Abstract
This paper classifies the vegetation types that comprise the habitat of the white-tailed deer
Introduction
The characterization, classification and evaluation of the habitat of the white-tailed deer
The white-tailed deer is the principal game species in North America, where its management and monitoring are common practices [1]. In contrast, few data exist concerning the ecology of this species in Neotropical habitats [10, 11]. In this region, the white-tailed deer is important because it is used by indigenous and rural people to complement their diet and, in some areas, as a commercial and game trophy [12]. However, intense hunting pressure and habitat loss have negatively affected the population of this deer in the tropics [13]. Monitoring of populations, habitat, and human factors that affect this species is therefore fundamental to appropriate management and conservation strategies. At specific sites, management practices are known by different names, depending on the country; for example, “
The white-tailed deer is one of the most prized species for subsistence and trophy hunting in different regions in Mexico [15–18]. In the UMAs scheme, this deer represents one of the main species exploited in the tropical regions [19–21]. This species inhabits the Tehuacan-Cuicatlan Biosphere Reserve (TCBR) [22, 23], which is highly important for biodiversity conservation [24]. In this region, both subsistence and trophy hunting are common practice. Previously, Ramos-Robles et al. [23] analyzed habitat-density relationships in four locations in the TCBR in order to determine the habitat (vegetation and topographic) and human variables associated with deer density. In this paper, we present: 1) a classification of vegetation types using multivariate cluster analysis and a Geographic Information System; 2) a comparison of the population density of white-tailed deer based on the maps used to classify vegetation types; and 3) suggestions for the management of this species in the study area, which is one of the first UMA in the TCBR where sustainable use of white-tailed deer is practiced.
Methods
Study area
The study was conducted on the communal lands of San Gabriel Casa Blanca in the municipality of San Antonio Nanahuatipam in the state of Oaxaca, Mexico (17° 39′ - 18° 53′ N, 96° 55′ - 97° 44′ W; Fig. 1a). The site is located within the region La Cañada on its border with the Oaxacan Mixteca. The community of approximately 5,700 ha forms part of the Tehuacan-Cuicatlan Biosphere Reserve (TCBR), which covers an area of 490,187 ha in the southeast of the state of Puebla and northeast of Oaxaca [24]. The community features a semi-desert climate with an average temperature of 24° C. Rainfall occurs in summer, and average annual precipitation is 438 mm. The municipality of San Antonio Nanahuatipam belongs to the Papaloapan region; the Salado and Calapa rivers are the largest watercourses, providing a permanent supply of water for the community and wildlife. The area has three permanent water channels, which are utilized for domestic use and irrigation. The site consists of rough terrain between canyons, hills, and mountains such as Nanahualtepec, Cihualtepec, and Petlanco. Land use in the municipality is composed of 8% agriculture, 1% urban, 63% tropical dry forest, and 28% crassicaule scrub. The land is distributed among small common and private property; 80% of the population is mainly engaged in cultivating sugar cane, tomatoes, corn, beans and melons, and raising cattle and goats [25].

Location of the Tehuacán-Cuicatlán Biosphere Reserve in Oaxaca and Puebla states, Mexico and studied site (a). Details of the vegetation classification by INEGI [26] in the studied location (b). Spatial distribution of the vegetation types and zones (A to D) according to GIS analysis and location of sampled transects (1-28) for plant description (c).
According to the Forest Series III inventory by INEGI [26], San Gabriel Casa Blanca has two main vegetation types: tropical dry forest and crassicaule scrub. Tropical dry forest is characterized by the dominance of woody plants of 8 to 10 m in height, with the dominant trees losing their foliage during the dry season (Fig. 1b). The most characteristic species are: tetcho (
Geographic Information System analysis
According to studies of land use and vegetation cover [24, 26], there are two vegetation types in the study area, but according to our objectives and field observations this information is inadequate. We therefore evaluated the patterns of land use with Landsat 8 images in the GIS Idrisi v. 16 using the modules
Vegetation sampling
During the dry season (March to May) of 2010, we established 28 transects (500 m) for vegetation characterization following the general procedures used in other studies of this deer species [23, 29–32]. Eleven sampling units were established every 50 m along each transect for a total of 308 sampling points. At each point, we sampled the trees using the point-centered quarter method to estimate: 1) tree variables (species composition, richness, Shannon diversity index, basal area, height and density) and 2) understory variables (species richness, and protection cover at 0–50 cm and 51–100 cm). We sampled in circular plots of radius 2 m in order to determine the number of species [31].
Multivariate analysis
To determine different vegetation types in the study area, we used hierarchical agglomerative clustering methods on the species abundance data per transect through multivariate cluster analyses. The original matrix was 28 transects per 58 tree species; however, for the purposes of the cluster analysis, we eliminated those species with fewer than 10 individuals and grouped the species of the genus
To interpret and compare the hierarchical clustering results, we used the cophenetic correlation, Shepard-like diagrams and Gower's distance index [9]. To identify interpretable clusters (k-groups), we decided at what level to cut the dendograms by plotting the fusion level values for each clustering method. In addition, we explored the silhouette width graphs as a measure of the degree of membership of each transect to its cluster, using the function
Statistical analysis
We present the results and graphs of the clustering that we considered the most ecologically interpretable according to the previous criteria. However, in some cases, two clustering methods and different dendrogram cutting values produced relatively different numbers of groups. The spatial plot of the clustering results was therefore superimposed on the vegetation type map obtained by GIS, in order to compare the k-groups (clusters) obtained with multivariate analysis. To test differences in vegetation variables (tree density, tree height, tree basal area, tree richness, understory cover and understory richness) among vegetation types (clusters), we used one-way analysis of variance [33]. We tested ANOVA assumptions using the Saphiro-test to confirm normality of the residuals and the Bartletttest to determine the homogeneity of variances. Where these tests were rejected, we used the non-parametric Kruskal-Wallis test. All analyses were run in R version 2.15.3 [34].
Deer population density
To estimate the spatial and temporal variation in population density of white-tailed deer, we used the indirect method of counting pellet-groups [35]. We surveyed 28 strip-transects (500 × 2 m) during the dry season of 2010. Pellet-groups could be collected following the fecal standing crop (FSC) or re-sampling the same plots two or more times (FAR, Fecal Accumulation Rate). Previously, Camargo-Sanabria & Mandujano [35] discussed the advantages and limitations of the FAR and FSC for census pellets in other tropical dry forests. In this study, we used the FSC method, visiting each sampling plot only once and counting the total standing crop of fecal material accumulated during the dry season. According to previous studies in tropical dry forests [19, 20, 23, 30, 32, 35], fecal loss is minimal during the dry season, whereas in the rainy season, the fecal decay rate is almost 100% from June to December. Moreover, visibility is low in the rainy season, as understory plants are at their most dense and the probability of detection of feces is therefore lower. Surveys were carried out by two or three persons.
To estimate population density, we employed PELLET version 2.1, which is a semi-automatized procedure in Excel ® [36]. PELLET approaches density calculation by assuming that density has a probabilistic distribution depending on variation in the parameters of defecation rate, time of persistence of the fecal pellet-groups, and the pattern or spatial distribution of the fecal pellet-groups. For statistical analysis, the density estimated in each transect was grouped according to criteria: the vegetation types defined by the INEGI [18] and the clusters obtained by multivariate analysis and SIG analysis. For each case, we compared the density using one-way ANOVA and the post-hoc Tukey HSD test in the program R.
Results
Vegetation map
Identification and mapping of different vegetation types were achieved through SIG considering the spatial distribution of the principal tree species, slopes, watercourses, human activities and other landscape elements (Fig. 1c). According to this SIG and the distribution of the principal trees, we defined the following vegetation types: tropical dry forest (TDF), dominated by

Landscape photographs of typical forest dominated by the columnar cactus tetecho (
Vegetation characterization
We counted a total of 1,232 trees of 58 species belonging to 20 plant families. The most frequently represented families were Cactaceae (12 spp.) and Burseraceae (10 spp.). Six families had two to four species, while 12 families were represented by a single species. The genera and species with the highest numbers of individuals were

Spatial distribution of the tree genus with the highest abundance (61.1% from 1,232 trees) in the 28 sampled transects, and slope variation in the studied site (right-down graphic). The size of the circle is proportional to the number of individuals of each species and slope (in red).
Vegetation classification
The four clustering methods (single linkage, complete-linkage, UPGMA and Ward) produced slightly different dendrograms. Calculation of the cophenetic distance coefficient suggests that UPGMA was the optimum clustering method for the matrix data. According to the final dendrogram, which produced four groups or clusters of transects (Fig. 4), the heat map of the double ordered matrix (Fig. 5), the spatial distribution of the 28 transects classified in the four groups (Fig. 6), the SIG analysis (Fig. 1c), and the abundance distribution of the principal genus (Fig. 3), the following is a general description of each cluster:
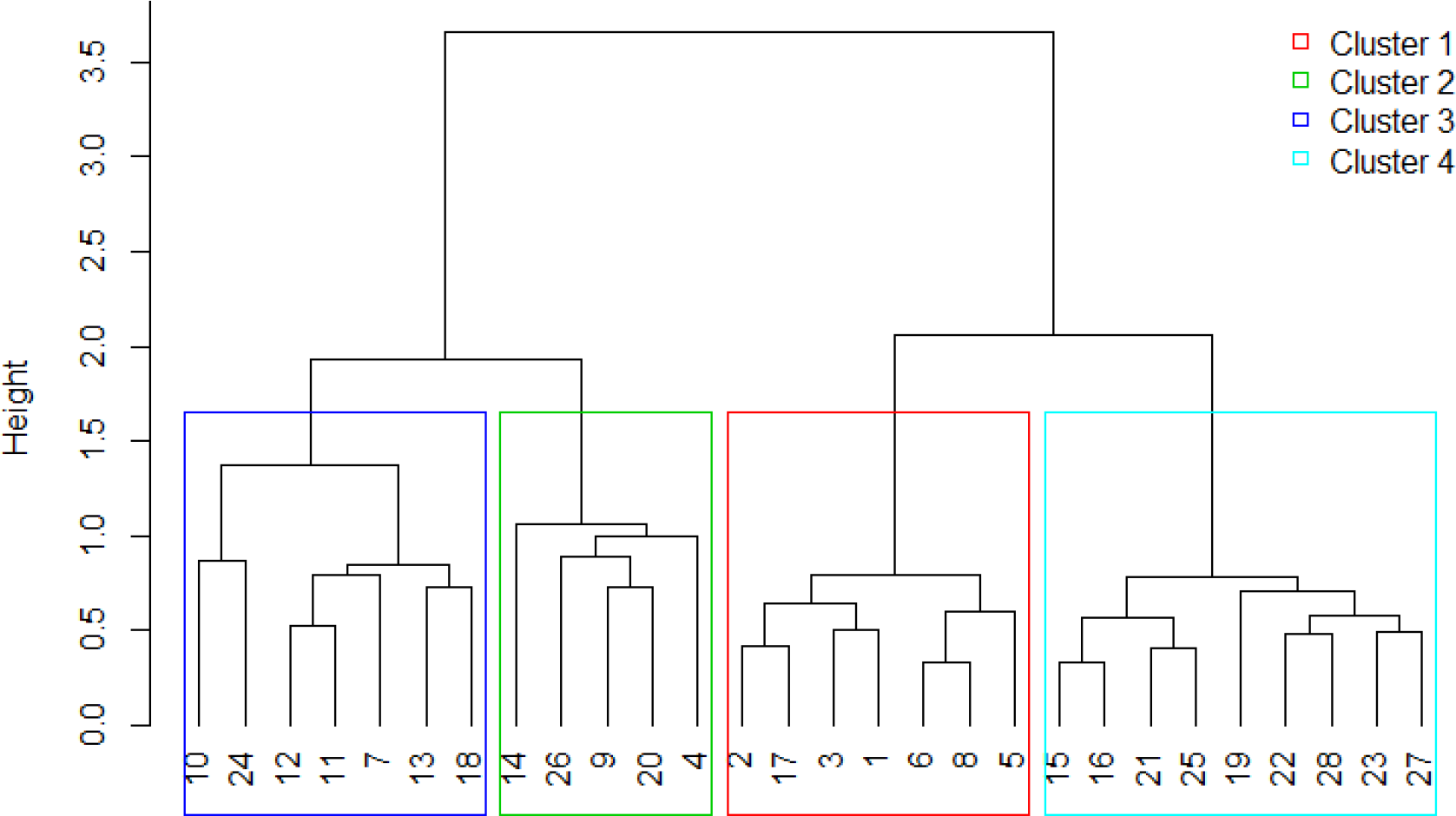
Final dendrogram with the boxes around the four classified groups of transects using the UPGMA average agglomerative clustering method.

Heat map of the double ordered matrix (28 sites × 21 species). Dark green represents higher abundance while light green lower. Abbreviations: escobillo

Map of the spatial distribution of the 28 transects classified in the four groups. Colors are according to the boxes in the Fig. 4.
Vegetation types differences
Vegetation variables varied significantly among the clusters (Fig. 7). Tree density was higher in clusters 2 and 4 (F = 3.857, df = 3, 24, P = 0.02). Tree height was higher in clusters 1 and 4 due to the presence of the columnar cactus as

Histograms of the evaluated variables in the clusters. Colors are according to the groups in previous figures. Statistical differences (ns, *, ***).
Deer population density
We counted 285 pellet-groups in 86% of the 28 transects sampled. Mean (±SE) density was 3.5 ± 0.6 ind/km2. When transects were classified according to the INEGI's classification, the densities were 4.1 and 2.7 ind/km2 for crassicaule scrub and tropical dry forest, respectively; these differences were significant (Fig. 8a;

Population density (ind/km2) of white-tailed deer grouping transects by vegetation according to the INEGI classification (a) and clusters obtained in multivariate analysis (b). The boxplot shows the quartiles and medians. Colors are according to the groups in previous figures. See the different maps of vegetation classification in the Fig. 1.
Discussion
A primary goal of vegetation classification is to arrange vegetation patterns into an ecologically meaningful set of types, and a secondary goal is its ability to serve as a guide for mapping [7]. Multivariate vegetation classification separated a large heterogeneous region into smaller and more homogeneous entities, in terms of land use [2]. According to the INEGI forest classification [26], the study area mainly presents crassicaule scrub while the tropical dry forest (TDF) is located in the hills of a portion of this site (see Fig. 1b). However, our results based on SIG and multivariate analysis of the field data suggest a completely different scenario (see Fig. 1c). The main finding is that crassicaule scrub is in fact limited to small patches, while the TDF dominated the study area and can be sub-classified into three or four types according to tree species composition and dominance, principally of the genera
To understand these results, it is important to compare these defined clusters with other community descriptions found in similar locations and how they are associated with the habitat and resources used by the white-tailed deer in tropical dry forests. According to the data, the 28 sampled transects were classified into four clusters depending on tree dominance. Clusters 1 and 4 correspond to vegetation dominated by
Cluster 2 is dominated by
In contrast, Cluster 3 is co-dominated by
The TDF dominated by
Importantly, because we sampled only during the dry season it is possible that the deer-habitat association could change during the rainy season. Pellet-group count is not appropriate for use during the rainy season due to the decomposition rate of feces [35]. However, preliminary analysis of both site occupation and activity patterns using cameratraps during the dry and rainy seasons suggests that deer use the same sites year-around [Mandujano et al. in preparation, 47]. Future studies using radio-tracking could give specific data of deer movements and changes in habitat use [see 48].
In comparison with other locations in the TCBR, the study site presents a relatively higher density of white-tailed deer [49]. However, comparison between the mean estimated density (3.5 ± 0.6 ind/km2) and data obtained in other tropical dry forests in the Central and Pacific region of Mexico [30, 32, 50] suggests that the TDF of the studied region in fact supports lower densities of white-tailed deer. The influence of the relatively low precipitation in the lower elevations of the TCBR could explain this pattern, as has been suggested in a large-scale geographical analysis by Mandujano and Naranjo [51]. Another possibility is that the actual density in the studied location could be lower than the carrying capacity (K) [50]. Therefore, it is necessary to evaluate biomass production and quality of the principal plant species consumed by this deer species [52].
Implications for conservation
The TDF is the dominant vegetation type that offers better habitat conditions and important food resources for the white-tailed deer in this location. Activities for the protection of deer have been conducted in the study site for several years, certainly benefitting both population and habitat. However, habitat management practices as water availability and food supplement in the critique period during the dry season, could be necessary to increase the habitat carrying capacity [53]. Our analysis of vegetation classification and the variation of white-tailed deer densities suggests that the study area could be a suitable site for practicing legal hunting, through the extensive model UMA [54].
Footnotes
Acknowledgements
The study received financial and logistical support of the Red de Biología y Conservación de Vertebrados del Instituto de Ecología A.C., PROCODES of CONANP-RBTC and CONACYT project CB-2009-01 No. 130702. The following people helped us in different aspects: J. C. Pizaña Soto, J. M. Salazar Torres, A. Vasquez, T. Pérez-Pérez, R. Rodriguez, J. C. Castillo, L. A. Escobedo-Morales, M. I. Ramos-Robles, A. Mendez, J. A. Vergara, E. López-Tello, B. Ramirez and C. López-Téllez. We thank the authorities and people of the commons of San Gabriel Casa Blanca. K. McMillan reviewed the English translation.
