Abstract
The state of Oaxaca is positioned in a rather unique biogeographical position with the highest diversity of vascular plants in Mexico. The isolation of xeric valleys surrounded by complex mountain ranges in Oaxaca supplies an excellent opportunity to investigate the influence of the Pleistocene events on xeric species. To test for the alternative hypotheses of Pleistocene glacial refugia, we used sequences of two chloroplast markers to examine the phylogeographic patterns of the endemic mistletoe species
Introduction
The Mexican highlands with a complex biogeographical, climatic, and geological history encompass one of the 25 world biodiversity hotspots (Mittermeier et al., 2005; Myers, Mittermeier, Mittermeier, da Fonseca, & Kent, 2000). Within these complex mountain systems, the state of Oaxaca is located in a rather unique biogeographical position with high diversity of endemic vascular plants (8.3% of the vascular flora of Oaxaca and 21% of the vascular flora of Mexico; García-Mendoza, Ordóñez, & Briones-Salas, 2004). The Valles Centrales, a region in the heart of the state of Oaxaca, is a unique geographical and cultural region consisting of several interconnected valleys surrounded by large rivers and high mountain ranges. These valleys and the adjacent Tehuacán-Cuicatlán Valley are arid and hot, with a dry-cold climate and a mean annual precipitation of 500 to 800 mm and an annual average temperature of 18℃ to 22℃ (García-Mendoza et al., 2004), and are especially rich in endemic plant species with very restricted distributions (Dávila et al., 1993; García-Mendoza et al., 2004; Sosa & De-Nova, 2012). The geological history and the glacial oscillations that occurred during the Pliocene and Pleistocene probably caused the fragmentation and long-term isolation of populations, subsequently leading to highly divergent lineages and endemism in this region (Bryson, García-Vázquez, & Riddle, 2011; Rojas-Soto, Espinosa de los Monteros, & Zink, 2007; Ruiz-Sanchez & Specht, 2013).
Integrative application of phylogeography and ENM has become a key approach for explaining intraspecific evolutionary patterns associated with climate oscillations (e.g., Bonatelli et al., 2014; Carstens & Richards, 2007; Ornelas et al., 2015; Perktas, Gür, & Ada, 2015; Peterson & Nyári, 2007; Waltari et al., 2007). Numerous studies using this approach indicate that the genetic differentiation of many extant taxa and species ranges were affected by climatic and environmental discontinuities as barriers and Pleistocene glacial/interglacial cycles, most notably in the temperate regions of northern Europe and North America (Carnaval, Hickerson, Haddad, Rodrigues, & Craig Moritz, 2009; Hewitt, 2000). Yet, Pleistocene climatic oscillations have had a profound effect on the distribution, demographic dynamics, and patterns of genetic variation of arid adapted species (Allal et al., 2011; Byrne, 2008; Meng, Gao, Huang, & Zhang, 2015; Riddle & Hafner, 2006; Turchetto-Zolet, Pinheiro, Salgueiro, & Palma-Silva, 2013). In the New World, cyclic range contractions and expansions shaped the current distribution of arid plants, their concomitant population dynamics and genetic differentiation, particularly during the Late Quaternary climatic changes (Collevatti, Rabelo, & Vieira, 2009; Rebernig et al., 2010). However, the influence of these climatic fluctuations on phylogeographic patterns of taxa occurring at the intertropical drylands remains largely unknown.
Phylogeographic studies of taxa from arid regions have revealed a pattern of lineage divergence associated to contraction to and expansion from
The distributions of organisms in xeric environments of Mexico underwent dramatic changes linked to Pleistocene climate cycles (Metcalfe, O'Hara, Caballero, & Davies, 2000; Shreve, 1942), and plant populations could have experienced repeated geographic isolation during the Pleistocene glacial/interglacial cycles approximately 2.4 to 0.01 million years ago (e.g., Angulo, Ruiz-Sanchez, & Sosa, 2012; Gándara & Sosa, 2014; Hernández-Hernández, Colorado, & Sosa, 2013; Rodríguez-Gómez & Ornelas, 2015; Ruiz-Sanchez, Rodríguez-Gómez, & Sosa, 2012; Vásquez-Cruz & Sosa, 2016). The effects of Pleistocene climate cycles between allopatric populations currently distributed in the southern portion of the Chihuahuan Desert and those distributed in the arid regions of the Valles Centrales and Tehuacan-Cuicatlán Valley of Oaxaca has been sparsely tested, with population divergence between these areas dated during the Pliocene (Gándara & Sosa, 2014) or during the Pleistocene, with demographic expansion during the LGM (Scheinvar, Gámez, Castellanos-Morales, Aguirre-Planter, & Eguiarte, 2017). A pattern of glacial survival into southern refugia followed by postglacial recolonization of northern regions seems to be ubiquitous for a variety of temperate taxa (e.g., Hewitt, 2000; Stewart, Lister, Barnes, & Dalén, 2010). The phylogeographic pattern of southern refugia for temperate species during glacial phases, which generally are confined to the southern portion of the species' distribution during warm climatic phases such as the current interglacial, is not followed by desert-adapted species (Angulo, Amarilla, Anton, & Sosa, 2017; Castellanos-Morales, Gámez, Castillo-Gámez & Eguiarte, 2016; Ruiz-Sanchez et al., 2012; Scheinvar et al., 2017). Phylogeographic patterns for studied species of the Chihuahuan Desert biota suggest the opposite pattern, a greatest range contraction during the LIG, with a later expansion to northern areas over the LGM. This pattern is accompanied with demographic expansion and a gradient of genetic diversity, with higher diversity values at southern latitudes (Angulo et al., 2017; Castellanos-Morales et al., 2016; Rebernig et al., 2010; Ruiz-Sanchez et al., 2012). Overall, results of current ENM and palaeomodeling show that the area currently occupied by studied species is substantially larger than it was during the LIG period and the LGM, suggesting that it has been gradually expanding northward since the LIG period. Previous phylogeographic studies that included plant populations of the Valles Centrales and the Tehuacán-Cuicatlán Valley have shown that their ranges contracted during interglacials and then expanded to colonize northern xeric regions during the glacial periods (the Meztitlán Valley and the Chihuahuan Desert; e.g., Loera, Ickert-Bond, & Sosa, 2017; Ruiz-Sanchez et al., 2012; Scheinvar et al., 2017) or expanded during the warm/humid LIG period and contracted to one or more southerly refugia during cold/dry glacial maxima (Cornejo-Romero et al., 2017).
Mistletoes are considered ecologically important key resources as they provide high-quality nutritional resources for a variety of obligate and partially dependent species, especially during periods of seasonal scarcity (Watson, 2001), and influence indirectly community structure in low productivity systems on bottom-up processes (e.g., Watson, 2009; Watson & Herring, 2014). Most of these parasitic plants depend on avian vectors for pollen and seed dispersal and obtain all of their water and minerals from the host (obligate hemiparasites) through a vascular connection termed haustorium (Calder & Bernhardt, 1983). Given their close connections with and dependence on their hosts (Cocoletzi, Angeles, Ceccantini, Patrón, & Ornelas, 2016; Kuijt, 2009), the geographical ranges of mistletoes are directly related to the availability of suitable host trees, and the geographic structuring of host populations might influence the genetic structuring of mistletoe populations (Norton & Carpenter, 1998; Ramírez-Barahona, González, González-Rodríguez, & Ornelas, 2017). Endemic species of mistletoes might be highly vulnerable to local extinctions, and specific conservation and management planning are needed to further develop strategies to support the persistence of multiple, interacting processes (climatic, hydrological, ecological, demographic, and genetic) of evolutionary refugia and ecological multiple interactions (e.g., host/parasite interaction) in an era of unprecedented environmental change (Davis, Pavlova, Thompson, & Sunnucks, 2013). Mistletoes infect a large number of tree species including commercially important coniferous and other hardwood timber stands (Mathiasen, Nickrent, Shaw, & Watson, 2008). Because of this, the current perception of mistletoes as invasive pests, damaging to individual trees and detrimental to forest health, and noxious weeds needs to be challenged and rather understood as an indicator of habitat health, or in superabundance as a signal of landscape perturbation (Norton & Reid, 1997; Watson, 2001).
In this study, we examine chloroplast DNA (cpDNA) sequence variation to explain genetic diversity in
Methods
Population Sampling
Geographic Information and Sample Sizes of the
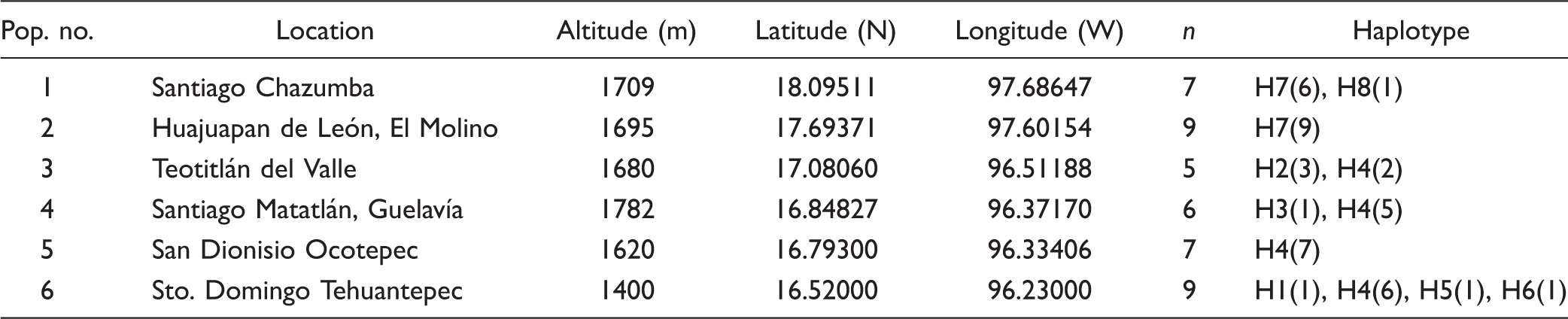
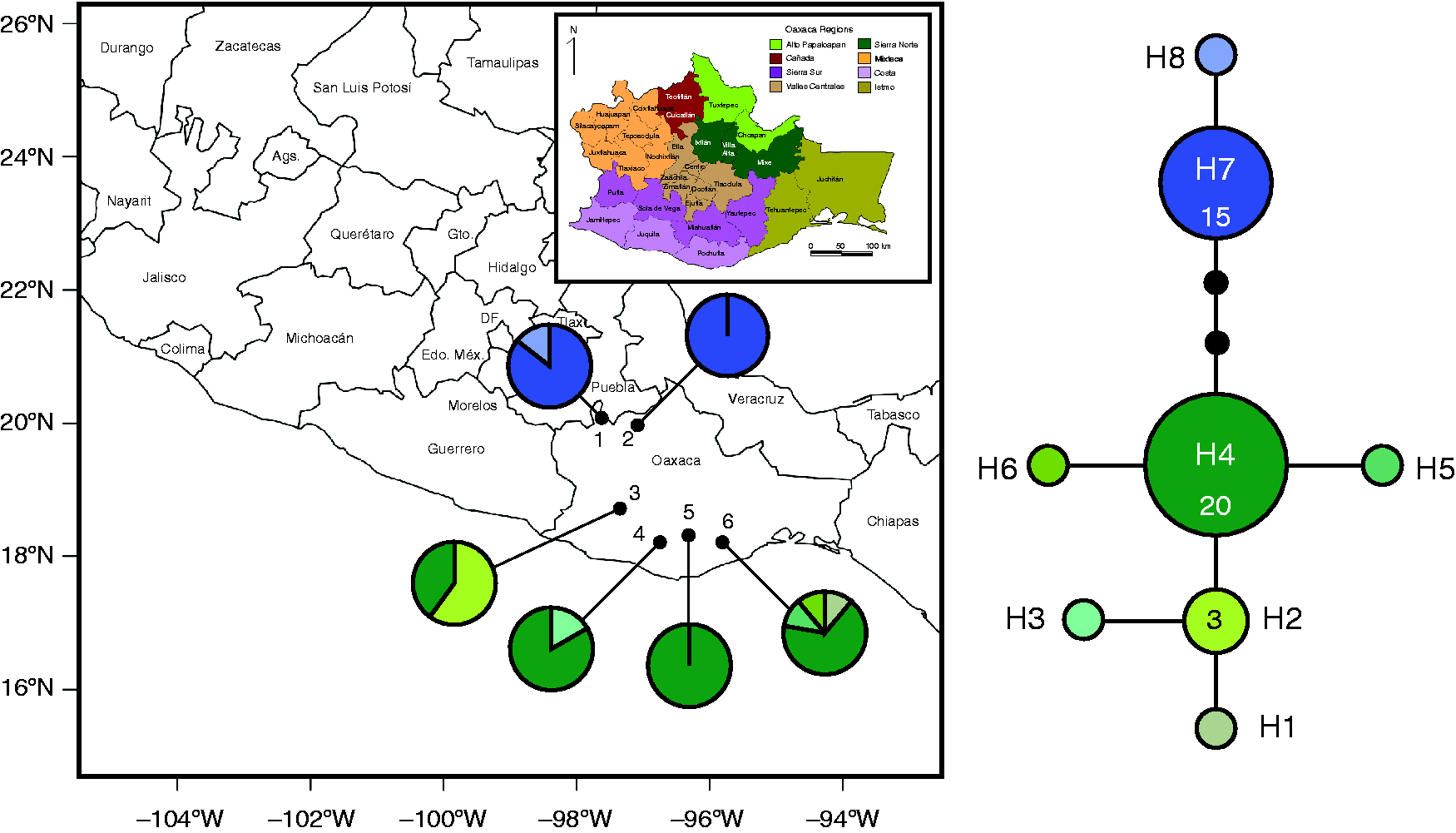
Collection localities (population numbers as in Table 1) and geographical distributions of 8 chloroplast haplotypes (
Chloroplast Sequencing
Total genomic DNA was extracted from silica-dried material using a modified 2×cetyl trimethyl ammonium bromide protocol (Doyle & Doyle, 1987) or the DNeasy Plant Mini kit (Qiagen, Valencia, CA, USA) using the manufacturer protocol. Sequences of noncoding spacer region that lies between the large subunit of ribulose-1, 5-bisphosphate-carboxyalse (rbcL) and the beta-subunit of the chloroplast ATP-synthase (atpB; hereafter
Haplotype Relationships
To infer genealogical relationships among haplotypes, a statistical parsimony network for the combined
Genetic Diversity and Population Structure
Average gene diversity within populations (
To determine whether or not populations are structured, two analyses of molecular variance (AMOVAs; Excoffier, Smouse, & Quattro, 1992) were performed based on pairwise differences using ARLEQUIN with populations treated as (a) a single group to determine the amount of variation partitioned among and within populations and (b) grouped into northern or southern according to parsimony network results. Locus-by-locus AMOVAs were performed using the Jukes and Cantor model for
Demographic History
Signatures of demographic changes or selection in the recent history of
Ecological Niche Modeling
The observed demographic patterns of arid land plant populations were tested for
We first constructed a model to the present using all climatic variables with 70% of the locality records as training data and the other 30% as testing data in addition to response curves, jackknife tests, logistic output format, and random seed parameters. We ran 1,000 iterations with no extrapolation in order to avoid artificial extrapolations from extreme values of the ecological variables; as such parameters are biased toward the environmental envelope of background points and occurrence data (Phillips et al., 2006). All other parameters in MAXENT were maintained at default settings. The logistic format was used to obtain the values of habitat suitability and the variable importance was determined comparing percentage contribution values and jackknife plots. Then, correlation coefficients between present variables were calculated using PAST 2.12 (Hammer, 2011) with the objective to identify and remove the variables that were highly correlated (correlation values ≥ 0.8) and less explanatory, having as a rule of decision, the relative contributions of each of them in the MAXENT model. After removing the highly correlated variables, five variables were used in the final analysis for the mistletoe (BIO6 = Minimum Temperature of Coldest Month, BIO14 = Precipitation of Driest Month, BIO15 = Precipitation Seasonality, BIO16 = Precipitation of Wettest Quarter, BIO19 = Precipitation of Coldest Quarter). The models were calibrated using the biogeographic provinces (Morrone, 2005, 2014) as hypothesis of the accessibility area of
Final models were constructed with 10 cross-validation replicates without clamping and extrapolation, and considering the average output grids as the final predictive models. To convert initial model outputs to binary predictions, we used the equal training sensitivity and specificity threshold over the average predicted suitability across replicates and we calculated the sensitivity (the proportion of observed presences in relation to those that were predicted, which quantifies the omission errors), the specificity (the proportion of observed absences compared to those that were predicted, which quantifies the commission errors), the overall accuracy, and the TSS (True Skill Statistic; Allouche, Tsoar, & Kadmon, 2006). The TSS test corrects the overall accuracy of the model prediction by the accuracy expected by chance. This test provides a score between −1 and +1, with values > 0.6 considered good, 0.2 to 0.6 fair to moderate, and <0.2 poor (Jones, Acker, & Halpern, 2010). We also used a threshold-independent method of model validation, the receiver operating characteristic (ROC) curve analysis. The ROC analysis characterizes the predictive performance of a model at all possible thresholds by a single number, the area under the curve (AUC; Phillips et al., 2006). The value of the AUC is between 0.5 and 1.0. If the value is 0.5, the model is no better than random; an area with a value equal or close to 1 indicates a good model (Fielding & Bell, 1997).
Environmental Variation
Measurements of temperature and precipitation (BIO 1–19 variables) were extracted for each location with occurrence of
Results
Haplotype Relationships
We obtained sequences of the chloroplast intergenic spacers
Statistical parsimony retrieved a well-resolved network in which two haplogroups could be distinguished for
Genetic Diversity and Population Structure
Differentiation among populations based on
Results of Locus by Locus AMOVA Models on
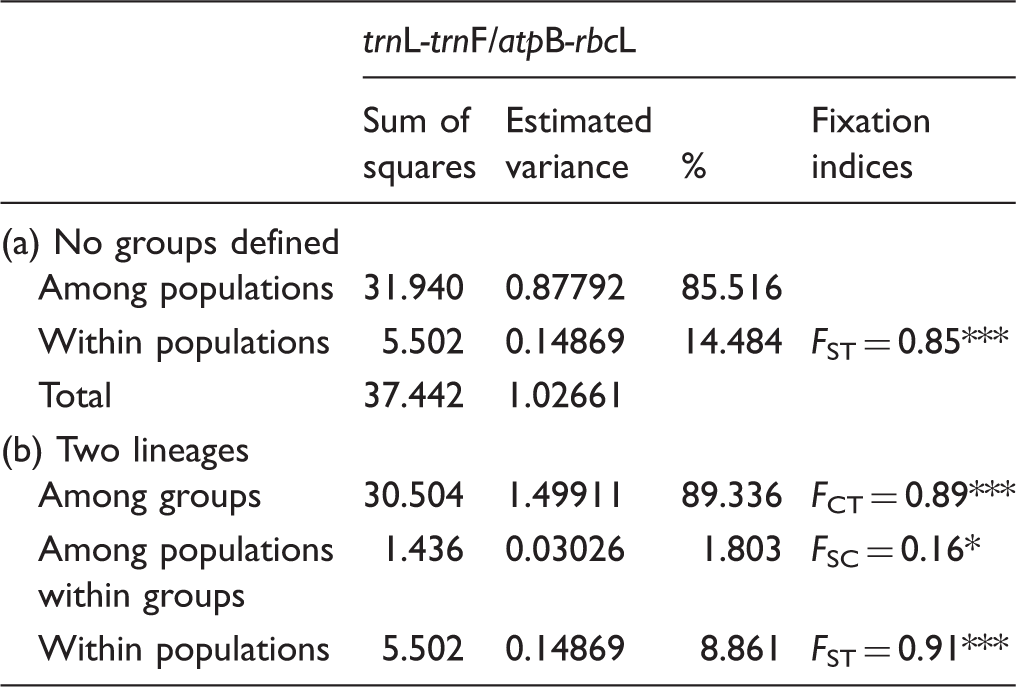
*
Summary Statistics of Neutrality Tests and Demographic Analysis of

Demographic History
When populations are treated as two groups (northern and southern), the Tajima's
Ecological Niche Modeling
The ENM yielded a good fit for the current geographic distribution of Results from the MAXENT analyses showing binary transformation of ecological niche models for 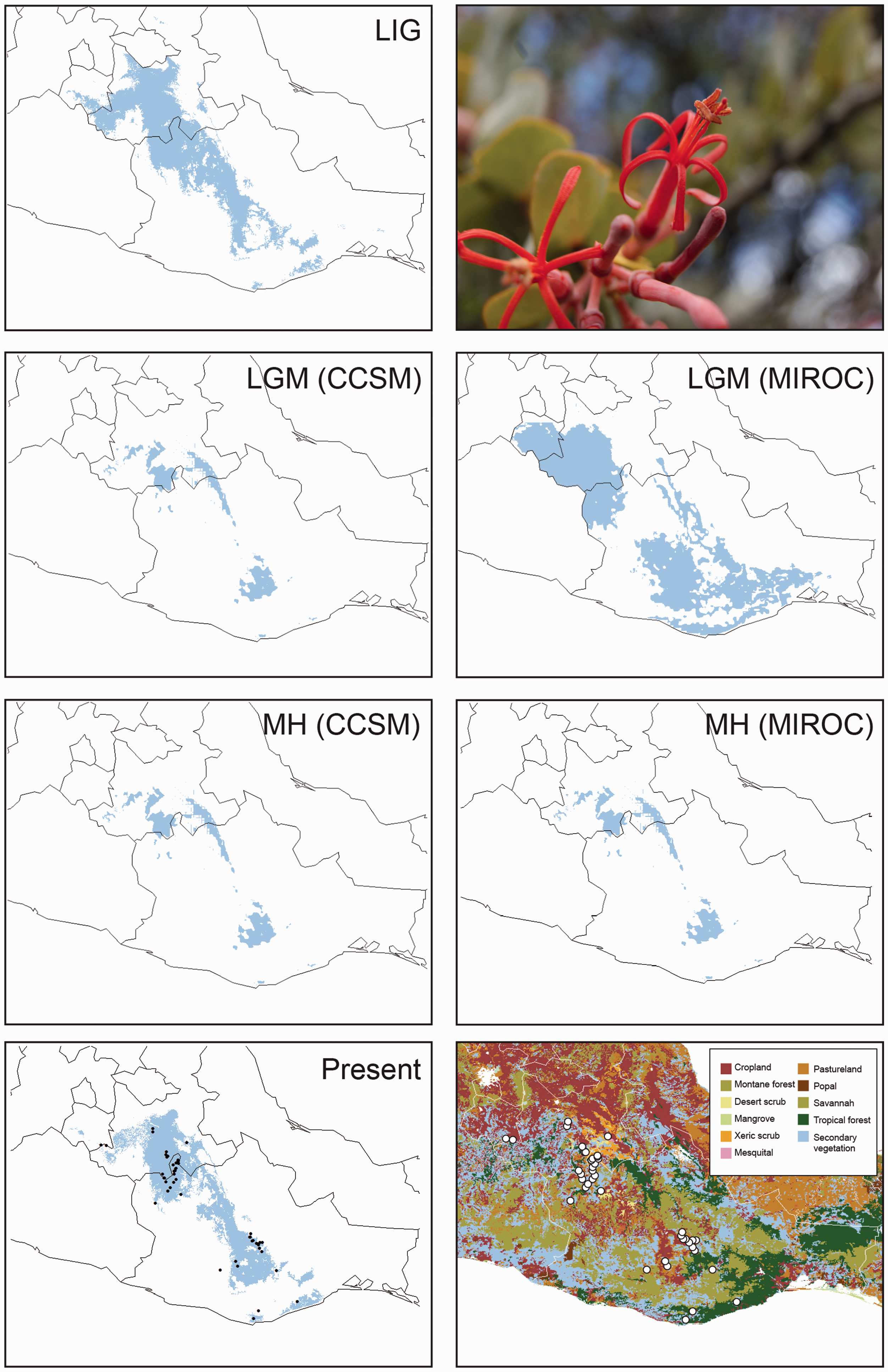
Neither CSSM nor MIROC scenarios for 2050 and 2070 predicted major future distributional change for Predicted distribution of 
Environmental Variation
Factor Loadings from the Principal Components Analysis of

Discussion
Genetic Diversity and Population Structure
Our survey of chloroplast sequence data in populations of
An alternative explanation is that the observed haplotype distribution in
Population Glacial Refugia
The levels of genetic differentiation found in
Palynological data from the Eocene Mequitongo Formation (Ramírez-Arriaga, Prámparo, Nieto-Samaniego, & Valiente-Banuet, 2017) and the Tehuacán Formation geochronologically dated as Middle Miocene (Ramírez-Arriaga et al., 2014) suggest that conditions were more humid than those that exist today in the Tehuacán-Cuicatlán Valley, a xeric region north of the central valleys of Oaxaca. The overall palynoflora suggests that climatic conditions led to the development of temperate
Implications for Conservation
Despite their positive role as ecological keystones in many forests and woodlands (Watson, 2001; Watson & Herring, 2014) and facilitative effects of hemiparasitic mistletoes on community structure (March & Watson, 2010; Ndagurwa, Dube, & Mlambo, 2014; Watson, 2015; Watson & Herring, 2012), the loss of mistletoe species should not represent intuitively a threat to associated hosts and could even provide benefit (Strona, 2015; Strona, Galli, & Fattorini, 2013). For instance, the extinction risk of
The results of the study are interesting from a conservation standpoint, as we provided the first estimate of genetic variability and structure of the short-range endemic
Footnotes
Acknowledgments
We thank Cristina Bárcenas, Andrés Ortíz-Rodríguez, Santiago Ramírez-Barahona, María José Pérez-Crespo, Etelvina Gándara, Alfonso Langle, Flor Rodríguez-Gómez, Sergio Díaz Infante, Hellen Martínez Roldán, and Sandra Rodríguez Mendieta who helped in obtaining samples and generated sequences for this work, and the municipal and community authorities of Santiago Matatlán, Oaxaca for logistic support. Permission to conduct our fieldwork was granted by the Mexican government (Instituto Nacional de Ecología, Secretaría del Medio Ambiente y Recursos Naturales, SGPA/DGGFS/712/1299/12).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by research funds from the Departamento de Biología Evolutiva, Instituto de Ecología, AC (20030/10563) and a competitive grant (grant number 155686) from the Consejo Nacional de Ciencia y Tecnología (CONACyT; ![]() ) awarded to Juan Francisco Ornelas. Y. Licona-Vera was supported by doctoral scholarship (262561) from CONACyT.
) awarded to Juan Francisco Ornelas. Y. Licona-Vera was supported by doctoral scholarship (262561) from CONACyT.
