Abstract
The secondary succession of tropical rainforest in abandoned agricultural fields modifies components of species diversity and processes of species colonization and replacement. In general, knowledge on invertebrate reassembly is lacking, even though invertebrate assemblages directly influence the maturation of forests. Ants are especially useful for testing hypotheses about the effects of resource diversity and microhabitat conditions. We experimentally assessed the effects of different successional stages on nest colonization. Then, we assessed whether nesting resources and microhabitat conditions (i.e., abiotic conditions) are potential mechanisms that influence the colonization pattern of twig-dwelling ants along a natural chronosequence for 1 year in the Lacandon region, Mexico. We found that ant species richness in twigs is correlated with total species found in leaf litter. The nest occupancy increased as succession progressed, but species richness did not change. Nests occupation increased to greater artificial nesting resource decomposition rates and lower soil pH. Meanwhile, species richness was increased only with lower soil compaction. Species composition changes among successional categories; the changes were greater with greater soil compaction and soil pH. The diversity of nesting resources did not drive twig-dwelling ant assembly. We conclude that the natural maturation of tropical forest enables greater colonization of twig-dwelling ants, but recovery of the species composition of assemblages after 30 years is incomplete, although it shows a recovery trend.
Introduction
At present, the rapid destruction and replacement of old-growth tropical forests by agriculture is one factor driving biodiversity loss (Gibson et al., 2011). However, in recent years, the cover of abandoned agricultural areas has increased in several tropical countries. This trend of land abandonment is likely to continue in the future (Arroyo-Rodríguez et al., 2015; Chazdon, 2014). Secondary succession may influence the colonization, replacement, and diversity patterns of arthropod species (Meloni & Varanda, 2015). Little is known about the reassembly of invertebrates after the abandonment of agricultural fields, even though invertebrate assemblages can improve the success of forest restoration through participating in several important ecosystem services (e.g., pollination, soil fertility, food, and plague control; Holmquist, Schmidt-Gengenbach, & Demetry, 2014).
Ants are highly diverse and abundant in the leaf litter of tropical forests, including secondary forests (Parr, 2012). Ant colonization and diversity are positively related to diversity of nesting resources, such as leaf litter and twigs (Armbrecht, Perfecto, & Vandermeer, 2004). Nest occupancy is also positively related to twig decomposition rates and vegetative composition (de Souza, Fernandes, Nascimento, Suguituru, & Morini, 2012). Furthermore, ants are sensitive to environmental variables and respond rapidly to anthropogenic changes (Del Toro, Ribbons, & Pelini, 2012). For instance, taxonomic and functional diversity of ants are lost during initial stages of secondary succession (Bihn, Verhaagh, Brändle, & Brandl, 2008; Rocha-Ortega et al., 2017) due to soil and vegetation condition changes (Schmidt, Ribas, & Schoereder, 2013). Given this context, ants are useful for testing hypotheses about secondary succession via the studying of nesting resource diversity changes (Murnen, Gonthier, & Philpott, 2013) and microhabitat conditions (Schmidt et al., 2013).
Among the microhabitat conditions that play significant roles in ant assembly, one of the most important is physical and chemical properties of soil and litter (Jacquemin, Drouet, Delsinne, Roisin, & Leponce, 2012). For instance, soil compaction determines free space among soil particles that act as a selective filter on ant fauna (Schmidt, Schoereder, & Caetano, 2017). Thus, low soil compaction and high soil moisture allow greater turnover of ground ant species along an environmental gradient (Rocha-Ortega & Castaño-Meneses, 2015). Moreover, litter quantity influences microclimatic conditions and soil pH in nest vicinity, which drive similar ant species compositions (Jílková, Matějíček, & Frouz Jan, 2011) among intermediate secondary and old-growth forests (OGFs; Osorio-Pérez, Barberena-Arias, & Aide, 2007). In turn, leaf-litter ants also play important roles in shaping soil structure and in promoting nutrient cycling via leaf-litter decomposition (Del Toro et al., 2012). Nutrient cycling is an important mechanism for maintaining and restoring tropical forest ecosystems (Robinson, van den Berg, Meirelles, & Ostle, 2015). In short, leaf-litter ants are sensitive to soil properties as well as participate in the formation of soil and the decomposition of organic matter and could help a quicker recovery of vegetation.
The response of ants to distinct impacts and to the subsequent recovery process has been evaluated (Parr, 2012), but the drivers behind nest colonization patterns of ant assemblages remain unexplained (Jiménez-Soto & Philpott, 2015). Over time, during tropical secondary succession, soil nutrients, litter layer (Robinson et al., 2015), and resource diversity increase (Uriarte et al., 2012), whereas microhabitat conditions are modified (Méndez-Bahena, 1999). However, floristic composition impoverished that occur during the initial stages of secondary succession (Chazdon et al., 2007) are particularly harmful for leaf-litter ants. For example, ants are dependent on tree parts (i.e., leaves and twigs) for nesting into cavities; therefore, diversity loss of this resource would affect ant nesting (Souza, Anjos, Carvalho, & Del-Claro, 2015). Also, leaf-litter ants require microhabitat conditions associated to OGF for suitable nesting; therefore, alterations of microhabitat conditions during succession stages may decrease species richness (Schmidt et al., 2013). The use of artificial nests can decouple the natural correlations among nesting resources and environmental changes (e.g., soil properties) during secondary succession. Therefore, it sheds light on the community assembly mechanisms, such as habitat filters, and nesting patterns that shape leaf-litter ant assemblages along the secondary succession.
In this study, we experimentally assessed the effects of different successional stages on nest colonization. Then, we assessed whether nesting resources and microhabitat conditions (i.e., abiotic conditions) are potential mechanisms that influence the colonization pattern of twig-dwelling ants along a natural chronosequence for 1 year in the Lacandon region, Mexico. Because of vegetation loss, we expected the scarcity of nesting resources and the unsuitable conditions for nesting in secondary forests. Therefore, we predicted that artificial nests would be more frequently occupied by ants in younger secondary forests than in OGFs. Also, we predicted that ant species richness would be higher in OGFs than in secondary forests.
Methods
Study Area and Site Selection
The Lacandon rainforest comprises parts of Belize, Guatemala, and Mexico and is one of the largest areas of tropical rainforest (ca. 800,000 ha) in Mesoamerica (Hernández-Ruedas et al., 2014). The study was performed in the Lacandon region near the village of Loma Bonita in Marqués de Comillas municipality (16° 04’ N, 90° 45’ W), south of the Montes Azules Biosphere Reserve (RBMA) in Chiapas, Mexico. Mean annual precipitation is about 3,000 mm, and average temperature is 24℃. A marked dry season occurs from February to April (Martínez-Ramos, Anten, & Ackerly, 2009). The research area mainly consists of three geomorphologic units defined according to soil and topographic criteria (Ibarra-Manríquez & Martínez-Ramos, 2002). This study was conducted within the most widespread geomorphologic unit in the region, which is characterized by topographical irregularity, an elevation of 115 to 300 m a.s.l., small hills and valleys, and sandy limestone soils of low pH (< 5.5). The dominant vegetation is lowland tropical rainforest (Ibarra-Manríquez & Martínez-Ramos, 2002). The regional landscape is composed of agricultural uses (65%), secondary forests (22%), and OGFs (13%; Zermeño-Hernández, Pingarroni, & Martínez-Ramos, 2016).
Within the study area, we selected eight permanent plots (0.5 ha) of secondary forest regenerating from abandoned cornfields (outside RBMA) with successional ages ranging from 6 to 27 years. These plots represented a natural gradient of forest recovery (Figure 1). For comparison purposes, we selected four plots of OGF within the RBMA. According to estimates, the OGFs were likely cut about 450 years ago; however, there is no conclusive evidence up to the present date (Zermeño-Hernández et al., 2016). The same plots used in the study of van Breugel, Martínez-Ramos, and Bongers (2006) were selected for the present study, as these authors chose plots with homogeneous environmental conditions in terms of soil, topography, and management history. Plots were located from 1 to 12 km apart. The secondary forest plots were embedded in remnants of 1 to 10 ha and may be influenced by the same effects of fragmentation (Table 1).
Map of the study area in the Lacandon region of Chiapas, Mexico, where sampled plots are labeled as secondary forests (SFs) according to the number of years since cornfield abandonment or as old-growth forests (OGFs). Number of Species and Colonies of Ants Found in Artificial Nests Across Eight Secondary Forests Abandoned at Different Times and Four Old-Growth Forest Plots in the Lacandon Region, Mexico. Note. OGF = old-growth forest; SC = successional category.

Experimental Design
Secondary forests largely differ in structure and composition compared with nearby OGFs (Letcher & Chazdon, 2009). Fallow age is one variable that may reflect community organization at a certain point in time along a successional trajectory. However, secondary succession is not necessarily unidirectional, and secondary forests will not necessarily resemble OGFs over time (van Breugel et al., 2006). Therefore, it is not possible to assign an age to OGFs. If an age is arbitrarily assigned to OGFs and included in a model, the model’s tendencies would be artificially influenced. In this respect, to evaluate the trajectory of change in ecosystem processes beyond a single site, chronosequence approaches in which multiple, independently restored sites of more or less the same age are considered appropriate (Dent & Wright, 2009; Gibson et al., 2011; Letcher & Chazdon, 2009; Matzek, Warren, & Fisher, 2016). For these reasons, the best means of comparing secondary forests with OGFs is through assigning successional categories. We classified the eight secondary forests into two continuous secondary successional categories: The first category included four forests abandoned 6 to 12 years ago (successional category 1 [SC1]), and the second category included four forests abandoned 13 to 27 years ago (SC2). The four OGFs were included in the final category of forest succession (OGF). The experiment was conducted from June 2010 to May 2011.
Nesting Resources
Artificial nests
To test whether the diversity of artificial nesting resources (ANRs) increased species richness and nest occupancy of twig-dwelling ants, we first selected twigs of similar length and diameter (approximately 2 cm in diameter and 30 cm long). Twigs were cut from living trees of the five most common species in the studied landscape (Hernández-Ruedas et al., 2014). These species are ordered from highest to lowest wood density: Dialium guianense (Aubl.), Byrsonima crassifolia (L.), Inga paterno Harms, Tabebuia rosea (DC.), and Trichospermum mexicanum (DC.). The density of wood has an important inverse effect on the rate of decomposition (i.e., wood of lower density has a higher decomposition rate; Jackson, Peltzer, & Wardle, 2013). Twigs with a high decomposition rate generate more inner spaces and facilitate ant colonization.
All twigs were dried in a drying chamber at 105℃ for 1 week. In each twig, a hole with a diameter of 1 cm and length of 5 cm was drilled lengthwise using a drill bit. Then, 5 twigs of the same species (conspecific trees) or a mix of twigs of 5 different species (heterospecific trees) were placed in a 35 × 15 cm mesh bag (0.5 cm of gap diameter), henceforth called ANR. In each plot, ANR diversity was tested using 10 conspecific and 10 heterospecific ANRs, which were placed alternately and flush with the soil surface every 6 m along the edge of a 50 × 6 m quadrat (Armbrecht et al., 2004). At the beginning of each period (i.e., prewet, wet, and postwet periods), a new set of 20 ANRs was placed and subsequently collected at the end of the period. Each period consisted of 120 days to maximize the chance of capturing maximum nest occupancy. The wet period occurred from June to September (precipitacion > 300 mm). Later, the postwet period was defined as the 120 days posterior to the wet season (precipitation from 100 to 300 mm). Finally, the prewet or dry period occured from February to May (precipiation < 60 mm). Ant colonization was considered successful when at least one of the twigs of the ANR was occupied by a fertile queen (queenright nest). We counted both occupied nests and species. First, voucher specimens of all ants were identified at the genus level following Mackay and Mackay (1989) and at species level according to Longino (2010). Then, the names of ant subfamilies, tribes, genera, and species were assigned according to AntCat classification, which is an online catalog containing the current taxonomy and phylogeny of the world’s ant (Bolton, 2018). A reference collection of species was deposited in the Entomological Collection of the Instituto de Ecología A.C. in Xalapa, Veracruz, Mexico (IEXA; Reg. SEMARNAT: Ver. IN.048.0198).
Decomposition rates
We estimated the decomposition rates for each ANR in each plot to characterize the cavity-entrance access. Because ants do not have specialized anatomical structures for making tunnels inside twigs, they depend on twig decomposition to greater space cavity entrance (see Powell, Costa, Lopes, & Vasconcelos, 2011). Before placement in the field, the ANRs were weighed to obtain the initial dry weight. At the end of each period, the ANRs were collected, dried, and weighed on a 0.01 -g precision scale. The mass loss of each ANR was calculated considering the initial weight as the total dry weight (100%). The decomposition percent was expressed as an average per plot (n = 60) per year in terms of twig mass per volume (g/cm3; Dickie, Fukami, Wilkie, Allen, & Buchanan, 2012).
Twigs with a high decomposition rate generate greater resource access, as ants do not have specialized anatomical structures for making tunnels inside wood (Powell et al., 2011).
Winkler Extractors, Pitfall Traps, and ANR Inventory
To compare assemblages naturally occurring on leaf litter and the ground with those of the ANRs, we used ant assemblage data collected using Winkler extractors and pitfall traps from a prior independent study conducted at the same plots (Rocha-Ortega & Favila, 2013).
Microhabitat Conditions
Description of Surrogate Variables for Nesting Resources and Microhabitat Conditions for Assessing Twig-Dwelling Ants Along a Forest Successional Gradient in the Lacandon Region of Chiapas in Southeastern Mexico.

Data Analyses
The number of nests occupied by a species in a plot during the 1-year sampling period was used as a measure of abundance. We calculated the inventory reliability for each heterospecific and conspecific ANR using the coverage estimator, which is a less biased estimator of sample completeness than nonparametric methods (Chao & Jost, 2012):
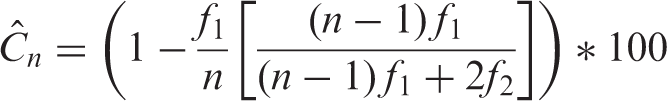
We performed correlations among the species richness and nest occupancy of ant assemblages collected in the ANRs and those of ground-foraging ants to determine their collinearity. This simple analysis suggested that environmental soil conditions during secondary succession may influence the sampled twig-dwelling assemblages in the same way that these influence ground-foraging ants. For this, we retrieved the ground-foraging ant inventory (collected by Winkler sacks and pitfall traps) of the same study plots in Rocha-Ortega and Favila (2013), which represented an independent sampling. Because the data (i.e., species richness or number of nests) did not meet the basic parametric assumptions, we used Spearman’s correlation coefficients.
Species richness and the number of occupied nests are commonly influenced by successional stages (Rocha-Ortega & Favila, 2013) and are likely influenced by nesting resources and environmental soil conditions, which are surrogate variables of successional stages. So, these microhabitat conditions likely influence the response variables of species richness and number of occupied nests. For this reason, we implemented generalized linear mixed models to assess the effects of nesting resources (ANR diversity and decomposition rate) and soil microhabitat conditions (compaction, pH, and moisture content of soil) on species richness and number of nests (Bolker et al., 2009). ANR diversity was measured as either heterospecific or conspecific. Decomposition rates were calculated as the average loss of twig mass per ANR diversity per plot per year (n = 30). Microhabitat conditions were measured next to each ANR and were expressed as average per plot per year (n = 60) to indicate the quality of soil conditions. We expected that greater diversity and decomposition rates would increase both ant species richness and nest occupancy. In addition, we expected that lower soil compaction and pH would also increase both ant species richness and nest occupancy, whereas lower soil moisture content would likely decrease both ant species richness and nest occupancy. In all models, we assumed a Poisson error distribution and specified plot as the random factor to control for any random effects. Next, to test whether the decomposition rate of the ANRs and the soil microhabitat conditions (i.e., soil compaction, soil pH, and soil moisture) varied among successional categories, we performed an analysis of variance of a generalized linear mixed-effect model with a Gaussian error distribution and specified plot as the random factor. To construct each model, we used both forward and backward stepwise selection of the explanatory variables according to their significance using the R package MASS 7.3. The predictive power of the models was evaluated according to the Akaike’s Information Criterion (AIC; Akaike, 1987); the ΔAIC of the null model was then compared with that of the aforementioned model. Models with ΔAIC ≤ 2 were considered equivalent. We performed post hoc analyses only when a factor (i.e., secondary succession and the surrogates of nesting resources and environmental soil conditions) exerted a significant effect on species richness or number of occupied nests. We applied a multiple comparison procedure, Dunnett’s C test (SC1, SC2 vs. OGF), to determine differences between each SC and the OGFs. All analyses were performed in the statistical software R 3.5 (R Development Core Team, 2016). The stepAIC function of the “MASS 7.3” package (Ripley et al., 2013), the glht function of the “multcomp” package (Hothorn et al., 2008), and the glmer and lmer functions of the “lme4” package were used during the analysis (Bates, Maechler, Bolker, & Walker, 2015).
Finally, we assessed the effects of SC and environmental soil conditions on species composition with a partial canonical correspondence analysis using the vegan package for R and tested the significance of the model and axes with a two-way analysis of variance (Oksanen et al., 2015).
Results
Mean ± SD of Species Richness and Number of Occupied Nests per SC and Diversity of Artificial Nesting Resources (ANRs).

Note. OG = old-growth; SC = successional category.
The sampling completeness of the heterospecific ANRs was 87.5% and of the conspecific ANRs was 79%. Meanwhile, sampling completeness was 76.8% in SC1, 85.2% in SC2, and 83.8 in OGF. Overall, ANRs had an inventory completeness of 90.6%, Winkler extractors of 100%, and pitfall traps of 96%. When comparing sampling completeness among different ANRs or sampling methods, accumulation curves did not show significant differences in species richness or sampling completeness (Figure 2), so these were considered comparable.
Rarefaction curves for ants nesting in twigs. (a, c) Species richness is plotted against sample coverage. (b, d) Sample coverage is plotted against the number of samples (artificial nesting resources [ANRs]) of conspecific ANRs (gray lines) and heterospecific ANRs (black lines) throughout the study. Both bars represent 95% confidence intervals.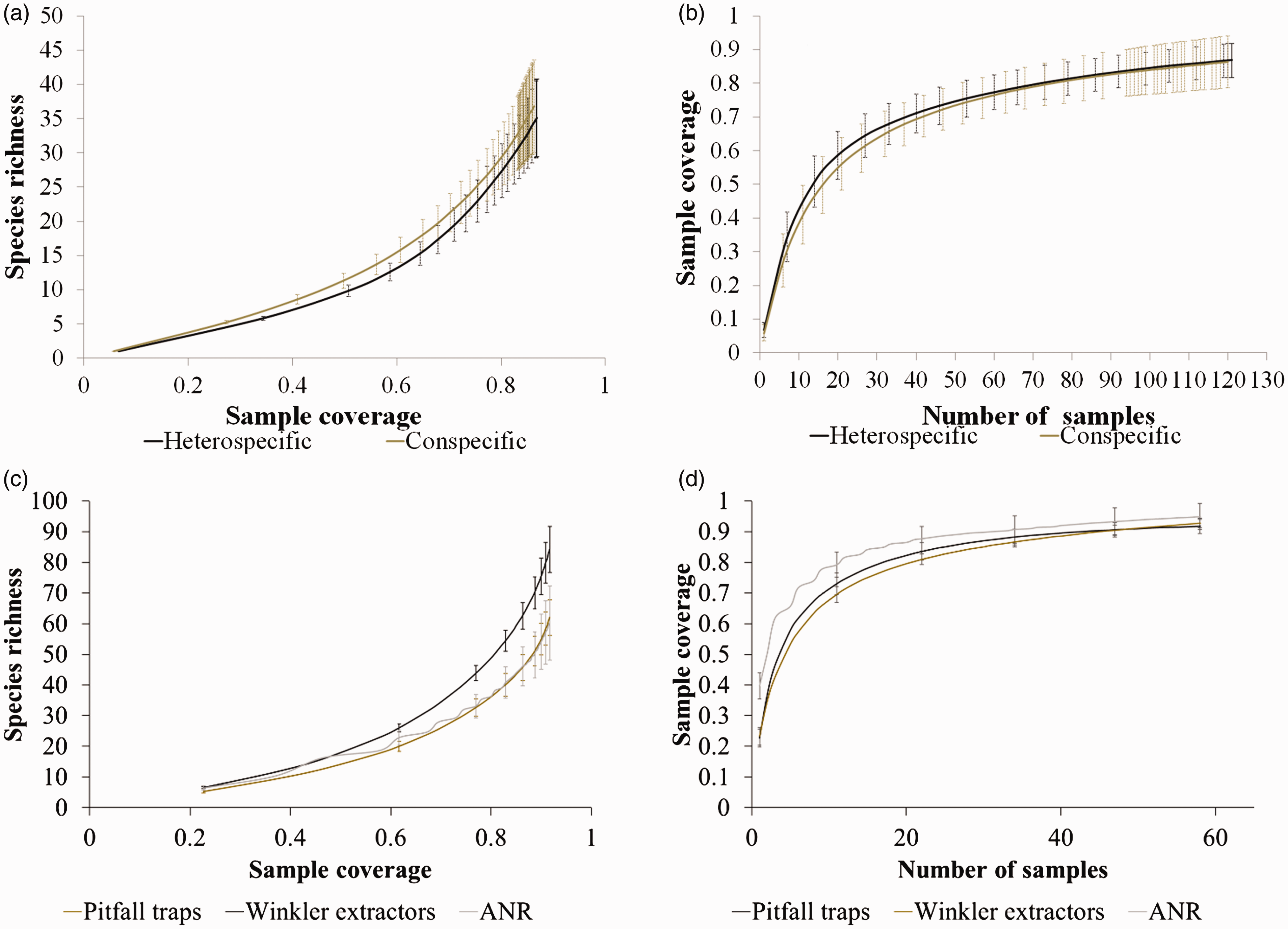
Correlations Between ANRs and Ground-Foraging Ant Assemblages
Species richness of ant assemblages in the ANRs was correlated with species richness of leaf-litter ants (i.e., Winkler traps; ρ = 117.5, p < .05) but not with species richness of ground ants (i.e., pitfall traps; ρ = 310.5; p = .74).
Surrogates of Nesting Resources and Microhabitat Conditions
Neither SC nor ANR diversity influenced the decomposition rate of the ANRs. Soil compaction, pH, and moisture differed among successional categories (F = 8.04, p = .02; F = 29.00, p < .001; F = 16.27, p = .001, respectively; Figure 3). Dunnett’s C post hoc test showed that soil compaction was greater in SC2 than OGF (p < .001); yet, no significant differences were found between SC1 and OGF. Soil pH in OGF was lower than in SC1 (p < .001) and SC2 (p < .001). Finally, soil moisture was higher in OGF than SC1 (p = .001) and SC2 (p < .001; Figure 3).
Variation in soil compaction and pH (mean ± SD) among abandoned cornfields (SC1 and SC2) and old-growth forests (OGFs). Bars labeled with the same letter are not significantly different.
Effects of Nesting Resources and Soil Conditions on Twig-Dwelling Ant Diversity
Results of Generalized Linear Mixed Model Testing for Effects of Nesting Resources and Soil Conditions on Alpha Diversity Metrics of Twig-Dwelling Ant Assemblages.

Note. Chi-square, df, and p values are given for likelihood ratio tests of the minimum adequate model against the null model, or the full model against the null model where no fixed factors were significant. ANR = artificial nesting resource.
p < 0.05.
Ant Species (Subfamily/Tribe) Collected in Old-Growth Forest (OGF) and Secondary Forest (SF) of Different Successional Ages in the Lacandon Region, the Largest Area of Tropical Rainforest in Mexico.

Note. Numbers listed indicate the observed species occurrences in the forests.
The number of occupied nests significantly varied among successional categories (Table 4). Post hoc tests showed that the number of nests was lower in SC1 than in OGF (p = .03). The number of occupied nests was similar between SC2 and OGF (p = .21). Regarding the effects of nesting resources and microhabitat conditions, the best model showed only that the number of nests increased with lower soil pH values and with higher soil compaction. Also, in the best model, soil moisture and interaction effects were not included. The decomposition rate also significantly and positively influenced the number of nests (Table 4).
Species composition was significantly associated with SC, F(1, 6) = 1.19, p = .004; soil compaction, F(1, 6) = 1.25, p = .01; and soil moisture, F(1, 6) = 1.11, p = .02. The first two canonical correspondence analysis axes explained for 63.3% of variation in species composition.
Discussion
Our study is novel in that it experimentally assessed the effects of secondary succession on ant nest colonization through two potential mechanisms of assembly: nest diversity and soil properties on nest colonization. The present study supports the importance of nesting colonization patterns for the recovery of twig-dwelling ant diversity in the Lacandon region of Chiapas, Mexico. Our results improve our understanding of the nesting resources and soil conditions that drive the recovery of twig-dwelling ants during secondary succession in tropical rainforest landscapes. We found that ant species richness in twigs is correlated with total species found in leaf litter. Nest occupation was regulated by SC, soil pH, and decomposition rate. Meanwhile, species richness only was regulated by soil compaction. Species turnover was driven by SC, soil pH, and compaction. Overall, in the studied successional chronosequence, reassembly of leaf-litter ants is more driven by soil conditions than nesting resources, particularly in younger forests.
Surprisingly, our results showed that succession stage significantly affected the nest occupation but not species richness. These results suggest that nest occupation by ants tends to increase with decreasing habitat disturbance (Armbrecht, Perfecto, & Silverman, 2006; Murnen et al., 2013), but species richness in twigs is not affected. We suggest two reasons why ant richness did not differ among successional stages. First, our chronosequence is embedded in a landscape with a high proportion of OGF (Zermeño-Hernández et al., 2016). This factor may enable species from OGFs to continually disperse into secondary forests independent of successional stage. Thus, local ant recovery might be regulated by landscape configuration more than local features (see Melo, Arroyo-Rodríguez, Fahrig, Martínez-Ramos, & Tabarelli, 2013). Second, local habitat complexity (e.g., vegetation structure) may not be important during colony founding. Likely, strategies such as colony claustral founding, in which case queens remain sealed in nest chambers and use only body reserves for energy (Brown & Bonhoeffer, 2003), may minimize the habitat effect on colony founding. In this case, the effects of habitat complexity would be more evident during colony growth (Souza et al., 2015).
Most studies on restoration approaches have focused on plants and have assumed that fauna returns as vegetation develops (Audino, Louzada, & Comita, 2014). However, faunal recovery also depends on other microhabitat conditions associated with secondary succession besides vegetation (Brudvig, 2011; Majer, 2009). The spatial distribution of trees and species-specific differences in leaf-litter production as well as the quality of secondary vegetation cause spatial heterogeneity in nutrient return to the forest floor and, therefore, in the redistribution of soil nutrients (see Uriarte, Turner, Thompson, & Zimmerman, 2015). Thus, spatial heterogeneity in nutrient could affect ant reassembly. In the present study, we found that decreasing soil compaction and pH are related to increasing diversity of twig-dwelling ants. These results evidence that the environmental soil conditions inherent to successional pathways likely drive ant diversity. In fact, environmental soil conditions are likely more influential on ant reassembly than nesting resource diversity.
Overall, we found that soil properties influence the species richness and nest occupancy, whereas ANR diversity did not drive ant assembly. Higher species richness was associated with lower soil compaction, while high occupancy was associated with lower soil pH values. Soil compaction could regulate the assembly of invertebrates via selecting for the body size required to move through small free spaces in soil particles, thus changing the composition of the invertebrate community that many ants rely upon for feeding (Ryder Wilkie, Mertl, & Traniello, 2007). Furthermore, a lower soil pH indicates a higher amount of organic matter and available nutrients (Frouz & Jilková, 2008), which enhance the suitability of nest sites (Schmidt et al., 2013). These combined results suggest that even when queenright colonies are not dependent on prey availability, queens likely select soil spots with certain conditions to favor colony growth. On the other hand, although low availability of nesting sites is associated with areas with less complex vegetation structures, the availability of nesting sites is naturally high for leaf-litter ant species (Souza et al., 2015). Therefore, soil conditions associated with suitability for nesting might limit the leaf-litter ant assembly more than nesting resources during secondary succession. In contrast, for arboreal ants that nest in trees, habitat disturbance is directly associated with the loss of tree species and availability of nesting sites (Jiménez-Soto & Philpott, 2015; Philpott & Foster, 2005; Souza et al., 2015). Thus, arboreal ants are more limited by nesting sites than leaf-litter ants.
Some additional evidence may support the idea that leaf-litter ants are weakly limited by nesting resources in secondary forests, at least during the colony founding stage. We found a high proportion of shared ant species among ANRs and Winkler extractors (60%). Our result suggests a low nesting resource specificity of leaf-litter ant species. Nesting strategies might explain this affirmation. For instance, claustral colony founding that is common in leaf-litter ants (Hölldobler & Wilson, 1990). In this case, queenright ants are not dependent on availability or unpredictability of nesting sites but rather on nest decay and new twigs, which drive recolonization (Byrne, 1994). Another nesting strategy present in leaf-litter ants is polydomy, wherein ant colonies occupy two or more spatially separated nests. Thus, ants may explore more resources over a larger area, maximizing their foraging efficiency and colony nutrient intake (Ellis & Robinson, 2014). Either of these two strategies could be minimizing ant species dependency on the specificity of nesting resources. While Armbrecht et al. (2004) showed the importance of a diverse mix of twigs for species richness, the driver in their study was not preference for different species of twigs but rather an emergent property: the diversity of twigs. In contrast, we found that factors such as suitable patches in soil properties can influence more the selection of nest site than mix of twigs.
Another mechanism that can be contributed to regulating the assembly of leaf-litter ants is positive interactions among species. We observed that 64% of total collected species shared their artificial nests with one or more species. Particularly, Hypoponera nitidula, Pheidole sp., Prionopelta modesta, Strumigenys gundlachi, and W. auropunctata. Sagata, Mack, Wright, and Lester (2010) reported similar results, indicating that 50% of twig-dwelling ant species shared both natural and artificial twig nests with another species. Plesiobiosis is the most rudimentary form of heterospecific associations and has been observed for ants. This type of association occurs between species pairs that differ in morphology, behavior, and taxonomy and denotes nesting in close proximity without biological interdependence (Kanizsai, Lrinczi, & Gallé, 2013). Accordingly, different ant species may coexist in the same twigs when coexisting species vary in prey specificity (see Longino, 2010). This also suggests that twig nests are not strongly partitioned mosaics. Sharing a nest may even represent an advantage, as coexisting ants may then engage in the mutual defense of twigs against shared predators (Sagata et al., 2010).
The decomposition rate of ANRs had a positive effect on nest occupancy and shift greater changes of species composition. This finding is consistent with Shik and Kaspari (2010), who found that nest occupancy in a tropical region increased with the decomposition of ephemeral nest resources and the subsequent conversion of detritus to food. In areas where twigs undergo accelerated decomposition, ant colonization may be directly favored by species’ metabolic requirements or indirectly favored by an increase in prey availability (Kaspari, Yanoviak, Dudley, Yuan, & Clay, 2009; McGlynn, 2006). Despite the high potential density of natural twig nests, perhaps only a subset is suitable for colonization (Byrne, 1994). Many twigs are probably uninhabitable, as both freshly fallen and thoroughly decayed twigs are not suitable for nesting (Herbers, 1986). Therefore, the decomposition rate of twigs (or ANRs in this study) may be a crucial factor that shapes the nests occupy by ants in tropical forests during secondary succession.
This study also indicates that the species composition of ant assemblages was significantly affected by secondary succession and was associated with soil compaction and moisture. Even so, secondary forests potentially attain species compositions similar to OGFs in the long term (Letcher & Chazdon, 2009). In our study, the composition of ant assemblages in different successional stages differed qualitatively from OGF sites, likely as a result of a strong founder effect (Wanner & Dunger, 2001). Thus, species richness is similar among successional stages, but the constituent species are different. Despite the species composition of secondary forests showing incomplete recovery after 30 years, these secondary forests still harbor many species associated with OGFs and nests in spots with certain soil properties (Frouz & Jilková, 2008; Jacquemin et al., 2012). Our results support that secondary tropical forests may become self-sustainable, functioning ecosystems and could be favored through the active introduction of ants via artificial nests.
Implications for Conservation
At present, natural secondary succession and restoration practices have mainly focused on plant recovery after disturbance and have assumed that fauna return as vegetation develops (Audino et al., 2014). However, faunal recovery also depends on other variables (Brudvig, 2011; Majer, 2009). Our results suggest that twig-dwelling ants may passively recolonize tropical secondary forests. Nevertheless, recovery of the species composition of assemblages after 30 years is incomplete, although it shows a recovery trend. The active introduction of ants via artificial nests (as the dry twigs used in this study) in secondary tropical forests may help to promote faster species composition recovery, as well as ant-mediated ecosystem processes.
Footnotes
Acknowledgments
We thank Dr. Miguel Martínez Ramos from the Institute for Ecosystem Research (Instituto de Investigaciones en Ecosistemas) of the National Autonomous University of Mexico, Morelia, México, for his assistance during fieldwork. We gratefully acknowledge the contributions of the anonymous referees and Dr. M. Favila, who contributed toward the first version of this article. We are grateful for Allison Jermain, MS, who kindly reviewed the article and gave useful suggestions. We also thank Dr. Helí Coronel-Arellano for providing maps for the region. We additionally express our gratitude to the Jamangape family for their assistance with fieldwork. Finally, this work is dedicated in loving memory of Ramón Rocha.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grants from the MABOTRO projects of SEMARNAT-CONACyT (grant 2002-01-0597), SEP-CONACyT (grant 2005-51043), and PAPIIT-UNAM IN227210. M. R.-O. received a doctoral fellowship (34925) from CONACyT during the duration of this study.
