function model:

Quadratic model:

Single-Factor Models of Soil Respiration and Temperature
As shown in Equations (4) to (8), five models that only consider the relationship between soil respiration and temperature were used to fit the relations. Their functions are as follows:
Linear model:

Exponential model:

Quadratic model:

Arrhenius model (Fang & Moncrieff, 2001):

Lloyd and Taylor model (Lloyd & Taylor, 1994):

Q10 Value
The temperature dependence of soil respiration, commonly referred to as the 

Mixed Models of Soil Respiration Considering Soil Temperature and Moisture
Equations (10) to (14) were used to study how the two factors of soil temperature and moisture together control soil respiration. Their functions are as follows:
Linear model 1:

Linear model 2:

Quadratic model:
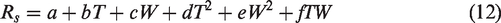
Power function model:

Exponential model:

Results
Seasonal Variation in Soil Respiration and Environmental Factors
Both soil temperature and moisture were measured in each field campaign, while simultaneously measuring soil respiration. As shown in Figure 2, seasonal patterns were observed in soil respiration, temperature, and moisture. Their annual average values were 2.52 ± 0.23 SE μmol CO2 m−2 · s−1, 24.02 ± 0.88 SE °C, and 12.88 ± 1.24 SE %, respectively.

Seasonal Dynamics of Soil Respiration (CO2 Flux) and Soil Temperature (
There is a significantly positive correlation between soil temperature and soil moisture at the depth of 5 cm (
Time series of soil respiration measurement varied greatly that ranged from 1.42 to 3.94 μmol CO2 m−2 · s−1 and peaked in June 2017. One-way analysis of variance showed significant variations in soil respiration rates during the rainy and dry seasons (
Diurnal Variation in Soil Respiration
Diurnal variation in soil respiration is shown in Figure 3. The mean rate of soil respiration was 1.97 μmol CO2 m−2 · s−1. Based on the measurements performed on March 18, 2019, no significant diurnal change was observed in soil respiration, and it was not related to soil temperature (

Diurnal Dynamics of Soil Respiration (CO2 Flux) and Soil Temperature (
Seasonal Controls of Soil Respiration
During the entire study period, the average soil respiration rate for different seasons was the highest in summer and lowest in winter. This was consistent with the change in soil temperature at 0–5 cm depth (Figure 4A). Meanwhile, the average soil respiration flux in the dry season (November to April) was smaller than that in the wet season (May to October), which was consistent with the change in soil moisture at 0 to 5 cm depth (Figure 4B).

Soil Respiration in Different Seasons Was Compared With Soil Temperature (A) and Soil Moisture (B).
Relationship Between Soil Respiration and Moisture
Pearson’s correlation analysis showed a significant positive correlation between soil CO2 fluxes and soil moisture levels in our study (
Fitted Relationships Between Soil Respiration and Soil Temperature and Moisture.


Different Models Fitting of Soil Respiration and Soil Moisture.
Relationship Between Soil Respiration and Temperature
Pearson’s correlation analysis showed a significant positive correlation between soil CO2 flux and soil temperature in our study (

Different Regression Models Fitting of Soil Respiration and Soil Temperature. A: Relationship between soil respiration and soil temperature fitted by different models throughout the total study period. B and C: Relationship between soil respiration and soil temperature fitted by different models in dry and wet season, respectively.
Effect of Different Regression Models on Q10
Despite the fact that most equations fitted the observed data well and provided similar estimates of soil respiration at different temperatures, the
Combined Effect of Soil Temperature and Moisture on Soil Respiration
The field campaign measurement of soil respiration reflects the combined effects of soil environmental factors. In these mixed models, the quadratic model had the best fit and performed better than any single-factor models (Table 2), which had the highest

Combined Effect of Soil Moisture and Temperature on Soil Respiration by Fitting With the Mixed Quadratic Model.
Discussion
Temporal Variations of Soil Respiration Rates
The soil respiration rates of the tropical lowland rainforest in Diaoluoshan, Hainan Island, exhibited seasonal variations (Figure 2). The temporal variations of soil respiration were sensitive to seasonal changes in soil environmental conditions (Figure 4). Emissions in the wet season were much greater because of the higher soil hydrothermal conditions which were beneficial to the life activities of soil microorganisms and plant roots than in the dry season. In addition, soil hydraulic condition is an important medium for the transfer of soil organic matter. Such patterns of soil respiration in tropical forests have also been observed at other sites such as Thailand (Adachi et al., 2009), Australia (Goodrick et al., 2016), Jianfengling (Jiang et al., 2016), Panama (Rubio & Detto, 2017), Xishuangbanna (Sha et al., 2005), and Amazon (Sotta et al., 2004). The annual soil respiration efflux at our site was 953 ± 87 g C m−2 · yr−1; this value is lower than that of a tropical lowland rainforest in Panama (1613 g C m−2 · yr−1; Rubio & Detto, 2017), Thailand (1724 g C m−2 · yr−1; Adachi et al., 2009), and Amazon (1487 g C m−2 · yr−1; Sotta et al., 2006), but higher than that of the tropical lowland rainforests in Xishuangbanna, China (831 g C m−2 · yr−1; Sha et al., 2005) and Hawaii (650–890 g C m−2 · yr−1; Raich, 1998). Soil is a complex and spatially heterogeneous mixture of minerals and organic pools, including litter, roots, and microorganisms. Each of these components responds differently to environmental variability (P. Li et al., 2013) and is uniquely coupled with other biotic processes, generating a broad spectrum of CO2 emission rates. Thus, it is not surprising that soil respiration rates vary considerably in different tropical forests.
Our results showed no diurnal fluctuations of soil respiration in Diaoluoshan tropical lowland rainforest between 7:00 and 19:00 h (Figure 3) and were broadly consistent with the results from other tropical forest ecosystems (Sha et al., 2005). On a short-term scale, temperature is the most important factor that determines the rate of soil respiration (Sotta et al., 2004) because temperature is a direct factor affecting root and microbial metabolic rates (Kuzyakov & Gavrichkova, 2010; Lükewille & Wright, 1997). However, soil temperature diurnal fluctuations in the tropics are small, especially in the areas covered by dense canopy, where little radiation reaches forest floors. Thus, in a tropical forest, no obvious diurnal fluctuations might be observed in soil respiration.
Effects of Environmental Factors on Soil Respiration
Soil temperature and moisture are the two most important soil parameters for soil CO2 emissions, since they control microbial activity and all related processes. In several ecosystems, soil CO2 fluxes increase exponentially with temperature and are often limited by available soil moisture (Carlyle & Than, 1988; Fang & Moncrieff, 2001; Xu & Shang, 2016). Nevertheless, because the temporal variation of soil temperature in the tropics lies usually within the optimum range of soil respiration, soil moisture is probably the most important abiotic factor influencing soil respiration within tropical forests (Y. Li et al., 2006; Sotta et al., 2006). Rubio and Detto (2017) claimed that soil moisture was the primary driver of temporal variability in tropical forests, with temperature playing a secondary role. However, as Davidson et al. (1998) pointed out, it is difficult, and perhaps impossible, to distinguish the effects of soil temperature and moisture separately. At our study site, the soil CO2 efflux exhibited a significant relationship with the interaction between soil temperature and soil water content (
On the other hand, low soil temperatures occurred simultaneously during the dry season in our study (Figure 2). Low soil temperatures resulted in low root and microbial metabolic rates. The soil water content at 0 to 5 cm depth decreased to 10%, probably resulting in very low root and microbial activity (Silletta et al., 2019). The decrease in soil water availability during the dry season affects several physiological processes, leading to plant dehydration and a substantial loss of root functionality in this ecosystem (Bucci et al., 2013; Scholz et al., 2012). This was also reflected in a decrease in plant transpiration during the drought period (Pereyra et al., 2017), and hence a decrease in CO2 capture. Furthermore, at low soil moisture conditions, decomposition is limited by soluble C availability (Davidson et al., 2012; Linn & Doran, 1984).
Selection of Soil Respiration Models
As the large temporal variations of soil water content, the single-factor models of soil moisture could not clearly explain and predict soil respiration (Table 2). The relationship between soil respiration rate and soil water content can be expressed as a quadratic function (Figure 5) at our study site. However, this relationship was not consistent in different studies. Adachi et al. (2009) and Sotta et al. (2006, 2007) studied different tropical forests and reported that soil respiration rate and soil water content could be described using a parabolic function. Chambers et al. (2004) reported that the relationship between soil respiration rate and soil water content is curvilinear.
In contrast, our data fitted better with the single-factor models of the effect of soil temperature on soil respiration (Table 2), though there might be large variations due to the higher water content in the wet season (Figure 6). It is difficult to identify the best model because of the close values of the coefficients of determination. A good fit of a model against the experimental data does not necessarily suggest the actual mechanism presumed by the model (Fang & Moncrieff, 2001).
The linear and quadratic models of the relationship between soil respiration and soil temperature are empirical and lack theoretical basis. The responses of soil respiration and mineralization processes to soil temperature are commonly described using exponential and Arrhenius equations. Both equations describe an exponential increase in respiration with increasing temperature, but with a different theoretical basis (Ellert & Bettany, 1992). An exponential increase in soil respiration with respect to temperature is commonly accepted (O’Connell, 1990; Thierron & Laudelout, 1996; Winkler et al., 1996). However, an exponential model could express the relationship between soil respiration and temperature under laboratory conditions, but not in field campaigns. At a high temperature, the sensitivity of soil respiration to temperature might be reduced in a field campaign. Enzymes might be deactivated or destroyed by a further high temperature.
Although the Arrhenius equation has a basis in thermodynamics, it might somewhat oversimplify the response mechanism of soil respiration to temperature (Ellert & Bettany, 1992). This equation uses the reciprocal of absolute temperature to predict the variation in respiration rate, suggesting that it might not be sensitive enough to the variation in soil respiration when temperature is low (Fang & Moncrieff, 2001). Lloyd and Taylor (1994) pointed out that a high value of activation energy under low temperature leads to an unbiased simulation of the equation.
The
Soil water content was significantly correlated with soil temperature at our site and covaried across seasons (Figure 2). It is very difficult to distinguish the response of soil respiration to soil temperature and water content, as the soils vary with seasons. Therefore, the model considering only one single factor might ignore the influence of another factor to a certain extent. However, the mixed models could reflect the effects of two factors on soil CO2 flux (Figure 7). Silletta et al. (2019) pointed out that the seasonal variations in soil respiration were mainly explained by the interaction between soil temperature and water content at a Patagonian site in Argentina. In our site, the warmer and wetter soil climate in the wet season increased soil respiration. However, when the moisture exceeds the optimal range, soil respiration becomes limited, offsetting the positive effects caused by the elevated soil temperature and reducing total respiration. At low soil temperatures, a clear trend of the soil respiration decreasing with increasing soil moisture was observed.
Implications for Conservation
The temporal variations and its determinants of soil respiration were investigated in a tropical lowland rainforest in Hainan Island. We found that (a) soil respiration varied significantly with the seasonal change in soil temperature and moisture; (b) soil respiration in the studied tropical lowland rainforest was co-controlled by the two factors of soil temperature and moisture, and the quadratic mixed model could best explain the effects. These two findings are highlighted as important for reducing errors in soil respiration evaluation and scaling up of soil C flux in climate change studies.
Supplemental Material
TRC914902 Supplementary material - Supplemental material for Controls of Temporal Variations on Soil Respiration in a Tropical Lowland Rainforest in Hainan Island, China
Supplemental material, TRC914902 Supplementary material for Controls of Temporal Variations on Soil Respiration in a Tropical Lowland Rainforest in Hainan Island, China by Yi-Bin Cui, Ji-Guang Feng, Li-Guo Liao, Rui Yu, Xiang Zhang, Yu-Hai Liu, Lian-Yan Yang, Jun-Fu Zhao and Zheng-Hong Tan in Tropical Conservation Science
Footnotes
Acknowledgments
We sincerely thank the national nature reserve of Diaoluoshan, especially Ms. Hai-yan Deng and Mr. Hai-wei Liu for their help with our field work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundations of China (grant nos. 41771099, 41861023, and 31660142) and C-Talent project of Hainan University (KYQD1626).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
