Abstract
Keywords
Introduction
Most descriptions of plant communities analyze species richness and abundance (Owen et al., 2019). As such, taxonomic diversity has generally been the basis for defining the role of natural protected areas (NPAs) in biodiversity conservation (Cadotte & Tucker, 2018). However, the number of species per se does not provide information about other key elements (e.g., reproduction, pollination, seed dispersal) that affect the capacity of plant communities to regenerate and persist in the long term (Larson & Funk, 2016). Relevant information on species’ natural history is increasingly necessary due to the constant loss and fragmentation of natural habitats (Sala et al., 2000). For example, in the selection of natural areas for conservation, it has recently been pointed out that it is necessary to consider other dimensions of biological diversity, such as phylogenetic and functional diversity, to conserve the evolutionary history and function of ecosystems and to understand their resilience and resistance to anthropogenic disturbance (Owen et al., 2019; Winter et al., 2013).
One way to assess the regenerative capacity of plant communities in NPAs is to characterize plant traits that influence their reproduction, survival, and ecosystem functions (Norden et al., 2009; Violle, 2007). The reproductive structures of plants, such as flowers and fruits, provide valuable information about their interactions with pollinators and seed dispersers, processes essential for maintaining diversity and gene flow within and between populations, and thus their long-term persistence (Aguilar et al., 2019; Neuschulz et al., 2016). For example, pollination is a key process for the reproduction of more than 85% of plant species native to the tropics and nearly 78% of cultivated plants (Ollerton et al., 2011; Quesada et al., 2009; Watanabe, 2014) and ensures their genetic variability in the populations (Aguilar et al., 2019). In addition, the regeneration of tropical forests depends on seeds reaching adequate places for their germination and establishment, which is closely related to how plants disperse their seeds (Howe, 2016). Therefore, fruit characteristics related to seed dispersal can provide significant information about the species’ dispersal capacity, which has been used to evaluate the resilience of plant communities to constant anthropogenic threats (e.g., land use change, habitat loss, climate change) (Escobar et al., 2020).
Interactions of plants with their pollinators and dispersers have mostly been evaluated in one or a few species at a time (Búrquez et al., 1987; Hoeltgebaum et al., 2018; Listabarth, 1993; Siefke & Bernal, 2004). However, analyzing community-level plant pollination and dispersal syndromes (e.g., different growth forms) is an important analytical strategy for understanding larger-scale patterns and identifying species assemblages vulnerable to the current environmental crisis (Cortés-Flores et al., 2017; Griz & Machado, 2001; Rosas-Guerrero et al., 2014). For example, sex expression is a pollination-related trait that can provide information about the plant’s vulnerability to habitat loss or fragmentation. Dioecious species, in which the female and the male individuals are spatially separated, are more vulnerable to fragmentation and isolation (Aguilar et al., 2019; Aguilar-Aguilar et al., 2023). Meanwhile, habitat loss has been shown to negatively impact populations of frugivores and seed dispersers, threatening the persistence of plants that depend on animals for dispersal (Lacher et al., 2019). In addition, placing ecological attributes of plants into a phylogenetic context can provide useful information to detect groups of species or even whole clades that may be particularly vulnerable to anthropic pressures (Velazco and Nevo, 2020; Zhang et al., 2024). However, few studies integrate taxonomic, functional, and phylogenetic dimensions of diversity to evaluate the biodiversity found in NPAs under the current scenario of accelerated diversity loss (Cui et al., 2024; Dehling et al., 2022; Lu et al., 2023; Manrique-Ascencio, 2024).
Central America is a region that hosts highly diverse tropical ecosystems, such as those established along elevational gradients in the mountains (e.g., tropical rainforest, montane forest, paramo) (Brummitt et al., 2021). However, this is one of the world’s most affected regions by land-use change, and at the same time one of the least studied in almost all areas of plant biology (except for Costa Rica, e.g., Raven et al., 2020). In this study, we updated the plant species records of two National Protected Areas (NPA) in Honduras and for each species we determined its risk category, as well as different reproductive traits related to pollination syndromes (flower color, size and sex expression) and dispersal (fruit color, size and type). Specifically, we evaluated the frequency of species with different growth forms and traits associated with pollination and seed dispersal syndromes and determined, whether the evaluated traits are more similar in the most phylogenetically related species. As in other tropical ecosystems, we hypothesize that we will find a high frequency of species with flower and fruit traits associated with pollination and seed dispersal by animals (Griz & Machado, 2001; Pereira et al., 2022; Sengupta, 2022). Given the close relationship between growth form and life history strategies of plants, growth form is expected to be similar in the most closely related species (Chazdon et al., 2003). In contrast, we hypothesize that the phylogenetic signal will be null for pollination and dispersal syndromes because these are sets of traits that have evolved convergently in different plant groups (Ashworth et al., 2015; Valenta & Nevo, 2020). In the context of our results, we highlight the importance of species conservation in these two Honduran NPAs from the perspectives of biotic interactions and ecosystem services in highly degraded landscapes.
Methods
Study Area
The present study was carried out in two NPAs in Honduras: Pico Bonito National Park (PBNP) and the Uyuca Biological Reserve (UBR), two protected natural areas surrounded by different anthropogenic land uses (Mora et al., 2013; REHDES, 2004). PBNP was decreed as a protected area with the objective of protecting cloud forests as a water catchment, at the same time as protecting its wild flora and fauna. PBNP is in northern Honduras, covers an area of 1,073 km2, and has an elevation gradient from 60 to 2,480 m a.s.l. The climate of this region is tropical rainy; annual precipitation ranges from 1,520–4,300 mm and occurs between June and February. The main ecosystems in PBNP are Tropical Rainforest, Montane Forest and Pine-Oak Forest. In the buffer area around PBNP the main activities are livestock grazing and industrial cultivation of banana (Musa acuminata, M. paradisiaca), pineapple (Ananas comosus) and African oil palm (Elaeis guineensis). Also, there are smaller-scale crops of cacao (Theobroma cacao), coffee (Coffea arabica) and rambutan (Nephelium lappaceum) (AFE-COHDEFOR, 2004; REHDES, 2004).
The Uyuca Biological Reserve (UBR) was declared a protected area in 1985. It has an approximate area of 9 km2 and the altitude range is 950 to 2,000 m a.s.l. The annual precipitation ranges from 1,500 to 2,400 mm and the rainy season occurs from May to November. UBR vegetation is dominated by Montane Forest and Pine-Oak Forest, mainly composed of shrubs and herbaceous vegetation. This is an important zone for the connectivity of the central forest corridor of Honduras. The land surrounding UBR has been used for the cultivation of potatoes (Solanum tuberosum), kale (Brassica oleracea var. capitata), carrots (Daucus carota), onions (Allium cepa) and staple crops such as maize (Zea mays) and beans (Phaseolus vulgaris), as well as for silvopastoral activities (Mora et al., 2013). In PBNP, agriculture, African oil palm and pineapple plantations, and the use of surface and groundwater for irrigation have threatened species diversity (AFE-COHDEFOR, 2004). On the other hand, the areas around UBR have been affected by wildfires, plant extraction, and urban growth (Mora et al., 2013). The loss of forest cover in these two NPAs could negatively impact species persistence, the stability of diverse biotic interactions, and the ecosystem services that benefit the communities surrounding these NPAs.
Data Collection
Review of Floristic Literature
A comprehensive review of local and regional floras, floristic inventories, management plans, and the database of Cyril Hardy Nelson Sutherland (TEFH) and Paul C. Standley Herbaria (EAP) of the PBNP and UBR was conducted (International Resources Group, 2006; Kukkonen & Hohnwald, 2009; Mora et al., 2013; REHDES, 2004). After initially listing the species, we implemented a curatorial process that involved two phases. First, we reviewed the names of the plant species present in the two NPAs, which were verified and updated following the “Angiosperm Phylogeny Group IV” (APG IV, 2016). Secondly, species with synonyms, dubious names, or incomplete identifications (i.e., to genus or family level) were corrected. In this phase, species for which no reliable identification data were available and for which it was not possible to access further information on their records or herbarium specimens were excluded ([insert Figure S1]). After thorough analysis, we were able to record 538 and 420 species in PBNP and UBR, respectively.
Species Attributes
Following the establishment of the definitive list of species, the information corresponding to each functional trait was assigned in accordance with the bibliography. In instances where no such information was found, traits were assigned based on observations of reproductive structures in herbarium specimens. The growth form of the plants was assigned considering the following categories: trees (woody plants ≥ 3 m height), shrubs (woody plants that branch from the base and ≤ 3 m height), herbs (non-woody annual and perennial plants), lianas (woody vines), and epiphytes (non-woody plants that grow on another plant) (Moreno, 1984). We assigned reproductive attributes based on the literature review and observation of the reproductive structures in the field and herbarium specimens. Sex expression of the flowers was assigned according to Bawa (1974) as sexually monomorphic flowers (hermaphroditic, monoecious, andromonoecious, or gynomonoecious) or sexually dimorphic (dioecious, androdioecious, or gynodioecious). Flower size was categorized according to the proposal by Rosas-Guerrero et al. (2014) as small (< 15 mm), medium (15-30 mm) or large (30-60 mm). Flower colors were classified (based on human vision) as green, pale (white, pale brown, pale pink) yellow, pink, orange, red, blue, or purple. Pollination syndromes were assigned according to flower morphology (color, shape and size) and consulting specialized literature (e.g., papers, regional floras). We classified species into the functional groups proposed by Frankie et al. (2004): plants pollinated by bees, butterflies, birds, bats, wind, or small insects (e.g., beetles, flies, ants, wasps, thrips).
We determined the fruit traits of each species based on reported in botanical descriptions in the World Flora Online database, and regional floras (Mesoamericana, Nicaragua). The fruit type was classified as achene, berry, schizocarp, strobilus, follicle, legume, nut, samara, or syconium (Glimn-Lacy & Kaufman, 2006). Fruit color was classified as green, red, black, brown, white, blue, orange, or yellow (Wheelwright & Janson, 1985). According to the color and type of fruit, we classified dispersal syndromes in zoochory (including epizoochory and endozoochory), autochory (including ballistic dispersal and barochory), or anemochory (Van der Pijl, 1972). Additionally, to assign the conservation status of the species recorded, we consulted the Red List databases (IUCN, 2023).
Data Analysis
We evaluated the difference in species frequency of reproductive traits and their association with growth form using log-linear models with a Poisson distribution in R software (Core Team R, 2021).
For each NPAs, we constructed a phylogenetic hypothesis based on all recorded plants. These phylogenetic trees included clustering of all vascular plants based on the ‘Open Tree of Life’ (Smith & Brown, 2018) using the megatree approach of the ‘U.PhyloMaker’ package (Jin & Qian, 2019) in R. The megatree is a well resolved tree, often based on phylogenies from already published phylogenies or compilations of phylogenetic information. This megatree is the basis for generating phylogenetic hypotheses of the set of species of interest. We use of GBOTB.extended.TPL.tre as megatree (Jin & Qian, 2022). The phylogenetic hypotheses of the two NPAs were generated using the scenario 3 reconstruction method, which consists of connecting the tip of a new genus or species to the midpoint of the family branch (Qian & Jin, 2021). This method is useful for generating hypotheses about diverse plant communities. However, it is not precise enough for studies requiring high phylogenetic resolution.
The multicotomies were resolved in the order in which they appear in the tree using the multi2di function of the ‘ape’ package (Paradis and Schliep, 2019) in R. To calculate the phylogenetic signal, the growth form, pollination, and dispersal syndromes were transformed into a series of binary characters. The conversion of multi-state attributes to binary characters is performed as follows: each state of the attributes is considered individually. A matrix of presence and absence is subsequently generated. In this matrix, 1 represents the presence of each attribute state, and 0 represents its absence. The D statistic of phylogenetic signal for binary characters was implemented with the function ‘phylo.d’ in the caper package in R (Fritz & Purvis, 2010). This algorithm is based on the differences between sister clades in each phylogeny. A D statistic value of 1 indicates that a binary trait has a random phylogenetic distribution, while a value of 0 or negative indicates that the trait is grouped and has evolved under the Brownian motion model. Phylogenetic signal was calculated using the Fritz and Purvis D, which is an approach to evaluating categorical attributes that considers the Brownian model of evolution to assess whether an attribute is more similar in phylogenetically closer species (Baeckens et al., 2023; Iglesias-Carrasco et al., 2022; Kamilar & Cooper, 2013). A categorical matrix was constructed with the different growth forms and pollination and dispersal syndromes for each species. This matrix was the basis for plotting each trait at the tips of the phylogenetic trees of each NPA. This procedure was carried out with the ‘ggtree’ package (Yu et al., 2017) in R.
Results
Species Composition and Conservation Status
The families with the largest number of species in PBNP were Rubiaceae (43), Melastomataceae (37), Orchidaceae (32), Fabaceae (28), Lauraceae (17) and Euphorbiaceae (16); while in UBR they were Asteraceae (51), Orchidaceae (49), Poaceae (45), Bromeliaceae (20) and Fabaceae (18) (Table S1 and S2). Within each NPA, the frequency of species by growth form differed significantly. In PBNP, the most common growth forms were trees (41%) and shrubs (29%), and the least common were herbs (21%), lianas (4%) and epiphytes (5%). In UBR, herbs were the most common (60%), followed by shrubs (16%) and trees (14%). Epiphytes (6%) and lianas (4%) were less common.
Species With the Highest Risk Categories Present in the Uyuca Biological Reserve (UBR) and Pico Bonito National Park (PBNP), According to the IUCN Red List. Critically Endangered (CR), Endangered (EN) and Vulnerable (VU)

Floral Attributes and Pollination Syndromes
Results of Log-Linear Models to Assess Differences in Species Frequencies for Each Attribute (Chi-Square for Goodness of Fit) and to Assess Whether Differences in Frequencies Depend on Growth Form (Chi-Square for Independence; Letters in Italicize). In Pico Bonito National Park (PBNP) and the Uyuca Biological Reserve (UBR), Honduras

Most species had monomorphic flowers in PBNP 87% (71 % hermaphroditic and 16% monoecious) and UBR 91% (81 % hermaphroditic and 10% monoecious); the remaining species were dioecious (13% and 9% of species in PBNP and UBR, respectively; [insert Figure 1(A), (B)]). In both NPAs, sex expression was significantly associated with growth form (Table 2). Shrubs (94% in PBNP and 98% in UBR), herbs (92% and 94%), and to a lesser extent trees (82% and 70%) were more likely to be hermaphroditic. In the UBR, a large number of tree species (30%) had dioecious flowers (Table 2 and [insert Figure 1(A), (B)]). Frequency of Species by Sex Expression (A-B), Flower Color (C-D) and Pollination Syndromes (E-F) in Different Growth Forms in the Pico Bonito National Park and the Uyuca Biological Reserve
Overall, pale-colored flowers (white, pale brown, pale pink) were the most frequent in both PBNP (74%) and UBR (64%). In both NPAs, flower color was related to growth form (Table 2). Purple flowers were common in herbs (PBNP 10% and UBR 15%), orange flowers were common in epiphytes in PBNP (66%), and yellow flowers were common in vines (PBNP 20% and UBR 16%; Table 2 and [insert Figure 1(C)-(D)]).
In both NPAs, we found that most plant species have pollination syndromes associated with biotic vectors, and these floral traits are significantly dependent on growth form (Table 2). In PBNP, the most frequent pollination syndromes were those associated with bees, small insects, and wind for trees; bees, small insects, birds, and butterflies for shrubs; and small insects and bees for herbs. In UBR, pollination by bees and wind were the most frequent syndromes for trees; bees and birds were the most common syndromes for shrubs; and wind, bee, and small insect pollination syndromes were the most common for herbs (Table 2 and [insert Figure 1(E)-(F)]).
Fruit Attributes and Dispersal Syndromes
We found that most species have fruit attributes related to dispersal by animals in the two NPAs. Brown fruits were the most frequent among the species in PBNP (36%), followed by purple (16%), black (13%) and red (12%). Species with brown fruits were also the most frequent in UBR (64%), with a lower proportion of species having black (9%), purple (8%), or red (6%) fruits. In both NPAs, fruit color was associated with growth form. Trees mostly had brown fruits, shrubs had black or red fruits, and herbs mostly had brown fruits in PBNP. In UBR, a large percentage of herbs, epiphytes, and shrubs had brown fruits, while most trees had black fruits (Table 2 and [insert Figure 2(A)-(B)]). Frequency of Species by Fruit Color (A-B), Fruit Type (C-D) and Dispersal Syndromes (E-F) in Different Growth Forms in the Pico Bonito National Park and the Uyuca Biological Reserve. The Category Other in Fruit Color Includes the Colors Blue, Pink, and White. The Category Other in Fruit Type Includes Achene, Nut, Samara, Schizocarp, Strobilus and Syconium
Overall, the most common fruit type among species in PBNP was drupes (32%), while in UBR it was capsules (31%). However, in both NPAs, fruit type was associated with growth form. In PBNP, the most common fruit types were drupes, capsules, and berries in trees, berries and drupes in shrubs, and drupes and capsules in herbs. In UBR, most trees had drupes, berries, or capsules; shrubs had a greater number of species with drupes, cypselas, or berries; most herb species had capsules, caryopses, or cypselas; and most epiphytes had legumes (Table 2 and [insert Figure 2(C)-(D)].
Zoochory was the most frequent dispersal syndrome among species in both PBNP (79%) and UBR (41%). In both NPAs, dispersal syndrome depended on growth form. Tree and shrub species dispersed their seeds mainly by zoochory, while herbs were dispersed by zoochory and anemochory in PBNP. In UBR, zoochory was also the main dispersal syndrome of trees and shrubs, while anemochory is the most frequent dispersal syndrome in herbs and vines (Table 2 and [insert Figure 2(E)-(F)]).
Species Attributes and Phylogeny
Phylogenetic Signal Values (D Metric) of Growth Form, Pollination Syndrome and Dispersal Syndrome of Plant Species Present in Pico Bonito National Park (PBNP) and Uyuca Biological Reserve (UBR) in Honduras. P (D > 0) Indicates the Probability That the Values of D Are Greater Than 0, and P (D < 1) Indicates the Probability That the Values of D Are Less Than 1. Therefore, Nonsignificant Values of P (D > 0) and Significant Values of P (D < 1) Indicate Phylogenetic Signal

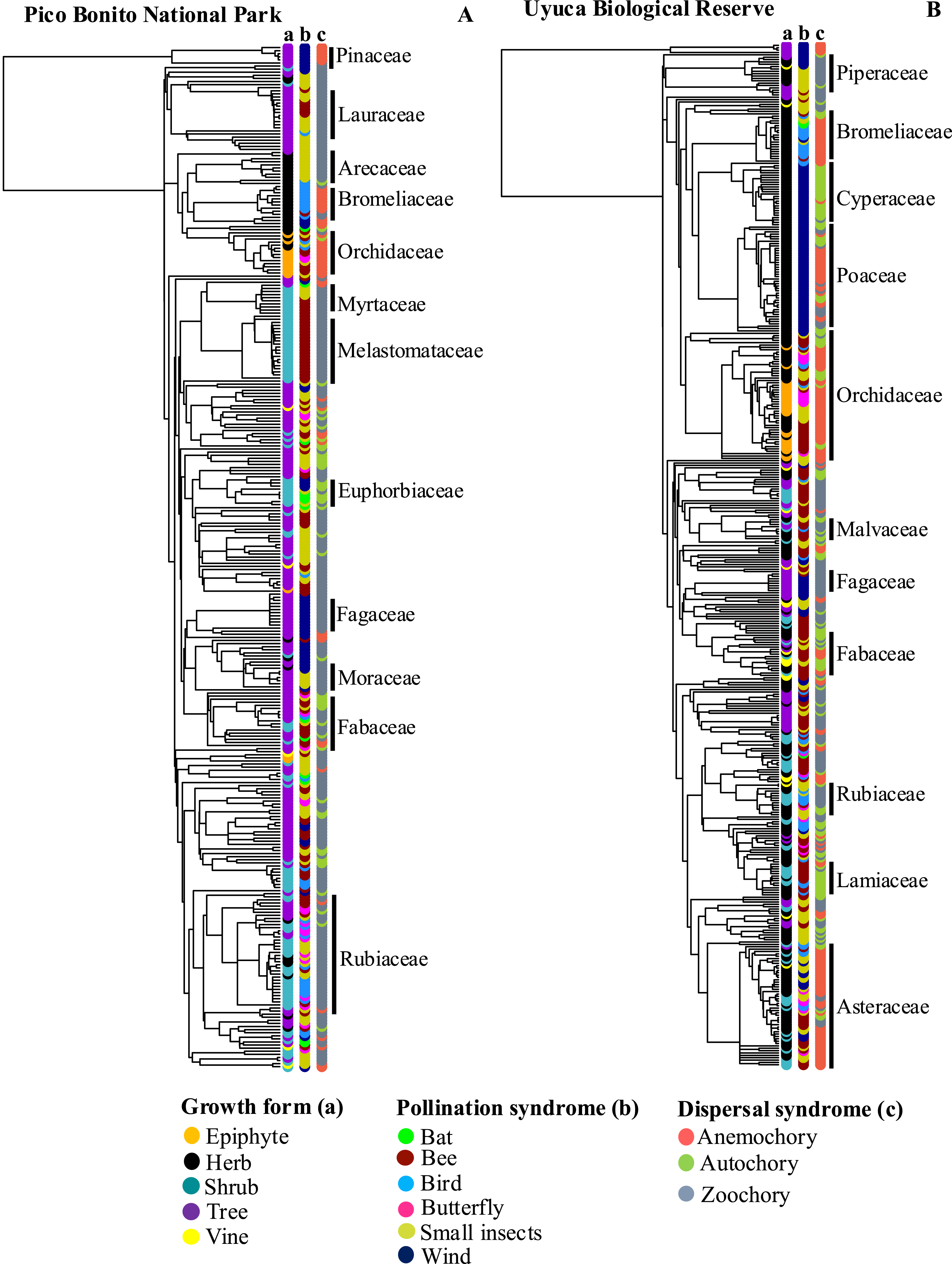
Growth Forms, Pollination Syndrome and Seed Dispersal Syndrome Plotted on the Tips of the Phylogenetic Trees of Pico Bonito National Park (A) and Uyuca Biological Reserve (B) in Honduras
Discussion
Species Composition and Conservation Status
The objective of this study was to demonstrate that beyond the number of species, the two NPAs studied safeguard phylogenetically conserved sets of attributes related to pollination and biotic seed dispersal. Thus, conserving these two NPAs could positively impact the conservation of key functional traits necessary for plant reproduction and regeneration, as well as the maintenance and recovery of ecosystems. One of the challenges in this study was collecting information on the reproductive traits and evolutionary histories of many species, especially in this understudied region of the planet. Thus, we relied on alternative sources of information besides vegetation ecology studies, such as floristic lists, regional floras, and herbarium records. We emphasized the importance of herbarium records, which are crucial for identifying flower and fruit traits. Previous studies have demonstrated the usefulness of reviewing herbarium specimens for determining plant traits and for conservation purposes (Daru, 2025).
In Honduras, nearly 7,524 plant species have been recorded (McCraine et al., 2018). Our study included 7.19% of these species in PBNP and 5.5% in UBR. We found that 29 species are in the most vulnerable conservation categories, 9 in UBR and 20 in PBNP. However, we found that no information was available for 61% and 33% of the species in the PBNP and UBR, respectively. This result highlights the lack of knowledge about the biology of plant species in protected ecosystems in Honduras. On the other hand, although most of the species on the IUCN Red List are listed as Least Concern, their conservation status may change negatively due to the constant anthropogenic pressures around and within these two protected areas. In this context, although the objective of both NPAs is to protect the biodiversity they harbor, human activities have negatively impacted the ecosystems in both NPAs (Batravil, 2015). In PBNP, agriculture, African oil palm and pineapple plantations, and the use of surface and groundwater for irrigation have threatened species diversity (AFE-COHDEFOR, 2004; Ferrufino-Acosta et al., 2018). On the other hand, the areas around UBR have been affected by wildfires, plant extraction, and urban growth (Mora et al., 2013). The loss of forest cover in these two NPAs could negatively impact species persistence, the stability of diverse biotic interactions, and the ecosystem services that benefit the communities surrounding these NPAs.
Floral Attributes and Pollination Syndromes
In line with our predictions in the two NPAs studied, we found that most species presented biotic pollination syndromes (PBNP 74% and UBR 85%), mainly related to bees and small insects (small wasps, bees, flies) (Figure 2). Bee pollination syndrome has been recorded as the most frequent syndrome in other Neotropical seasonal forests (e.g., Caatinga, Restinga, Cerrado and Pine Forest) (Cortes-Flores et al., 2017; Martins & Batalha, 2006; Quirino & Machado, 2014).
Most species in both NPAs had small white or yellow hermaphroditic flowers (e.g., Calea, Clethra, Cyperus, Dendropanax, Eleocharis, Paspalum and Saurauia), which are generally pollinated by bees or small insects. Small flowers in two NPAs are often organized as dense inflorescences, as occurs in species of Arecaceae, Araliaceae, Asteraceae, Boraginaceae, Burseraceae, Melastomataceae, and provide pollen and nectar for small, medium, and large bees (Bawa et al., 1985; Bawa & Opler, 1975). Species with small or medium hermaphroditic pink flowers were also frequent, mainly in UBR. These include Melochia tomentosa, which is frequently visited by bees (Apis mellifera, Centris spp. and Xylocopa spp.) (Frankie et al., 2004; Machado & Sazima, 2008). Thus, it seems likely that the high frequency of small flowers in the two NPAs analyzed represents an important source of floral resources for different groups of bees.
Large, hermaphroditic, red flowers were mainly present in shrub species and are associated with pollination by birds. For example, Lobelia laxiflora is geitonogamous and depends on pollination by birds such as the hummingbirds Lampornis amethystinus and Saucerottia cyanocephala. Similarly, Tillandsia deppeana is pollinated by Campylopterus curvipennis, Amazilia yucatanensis and Amazilia cyanocephala (Feinsinger, 1978; Lara & Ornelas, 2002). Other examples include the protandrous Moussonia deppeana, which is pollinated by Lampornis amethystinus and Palicourea padifolia, which is a distylous species pollinated by Eugenes fulgens and Amazilia cyanocephala (Ornelas et al., 2007).
Although many hermaphroditic plants were recorded in the studied NPAs, it is important to point out that some species described as hermaphroditic show combined sexual systems. For example, there are andromonoecious systems such as Boehmeria cylindrica (with a wind pollination syndrome), Oreopanax xalapensis (bee syndrome) and Chamaedorea graminifolia (fly syndrome). There are also gynodioecious species such as Valeriana scandens (small insect syndrome), and polygamous flowers like Quiina macrophylla (moth syndrome) or Chrysochlamys nicaraguensis, Simarouba glauca and Sanicula liberta (all pollinated by small insects).
Fruit Attributes and Dispersal Syndromes
As expected, the frequency of species with fruit traits related to animal dispersal was higher in the two NPAs. The dominance of animal-dispersed species is a pattern that has been documented in other tropical rainforests, where the high frequency of species with fleshy fruits has been hypothesized to be related to high water availability (Almeida-Neto et al., 2008). In PBNP, most of the plants have fleshy fruits that are dispersed mainly by vertebrates, including many groups of birds and mammals (Galetti et al., 2011). Many birds feed on the fruits of plant species included in this study. For example, Euphonia affinis consumes the berries of Conostegia xalapensis, and Pteroglossus torquatus (Collared Aracari) feeds on the drupes of Bactris trichophylla (Escoto, 2021) in PBNP. Meanwhile, phyllostomid bats consume fruits of plants of the genera Cecropia and Piper (Casallas-Pabón et al., 2017; Olea-Wagner et al., 2007), as well as Vismia and Ficus (Casallas-Pabón et al., 2017). The medium-sized mammal Eira barbara has also been observed feeding on Licania platypus “zapotillo” in Honduras (Adams, 2021; 2017), as well as in the tropical rainforest of the Montes Azules Biosphere Reserve in Chiapas, Mexico (Camargo-Sanabria & Delgado-Martínez, 2016).
The high number of zoochorous species may indicate the importance of NPAs as food sources for different bird and mammal communities. It has also been suggested that zoochorous diaspores have a greater chance of reaching different habitats due to the constant movement of animals (Howe & Smallwood, 1982; Pereira et al., 2022). For example, the UBR contains nearly 180 species of birds that could be considered seed dispersers, including Pharomachrus mocinno, Ramphastos sulfuratus and Pteroglossus torquatus (https://www.ebird.org). In addition, species of phyllostomid bats of the genera Artibeus, Sturnira, Carollia have been reported as possible seed dispersers in the area (Mora et al., 2013). In PBNP, 150 species of birds have been recorded as potential seed dispersers, including highly efficient seed dispersers like toucans (Aulacorhynchus prasinus, Ramphastos sulfuratus, Pteroglossus torquatus, Selenidera spectabilis), large cracids (Crax rubra), tinamous (Tinamous major), trogons (Trogon spp.), quetzals (Pharomachrus mocinno), cotingas (Cotinga amabilis), icterids (Psarocolius montezuma, Icterus spp.) and at least 16 species of doves.
Finally, autochory and anemochory were syndromes present mainly in herbaceous species of the families Asteraceae, Orchidaceae, Poaceae, Cyperaceae, and others in the UBR. The presence of dry fruits (caryopses, achenes, capsules) and their dispersal by wind and barochory in herbs has also been a pattern in other studies of seasonal ecosystems like the Caatinga, Cerrado and tropical mountain forests (Cortés-Flores et al., 2013, 2019; Griz & Machado, 2001). The high number of anemochorous and autochorous species in UBR may facilitate the colonization of regenerating areas since in this reserve there is an open canopy where the wind favors the movement of diaspores (Howe & Smallwood, 1982; Pereira et al., 2022).
Species Attributes and Phylogeny
Our results are like that of findings in other studies, where growth form and dispersal syndromes were more similar among species that were closer phylogenetically (Chazdon et al., 2003; Wang et al., 2022). Plant growth form is related to other functional traits, such as fruit type, when trees are related to fleshy fruits, and herbs are related to dry fruits (Chen et al., 2004). In URB, we identified groups such as the Asteraceae and Ochidaceae, which are predominantly herbaceous species with dry fruits dispersed by autochory and anemochory, respectively. In PBNP, a notable phylogenic pattern was the presence of fleshy fruits dispersed by animals of trees (Lauraceae, Moraceae and Myrtaceae) and shrub species (Melastomataceae, Piperaceae, and Rubiaceae). The results of this study align with the proposal that more basal families, such as Lauraceae, Araceae, and Myrtaceae, produce fleshy fruits that are dispersed by animals (Fleming & Kress, 2011). The presence of similar dispersal syndromes in more closely related species has been explained from the perspective of the morphological characteristics of the fruits and the predominant environment in which the species have evolved (Chen et al., 2017; Wang et al., 2022).
The results of the study did not reveal a consistent pattern of phylogenetic signal associated with pollination syndromes. It has been posited that traits associated with pollinator attraction exhibit considerable variation in their phylogenetic conservatism, and that the magnitude of phylogenetic signal may differ at the level of pollinator functional groups (Dellinger, 2020; Rosas-Guerrero et al., 2014; Van der Niet & Johnson 2012). Our research revealed the presence of phylogenetic signal in bird, wind, and bats syndromes in PBNP, as well as bat, butterfly, and wind pollination syndromes in UBR. Wind pollination syndrome has been observed in the families Fagaceae, Pinaceae, and Poaceae. Furthermore, pollination by birds was observed in species belonging to the Bromeliaceae family, while pollination by bats was documented in species of the Cactaceae family. The variation in the phylogenetic signal in pollination-related attributes is probably associated with processes of competition for pollinators among coexisting plant species in the communities (Sargent & Ackerly, 2008). Conversely, the presence of unclear patterns of phylogenetic signal in pollination syndromes has been associated with convergent floral adaptations to specific functional groups of pollinators (Rosas-Guerrero et al., 2014).
Our analysis revealed distinct phylogenetic patterns at the family level within the phylogenetic trees of each NPA. It has been observed that certain families of plants possess a similar growth form, as well as the same pollination and dispersal syndromes (e.g., Bromeliaceae, Fagaceae, Pinaceae, Melastomataceae). Conversely, there are families with high variation in the traits analyzed, such as Asteraceae, Euphorbiaceae, Fabaceae, Rubiaceae, and Malvaceae. The documented patterns can serve as a preliminary approach for identifying groups of species that may be particularly vulnerable to anthropogenic disturbance. A comprehensive understanding of the evolutionary history of species assemblages in communities, along with attributes relevant to reproduction and survival, may prove to be a critical element in elucidating which species may show greater resilience or susceptibility to current severe environmental changes (Tucke et al., 2019).
Implications for Conservation
The present study is the first to evaluate the diversity of plant species found within the two NPAs in Central America from the perspective of the functional reproductive attributes of flowers and fruits and species phylogeny. Since most of the plants in the two NPAs had biotic pollination and seed dispersal syndromes, the conservation of these forests could contribute to the maintenance of pollination and seed dispersal services not only in the NPAs themselves but also in the areas surrounding them. Several of the main crops in the areas surrounding the NPAs are dependent on biotic pollinators. For example, pineapple crops are pollinated by hummingbirds (Westerkamp & Gottsberger, 2000), and avocado is pollinated by bees and flies (Afik et al., 2006; Díaz & Ferrufino-Acosta, 2023). In addition, cacao, which is one of the most important crops in the buffer zone of PBNP, depends on pollination by dipteran species of the family Ceratopogonidae (e.g., Atrichopogon, Dasyhelea, Forcipomyia; Córdoba et al., 2013). Ensuring the proximity and diversity of NPAs near crop fields have even been considered an important measure to guarantee the presence of pollinators to improve crop yields (Garibaldi et al., 2017; Hipolito et al., 2018). Although our results do not provide a direct assessment of dispersal by animals, the presence of a high number of zoochorous species highlights the importance of the conservation of NPAs as a source of food for different communities of birds and mammals. In addition, the different syndromes of seed dispersal may represent different pathways for the arrival of propagules in the fragmented areas surrounding the NPAs.
Analysis of phylogenetic patterns provides information on the ability of species to evolve in response to environmental change (Winter et al., 2013). Therefore, the phylogenetic patterns found in our study can serve as a basis for assessing the vulnerability of species groups to habitat fragmentation and loss. Our results indicate that assessing other components of biodiversity beyond species numbers can highlight the importance of protected areas not only for species conservation, but also as a source of functions and evolutionary histories for restoring areas in heavily human-modified landscapes. Future studies should evaluate how much taxonomic, functional, and phylogenetic diversity is conserved in NPAs as well as evaluate the ecosystem services of the NPAs from an integrative perspective (ecological, social, and economic).
Supplemental Material
Supplemental Material - Beyond Species Richness: Conserving Species and Reproductive Functions of Honduran Flora in Protected Natural Areas
Supplemental Material for Beyond Species Richness: Conserving Species and Reproductive Functions of Honduran Flora in Protected Natural Areas by Lilian Ferrufino-Acosta, Vivian Cabrera, Nereyda Estrada, German Sandoval, Armando Aguirre-Jaimes, Mauricio Quesada, Jorge Cortés-Flores in Tropical Conservation Science
Supplemental Material
Supplemental Material - Beyond Species Richness: Conserving Species and Reproductive Functions of Honduran Flora in Protected Natural Areas
Supplemental Material for Beyond Species Richness: Conserving Species and Reproductive Functions of Honduran Flora in Protected Natural Areas by Lilian Ferrufino-Acosta, Vivian Cabrera, Nereyda Estrada, German Sandoval, Armando Aguirre-Jaimes, Mauricio Quesada, Jorge Cortés-Flores in Tropical Conservation Science
Supplemental Material
Supplemental Material - Beyond Species Richness: Conserving Species and Reproductive Functions of Honduran Flora in Protected Natural Areas
Supplemental Material for Beyond Species Richness: Conserving Species and Reproductive Functions of Honduran Flora in Protected Natural Areas by Lilian Ferrufino-Acosta, Vivian Cabrera, Nereyda Estrada, German Sandoval, Armando Aguirre-Jaimes, Mauricio Quesada, Jorge Cortés-Flores in Tropical Conservation Science
Footnotes
Acknowledgments
J.C.F. thanks DGAPA-UNAM (Dirección General de Asuntos del Personal Académico-Universidad Nacional Autónoma de México) for the award of a postdoctoral fellowship grant. This work was supported by Universidad Nacional Autónoma de México Postdoctoral Program (POSDOC). We thank the biologist Federico Reconco for the search of information for the databases. Betsabe Ruiz-Guerra (Instituto de Ecología A.C.) helped with the databases as well as editing of figures. Thanks to Rosamond Coates (IB-UNAM) for the grammatical review of English text.
Funding
This study was supported by a grant from Universidad Nacional Autónoma de México (UNAM) (PAPIIT IV200418, IN224920, IN219021, IA203521), SADER-CONACHyT 291333, CONACHyT-National Repositories 271432, CONAHCYT-UNAM-UAGro-UMSH to Laboratorio Nacional de Análisis y Síntesis Ecológica LANASE (2015-LN250996, 2016-LN271449, 2017-LN280505, 2018-LN293701, 2019-LN299033, 2020-LN314852, 2021-LN 315810), LANASE-UNAM 2015-2022, and Programa Iberoamericano de Ciencia y Tecnología para el Desarrollo RED CYTED SEPODI (417RT0527).
Declaration of Conflicting Interests
The Authors declare that there is no conflict of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
